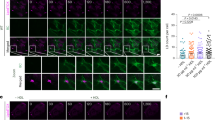
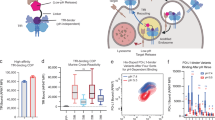
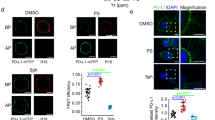
Abstract
N-terminal signal peptides (SPs) are traditionally considered as drivers of co-translational translocation of newly synthesised proteins into the endoplasmic reticulum (ER). However, growing evidences suggest that proteins with SPs can also undergo post-translational insertion into the ER membrane after synthesis is complete. Recently, an intermediate third mechanism has been uncovered where proteins with marginally hydrophobic or suboptimal SPs are translocated following an initial delay after translation initiation. Here, we show that this “delayed translocation” allows a temporary exposure of the nascent chain to the cytosolic environment, enabling exoplasmic domain modifications by cytosolic enzymes. We report that programmed death ligand-1 (PD-L1) follows this pathway, featuring a suboptimal SP that exposes its extracellular domain to the cytosol, enabling AMPK-dependent regulation of PD-L1 function. Importantly, optimising the SP of PD-L1 eliminates the cytosolic exposure, disrupting PD-L1’s trafficking and maturation, highlighting the physiological importance of the delayed translocation mechanism.
Similar content being viewed by others
Data availability
All data are available in the article and supplementary information. The source data for all data presented in the figures are provided with this paper as a Source Data file. Original uncropped gels are provided in the supplementary information file. Material is available upon request. The presented research complies with all relevant ethical regulations. Source data are provided with this paper.
References
Juszkiewicz, S. & Hegde, R. S. Quality control of orphaned proteins. Mol. Cell 71, 443–457 (2018).
Wenzell, N. A. et al. Global signal peptide profiling reveals principles of selective Sec61 inhibition. Nat. Chem. Biol. 20, 1154–1163 (2024).
von Heijne, G. Signal sequences. limits Var. J. Mol. Biol. 184, 99–105 (1985).
Zimmermann, R., Eyrisch, S., Ahmad, M. & Helms, V. Protein translocation across the ER membrane. Biochim Biophys. Acta 1808, 912–924 (2011).
Hosomi, A. et al. The ER-associated protease Ste24 prevents N-terminal signal peptide-independent translocation into the endoplasmic reticulum in Saccharomyces cerevisiae. J. Biol. Chem. 295, 10406–10419 (2020).
Sun, S., Li, X. & Mariappan, M. Signal sequences encode information for protein folding in the endoplasmic reticulum. J. Cell Biol. 222, e202203070 (2023).
Perara, E., Rothman, R. E. & Lingappa, V. R. Uncoupling translocation from translation: implications for transport of proteins across membranes. Science 232, 348–352 (1986).
Kim, S. J., Mitra, D., Salerno, J. R. & Hegde, R. S. Signal sequences control gating of the protein translocation channel in a substrate-specific manner. Dev. Cell 2, 207–217 (2002).
Ainger, K. J. & Meyer, D. I. Translocation of nascent secretory proteins across membranes can occur late in translation. EMBO J. 5, 951–955 (1986).
Hatsuzawa, K., Tagaya, M. & Mizushima, S. The hydrophobic region of signal peptides is a determinant for SRP recognition and protein translocation across the ER membrane. J. Biochem 121, 270–277 (1997).
Pardoll, D. M. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer 12, 252–264 (2012).
Li, C.-W. et al. Glycosylation and stabilization of programmed death ligand-1 suppresses T-cell activity. Nat. Commun. 7, 12632 (2016).
Cha, J.-H. et al. Metformin Promotes Antitumor Immunity via Endoplasmic-Reticulum-Associated Degradation of PD-L1. Mol. Cell 71, 606–620.e7 (2018).
Chan, L.-C. et al. IL-6/JAK1 pathway drives PD-L1 Y112 phosphorylation to promote cancer immune evasion. J. Clin. Invest 129, 3324–3338 (2019).
Zhang, X. et al. NEK2 inhibition triggers anti-pancreatic cancer immunity by targeting PD-L1. Nat. Commun. 12, 4536 (2021).
Hsu, J.-M., Li, C.-W., Lai, Y.-J. & Hung, M.-C. Posttranslational Modifications of PD-L1 and Their Applications in Cancer Therapy. Cancer Res 78, 6349–6353 (2018).
Zheng, T. & Nicchitta, C. V. Structural determinants for signal sequence function in the mammalian endoplasmic reticulum. J. Biol. Chem. 274, 36623–36630 (1999).
Nilsson, I. et al. The code for directing proteins for translocation across ER membrane: SRP cotranslationally recognizes specific features of a signal sequence. J. Mol. Biol. 427, 1191–1201 (2015).
Celińska, E., Borkowska, M., Białas, W., Korpys, P. & Nicaud, J.-M. Robust signal peptides for protein secretion in Yarrowia lipolytica: identification and characterization of novel secretory tags. Appl Microbiol Biotechnol. 102, 5221–5233 (2018).
Barrette-Ng, I. H., Wu, S.-C., Tjia, W.-M., Wong, S.-L. & Ng, K. K. S. The structure of the SBP-Tag–streptavidin complex reveals a novel helical scaffold bridging binding pockets on separate subunits. Acta Crystallogr D. Biol. Crystallogr 69, 879–887 (2013).
White, S. H. & Von Heijne, G. How Translocons Select Transmembrane Helices. Annu. Rev. Biophys. 37, 23–42 (2008).
Lakkaraju, A. K. K. et al. Efficient secretion of small proteins in mammalian cells relies on Sec62-dependent posttranslational translocation. Mol. Biol. Cell 23, 2712–2722 (2012).
Haßdenteufel, S., Nguyen, D., Helms, V., Lang, S. & Zimmermann, R. ER import of small human presecretory proteins: components and mechanisms. FEBS Lett. 593, 2506–2524 (2019).
Zhou, G. et al. Role of AMP-activated protein kinase in mechanism of metformin action. J. Clin. Invest. 108, 1167–1174 (2001).
Cool, B. et al. Identification and characterization of a small molecule AMPK activator that treats key components of type 2 diabetes and the metabolic syndrome. Cell Metab. 3, 403–416 (2006).
Dai, R. Y. et al. Implication of transcriptional repression in compound C-induced apoptosis in cancer cells. Cell Death Dis. 4, e883 (2013).
Kai, Y., Kawano, Y., Yamamoto, H. & Narahara, H. A possible role for AMP-activated protein kinase activated by metformin and AICAR in human granulosa cells. Reprod. Biol. Endocrinol. 13, 27 (2015).
Pascarella, A. et al. Vacuolated PAS-positive lymphocytes as an hallmark of Pompe disease and other myopathies related to impaired autophagy. J. Cell Physiol. 233, 5829–5837 (2018).
Dite, T. A. et al. AMP-activated protein kinase selectively inhibited by the type II inhibitor SBI-0206965. J. Biol. Chem. 293, 8874–8885 (2018).
Branon, T. C. et al. Efficient proximity labeling in living cells and organisms with TurboID. Nat. Biotechnol. 36, 880–887 (2018).
Goder, V. & Spiess, M. Molecular mechanism of signal sequence orientation in the endoplasmic reticulum. EMBO J. 22, 3645–3653 (2003).
Teufel, F. et al. SignalP 6.0 predicts all five types of signal peptides using protein language models. Nat. Biotechnol. 40, 1023–1025 (2022).
Käll, L., Krogh, A. & Sonnhammer, E. L. L. A combined transmembrane topology and signal peptide prediction method. J. Mol. Biol. 338, 1027–1036 (2004).
Choo, K. H. & Ranganathan, S. Flanking signal and mature peptide residues influence signal peptide cleavage. BMC Bioinforma. 9, S15 (2008).
Boncompain, G. et al. Synchronization of secretory protein traffic in populations of cells. Nat. Methods 9, 493–498 (2012).
Gasparotto, D. et al. Mapping cryptic phosphorylation sites in the human proteome. EMBO J. 44, 6704–6731 (2025).
Walter, P. & Blobel, G. Translocation of proteins across the endoplasmic reticulum III. Signal recognition protein (SRP) causes signal sequence-dependent and site-specific arrest of chain elongation that is released by microsomal membranes. J. Cell Biol. 91, 557–561 (1981).
Lakkaraju, A. K. K., Mary, C., Scherrer, A., Johnson, A. E. & Strub, K. SRP keeps polypeptides translocation-competent by slowing translation to match limiting ER-targeting sites. Cell 133, 440–451 (2008).
Chartron, J. W., Hunt, K. C. L. & Frydman, J. Cotranslational signal-independent SRP preloading during membrane targeting. Nature 536, 224–228 (2016).
Meyer, H. A. et al. Mammalian Sec61 is associated with Sec62 and Sec63. J. Biol. Chem. 275, 14550–14557 (2000).
Fons, R. D., Bogert, B. A. & Hegde, R. S. Substrate-specific function of the translocon-associated protein complex during translocation across the ER membrane. J. Cell Biol. 160, 529–539 (2003).
Tyedmers, J. et al. Homologs of the yeast Sec complex subunits Sec62p and Sec63p are abundant proteins in dog pancreas microsomes. Proc. Natl. Acad. Sci. USA 97, 7214–7219 (2000).
Haßdenteufel, S. et al. Chaperone-mediated sec61 channel gating during er import of small precursor proteins overcomes sec61 inhibitor-reinforced energy barrier. Cell Rep. 23, 1373–1386 (2018).
Ziska, A. et al. The signal peptide plus a cluster of positive charges in prion protein dictate chaperone-mediated Sec61 channel gating. Biol. Open 8, bio040691 (2019).
Nguyen, D. et al. Proteomics reveals signal peptide features determining the client specificity in human TRAP-dependent ER protein import. Nat. Commun. 9, 3765 (2018).
Schorr, S. et al. Identification of signal peptide features for substrate specificity in human Sec62/Sec63-dependent ER protein import. FEBS J. 287, 4612–4640 (2020).
Gamerdinger, M., Hanebuth, M. A., Frickey, T. & Deuerling, E. The principle of antagonism ensures protein targeting specificity at the endoplasmic reticulum. Science 348, 201–207 (2015).
Ullers, R. S. et al. Interplay of signal recognition particle and trigger factor at L23 near the nascent chain exit site on the Escherichia coli ribosome. J. Cell Biol. 161, 679–684 (2003).
Lee, H. C. & Bernstein, H. D. Trigger factor retards protein export in Escherichia coli. J. Biol. Chem. 277, 43527–43535 (2002).
Patzelt, H. et al. Binding specificity of Escherichia coli trigger factor. Proc. Natl. Acad. Sci. Usa. 98, 14244–14249 (2001).
Qin, W. et al. The diverse function of pd-1/pd-l pathway beyond cancer. Front Immunol. 10, 2298 (2019).
Besemer, J. et al. Selective inhibition of cotranslational translocation of vascular cell adhesion molecule 1. Nature 436, 290–293 (2005).
Vitale, F. et al. A light-resuming strategy as a screening method for selecting Sec61 inhibitors down-modulating PD-L1 expression. Nat. Commun. 16, 7243 (2025).
Kroonen, J. et al. Human glioblastoma-initiating cells invade specifically the subventricular zones and olfactory bulbs of mice after striatal injection. Int J. Cancer 129, 574–585 (2011).
Brodsky, J. L., Goeckeler, J. & Schekman, R. BiP and Sec63p are required for both co- and posttranslational protein translocation into the yeast endoplasmic reticulum. Proc. Natl. Acad. Sci. USA 92, 9643–9646 (1995).
Jordan, M., Schallhorn, A. & Wurm, F. M. Transfecting mammalian cells: optimization of critical parameters affecting calcium-phosphate precipitate formation. Nucleic Acids Res. 24, 596–601 (1996).
D’Arrigo, P. et al. A regulatory role for the co-chaperone FKBP51s in PD-L1 expression in glioma. Oncotarget 8, 68291–68304 (2017).
Abraham, O. et al. Control of protein trafficking by reversible masking of transport signals. MBoC 27, 1310–1319 (2016).
D’Agostino, M. et al. ER reorganization is remarkably induced in cos-7 cells accumulating transmembrane protein receptors not competent for export from the endoplasmic reticulum. J. Membr. Biol. 247, 1149–1159 (2014).
Caiazza, C. et al. Effects of long-term citrate treatment in the pc3 prostate cancer cell line. IJMS 20, 2613 (2019).
Allocca, S. et al. An αB-Crystallin Peptide Rescues Compartmentalization and Trafficking Response to Cu Overload of ATP7B-H1069Q, the Most Frequent Cause of Wilson Disease in the Caucasian Population. IJMS 19, 1892 (2018).
D’Agostino, M. et al. Unconventional secretion of α-Crystallin B requires the Autophagic pathway and is controlled by phosphorylation of its serine 59 residue. Sci. Rep. 9, 16892 (2019).
D’Agostino, M., Risselada, H. J., Endter, L. J., Comte-Miserez, V. & Mayer, A. SNARE -mediated membrane fusion arrests at pore expansion to regulate the volume of an organelle. EMBO J. 37, e99193 (2018).
Xie, W. et al. A Luciferase Reporter Gene System for High-Throughput Screening of γ-Globin Gene Activators. in High Throughput Screening (ed. Janzen, W. P.) vol. 1439 207–226 (Springer New York, New York, NY, 2016).
Kyte, J. & Doolittle, R. F. A simple method for displaying the hydropathic character of a protein. J. Mol. Biol. 157, 105–132 (1982).
Acknowledgements
We acknowledge Bruno Goud (Institut Curie, Paris), Maria Antonietta De Matteis (Tigem, Pozzuoli), Stefano Bonatti and Tommaso Russo (DMMBM, UNINA) for sharing reagents, scientific support and helpful discussions. We acknowledge Alice Ting for the plasmid V5-TurboID-NES_pCDNA3 (Addgene plasmid #107169). We thank the Cell and Tissue Imaging (PICT-IBiSA, Institut Curie), a member of the French National Research Infrastructure France-BioImaging (ANR10-INBS-04). This work in the M.D.A. lab has been funded by the Italian Minister for Research and University (PRIN B53D2300249006), the Italian Minister for Research and University (PRIN 20177XJCHX) to M.R., and the STAR-Junior Principal Investigator Grants 2018 to S.R. The work in F.P. lab has been supported by the Fondation pour la Recherche Médicale (EQU201903007925), by the Agence Nationale de la Recherche (ANR-19-CE13- 0006-03; ANR-20-CE14-0017-02; ANR-19-CE13-0002-03; ANR-11-LABX-0038), and has also received support under the program «Investissements d’Avenir» launched by the French Government and implemented by ANR with the references CelTisPhyBio (11-LBX-0038), ANR-10-IDEX-0001-02 PSL. The work of M.C.S. has been supported by grants ANR-11-LABX-0038 and ANR-10-IDEX-0001-02.
Author information
Authors and Affiliations
Contributions
Conceptualisation: M. D. A., F. P. and M. C. S. Experiments design: G. S., M. C. S., F. V., S. R., L. M., M. R., F. P., M. D. A. Investigation: G. S., M. C. S., F. V., R. B., P. P., M. G. C., S. R. and L. M. Funding acquisition: M. D. A., F. P. and M. R. Writing – original draft: M. D. A., F. P. and M. C. S. Writing – review & editing: All the authors read, revised and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cannata Serio, M., Vitale, F., Scerra, G. et al. A delayed translocation into the endoplasmic reticulum controls the post-translational modifications of PD-L1. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71760-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71760-x