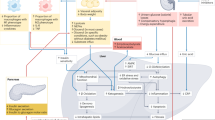
Abstract
Cardiovascular-kidney-metabolic (CKM) syndrome, driven by interlinked metabolic, renal and cardiovascular dysfunction, remains therapeutically challenging due to its multi-organ complexity. Although sodium-glucose cotransporter 2 (SGLT2) inhibitors such as empagliflozin (EMPA) confer cardiorenal benefits, their efficacy is limited by poor renal specificity and systemic off-target exposure. Here, we report a kidney-targeted and urea-responsive nanocarrier (T-PAAD NPs) that enables renal tubule-selective release of EMPA in response to pathological urea concentrations. This delivery strategy, rarely explored in nanomedicine, synergistically enhances therapeutic precision while integrating reactive oxygen species (ROS) scavenging to mitigate oxidative stress. In male mouse models of CKM, T-PAAD NPs/EMPA effectively reprogram cardiac and renal energy metabolism, restore filtration and contractile function, and achieve superior glycemic, renal, and cardiovascular outcomes compared to free EMPA. By coupling bioresponsive controlled release with metabolic modulation, this nanoplatform provides a promising approach to treat CKM and other systemic metabolic disorders.
Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its Supplementary Information. Source data are provided with this paper. The transcriptome sequencing data generated in this study have been deposited in the NCBI Sequence Read Archive under accession code PRJNA1390588 and its corresponding hyperlink: https://www.ncbi.nlm.nih.gov/sra/PRJNA1390588.
The metabolomic data of kidney and heart have been uploaded into the MetaboLights database (https://www.ebi.ac.uk/metabolights/) (Accession Number: MTBLS13988 (kidney)[https://www.ebi.ac.uk/metabolights/MTBLS13988], MTBLS13991(heart) [https://www.ebi.ac.uk/metabolights/MTBLS13991]). Source data are provided with this paper.
References
Marassi, M. & Fadini, G. P. The cardio-renal-metabolic connection: a review of the evidence. Cardiovasc. Diabetol. 22, 195 (2023).
Khan, S. S. et al. Novel prediction equations for absolute risk assessment of total cardiovascular disease incorporating cardiovascular-kidney-metabolic health: a scientific statement from the American Heart Association. Circulation 148, 1982–2004 (2023).
Ndumele, C. E. et al. A synopsis of the evidence for the science and clinical management of cardiovascular-kidney-metabolic (CKM) syndrome: a scientific statement from the American Heart Association. Circulation 148, 1636–1664 (2023).
Xie, Z. et al. Global burden of the key components of cardiovascular-kidney-metabolic syndrome. J. Am. Soc. Nephrol. 36, 1572–1584 (2025).
Mayne, K. J. et al. Cardiovascular-kidney-metabolic syndrome and mortality in a prospective UK cohort study. Eur. J. Prev. Cardiol. zwaf514 https://doi.org/10.1093/eurjpc/zwaf514 (2025).
Ndumele, C. E. et al. Cardiovascular-kidney-metabolic health: a presidential advisory from the American Heart Association. Circulation 148, 1606–1635 (2023).
Quaggin, S. E. & Magod, B. A united vision for cardiovascular-kidney-metabolic health. Nat. Rev. Nephrol. 20, 273–274 (2024).
Claudel, S. E. & Verma, A. Cardiovascular-kidney-metabolic syndrome: a step toward multidisciplinary and inclusive care. Cell Metab. 35, 2104–2106 (2023).
O’Hara, D. V. et al. Applications of SGLT2 inhibitors beyond glycaemic control. Nat. Rev. Nephrol. 20, 513–529 (2024).
Avogaro, A., Fadini, G. P. & Del Prato, S. Reinterpreting cardiorenal protection of renal sodium-glucose cotransporter 2 inhibitors via cellular life history programming. Diab. Care 43, 501–507 (2020).
Lin, D. S. H., Lee, J.-K., Huang, K.-C., Lin, T.-T. & Lo, H.-Y. Effects of sodium-glucose cotransporter 2 inhibitors on cardiovascular and renal outcomes in people with diabetes and advanced chronic kidney disease. Diabetologia 67, 2459–2470 (2024).
Preda, A. et al. SGLT2 inhibitors: from glucose-lowering to cardiovascular benefits. Cardiovasc. Res. 120, 443–460 (2024).
Cowie, M. R. & Fisher, M. SGLT2 inhibitors: mechanisms of cardiovascular benefit beyond glycaemic control. Nat. Rev. Cardiol. 17, 761–772 (2020).
Packer, M. Critical reanalysis of the mechanisms underlying the cardiorenal benefits of SGLT2 inhibitors and reaffirmation of the nutrient deprivation signaling/autophagy hypothesis. Circulation 146, 1383–1405 (2022).
Wanner, C. et al. Empagliflozin and progression of kidney disease in type 2 diabetes. N. Engl. J. Med. 375, 323–334 (2016).
Mahmud, F. H. et al. Adjunct-to-insulin therapy using SGLT2 inhibitors in youth with type 1 diabetes: a randomized controlled trial. Nat. Med. 31, 2317–2324 (2025).
Gao, Y.-M. et al. Cardiorenal protection of SGLT2 inhibitors-perspectives from metabolic reprogramming. EBioMedicine 83, 104215 (2022).
Vallon, V. & Verma, S. Effects of SGLT2 inhibitors on kidney and cardiovascular function. Annu. Rev. Physiol. 83, 503–528 (2021).
Wheeler, D. C. et al. Effects of dapagliflozin on major adverse kidney and cardiovascular events in patients with diabetic and non-diabetic chronic kidney disease: a prespecified analysis from the DAPA-CKD trial. Lancet Diab. Endocrinol. 9, 22–31 (2021).
Romagnani, P. SGLT2 inhibitors in CKD: are they really effective in all patients? Nephrol. Dial. Transplant. 40, 1838–1842 (2025).
Hammad, R. W., Sanad, R. A.-B., Abdelmalak, N. S. & Latif, R. Cubosomal functionalized block copolymer platform for dual delivery of linagliptin and empagliflozin: recent advances in synergistic strategies for maximizing control of high-risk type II diabetes. Drug Deliv. Transl. Res. 14, 678–695 (2024).
Roointan, A., Xu, R., Corrie, S., Hagemeyer, C. E. & Alt, K. Nanotherapeutics in kidney disease: innovations, challenges, and future directions. J. Am. Soc. Nephrol. 36, 500–518 (2025).
Huang, Y., Wang, J., Jiang, K. & Chung, E. J. Improving kidney targeting: the influence of nanoparticle physicochemical properties on kidney interactions. J. Control. Rel. 334, 127–137 (2021).
Tsai, W.-H. et al. Risk of fracture caused by anti-diabetic drugs in individuals with type 2 diabetes: a network meta-analysis. Diab. Res. Clin. Pract. 192, 110082 (2022).
Musso, G., Saba, F., Cassader, M. & Gambino, R. Diabetic ketoacidosis with SGLT2 inhibitors. BMJ 371, m4147 (2020).
Dawwas, G. K., Flory, J. H., Hennessy, S., Leonard, C. E. & Lewis, J. D. Comparative safety of sodium-glucose cotransporter 2 inhibitors versus dipeptidyl peptidase 4 inhibitors and sulfonylureas on the risk of diabetic ketoacidosis. Diab. Care 45, 919–927 (2022).
Ljungberg, C. et al. Risk of urogenital infections in people with type 2 diabetes initiating SGLT2is versus GLP-1RAs in routine clinical care: a Danish cohort study. Diab. Care 48, 945–954 (2025).
Li, X. et al. Empagliflozin prevents oxidative stress in human coronary artery endothelial cells via the NHE/PKC/NOX axis. Redox Biol. 69, 102979 (2024).
Kasperova, B. J. et al. Sodium-glucose cotransporter 2 inhibitors induce anti-inflammatory and anti-ferroptotic shift in epicardial adipose tissue of subjects with severe heart failure. Cardiovasc. Diabetol. 23, 223 (2024).
Matsui, T., Sotokawauchi, A., Nishino, Y., Koga, Y. & Yamagishi, S.-I. Empagliflozin ameliorates renal and metabolic derangements in obese type 2 diabetic mice by blocking advanced glycation end product-receptor axis. Mol. Med. 31, 88 (2025).
Elbadr, M. et al. Protective and therapeutic effects of empagliflozin in nephrotoxicity induced by 5-Fluorouracil in rats: Role of caspase-3, inflammation and oxidative stress. J. Adv. Vet. Res. 14, 1201–1207 (2024).
Osman, A. T., Sharkawi, S. M. Z., Hassan, M. I. A., Abo-Youssef, A. M. & Hemeida, R. A. M. Empagliflozin and neohesperidin protect against methotrexate-induced renal toxicity via suppression of oxidative stress and inflammation in male rats. Food Chem. Toxicol. 155, 112406 (2021).
Machtakova, M., Thérien-Aubin, H. & Landfester, K. Polymer nano-systems for the encapsulation and delivery of active biomacromolecular therapeutic agents. Chem. Soc. Rev. 51, 128–152 (2022).
Beach, M. A. et al. Polymeric nanoparticles for drug delivery. Chem. Rev. 124, 5505–5616 (2024).
Sabiu, G. et al. Targeted nanotherapy for kidney diseases: a comprehensive review. Nephrol. Dial. Transplant. 38, 1385–1396 (2023).
Huang, Y. et al. Physiological principles underlying the kidney targeting of renal nanomedicines. Nat. Rev. Nephrol. 20, 354–370 (2024).
Kamaly, N., He, J. C., Ausiello, D. A. & Farokhzad, O. C. Nanomedicines for renal disease: current status and future applications. Nat. Rev. Nephrol. 12, 738–753 (2016).
Klein, J. D., Blount, M. A. & Sands, J. M. Urea transport in the kidney. Compr. Physiol. 1, 699–729 (2011).
Fenton, R. A. & Knepper, M. A. Urea and renal function in the 21st century: insights from knockout mice. J. Am. Soc. Nephrol. 18, 679–688 (2007).
Huang, S.-M. et al. Structural insights into the mechanisms of urea permeation and distinct inhibition modes of urea transporters. Nat. Commun. 15, 10226 (2024).
Vanholder, R. et al. Review on uremic toxins: classification, concentration, and interindividual variability. Kidney Int. 63, 1934–1943 (2003).
Duranton, F. et al. Normal and pathologic concentrations of uremic toxins. J. Am. Soc. Nephrol. 23, 1258–1270 (2012).
Ren, X. et al. UCST polymeric H2S-donor fabricated nanomedicine managing atherosclerosis by gas-photothermal therapy involving a “turning waste into treasure” strategy. Adv. Funct. Mater. 35, 2419295 (2025).
Yang, Z. et al. Mitochondrial-uncoupling nanomedicine for self-heating and immunometabolism regulation in cancer cells. Biomaterials 314, 122883 (2025).
Huang, Y., Jiang, K., Zhang, X. & Chung, E. J. The effect of size, charge, and peptide ligand length on kidney targeting by small, organic nanoparticles. Bioeng. Transl. Med. 5, e10173 (2020).
Janzer, M. et al. Drug conjugation affects pharmacokinetics and specificity of kidney-targeted peptide carriers. Bioconjugate Chem. 27, 2441–2449 (2016).
Shang, S. et al. Targeted therapy of kidney disease with nanoparticle drug delivery materials. Bioact. Mater. 37, 206–221 (2024).
Ostrominski, J. W. et al. Prevalence and overlap of cardiac, renal, and metabolic conditions in US Adults, 1999-2020. JAMA Cardiol. 8, 1050–1060 (2023).
McCullough, P. A., Amin, A., Pantalone, K. M. & Ronco, C. Cardiorenal nexus: a review with focus on combined chronic heart and kidney failure, and insights from recent clinical trials. J. Am. Heart Assoc. 11, e024139 (2022).
Hatamizadeh, P. et al. Cardiorenal syndrome: pathophysiology and potential targets for clinical management. Nat. Rev. Nephrol. 9, 99–111 (2013).
Suh, S. H. & Kim, S. W. Dyslipidemia in patients with chronic kidney disease: an updated overview. Diab. Metab. J. 47, 612–629 (2023).
Ferro, C. J. et al. Lipid management in patients with chronic kidney disease. Nat. Rev. Nephrol. 14, 727–749 (2018).
Sarafidis, P. A. & Tsapas, A. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N. Engl. J. Med. 374, 1092 (2016).
Kuchay, M. S. et al. Effect of empagliflozin on liver fat in patients with type 2 diabetes and nonalcoholic fatty liver disease: a randomized controlled trial (E-LIFT Trial). Diab. Care 41, 1801–1808 (2018).
Acknowledgements
This work was supported by the Young Top Talents Project of Shaanxi Sanqin Talents Special Support Program (2023STZZK09), the Fundamental Research Funds for the Central Universities (xzy012023002), the Shaanxi Fundamental Science Research Project for Chemistry & Biology (22JHQ072), the National Natural Science Foundation of China (82370726), Natural Science Foundation of Shaanxi Province (2024JC-YBMS-272), the Postdoctoral Science Foundation of Shaanxi Province (2023BSHYDZZ05), the Postdoctoral Fellowship Program of CPSF (GZC20232112). We appreciate the help from Kexin Sun (Laboratory Animal Center (LAC), Xi’an Jiaotong University, China) for assistance with echocardiographic measurement and renal doppler ultrasonography.
Author information
Authors and Affiliations
Contributions
Zhe Yang and Zhongmin Tian conceived the concept and directed the research. Xuechun Ren and Di Gao carried out material synthesis. Xuechun Ren, Di Gao, Rong Yun and Xinyang Liu performed material characterization and electrochemical tests. Xuechun Ren, Rong Yun, Chenna Di, Zeyu Hu and Xinyuan Zhang conducted the cell and animal experiments. Zhe Yang and Zhongmin Tian wrote the paper. All authors discussed the results and commented on the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Sabu Thomas, Kengo Kidokoro, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ren, X., Gao, D., Yun, R. et al. A urea-activated nanocarrier for site-specific SGLT2 inhibition and metabolic rescue against cardiovascular-kidney-metabolic syndrome. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71424-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71424-w