Abstract
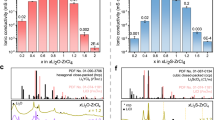
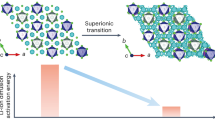
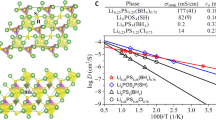
The design of superionic conductors has been largely focused on static structural features, with the dynamic ion transport mechanism less explored. Here, we explore a paradigm that harnesses the polyanion rotations to trigger the dynamically disordered Li sublattice as well as the liquid-like cation diffusion for superior ionic conductivity in crystals. A descriptor called rotation tolerance factor was proposed as a predictive metric for identifying the potential fast-rotating anion clusters with the low mass and reduced valence charge for given structural frameworks. Guided by this factor, halides with rotational polyanions, namely Li3Y(SH)6, Li3Y(NH2)6, Li2Zr(NH2)6, and an oxide (Li6.5La3Zr2O11.5(NH2)0.5) have been designed with synergistic polyanion rotation and Li⁺ sublattice disorder, which lead to enhanced Li ionic conductivities at room temperature compared to the counterparts without polyanions. The experimentally synthesized NH2- incorporated Li2ZrCl5.92(NH2)0.08 demonstrates a four-fold higher conductivity over Li2ZrCl6 control, enabling all-solid-state Li-In | |LiCoO2 and Li-In | |LiNi0.88Co0.09Mn0.03O2 cells with 96.5% and 97.4% capacity retention after 190 cycles at 140 and 200 mA g-1, respectively. This work provides an insight that flexible anion rotations could promote the dynamically disordered lithium sublattice distribution as well as the ionic conductivity.
Similar content being viewed by others
Data availability
The data generated in this study are provided in the Supplementary Information/Source Data file. Source data provided with this paper. Source data are provided with this paper.
Code availability
The machine learning molecular dynamics simulations were performed using open-source software packages, including LAMMPS, MLIP, Pymatgen and MAML, available at https://www.lammps.org, https://instadeepai.github.io/mlip/http://pymatgen.org and https://github.com/materialyzeai/maml. Additional scripts, structures, and relevant details are available at https://doi.org/10.5281/zenodo.18802601.
References
Jun, K. et al. Lithium superionic conductors with corner-sharing frameworks. Nat. Mater. 21, 924–931 (2022).
Restle, T. M. F. et al. Fast lithium ion conduction in lithium phosphidoaluminates. Angew. Chem. 59, 5665–5674 (2020).
Liu, H. et al. Copper ion liquid-like thermoelectrics. Nat. Mater. 11, 422–425 (2012).
Zhang, L. et al. Recent advances of Li7La3Zr2O12-based solid-state lithium batteries towards high energy density. Energy Storage Mater. 49, 299–338 (2022).
Kato, Y. et al. High-power all-solid-state batteries using sulfide superionic conductors. Nat. Energy 1, 16030 (2016).
Yin, Y.-C. et al. A LaCl3-based lithium superionic conductor compatible with lithium metal. Nature 616, 77–83 (2023).
Wang, Y. et al. Liquid-like solid-state diffusion of lithium ions in super-halide-rich argyrodite. Cell Rep. Phys. Sci. 5, 102314 (2024).
Wu, J. F. et al. Liquid-like Li-ion conduction in oxides enabling anomalously stable charge transport across the Li/electrolyte interface in all-solid-state batteries. Adv. Mater. 35, e2303730 (2023).
Wang, S. et al. Lithium chlorides and bromides as promising solid-state chemistries for fast ion conductors with good electrochemical stability. Angew. Chem. 58, 8039–8043 (2019).
Wang, Q. et al. Designing lithium halide solid electrolytes. Nat. Commun. 15, 1050 (2024).
Xu, Z. & Zhu, H. Anion charge and lattice volume maps for searching lithium superionic conductors. Chem. Mater. 32, 4618–4626 (2020).
Wang, Y. et al. Design principles for solid-state lithium superionic conductors. Nat. Mater. 14, 1026–1031 (2015).
Xu, Z., Chen, X., Chen, R., Li, X. & Zhu, H. Anion charge and lattice volume dependent lithium ion migration in compounds with fcc anion sublattices. npj Comput. Mater. 6, 47 (2020).
Wang, C., Cheng, R. & Chen, Y. Theoretical evaluation of the persistence of transverse phonons across a liquid-like transition in superionic conductor KAg3Se2. Chem. Mater. 35, 1780–1787 (2023).
Ding, J. et al. Liquid-like dynamics in a solid-state lithium electrolyte. Nat. Phys. 21, 118–125 (2025).
Nield, V. M., Keen, D. A., Hayes, W. & McGreevy, R. L. Structure and fast-ion conduction in α-AgI. Solid State Ion. 66, 247–258 (1993).
Vivas, E. I., Peña Lara, D. & Alvaro, G. M. Structural and transport properties for the superionic conductors AgI and RbAg4I5 through molecular dynamic simulation. Ionics 27, 781–788 (2020).
Bernges, T. et al. Considering the role of ion transport in diffuson-dominated thermal conductivity. Adv. Energy Mater. 12, 2200717 (2022).
Morscher, A. et al. Control of ionic conductivity by lithium distribution in cubic oxide argyrodites Li6+xP1-xSixO5Cl. J. Am. Chem. Soc. 144, 22178–22192 (2022).
Pogodin, A. I. et al. The copper argyrodites Cu7-nPS6-nBrn: crystal growth, structures and ionic conductivity. Solid State Ion. 341, 115023 (2019).
Zeng, Y. et al. High-entropy mechanism to boost ionic conductivity. Science 378, 1320–1324 (2022).
Wang, S. et al. High-entropy strategy flattening lithium ion migration energy landscape to enhance the conductivity of garnet-type solid-state electrolytes. Adv. Funct. Mater. 35, 2416389 (2024).
Matsui, N. et al. Effect of Pb 6s2 lone pair on the potential flattening of fluoride-ion conduction in perovskite-type fluoride. J. Mater. Chem. A 12, 3989–3996 (2024).
Schwietert, T. K. et al. Understanding the role of aliovalent cation substitution on the li-ion diffusion mechanism in Li6+xP1-xSixS5Br argyrodites. Mater. Adv. 5, 1952–1959 (2024).
Hallett, J. E., Turci, F. & Royall, C. P. Local structure in deeply supercooled liquids exhibits growing lengthscales and dynamical correlations. Nat. Commun. 9, 3272 (2018).
Xu, Z. et al. Machine learning molecular dynamics simulation identifying weakly negative effect of polyanion rotation on Li-ion migration. npj Comput. Mater. 9, 105 (2023).
Guan, C. et al. Unlocking the chemical space in anti-perovskite conductors by incorporating anion rotation dynamics. Energy Storage Mater. 62, 102936 (2023).
Wang, C., Wu, Y., Pei, Y. & Chen, Y. Dynamic disorder phonon scattering mediated by Cu atomic hopping and diffusion in Cu3SbSe3. npj Comput. Mater. 6, 155 (2020).
Wan, T. H. & Ciucci, F. Ab initio study of the defect chemistry and substitutional strategies for highly conductive Li3YX6 (X = F, Cl, Br, and I) electrolyte for the application of solid-state batteries. ACS Appl. Energy Mater. 4, 7930–7941 (2021).
Zhang, H. et al. Li-richening strategy in Li2ZrCl6 lattice towards enhanced ionic conductivity. J. Energy Chem. 79, 348–356 (2023).
Jun, K., Lee, B., Kam, R. & Ceder, G. The nonexistence of a paddlewheel effect in superionic conductors. Proc. Natl. Acad. Sci. USA 121, e2316493121 (2024).
Wang, K. et al. A cost-effective and humidity-tolerant chloride solid electrolyte for lithium batteries. Nat. Commun. 12, 4410 (2021).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. b 54, 11169–11186 (1996).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Brehm, M., Thomas, M., Gehrke, S. & Kirchner, B. TRAVIS-A free analyzer for trajectories from molecular simulation. J. Chem. Phys. 152, 164105 (2020).
de Klerk, N. J. J., van der Maas, E. & Wagemaker, M. Analysis of diffusion in solid-state electrolytes through MD simulations, improvement of the Li-ion conductivity in beta-Li3PS4 as an example. ACS Appl. Energy Mater. 1, 3230–3242 (2018).
de Klerk, N. J. J., Rosłoń, I. & Wagemaker, M. Diffusion mechanism of Li argyrodite solid electrolytes for Li-Ion batteries and prediction of optimized halogen doping: the effect of Li vacancies, halogens, and halogen disorder. Chem. Mater. 28, 7955–7963 (2016).
Ong, S. P. et al. Python Materials Genomics (pymatgen): a robust, open-source python library for materials analysis. Comput. Mater. Sci. 68, 314–319 (2013).
He, X., Zhu, Y., Epstein, A. & Mo, Y. Statistical variances of diffusional properties from ab initio molecular dynamics simulations. npj Comput. Mater. 4, 18 (2018).
Qi, J. et al. Bridging the gap between simulated and experimental ionic conductivities in lithium superionic conductors. Mater. Today Phys. 21, 100463 (2021).
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (52573251, 523B2106, 52072240), Science and Technology Commission of Shanghai Municipality (23DZ1200800), the National Key R&D Program (2025YFF0516300) and the Materials Genome Initiative Center at Shanghai Jiao Tong University. All simulations were carried out with computational resources from Shanghai Jiao Tong University High Performance Computing Center.
Author information
Authors and Affiliations
Contributions
C.G., H.Z. conceived the project and designed the simulations and experiments. J.Z, J.L. and Y.Z. conducted the experiments. C.G. carried out DFT simulations and data analysis. R.O. assisted with the analyses. C.G. wrote the manuscript. F.H. and H.Z. supervised the project. All authors contributed to the review and editing of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Shumin Zhang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Guan, C., Zong, J., Li, J. et al. Triggering dynamically disordered lithium sublattice in superionic conductors. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71304-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71304-3