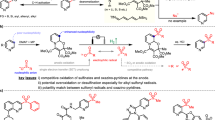
Abstract
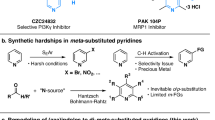
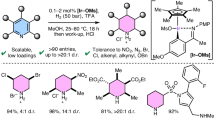
Direct and selective functionalization of ubiquitous pyridine scaffold is of paramount importance across numerous fields, yet remains challenging due to the inherent inertness of pyridines and the presence of competing reactive sites. Here, using an undivided cell equipped with a zinc cathode, a graphite anode, and a TEMPO mediator, we report a room-temperature electrochemical strategy that enables the direct hydroxyalkylation of diverse pyridines with readily available carbonyl compounds. This method features a broad substrate scope, operational simplicity, metal-free conditions, and high step/atom economy. It overcomes the limitations of conventional approaches requiring pre-functionalized substrates or stoichiometric activators, establishing a practical platform for direct access to C4- or C2-hydroxyalkyl pyridines, depending on the substitution pattern of the pyridine substrates. Mechanistic studies reveal that acetic acid activates both reactants, and the products are formed via cross-coupling of cathodic reduction-induced pyridyl radicals and hydroxy α-radicals followed by protonation and anodic oxidative aromatization.
Similar content being viewed by others
Data availability
All NMR spectra, mass spectrometry data, and experimental procedures generated in this study are provided in the Supplementary Information. The computational coordinate data generated in this study are provided in the Source Data, The X-ray crystallographic coordinates for structures reported in this study have been deposited at the Cambridge Crystallographic Data Centre (CCDC), under deposition numbers of CCDC 2416007 (C21). These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif. All data are available from the corresponding author upon request. Source data are provided with this paper.
References
Marshall, C. M., Federice, J. G., Bell, C. N., Cox, P. B. & Njardarson, J. T. An update on the Nitrogen heterocycle compositions and Properties of U.S. FDA-Approved Pharmaceuticals (2013–2023). J. Med. Chem. 67, 11622–11655 (2024).
Feng, J., Geng, W. C., Jiang, H. & Wu, B. Recent advances in biocatalysis of nitrogen-containing heterocycles. Biot. Adv. 54, 107813 (2022).
Cernak, T., Dykstra, K. D., Tyagarajan, S., Vachal, P. & Krska, S. W. The medicinal chemist’s toolbox for late stage functionalization of drug-like molecules. Chem. Soc. Rev. 45, 546–576 (2016).
Baumann, M. & Baxendale, I. R. An overview of the synthetic routes to the best selling Drugs Containing 6-membered Heterocycles. Beilstein J. Org. Chem. 9, 2265–2319 (2013).
Sevov, C. S. et al. Evolutionary design of low molecular weight organic anolyte materials for applications in nonaqueous redox flow batteries. J. Am. Chem. Soc. 137, 14465–14472 (2015).
Heravi, M. M. & Zadsirjan, V. Prescribed drugs containing nitrogen heterocycles: an overview. RSC Adv. 10, 44247–44311 (2020).
Meyer, E. A. et al. Discovery of the clinical candidate IDOR-1117-2520: A Potent and selective antagonist of CCR6 for autoimmune diseases. J. Med. Chem. 67, 8077–8098 (2024).
Hu, Z. et al. Design, Synthesis and antitumor activity of a novel FGFR2-selective degrader to overcome resistance of the FGFR2V564F Gatekeeper mutation based on a pan-FGFR inhibitor. Eur. J. Med. Chem. 275, 116612 (2024).
Ghosh, A. K. et al. Exploration of P1 and P4 Modifications of Nirmatrelvir: design, synthesis, biological evaluation, and X-ray structural studies of SARS-CoV-2 Mpro inhibitors. Eur. J. Med. Chem. 267, 116132 (2024).
Wu, Y., Lu, M., Kuo, S., Chu, P. & Chang, C. Synthesis and SAR Investigation of Biphenylaminoquinoline derivatives with Benzyloxy Substituents as promising anticancer agents. Chem. Biol. Drug Des. 103, e14509 (2024).
Calderón-Ospina, C. A. & Nava-Mesa, M. O. B Vitamins in the nervous system: current knowledge of the biochemical modes of action and synergies of thiamine, pyridoxine, and cobalamin. CNS Neurosci. Ther. 26, 5–13 (2019).
Ye, S., Hosono, N. & Uemura, T. Polymer-Grafting from MOF Nanosheets for the Fabrication of Versatile 2D Materials. Adv. Funct. Mater. 34, 2312265 (2023).
Scharf, S. et al. Synthesis and Twin Polymerization of Si(OCH2py)4 for Nitrogen-containing Carbon Materials. Eur. J. Inorg. Chem. 27, e202300656 (2024).
Biyiklioglu, Z. et al. New Fluorescent Manganese(III) Phthalocyanines Bearing Non-peripherally Octa-(3-pyridin-3-ylpropoxy) and (4-pyridin-3-ylpropoxy) for the Sensitive Determination of Pd2+ in Real Water Samples. Inorg. Chem. Commun. 159, 111824 (2024).
Abell, J. C. et al. Synthesis of Dihydropyridine Spirocycles by Semi-Pinacol-Driven Dearomatization of Pyridines. Org. Lett. 25, 400–404 (2023).
Wei, K., Yang, T., Chen, Q., Liang, S. & Yu, W. Iron-catalysed 1,2-aryl Migration of Tertiary Azides. Chem. Commun. 56, 11685–11688 (2020).
Proctor, R. S. J. & Phipps, R. J. Recent Advances in Minisci-Type Reactions. Angew. Chem. Int. Ed. 58, 13666–13699 (2019).
Proctor, R. S. J., Davis, H. J. & Phipps, R. J. Catalytic Enantioselective Minisci-type Addition to Heteroarenes. Science 360, 419–422 (2018).
Colgan, A. C. et al. Hydrogen Atom Transfer Driven Enantioselective Minisci Reaction of Alcohols. Angew. Chem. Int. Ed. 61, e202200266 (2022).
Xu, P., Chen, P. & Xu, H. Scalable Photoelectrochemical Dehydrogenative Cross-Coupling of Heteroarenes with Aliphatic C−H Bonds. Angew. Chem. Int. Ed. 59, 14275–14280 (2020).
Lai, X., Shu, X., Song, J. & Xu, H. Electrophotocatalytic Decarboxylative C−H functionalization of heteroarenes. Angew. Chem. Int. Ed. 59, 10626–10632 (2020).
Quevedo-Flores, B., Bosque, I. & Gonzalez-Gomez, J. C. Electrophotocatalytic Hydroxymethylation of Azaarenes with Methanol. Org. Lett. 26, 7447–7451 (2024).
Zhang, T. et al. A Directive Ni catalyst overrides conventional site selectivity in Pyridine C–H Alkenylation. Nat. Chem. 13, 1207–1213 (2021).
Cao, H., Cheng, Q. & Studer, A. Radical and ionic meta-C–H functionalization of pyridines, quinolines, and isoquinolines. Science 378, 779–785 (2022).
Cao, H., Cheng, Q. & Studer, A. meta-Selective C−H Functionalization of Pyridines. Angew. Chem. Int. Ed. 62, e202302941 (2023).
Cai, C. Y., Chen, S. J., Merchant, R. R., Kanda, Y. & Qin, T. C3 Selective Hydroxylation of Pyridines via Photochemical Valence Isomerization of Pyridine N-Oxides. J. Am. Chem. Soc. 146, 24257–24264 (2024).
Li, S. et al. C3 Selective Chalcogenation and Fluorination of Pyridine Using Classic Zincke Imine Intermediates. Nat. Commun. 15, 7420–7428 (2024).
Boyle, B. T., Levy, J. N., de Lescure, L., Paton, R. S. & McNally, A. Halogenation of the 3-position of Pyridines Through Zincke Imine Intermediates. Science 378, 773–779 (2022).
Qin, S. et al. Electrochemical Meta-C–H Sulfonylation of Pyridines with Nucleophilic Sulfinates. Nat. Commun. 15, 7428–7435 (2024).
Hart, M. A., Uhlenbruck, B. J. H., Levy, J. N. & McNally, A. 3-Selective Pyridine Fluorination via Zincke Imine Intermediates. J. Am. Chem. Soc. 147, 18406–18411 (2025).
Li, B. J. & Shi, Z. J. Ir-catalyzed highly selective addition of Pyridyl C–H Bonds to Aldehydes Promoted by Triethylsilane. Chem. Sci. 2, 488–493 (2011).
Liu, Z. et al. Borane-Catalyzed C3-Alkylation of Pyridines with Imines, Aldehydes, or Ketones as Electrophiles. J. Am. Chem. Soc. 144, 4810–4818 (2022).
Friedrich, M. et al. Direct C–H-sulfonylation of 6-membered Nitrogen-heteroaromatics. Tetrahedron Chem. 1, 100003 (2022).
Jung, S., Lee, H., Moon, Y., Jung, H. Y. & Hong, S. Site-Selective C–H Acylation of Pyridinium Derivatives by Photoredox Catalysis. ACS Catal. 9, 9891–9896 (2019).
Nagase, M., Kuninobu, Y. & Kanai, M. 4-Position-Selective C–H Perfluoroalkylation and Perfluoroarylation of Six-Membered Heteroaromatic Compounds. J. Am. Chem. Soc. 138, 6103–6106 (2016).
Choi, J., Laudadio, G., Godineau, E. & Baran, P. S. Practical and regioselective synthesis of C-4-Alkylated Pyridines. J. Am. Chem. Soc. 143, 11927–11933 (2021).
Moon, Y. et al. Visible light induced alkene aminopyridylation using n-aminopyridinium salts as bifunctional reagents. Nat. Commun. 10, 4117–4125 (2019).
Obradors, C. & List, B. Azine activation via silylium catalysis. J. Am. Chem. Soc. 143, 6817–6822 (2021).
Zhang, X. et al. Phosphorus-mediated Sp2–sp3 Couplings for C–H Fluoroalkylation of Azines. Nature 594, 217–222 (2021).
Fricke, P. J., Dolewski, R. D. & McNally, A. Four-selective pyridine alkylation via wittig olefination of dearomatized pyridylphosphonium ylides. Angew. Chem. Int. Ed. 60, 21283–21288 (2021).
Levy, J. N., Alegre-Requena, J. V., Liu, R., Paton, R. S. & McNally, A. Selective halogenation of pyridines using designed phosphine reagents. J. Am. Chem. Soc. 142, 11295–11305 (2020).
Koniarczyk, J. L., Greenwood, J. W., Alegre-Requena, J. V., Paton, R. S. & McNally, A. A Pyridine–pyridine cross-coupling reaction via dearomatized radical intermediates. Angew. Chem. Int. Ed. 58, 14882–14886 (2019).
Zhang, X. & McNally, A. Cobalt-catalyzed alkylation of drug-like molecules and pharmaceuticals using heterocyclic phosphonium salts. ACS Catal. 9, 4862–4866 (2019).
Zhang, X. & McNally, A. Phosphonium salts as pseudohalides: regioselective nickel-catalyzed cross-coupling of complex pyridines and diazines. Angew. Chem. Int. Ed. 56, 9833–9836 (2017).
Hilton, M. C. et al. Heterobiaryl Synthesis by Contractive C–C Coupling via P(V) Intermediates. Science 362, 799–804 (2018).
Hilton, M. C., Dolewski, R. D. & McNally, A. Selective functionalization of pyridines via heterocyclic phosphonium salts. J. Am. Chem. Soc. 138, 13806–13809 (2016).
Li, C. et al. Regioselective synthesis of 4-functionalized pyridines. Chem 10, 628–643 (2024).
Ma, C., Guo, J. F., Xu, S. S. & Mei, T. S. Recent advances in Asymmetric Organometallic Electrochemical Synthesis (AOES). Acc. Chem. Res. 58, 399–414 (2025).
Wang, Y. et al. Electrochemical late-stage functionalization. Chem. Rev. 123, 11269–11335 (2023).
Yuan, Y., Yang, J. & Lei, A. Recent advances in electrochemical oxidative cross-coupling with hydrogen evolution involving radicals. Chem. Soc. Rev. 50, 10058–10086 (2021).
Wang, Y., Wang, C., Li, M., Yu, Y. & Zhang, B. Nitrate electroreduction: mechanism insight, in situ characterization, performance evaluation, and challenges. Chem. Soc. Rev. 50, 6720–6733 (2021).
Novaes, L. F. T. et al. Electrocatalysis as an enabling technology for organic synthesis. Chem. Soc. Rev. 50, 7941–8002 (2021).
Bu, F. et al. Electrocatalytic reductive deuteration of arenes and heteroarenes. Nature 634, 592–599 (2024).
Michiyuki, T., Homölle, S. L., Pandit, N. K. & Ackermann, L. Electrocatalytic formal C(sp2)−H Alkylations via Nickel-Catalyzed cross-electrophile coupling with versatile Arylsulfonium Salts. Angew. Chem. Int. Ed. 63, e202401198 (2024).
Röckl, J. L., Pollok, D., Franke, R. & Waldvogel, S. R. A Decade of Electrochemical Dehydrogenative C,C-Coupling of Aryls. Acc. Chem. Res. 53, 45–61 (2019).
Sun, G. Q., Liao, L. L., Ran, C. K., Ye, J. H. & Yu, D. G. Recent advances in electrochemical carboxylation with CO2. Acc. Chem. Res. 57, 2728–2745 (2024).
Liao, L. L. et al. Electrochemical ring-opening dicarboxylation of strained carbon–carbon single bonds with CO2 facile synthesis of diacids and derivatization into Polyesters. J. Am. Chem. Soc. 144, 2062–2068 (2022).
Harwood, S. J. et al. Modular terpene synthesis enabled by mild electrochemical couplings. Science 375, 745–752 (2022).
Zhang, B. et al. Ni-electrocatalytic Csp3–Csp3 doubly decarboxylative coupling. Nature 606, 313–318 (2022).
Bu, F. et al. Electrocatalytic alkene hydrogenation/deuteration. J. Am. Chem. Soc. 147, 5785–5795 (2025).
Kumar, G. S. et al. Nickel-catalyzed chain-walking cross-electrophile coupling of alkyl and aryl halides and olefin hydroarylation enabled by electrochemical reduction. Angew. Chem. Int. Ed. 59, 6513–6519 (2020).
Hamby, T. B., LaLama, M. J. & Sevov, C. S. Controlling Ni redox states by dynamic ligand exchange for electroreductive Csp3–Csp2 coupling. Science 376, 410–416 (2022).
Su, S., Guo, Y., Parnitzke, B., Poerio, T. & Derosa, J. A Voltage-Controlled Strategy for Modular Shono-Type Amination. J. Am. Chem. Soc. 146, 28663–28668 (2024).
Zeng, W., Wang, Y., Peng, C. & Qiu, Y. Organo-mediator enabled electrochemical transformations. Chem. Soc. Rev. 54, 4468–4501 (2025).
Xiong, P. & Xu, H. C. Molecular photoelectrocatalysis for radical reactions. Acc. Chem. Res. 58, 299–311 (2025).
Shi, S. H., Liang, Y. & Jiao, N. Electrochemical oxidation induced selective C–C bond cleavage. Chem. Rev. 121, 485–505 (2020).
Cai, C. Y. et al. Photoelectrochemical Asymmetric Catalysis Enables Site- and Enantioselective Cyanation of Benzylic C–H Bonds. Nat. Catal. 5, 943–951 (2022).
Huang, C. & Xu, H. Scalable and practical electrooxidation of electron-deficient methylarenes to access aromatic aldehydes. Angew. Chem. Int. Ed. 64, e202504612 (2025).
Zeng, W., Peng, C. & Qiu, Y. Electrochemical Benzylic C–H Carboxylation. J. Am. Chem. Soc. 147, 13461–13470 (2025).
Zhang, C., Tang, H., Zhao, X., Shen, X. & Qiu, Y. Electrochemical Direct Hydroxylation of Benzylic C–H Bonds Assisted by HFIP. J. Am. Chem. Soc. 147, 23297–23307 (2025).
Zhang, W. et al. Electrochemically driven cross-electrophile coupling of alkyl halides. Nature 604, 292–297 (2022).
Sun, G. Q. et al. Electrochemical reactor dictates site selectivity in N-heteroarene carboxylations. Nature 615, 67–72 (2023).
Wang, M. et al. Room temperature construction of vicinal amino alcohols via electroreductive cross-coupling of N-heteroarenes and carbonyls. J. Am. Chem. Soc. 145, 10967–10973 (2023).
Nie, L. et al. Linear paired electrolysis enables redox-Neutral (3 + 2) annulation of benzofuran with vinyldiazo compounds. J. Am. Chem. Soc. 146, 31330–31338 (2024).
Huang, C., Tao, Y., Cao, X., Zhou, C. & Lu, Q. Asymmetric paired electrocatalysis: enantioselective olefin–sulfonylimine coupling. J. Am. Chem. Soc. 146, 1984–1991 (2023).
Tao, Y., Ma, W., Sun, R., Huang, C. & Lu, Q. Asymmetric paired electrolysis: enantioselective alkylation of Sulfonylimines via C(sp3)−H functionalization. Angew. Chem. Int. Ed. 63, e202409222 (2024).
Simmons, E. M. & Hartwig, J. F. On the interpretation of deuterium kinetic isotope effects in C-H bond functionalizations by transition-metal complexes. Angew. Chem. Int. Ed. 51, 3066–3072 (2012).
Zhao, Z. et al. Electrochemical CzH deuteration of pyridine derivatives with D2O. Nat. Commun. 15, 3832–3840 (2024).
Fang, X., Hu, X., Li, Q., Ni, S. & Ruan, Z. Paired Electro-Synthesis of Remote Amino Alcohols With/in H2O. Angew. Chem. Int. Ed. 64, e202418277 (2024).
Li, P. et al. Facile and general electrochemical deuteration of unactivated alkyl halides. Nat. Commun. 13, 3774 (2002).
Hamlin, T. A. et al. Toward a unified mechanism for oxoammonium salt-mediated oxidation reactions: a theoretical and experimental study using a hydride transfer model. J. Org. Chem. 80, 8150–8167 (2015).
Lambert, K. M., Bobbitt, J. M., Eldirany, S. A., Wiberg, K. B. & Bailey, W. F. Facile oxidation of primary amines to nitriles using an oxoammonium salt. Org. Lett. 16, 6484–6487 (2014).
Bailey, W. F., Bobbitt, J. M. & Wiberg, K. B. Mechanism of the oxidation of alcohols by oxoammonium cations. J. Org. Chem. 72, 4504–4509 (2007).
Zhang, J. Y. & Zhu, X. Q. Comparison Between 1,2-Dihydropyridine and 1,4-Dihydropyridine on Hydride-Donating Ability and Activity. Molecules 27, 5382 (2022).
Zhu, X. et al. Thermodynamics and kinetics of the hydride-transfer cycles for 1-Aryl-1,4-dihydronicotinamide and Its 1,2-Dihydroisomer. Chem. A Eur. J. 9, 3937–3945 (2003).
Xia, K., Shen, G. B. & Zhu, X. Q. Thermodynamics of various F420 Coenzyme models as sources of electrons, hydride ions, hydrogen atoms and protons in acetonitrile. Org. Biomol. Chem. 13, 6255–6268 (2015).
Acknowledgements
The authors are grateful for the financial support of the National Natural Science Foundation of China (22471080), Natural Science Foundation of Guangdong Province (2025A1515012079), Guangdong Basic and Applied Basic Research Foundation (2024B1515040027), and the Postdoctoral Fellowship Program of CPSF (GZB20250265).
Author information
Authors and Affiliations
Contributions
M.Z. conceived the idea, analyzed the data, directed the project, and wrote the manuscript. C.-Q.Z. and M.-R.W. carried out all the catalytic experiments. C.-Q.Z. drew the structures of all the obtained compounds, analyzed the single crystal structures, synthesized the raw materials and carried out NMR tests. M.-R. W. performed the DFT calculations. P.H.D. discussed the mechanistic aspects and revised the manuscript. All the authors have read the manuscript and agree with its content. All the authors contributed to the discussion during the whole project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Youai Qiu and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, C., Wang, M., Dixneuf, P.H. et al. Room-temperature electrochemical hydroxyalkylation of pyridines. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71858-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71858-2