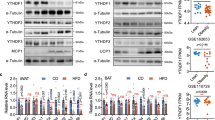
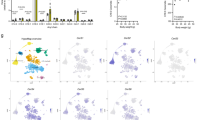
Abstract
METTL14 mediates N6-methyladenosine (m6A) RNA modification, while YTHDC1 and YTHDF2 specifically bind m6A-methylated RNA to regulate RNA fate. POMC neurons constitute the core of the central melanocortin circuit, and POMC deficiency causes obesity in both mice and humans. However, how m6A-based epitranscriptomics regulates melanocortin circuit function remains unclear. Here, we generated and characterized POMC neuron-specific knockout mice lacking Mettl14 (Mettl14ΔPOMC), Ythdc1 (Ythdc1ΔPOMC), or Ythdf2 (Ythdf2 ΔPOMC). Mettl14ΔPOMC and Ythdc1ΔPOMC mice develop hyperphagia, obesity, glucose intolerance, insulin resistance, and hepatic steatosis in both sexes under standard chow conditions, accompanied by POMC downregulation. Conversely, POMC neuron-specific overexpression of METTL14 or YTHDC1 protects against diet-induced obesity. In contrast, Ythdf2 ΔPOMC mice are resistant to obesity, revealing an m6A-dependent balance between YTHDC1 and YTHDF2. Mechanistically, the METTL14/YTHDC1 pathway is indispensable for embryonic POMC neurogenesis, while in adults YTHDC1 maintains melanocortin circuit integrity/function. METTL14 and YTHDC1 directly target POMC and ISL1 transcripts to regulate protein expression. POMC neuron-specific restoration of POMC reverses obesity and metabolic phenotypes in Mettl14ΔPOMC and Ythdc1ΔPOMC mice, defining an anti-obesity METTL14/m6A/YTHDC1/POMC axis. These findings identify METTL14 as the m6A writer for POMC/ISL1 and YTHDC1 and YTHDF2 as their readers, uncovering a critical role of m6A epitranscriptomic regulation in melanocortin circuit development and maintenance.
Similar content being viewed by others
Data availability
Source data were provided within this paper as a Source Data file. Source data are provided with this paper.
References
Toda, C., Santoro, A., Kim, J. D. & Diano, S. POMC neurons: from birth to death. Annu. Rev. Physiol. 79, 209–236 (2017).
Quarta, C., Fioramonti, X. & Cota, D. POMC neurons dysfunction in diet-induced metabolic disease: hallmark or mechanism of disease? Neuroscience 447, 3–14 (2019).
Huszar, D. et al. Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell 88, 131–141 (1997).
Chen, A. S. et al. Inactivation of the mouse melanocortin-3 receptor results in increased fat mass and reduced lean body mass. Nat. Genet. 26, 97–102 (2000).
Cone, R. D. Studies on the physiological functions of the melanocortin system. Endocr. Rev. 27, 736–749 (2006).
Mercer, A. J., Hentges, S. T., Meshul, C. K. & Low, M. J. Unraveling the central proopiomelanocortin neural circuits. Front. Neurosci. 7, 19 (2013).
Nasif, S. et al. Islet 1 specifies the identity of hypothalamic melanocortin neurons and is critical for normal food intake and adiposity in adulthood. Proc. Natl. Acad. Sci. USA. 112, E1861–E1870 (2015).
Herb, B. R. et al. Single-cell genomics reveals region-specific developmental trajectories underlying neuronal diversity in the human hypothalamus. Sci. Adv. 9, eadf6251 (2023).
Orquera, D. P. et al. The Homeodomain Transcription Factor NKX2.1 Is Essential for the Early Specification of Melanocortin Neuron Identity and Activates Pomc Expression in the Developing Hypothalamus. J. Neurosci. 39, 4023–4035 (2019).
Padilla, S. L., Carmody, J. S. & Zeltser, L. M. Pomc-expressing progenitors give rise to antagonistic neuronal populations in hypothalamic feeding circuits. Nat. Med. 16, 403–405 (2010).
Murakami, S. & Jaffrey, S. R. Hidden codes in mRNA: control of gene expression by m(6)A. Mol. Cell 82, 2236–2251 (2022).
Shi, H., Wei, J. & He, C. Where, When, and How: context-dependent functions of RNA methylation writers, readers, and erasers. Mol. Cell 74, 640–650 (2019).
Geula, S. et al. Stem cells. m6A mRNA methylation facilitates resolution of naive pluripotency toward differentiation. Science 347, 1002–1006 (2015).
Meng, T. G. et al. Mettl14 is required for mouse postimplantation development by facilitating epiblast maturation. FASEB J. 33, 1179–1187 (2018).
Kasowitz, S. D. et al. Nuclear m6A reader YTHDC1 regulates alternative polyadenylation and splicing during mouse oocyte development. PLoS Genet. 14, e1007412 (2018).
Li, M. et al. Ythdf2-mediated m(6)A mRNA clearance modulates neural development in mice. Genome Biol. 19, 69 (2018).
Yaswen, L., Diehl, N., Brennan, M. B. & Hochgeschwender, U. Obesity in the mouse model of pro-opiomelanocortin deficiency responds to peripheral melanocortin. Nat. Med. 5, 1066–1070 (1999).
Berglund, E. D. et al. Serotonin 2C receptors in pro-opiomelanocortin neurons regulate energy and glucose homeostasis. J. Clin. Investig. 123, 5061–5070 (2013).
Kang, Q. et al. Adipose METTL14-elicited N(6) -methyladenosine promotes obesity, insulin resistance, and NAFLD through suppressing beta adrenergic signaling and lipolysis. Adv. Sci. 10, e2301645 (2023).
Zheng, Q. et al. METTL14-induced M(6)A methylation increases G6pc biosynthesis, hepatic glucose production and metabolic disorders in obesity. Adv. Sci. 12, e2417355 (2025).
Xu, Y. et al. Lateral septum as a melanocortin downstream site in obesity development. Cell Rep. 42, 112502 (2023).
Croizier, S. & Bouret, S. G. Molecular control of the development of hypothalamic neurons involved in metabolic regulation. J. Chem. Neuroanat. 123, 102117 (2022).
Quarta, C. et al. Functional identity of hypothalamic melanocortin neurons depends on Tbx3. Nat. Metab. 1, 222–235 (2019).
Lee, B. et al. Dlx1/2 and Otp coordinate the production of hypothalamic GHRH- and AgRP-neurons. Nat. Commun. 9, 2026 (2018).
Sanz, E. et al. Fertility-regulating Kiss1 neurons arise from hypothalamic POMC-expressing progenitors. J. Neurosci. 35, 5549–5556 (2015).
Shen, Y. et al. m(6)A deficiency impairs hypothalamic neurogenesis of feeding-related neurons in mice and human organoids and leads to adult obesity in mice. Cell Stem Cell 32, 727–743.e8 (2025).
Liu, J. et al. METTL14 is essential for beta-cell survival and insulin secretion. Biochim Biophys. Acta Mol. Basis Dis. 1865, 2138–2148 (2019).
Li, Y. et al. SH2B1 defends against energy imbalance, obesity, and metabolic disease via a paraventricular hypothalamus–>dorsal raphe nucleus neurocircuit. Adv. Sci. 11, e2400437 (2024).
Acknowledgements
We thank Feng Jiang, Qiantao Zheng, Ruoyu Zhou, Lorelei Baron, Martin Myers, David Olson, Hui Yu, Ling Qi, Wei-Zhen Zhang, and Christin Carter-Su (University of Michigan) for their advice. We thank Malcolm J. Low (University of Michigan) and Joel K Elmquist (University of Texas Southwestern Medical Center) for providing POMC-CreERT2 mice. We thank Qingchun Tong (University of Texas Health Science Center at Houston) for providing AAV-DIO-POMC and AAV-DIO-α-MSH vectors. We thank Dr. Nathan Qi (University of Michigan) for discussion about CLAMS data analysis. This study was supported by grants R01 DK114220, R01 DK130111, and R01 DK141559 (LR) from the National Institutes of Health and 20POST35210557 (YL) from the American Heart Association (AHA). This work utilized the cores supported by the Michigan Diabetes Research and Training Center (NIH DK020572), Michigan Metabolomics and Obesity Center (DK089503), and the University of Michigan Center for Gastrointestinal Research (NIDDK P30DK034933).
Author information
Authors and Affiliations
Contributions
YL conducted the experiments, YL, MHK, DR, and LR analyzed the data, DR provided Mettl14f/f, Ythdc1f/f, and Ythdf2f/f mice, YL and LR designed the experiments and wrote the paper, YL, MHK, DR, and LR edited the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, Y., Kim, MH., Ren, D. et al. POMC neuron METTL14/m6A/YTHDC1/YTHDF2 pathways safeguard energy balance, body weight, and metabolism. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71672-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71672-w