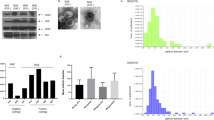
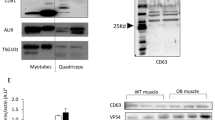
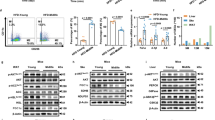
Abstract
Extracellular vesicles (EVs) are nano-sized, membrane-delimited, particles released by cells that carry signaling macromolecules. A major pathway of EV production is potentiated by neutral sphingomyelinase 2 (SMPD3/nSMAse2), an enzyme that generates ceramide from sphingomyelin. In our attempt to study this pathway in adipocytes of male mice, we discover that the elimination of SMPD3 from adipocytes in vivo triggers a signal to surrounding immune cell-like preadipocytes to release EVs that carry SMPD3 mRNA. This results in a widespread increase in SMPD3 mRNA in purified null adipocytes without a change in the transcripts of other enzymes involved in ceramide metabolism. These results point to a selective mechanism by which specific mRNA molecules are acquired from the microenvironment to a level that can restore expression of mRNA and protein in a cell that is depleted of the corresponding genetic information.
Similar content being viewed by others
Data availability
All data supporting the results of this study can be found in the article, supplementary information, and Source Data File. The sequencing data has been deposited in the GEO database under accession numbers GSE319159 (scRNAseq) and GSE319160 (RNAseq). Source data are provided with this paper.
References
van Niel, G., D’Angelo, G. & Raposo, G. Shedding light on the cell biology of extracellular vesicles. Nat. Rev. Mol. Cell Biol. 19, 213–228 (2018).
Fu, S. et al. Extracellular vesicles in cardiovascular diseases. Cell Death Discov. 6, 68 (2020).
Raghav, A. et al. Extracellular vesicles in neurodegenerative diseases: A systematic review. Front Mol. Neurosci. 15, 1061076 (2022).
Isaac, R., Reis, F. C. G., Ying, W. & Olefsky, J. M. Exosomes as mediators of intercellular crosstalk in metabolism. Cell Metab. 33, 1744–1762 (2021).
Xu, R. et al. Extracellular vesicles in cancer - implications for future improvements in cancer care. Nat. Rev. Clin. Oncol. 15, 617–638 (2018).
Crewe, C. & Scherer, P. E. Intercellular and interorgan crosstalk through adipocyte extracellular vesicles. Rev. Endocr. Metab. Disord. 23, 61–69 (2022).
Crewe, C. et al. Deficient caveolin-1 synthesis in adipocytes stimulates systemic insulin-independent glucose uptake via extracellular vesicles. Diabetes 71, 2496–2512 (2022).
Crewe, C. et al. Extracellular vesicle-based interorgan transport of mitochondria from energetically stressed adipocytes. Cell Metab. 33, 1853–1868 e1811 (2021).
Trajkovic, K. et al. Ceramide triggers budding of exosome vesicles into multivesicular endosomes. Science 319, 1244–1247 (2008).
Crewe, C. et al. An endothelial-to-adipocyte extracellular vesicle axis governed by metabolic State. Cell 175, 695–708 e613 (2018).
O’Brien, K., Breyne, K., Ughetto, S., Laurent, L. C. & Breakefield, X. O. RNA delivery by extracellular vesicles in mammalian cells and its applications. Nat. Rev. Mol. Cell Biol. 21, 585–606 (2020).
Bonsergent, E. et al. Quantitative characterization of extracellular vesicle uptake and content delivery within mammalian cells. Nat. Commun. 12, 1864 (2021).
Merrick, D. et al. Identification of a mesenchymal progenitor cell hierarchy in adipose tissue. Science 364, https://doi.org/10.1126/science.aav2501 (2019).
Prieto-Vila, M., Yoshioka, Y. & Ochiya, T. Biological functions driven by mrnas carried by extracellular vesicles in cancer. Front Cell Dev. Biol. 9, 620498 (2021).
Tomita, T. et al. Extracellular mRNA transported to the nucleus exerts translation-independent function. Nat. Commun. 12, 3655 (2021).
Ratajczak, J. et al. Embryonic stem cell-derived microvesicles reprogram hematopoietic progenitors: evidence for horizontal transfer of mRNA and protein delivery. Leukemia 20, 847–856 (2006).
Dellar, E. R., Hill, C., Melling, G. E. & Carter, D. R. & Baena-Lopez, L. A. Unpacking extracellular vesicles: RNA cargo loading and function. J. Extracell. Biol. 1, e40 (2022).
Wang, Z. V., Deng, Y., Wang, Q. A., Sun, K. & Scherer, P. E. Identification and characterization of a promoter cassette conferring adipocyte-specific gene expression. Endocrinology 151, 2933–2939 (2010).
Ianevski, A., Giri, A. K. & Aittokallio, T. Fully-automated and ultra-fast cell-type identification using specific marker combinations from single-cell transcriptomic data. Nat. Commun. 13, 1246 (2022).
Acknowledgements
Funding: National Institutes of Health grants R00-DK122019, 1R01DK137791, 1R21EB035738, DRC at Washington University (NIH P30DK020579), American Heart Association (AHA 23IPA1054013) (CC). R01-DK55758, R01-DK099110, R01-DK127274, R01-DK143576, R01-DK131537, P01-AG051459, the UTSW NORC P30-DK127984 (P.E.S.). R01-DK114036, R01-DK130892, R01-DK136592 (CL). The authors thank the UT Southwestern Metabolic Phenotyping Core (RRID: SCR_026404) for its assistance with metabolic phenotyping.
Author information
Authors and Affiliations
Contributions
Conceptualization: C.C. and P.E.S. Investigation: C.C., C.M.G., T.O., S.C., J.B.F., M.Y.W., S.T., Y.L.P., M.R., S.A.P., Z.Y., Y.C.Z., A.L., L.G., C.K.C., and C.L. Data curation: C.C., Z.Y., C.X., and C.K.C. Formal analysis: C.C., Z.Y., C.X., A.L. Visualization: C.C., Z.Y., C.X., T.O., and P.E.S. Funding acquisition: C.C. and P.E.S. Supervision: C.C. and P.E.S. Writing-original draft: CC. Writing - review and editing C.C. and P.E.S.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Baisong Lu who co-reviewed with Balqees Khader; and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Crewe, C., Gliniak, C.M., Onodera, T. et al. Adipocytes signal to recruit specific mRNAs from surrounding cells to restore expression deficits. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71740-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71740-1