Abstract
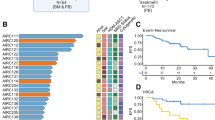
Daratumumab is approved for patients with multiple myeloma (MM) and high-risk smoldering MM (HR-SMM). However, HR-SMM is often as genomically complex as MM, suggesting it may be too advanced for single-agent intervention. We report on a Phase II trial of single-agent daratumumab in patients with earlier-stage disease, including high-risk monoclonal gammopathy of undetermined significance and low-risk SMM, to test if earlier treatment can induce deep responses and prevent progression to MM (D-PRISM/NCT03236428, n = 41). As primary outcome, the rate of Very Good Partial Response or better is 17% (95% CI: 7–32), which is comparable to what was observed in HR-SMM and does not meet the study’s primary endpoint. The overall response rate is 54%, with two patients developing MM and 51% biochemical progression. Grade 3 or higher toxicities include hypertension (7%), diarrhea (2%), flu-like symptoms (2%), and headache (2%). Genomic and immune variables associated with biochemical progression are identified in exploratory analyses leveraging whole-genome and single-cell RNA-sequencing. This study demonstrates that, although less effective than expected, daratumumab is safe and can induce deep responses in certain early-stage patients, highlighting the importance of adopting genomic and immune profiling to improve patient selection and maximize the benefit/risk ratio in trials of early intervention.
Similar content being viewed by others
Data availability
Data generated for this study were deposited in a controlled-access repository (dbGaP) in accordance with privacy requirements set forth in the informed consent forms signed by study participants. Access to this dataset can be obtained by registering the investigator’s institution in eRA Commons, establishing an eRA Commons account for the investigator and submitting a Data Access Request through the dbGaP Authorized Access website; following authorization by the requesting institution’s signing official and review by NIH staff, the request may be approved for data download. Raw and processed single-cell RNA-sequencing data are deposited in dbGaP (phs004127.v1.p1, https://www.ncbi.nlm.nih.gov/projects/gap/cgi-bin/study.cgi?study_id=phs004127.v1.p1). Raw WGS data used for cytogenetic classification are also deposited in dbGaP (phs003846.v1.p1, https://www.ncbi.nlm.nih.gov/projects/gap/cgi-bin/study.cgi?study_id=phs003846.v1.p1)33. Outcome and toxicity data for all patients in the trial are provided in Supplementary Data 1 and the study protocol is provided in the Supplementary Information file. Source data are provided with this paper.
Code availability
R code used to conduct scRNAseq analyses has been deposited to Github (https://github.com/ghobriallab/Nat_Comm_2026_D-PRISM_STUDY) and accessioned on Zenodo58.
References
Kyle, R. A. et al. Long-term follow-up of monoclonal gammopathy of undetermined significance. N. Engl. J. Med. 378, 241–249 (2018).
Kyle, R. A. et al. A long-term study of prognosis in monoclonal gammopathy of undetermined significance. N. Engl. J. Med. 346, 564–569 (2002).
Kyle, R. A. et al. Clinical course and prognosis of smoldering (asymptomatic) multiple myeloma. N. Engl. J. Med. 356, 2582–2590 (2007).
Kyle, R. A. et al. Prevalence of monoclonal gammopathy of undetermined significance. N. Engl. J. Med. 354, 1362–1369 (2006).
Rajkumar, S. V. et al. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol. 15, e538–e548 (2014).
Santockyte, R. et al. Sensitive multiple myeloma disease monitoring by mass spectrometry. Blood Cancer J. 11, 78 (2021).
Murray, D. et al. Detection and prevalence of monoclonal gammopathy of undetermined significance: a study utilizing mass spectrometry-based monoclonal immunoglobulin rapid accurate mass measurement. Blood Cancer J. 9, 102 (2019).
Barnidge, D. R. et al. Using mass spectrometry to monitor monoclonal immunoglobulins in patients with a monoclonal gammopathy. J. Proteome Res. 13, 1419–1427 (2014).
Landgren, O. et al. Prevalence of myeloma precursor state monoclonal gammopathy of undetermined significance in 12372 individuals 10-49 years old: a population-based study from the National Health and Nutrition Examination Survey. Blood Cancer J. 7, e618 (2017).
Landgren, O. et al. Racial disparities in the prevalence of monoclonal gammopathies: a population-based study of 12,482 persons from the National Health and Nutritional Examination Survey. Leukemia 28, 1537–1542 (2014).
El-Khoury, H. et al. Prevalence of monoclonal gammopathies and clinical outcomes in a high-risk US population screened by mass spectrometry: a multicentre cohort study. Lancet Haematol. 9, e340–e349 (2022).
Rajkumar, S. V. & Kumar, S. Monoclonal gammopathy of undetermined significance. N. Engl. J. Med. 393, 1315–1326 (2025).
Rajkumar, S. V. et al. Serum free light chain ratio is an independent risk factor for progression in monoclonal gammopathy of undetermined significance. Blood 106, 812–817 (2005).
Mateos, M. V. et al. International Myeloma Working Group risk stratification model for smoldering multiple myeloma (SMM). Blood Cancer J. 10, 102 (2020).
Lakshman, A. et al. Risk stratification of smoldering multiple myeloma incorporating revised IMWG diagnostic criteria. Blood Cancer J. 8, 59 (2018).
Rajkumar, S. V. et al. Impact of primary molecular cytogenetic abnormalities and risk of progression in smoldering multiple myeloma. Leukemia 27, 1738–1744 (2013).
Rajkumar, S. V., Landgren, O. & Mateos, M. V. Smoldering multiple myeloma. Blood 125, 3069–3075 (2015).
Bustoros, M. et al. Genomic profiling of smoldering multiple myeloma identifies patients at a high risk of disease progression. J. Clin. Oncol. 38, 2380–2389 (2020).
Misund, K. et al. MYC dysregulation in the progression of multiple myeloma. Leukemia 34, 322–326 (2020).
Boyle, E. M. et al. The molecular make up of smoldering myeloma highlights the evolutionary pathways leading to multiple myeloma. Nat. Commun. 12, 293 (2021).
Bolli, N. et al. Genomic patterns of progression in smoldering multiple myeloma. Nat. Commun. 9, 3363 (2018).
Kazandjian, D. et al. Genomic profiling to contextualize the results of intervention for smoldering multiple myeloma. Clin. Cancer Res. 30, 4482–4490 (2024).
Oben, B. et al. Whole-genome sequencing reveals progressive versus stable myeloma precursor conditions as two distinct entities. Nat. Commun. 12, 1861 (2021).
Go, R. S. & Rajkumar, S. V. How I manage monoclonal gammopathy of undetermined significance. Blood 131, 163–173 (2018).
Visram, A., Cook, J. & Warsame, R. Smoldering multiple myeloma: evolving diagnostic criteria and treatment strategies. Hematology 2021, 673–681 (2021).
Rajkumar, S. V. et al. Smoldering multiple myeloma current treatment algorithms. Blood Cancer J. 12, 129 (2022).
Mateos, M. V. et al. Lenalidomide plus dexamethasone versus observation in patients with high-risk smouldering multiple myeloma (QuiRedex): long-term follow-up of a randomised, controlled, phase 3 trial. Lancet Oncol. 17, 1127–1136 (2016).
Mateos, M. V. et al. Lenalidomide-dexamethasone versus observation in high-risk smoldering myeloma after 12 years of median follow-up time: a randomized, open-label study. Eur. J. Cancer 174, 243–250 (2022).
Lonial, S. et al. Randomized trial of lenalidomide versus observation in smoldering multiple myeloma. J. Clin. Oncol. 38, 1126–1137 (2020).
Nadeem, O. & Ghobrial, I. M. Early intervention with daratumumab improves survival for patients with high-risk smouldering myeloma. Nat. Rev. Clin. Oncol. 22, 159–160 (2025).
Dimopoulos, M. A. et al. Daratumumab or active monitoring for high-risk smoldering multiple myeloma. N. Engl. J. Med. 392, 1777–1788 (2024).
Ghobrial, I. M. et al. Round table discussion on optimal clinical trial design in precursor multiple myeloma. Blood Cancer Discov. 5, 146–152 (2024).
Alberge, J. B. et al. Genomic landscape of multiple myeloma and its precursor conditions. Nat. Genet 57, 1493–1503 (2025).
Maura, F. et al. Genomics define malignant transformation in myeloma precursor conditions. J. Clin. Oncol. 44, 188–199 (2026).
Mateos, M. V. et al. Curative strategy for high-risk smoldering myeloma: carfilzomib, lenalidomide, and dexamethasone (KRd) followed by transplant, krd consolidation, and rd maintenance. J. Clin. Oncol. 42, 3247–3256 (2024).
Kazandjian, D. et al. Carfilzomib, lenalidomide, and dexamethasone followed by lenalidomide maintenance for prevention of symptomatic multiple myeloma in patients with high-risk smoldering myeloma: a phase 2 nonrandomized controlled trial. JAMA Oncol. 7, 1678–1685 (2021).
Nadeem, O. et al. Deeper response predicts better outcomes in high-risk-smoldering-myeloma: results of the I-PRISM phase II clinical trial. Nat. Commun. 16, 358 (2025).
Avet-Loiseau, H. & Bahlis, N. J. Smoldering multiple myeloma: Taking the narrow over the wide path?. Blood 143, 2025–2028 (2024).
Dispenzieri, A. et al. International Myeloma Working Group guidelines for serum-free light chain analysis in multiple myeloma and related disorders. Leukemia 23, 215–224 (2009).
Casneuf, T. et al. Effects of daratumumab on natural killer cells and impact on clinical outcomes in relapsed or refractory multiple myeloma. Blood Adv. 1, 2105–2114 (2017).
Landgren, C. O. et al. Daratumumab monotherapy for patients with intermediate-risk or high-risk smoldering multiple myeloma: a randomized, open-label, multicenter, phase 2 study (CENTAURUS). Leukemia 34, 1840–1852 (2020).
Landgren, O. et al. Efficacy and Safety of Daratumumab (DARA) monotherapy in patients with intermediate-risk or high-risk smoldering multiple myeloma (SMM): final analysis of the phase 2 centaurus study. Blood 142, 210–210 (2023).
Sklavenitis-Pistofidis, R. et al. Immune biomarkers of response to immunotherapy in patients with high-risk smoldering myeloma. Cancer Cell 40, 1358–1373.e8 (2022).
Korst, C. L. B. M. et al. The bone marrow NK cell profile predicts MRD negativity in patients with multiple myeloma treated with daratumumab-based therapy. Blood 145, 3007–3014 (2025).
Casneuf, T. et al. Deep immune profiling of patients treated with lenalidomide and dexamethasone with or without daratumumab. Leukemia 35, 573–584 (2021).
Verkleij, C. P. M. et al. NK cell phenotype is associated with response and resistance to daratumumab in relapsed/refractory multiple myeloma. Hemasphere 7, e881 (2023).
Shemesh, A. et al. Diminished cell proliferation promotes natural killer cell adaptive-like phenotype by limiting FcepsilonRIgamma expression. J. Exp. Med. 219, e20220551 (2022).
Bigley, A. B. et al. FcepsilonRIgamma-negative NK cells persist in vivo and enhance efficacy of therapeutic monoclonal antibodies in multiple myeloma. Blood Adv. 5, 3021–3031 (2021).
Liu, W. et al. FcRgamma gene editing reprograms conventional NK cells to display key features of adaptive human NK Cells. iScience 23, 101709 (2020).
Kumar, S. et al. International Myeloma Working Group consensus criteria for response and minimal residual disease assessment in multiple myeloma. Lancet Oncol. 17, e328–e346 (2016).
Zheng, G. X. Y. et al. Massively parallel digital transcriptional profiling of single cells. Nat. Commun. 8, 14049 (2017).
Hao, Y. et al. Dictionary learning for integrative, multimodal and scalable single-cell analysis. Nat. Biotechnol. 42, 293–304 (2024).
Zavidij, O. et al. Single-cell RNA sequencing reveals compromised immune microenvironment in precursor stages of multiple myeloma. Nat. Cancer 1, 493–506 (2020).
Sklavenitis-Pistofidis, R. et al. Single-cell RNA sequencing defines distinct disease subtypes and reveals hypo-responsiveness to interferon in asymptomatic Waldenstrom’s Macroglobulinemia. Nat. Commun. 16, 1480 (2025).
Boiarsky, R. et al. Single cell characterization of myeloma and its precursor conditions reveals transcriptional signatures of early tumorigenesis. Nat. Commun. 13, 7040 (2022).
Sklavenitis-Pistofidis, R. et al. Large-scale dependency and drug screens to characterize the therapeutic vulnerabilities of multiple myeloma with 1q+. Blood 146, 89–103 (2025).
Dutta A.K., et al: MinimuMM-seq: genome sequencing of circulating tumor cells for minimally invasive molecular characterization of multiple myeloma pathology. Cancer Discov. 13, 348–363 (2022).
Aranha M.P. ghobriallab/Nat_Comm_2026_D-PRISM_STUDY: study published 2026 (Version 20260209) Zenodo. https://doi.org/10.5281/zenodo.18566827 (2026).
Acknowledgements
The authors thank the patients and their families for participating in this study. Anna V. Justis, Ph.D., a medical writer employed by Dana-Farber Cancer Institute, edited this manuscript. Johnson and Johnson provided support for the clinical trial. This funder reviewed the final manuscript and approved its publication; they were not involved in the conceptualization, design, or conduct of the trial or preparation of the manuscript. Funding was also provided by the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation, Cancer Research UK, and the NIH (R35CA263817 awarded to I.M.G.).
Author information
Authors and Affiliations
Contributions
O.N. and I.M.G. conceptualized the study. O.N., M.K., J.V.M., A.J.Y., J.A.Z., A.K., C.D., C.R., F.A., M.M., A.B., J.R., E.K.O’D., J.P.L., and P.G.R. conducted the clinical trial. F.C., E.D.L., A.K.D., and M.R. conducted laboratory analyses and collected data. S.M., J.P., R.S-P., and T.H.M., collected clinical data and participated in data curation. J.P. supervised data curation. A.S. supervised clinical trial conduct and data collection. I.M.G. acquired funding for this study. M.P.A., R.A.R., T.W., F.C., J-B.A., J.T., L.T., L.P., and R.S-P. conducted formal analyses, and G.G. and R.S-P. supervised analyses. M.P.A., O.N., and R.S-P. wrote the original draft of this manuscript. All authors reviewed and approved the final version for publication. O.N., G.G., R.S-P., and I.M.G. supervised this study.
Corresponding authors
Ethics declarations
Competing interests
O.N. reports research support from Takeda and Janssen; advisory board participation for Bristol-Myers Squibb, Janssen, Sanofi, Takeda, Kite and GPCR therapeutics; and honorarium from Pfizer. M.B. reports consultancy for Janssen, BMS, Takeda, Epizyme, Karyopharm, Menarini Biosystems, and Adaptive Biotechnology. A.J.Y reports consulting for AbbVie, Adaptive Biotechnologies, Amgen, BMS, Celgene, GSK, Johnson & Johnson, Karyopharm, Oncopeptides, Pfizer, Prothena, Regeneron, Sanofi, Sebia, Takeda. Research funding to institution from Amgen, BMS, GSK, Johnson & Johnson, Sanofi. P.G.R. is a consultant for Celgene/Bristol-Myers Squibb, GlaxoSmithKline, Karyopharm, Oncopeptides, Regeneron, and Sanofi, and has received research support from Oncopeptides. T.H.M. reports consulting for Sanofi. G.G. is a consultant, current equity holder, and co-founder for Scorpion Therapeutics and Predicta Biosciences, is receiving research funds from IBM, Pharmacyclics, and is a holder of patents and royalties for SignatureAnalyzer-GPU (US 2021/0358574, published), MSMuTect and MSMutSig (US 11,608,533, issued), MSIDetect (US 2023/0332246, published), and POLYSOLVER (US 11,725,237, issued). None of these patents are related to this work. R.S-P. is a consultant, private equity holder, and co-founder of Predicta Biosciences. I.M.G. has consulted for Bristol-Myers Squibb, AstraZeneca, Amgen, BioSkryb, Clinical Care Options, Curio Science, Sanofi, Janssen, Pfizer, Menarini Silicone Biosystems, Aptitude Health, GlaxoSmithKline, AbbVie, Adaptive Biotechnologies, Window Therapeutics, and Regeneron. She has received honoraria or speaker fees from Vor Biopharma, Janssen, MJH Life Sciences, Novartis, Takeda, Amgen, Regeneron, Curio Science, Standard Biotools, and Physicians’ Education Resource. She is a founder and board member of and holds private equity in Predicta Biosciences. Her spouse is the chief medical officer of and holds private equity in Disc Medicine. All other authors declare no conflicts of interest.
Peer review
Peer review information
Nature Communications thanks Rajshekhar Chakraborty, Elias Mai and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Nadeem, O., Aranha, M.P., Redd, R.A. et al. Daratumumab in high-risk MGUS and low-risk smoldering myeloma: results of the Phase II D-PRISM study. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71483-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71483-z