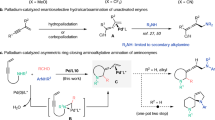
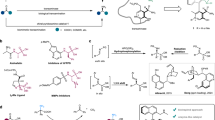
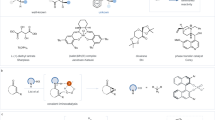
Abstract
Chiral amino acids are essential building blocks in asymmetric synthesis and drug discovery, yet their efficient preparation from racemic mixtures remains challenging. Here we show that a rationally designed phosphine oxide catalyst derived from L-pyroglutaminol enables the highly efficient kinetic resolution of racemic amino acids under mild conditions. Using L-pyroglutaminol as the esterification reagent, this catalytic system delivers a broad range of chiral esters and recovered amino acids with excellent stereoselectivities (s > 1057). Mechanistic studies suggest that the superior stereocontrol arises from a cooperative double hydrogen-bonding interaction between the catalyst and the pyroglutaminol core. This work provides a practical and scalable approach to enantioenriched amino acids, highlighting the potential of dual chiral cooperative catalysis in asymmetric synthesis.
Similar content being viewed by others
Data availability
The authors declare that the data supporting the findings of this study are available within the paper and its Supplementary Information and Supplementary Data files. Should any raw data files be needed in another format they are available from the corresponding author upon request.
References
Walsh, C. T., O’Brien, R. V. & Khosla, C. Nonproteinogenic amino acid building blocks for nonribosomal peptide and hybrid polyketide scaffolds. Angew. Chem. Int. Ed. 52, 7098–7124 (2013).
Blaskovich, M. A. T. Unusual amino acids in medicinal chemistry. J. Med. Chem. 59, 10807–10836 (2016).
Almhjell, P. J., Boville, C. E. & Arnold, F. H. Engineering enzymes for noncanonical amino acid synthesis. Chem. Soc. Rev. 47, 8980–8997 (2018).
Marchand, J. A. et al. Discovery of a pathway for terminal-alkyne amino acid biosynthesis. Nature 567, 420–424 (2019).
Li, M.-L., Yu, J.-H., Li, Y.-H., Zhu, S.-F. & Zhou, Q.-L. Highly enantioselective carbene insertion into N-H bonds of aliphatic amines. Science 366, 990–994 (2019).
Saleh, A. M., Wilding, K. M., Calve, S., Bundy, B. C. & Kinzer-Ursem, T. L. Kinzer-ursem, non-canonical amino acid labeling in proteomics and biotechnology. J. Biol. Eng. 13, 43 (2019).
Hedges, J. B. & Ryan, K. S. Biosynthetic pathways to non-proteinogenic α-amino acids. Chem. Rev. 120, 3161–3209 (2020).
Hickey, J. L., Sindhikara, D., Zultanski, S. L. & Schultz, D. M. Beyond 20 in the 21st century: Prospects and challenges of non-canonical amino acids in peptide drug discovery. ACS Med. Chem. Lett. 14, 557–565 (2023).
Eftekhari-Sis, B. & Zirak, M. α-imino esters in organic synthesis: Recent advances. Chem. Rev. 117, 8326–8419 (2017).
Cabré, A., Verdaguer, X. & Riera, A. Recent advances in the enantioselective synthesis of chiral amines via transition metal-catalyzed asymmetric hydrogenation. Chem. Rev. 122, 269–339 (2022).
Li, Y., Yu, Y.-N. & Xu, M.-H. Simple open-chain phosphite-olefin as ligand for Rh-catalyzed asymmetric arylation of cyclic ketimines: Enantioselective access to gem-diaryl α-amino acid derivatives. ACS Catal. 6, 661–665 (2016).
Huo, X., Zhang, J., Fu, J., He, R. & Zhang, W. Ir/Cu dual catalysis: Enantio- and diastereodivergent access to α,α-disubstituted α-amino acids bearing vicinal stereocenters. J. Am. Chem. Soc. 140, 2080–2084 (2018).
Hu, B. & Deng, L. Catalytic asymmetric synthesis of trifluoromethylated γ-amino acids through the umpolung addition of trifluoromethyl imines to carboxylic acid derivatives. Angew. Chem. Int. Ed. 57, 2233–2237 (2018).
Hu, Y. et al. Nickel-catalyzed asymmetric hydrogenation of 2-amidoacrylates. Angew. Chem Int. Ed. 59, 5371–5375 (2020).
Yao, P. et al. Asymmetric synthesis of N-substituted a-amino Es- ters from a-ketoesters via imine reductase-catalyzed reductive ami-nation. Angew. Chem. Int. Ed. 60, 8717–8721 (2021).
Hu, L., Wang, Y.-Z., Xu, L., Yin, Q. & Zhang, X. Highly enan- tioselective synthesis of N-unprotected unnatural α-amino acid de- rivatives by ruthenium-catalyzed direct asymmetric reductive ami- nation. Angew. Chem. Int. Ed. 61, e202202552 (2022).
Wu, X. et al. Modular α-tertiary amino ester synthesis through cobalt-catalysed asymmetric aza-barbier reaction. Nat. Chem. 16, 398–407 (2024).
Liu, S., Gao, J., Zou, Y. & Hai, Y. Enzymatic synthesis of unprotected α,β-diamino acids via direct asymmetric mannich reactions. J. Am. Chem. Soc. 146, 20263–20269 (2024).
Zhu, S. F. & Zhou, Q. L. Transition-metal-catalyzed enantioselective heteroatom-hydrogen bond insertion reactions. Acc. Chem. Res. 45, 1365–1377 (2012).
Gillingham, D. & Fei, N. Catalytic X-H insertion reactions based on carbenoids. Chem. Soc. Rev. 42, 4918–4931 (2013).
He, J. et al. Ligand-controlled C(sp3)-H arylation and olefination in synthesis of unnatural chiral α-amino acids. Science 343, 1216–1220 (2014).
Jin, L.-M., Xu, P., Xie, J. & Zhang, X. P. Enantioselective intermolecular radical C-H amination. J. Am. Chem. Soc. 142, 20828–20836 (2020).
Yang, Z.-P., Freas, D. J. & Fu, G. C. Asymmetric synthesis of protected unnatural α-amino acids via enantioconvergent Nickel-catalyzed cross-coupling. J. Am. Chem. Soc. 143, 8614–8618 (2021).
Che, C., Li, Y.-N., Cheng, X., Lu, Y.-N. & Wang, C.-J. Visible-light-enabled enantioconvergent synthesis of a-amino acid derivatives via synergistic Brønsted acid/photoredox. Catal. Angew. Chem., Int. Ed. 60, 4698–4704 (2021).
Cheng, L. et al. Stereoselective amino acid synthesis by synergistic photoredox-pyridoxal radical biocatalysis. Science 381, 444–451 (2023).
Wang, T.-C. et al. Stereoselective amino acid synthesis by photobiocatalytic oxidative coupling. Nature 629, 98–104 (2024).
Ouyang, Y., Page, C. G., Bilodeau, C. & Hyster, T. K. Synergistic photoenzymatic catalysis enables synthesis of a‑tertiary amino acids using threonine aldolases. J. Am. Chem. Soc. 146, 13754–13759 (2024).
Ye, C.-X., Shen, X., Chen, S. & Meggers, E. Stereocontrolled 1,3-nitrogen migration to access chiral α-amino acids. Nat. Chem. 14, 566–573 (2022).
Yamamoto, E. et al. Dynamic kinetic resolution of N‑protected amino acid esters via phase-transfer catalytic base hydrolysis. ACS Catal. 8, 5708–5713 (2018).
Vedejs, E. & Jure, M. Efficiency in nonenzymatic kinetic resolution. Angew. Chem., Int. Ed. 44, 3974–4001 (2005).
Pellissier, H. Catalytic non-enzymatic kinetic resolution. Adv. Synth. Catal. 353, 1613–1666 (2011).
Peng, T., Li, S., Yang, D. & Wang, L. Recent advances in iintramolecular kinetic resolution rreactions. Org. Chem. Front. 10, 3401–3428 (2023).
Tang, M., Gu, H., He, S., Rajkumar, S. & Yang, X. Asymmetric enamide-imine tautomerism in the kinetic resolution of tertiary alcohols. Angew. Chem., Int. Ed. 60, 21334–21339 (2021).
Fang, S. et al. Enantiodivergent kinetic resolution of 1,1’-biaryl-2,2’-diols and amino alcohols by dipeptide-phosphonium salt catalysis inspired by the Atherton-Todd reaction. Angew. Chem., Int. Ed. 60, 14921–14930 (2021).
Hua, Y. et al. Kinetic resolution of tertiary benzyl alcohols via palladium/chiral norbornene cooperative catalysis. Angew. Chem., Int. Ed. 60, 12824–12828 (2021).
Westwood, M. T. et al. Isothiourea-catalysed acylative kinetic resolution of tertiary pyrazolone alcohols. Angew. Chem Int. Ed. 63, e202407983 (2024).
An, H. et al. Kinetic resolution of acyclic tertiary propargylic alcohols by NHC-catalyzed enantioselective acylation. Org. Lett. 26, 702–707 (2024).
Gou, F.-H., Ren, F., Wu, Y. & Wang, P. Catalytic kinetic resolution of monohydrosilanes via rhodium-catalyzed enantioselective intramolecular hydrosilylation. Angew. Chem. Int. Ed. 63, e202404732 (2024).
Shen, Q. et al. Peroxygenase-enabled reductive kinetic resolution for the enantioenrichment of organoperoxides. Angew. Chem Int. Ed. 63, e202401590 (2024).
Cao, M. et al. Catalytic enantioselective hydroxylation of tertiary propargylic C(sp3)-H bonds in acyclic systems: A kinetic resolution study. J. Am. Chem. Soc. 146, 18396–18406 (2024).
Chen, Y.-B. et al. Enantioselective functionalization of unactivated C(sp3)-H bonds through copper-catalyzed diyne cyclization by kinetic resolution. Nat. Commun. 15, 2232 (2024).
Li, L. et al. Kinetic resolution of β-sulfonyl ketones through enantioselective β-elimination using a cation-binding polyether catalyst. Angew. Chem., Int. Ed. 55, 331–335 (2016).
Liu, W., Jiang, Q. & Yang, X. A versatile method for kinetic resolution of protecting-group-free BINAMs and NOBINs through chiral phosphoric acid catalyzed triazane formation. Angew. Chem., Int. Ed. 59, 23598–23602 (2020).
Pan, Y.-L. et al. Kinetic resolution of 2H-azirines by asymmetric allylation reactions. ACS Catal. 11, 13752–13760 (2021).
Dong, S. et al. Organocatalytic kinetic resolution of sulfoximines. J. Am. Chem. Soc. 138, 2166–2169 (2016).
Tang, M. et al. Kinetic resolution of sulfoximines via asymmetric organocatalyzed formation of benzothiadiazine-1-oxides. Org. Lett. 26, 1914–1919 (2024).
Vastakaite, G., Budinská, A., Bögli, C. L., Boll, L. B. & Wennemers, H. Kinetic resolution of β‑branched aldehydes through peptide-catalyzed conjugate addition reactions. J. Am. Chem. Soc. 146, 19101–19107 (2024).
Dai, Q., Liu, L. & Zhang, J. Palladium/xiao-phos-catalyzed kinetic resolution of sec-phosphine oxides by P-benzylation. Angew. Chem Int. Ed. 60, 27247–27252 (2021).
Yin, L. et al. Asymmetric synthesis of P-stereogenic phosphindane oxides via kinetic resolution and their biological activity. Nat. Commun. 15, 2548 (2024).
Chen, Y., Zhu, C., Guo, Z., Liu, W. & Yang, X. Asymmetric synthesis of hydroquinolines with a,a-disubstitution through organocatalyzed kinetic resolution. Angew. Chem. Int. Ed. 60, 5268–5272 (2021).
Fang, Y. et al. Axially chiral bridged biaryls by Ni-catalyzed kinetic asymmetric C-O bond cleavage. ACS Catal. 14, 8176–8183 (2024).
Guo, X., Xu, G., Yang, R. & Wang, Q. Specific discrimination polymerization for highly isotactic polyesters synthesis. J. Am. Chem. Soc. 146, 9084–9095 (2024).
Li, L. et al. Dinuclear titanium(III)-catalyzed radical-type kinetic resolution of epoxides for the enantioselective synthesis of cis-glycidic esters. J. Am. Chem. Soc. 146, 13546–13557 (2024).
Walton, C. J. W. et al. Engineered aminotransferase for the production of D-phenylalanine derivatives using biocatalytic cascades. ChemCatChem 10, 470–474 (2018).
Han, S.‑W. & Shin, J.‑S. One-pot preparation of D-amino acids through biocatalytic deracemization using alanine dehydrogenase and ω-transaminase. Catal. Lett. 148, 3678–3684 (2018).
Zhu, L. et al. One-pot enzymatic synthesis of D-arylalanines using phenylalanine ammonia lyase and L-amino acid deaminase. Appl. Biochem. Biotechnol. 187, 75–89 (2019).
Ishida, C. et al. Reconstruction of hyper-thermostable ancestral L-amino acid oxidase to perform deracemization to D-amino acids. CheCatChem 13, 5228–5235 (2021).
Yu, J., Darù, A., Deng, M. & Blackmond, D. G. Prebiotic access to enantioenriched amino acids via peptide-mediated transamination reactions. Proc. Natl. Acad. Sci. USA. 121, e2315447121 (2024).
Masaki, M. & Fukui, K. Reaction of tertiary phosphine dichlorides with thiols in the presence of triethylamine. A convenient method for the reduction of phosphine oxides to phosphines. Chem. Lett. 6, 151–152 (1977).
Shipilovskikh, S. A., Vaganov, V. Y., Denisova, E. I., Rubtsov, A. E. & Malkov, A. V. Dehydration of amides to nitriles under conditions of a catalytic appel reaction. Org. Lett. 20, 728–731 (2018).
Zhu, H., Qu, Z. W. & Grimme, S. Reduction of phosphine oxide by using chlorination reagents and dihydrogen: DFT mechanistic insights. Chem. Eur. J. 25, 4670–4672 (2019).
Bornemann, D. et al. Deoxygenative fluorination of phosphine oxides: A general route to fluorinated organophosphorus(V) compounds and beyond. Angew. Chem. Int. Ed. 59, 22790–22795 (2020).
Ren, J.-W., Tong, M.-N., Zhao, Y.-F. & Ni, F. Synthesis of dipeptide, amide, and ester without racemization by oxalyl chloride and catalytic triphenylphosphine oxide. Org. Lett. 23, 7497–7502 (2021).
Kurtz, K. C. M., Hsung, R. P. & Zhang, Y. A ring-closing Yne-carbonyl metathesis of ynamides. Org. Lett. 8, 231–234 (2006).
Hiraoka, S., Matsumoto, T., Matsuzaka, K., Sato, T. & Chida, N. Approach to fully substituted cyclic nitrones from N-hydroxylactam derivatives: Development and application to the total synthesis of cylindricine. C. Angew. Chem., Int. Ed. 58, 4381–4385 (2019).
Lind, F., Markelov, K. & Studer, A. Benzoyldiisopropylchlorosilane: A visible light photocleavable alcohol protecting group. Chem. Sci. 14, 12615–12620 (2023).
Moghadam, F. A. et al. Formation of all-carbon quaternary centers via enantioselective Pd-catalyzed α-vinylation of γ-lactams. Org. Lett. 26, 7551–7554 (2024).
Zhao, L. et al. Electrochemical dehydroxymethylative functionalization of alkanols for forging C(sp3)-heteroatom bonds. Green. Chem. 26, 4733–4741 (2024).
Acknowledgements
We gratefully acknowledge the financial support from the National Natural Science Foundation of China (No. 22508288, J.-W. Ren), Shandong Provincial Natural Science Foundation (No. ZR2023QB153, J.-W. Ren), Science and Technology Development Project of Tai’an City (No. 2022GX063, J.-W. Ren), and Taishan University.
Author information
Authors and Affiliations
Contributions
J.-W. Ren and J.-H. Sun conceived the idea. J.-W. Ren directed the project. K.-H. Li, M.-R. Lin, J.-L. Zeng and X.-M. Ai performed the experiments. J.-W. Ren wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Thomas Hansen, and the other, anonymous, reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ren, JW., Sun, JH., Li, KH. et al. Kinetic resolution of amino acids by phosphine oxide catalyzed enantioselective esterification. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71469-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71469-x