Abstract
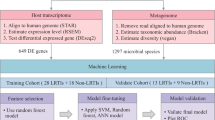
Distinguishing lower respiratory tract infection (LRTI) from incidental pathogen carriage (IPC) is clinically challenging. The immunologic and microbial factors defining the states of LRTI and IPC are poorly understood. Here, we perform host-microbe metatranscriptomic profiling of tracheal aspirates from 326 mechanically ventilated children with clinically adjudicated LRTI (n = 207), IPC (n = 70), or non-infectious respiratory failure (n = 49). In the airway microbiome, LRTI shows reduced alpha diversity and taxonomic richness, while IPC displays greater bacterial abundance, enrichment in respiratory anaerobes, and increased metabolic activity. At the host level, patients with LRTI exhibit a distinct lower airway transcriptional signature of innate and adaptive immune activation compared to those with IPC, who have similar transcriptional profiles to uninfected controls. Mediation analyses suggest the airway microbiome influences the host response to pathogens. An integrated host-microbe metatranscriptomic classifier accurately discriminates LRTI from IPC and controls (AUC = 0.89, 95% confidence interval (CI) 0.85–0.92). The single gene FABP4, encoding a macrophage-associated lipid chaperone and recently described pneumonia biomarker, performs similarly when combined with alpha diversity; FABP4 protein alone achieves an AUC = 0.88 (95% CI 0.82–0.93). Together, our findings reveal distinct ecological and immunologic archetypes defining LRTI and IPC, and support data-driven, biology-informed LRTI diagnostics incorporating host and microbial features.
Similar content being viewed by others
Data availability
FASTQ files containing the microbial sequencing reads following subtraction of reads aligning to the human genome have been deposited in the NCBI Sequence Read Archive under BioProject accession PRJNA748764. Processed host gene counts, microbial taxon counts, and deidentified clinical metadata are available in the GitHub repository associated with this work: https://github.com/infectiousdisease-langelier-lab/Incidental_pathogen_carriage (https://doi.org/10.5281/zenodo.19078486)87. Source data for each of the figures is provided in the Supplementary information. Source data are provided with this paper.
Code availability
The code used for data processing, statistical analyses, and figure generation is publicly available at: https://github.com/infectiousdisease-langelier-lab/Incidental_pathogen_carriage (https://doi.org/10.5281/zenodo.19078486)87.
References
Man, W. H., de Steenhuijsen Piters, W. A. A. & Bogaert, D. The microbiota of the respiratory tract: gatekeeper to respiratory health. Nat. Rev. Microbiol. 15, 259–270 (2017).
Robinson, J. Colonization and infection of the respiratory tract: what do we know?. Paediatr. Child Health 9, 21–24 (2004).
Vo, P. & Kharasch, V. S. Respiratory failure. Pediatrics Rev. 35, 476–486 (2014).
Panetti, B. et al. Acute respiratory failure in children: a clinical update on diagnosis. Children 11, 1232 (2024).
Fàbregas, N. et al. Histopathologic and microbiologic aspects of ventilator-associated pneumonia. Anesthesiology 84, 760–771 (1996).
Jeffrey, M., Denny, K. J., Lipman, J. & Conway Morris, A. Differentiating infection, colonisation, and sterile inflammation in critical illness: the emerging role of host-response profiling. Intensive Care Med. 49, 760–771 (2023).
Lydon, E. C., Ko, E. R. & Tsalik, E. L. The host response as a tool for infectious disease diagnosis and management. Expert Rev. Mol. Diagnostics 18, 723–738 (2018).
Vaughn, V. M. et al. Excess antibiotic treatment duration and adverse events in patients hospitalized with pneumonia: a multihospital cohort study. Ann. Intern. Med. 171, 153–163 (2019).
Gupta, A. B. et al. Inappropriate diagnosis of pneumonia among hospitalized adults. JAMA Intern. Med. 184, 548–556 (2024).
Anadol, D., Aydin, Y. Z. & Göçmen, A. Overdiagnosis of pneumonia in children. Turk. J. Pediatr. 43, 205–209 (2001).
Pan, H., Cui, B., Huang, Y., Yang, J. & Ba-Thein, W. Nasal carriage of common bacterial pathogens among healthy kindergarten children in Chaoshan region, southern China: a cross-sectional study. BMC Pediatr. 16, 161 (2016).
Regev-Yochay, G. et al. Nasopharyngeal carriage of Streptococcus pneumoniae by adults and children in community and family settings. Clin. Infect. Dis. 38, 632–639 (2004).
Nokso-Koivisto, J., Kinnari, T. J., Lindahl, P., Hovi, T. & Pitkäranta, A. Human picornavirus and coronavirus RNA in nasopharynx of children without concurrent respiratory symptoms. J. Med. Virol. 66, 417–420 (2002).
Parker, A. M. et al. Upper respiratory Streptococcus pneumoniae colonization among working-age adults with prevalent exposure to overcrowding. Microbiol. Spectr. 12, e00879-24 (2024).
Desai, A. P. et al. Decline in pneumococcal nasopharyngeal carriage of vaccine serotypes after the introduction of the 13-valent pneumococcal conjugate vaccine in children in Atlanta, Georgia. Pediatr. Infect. Dis. J. 34, 1168–1174 (2015).
Bogaert, D., De Groot, R. & Hermans, P. W. M. Streptococcus pneumoniae colonisation: the key to pneumococcal disease. Lancet Infect. Dis. 4, 144–154 (2004).
Huang, S. S. et al. Healthcare utilization and cost of pneumococcal disease in the United States. Vaccine 29, 3398–3412 (2011).
Vaneechoutte, M., Verschraegen, G., Claeys, G., Weise, B. & Van den Abeele, A. M. Respiratory tract carrier rates of Moraxella (Branhamella) catarrhalis in adults and children and interpretation of the isolation of M. catarrhalis from sputum. J. Clin. Microbiol. 28, 2674–2680 (1990).
Faden, H., Harabuchi, Y. & Hong, J. J. Epidemiology of Moraxella catarrhalis in children during the first 2 years of life: relationship to otitis media. J. Infect. Dis. 169, 1312–1317 (1994).
Ejlertsen, T., Thisted, E., Ebbesen, F., Olesen, B. & Renneberg, J. Branhamella catarrhalis in children and adults. A study of prevalence, time of colonisation, and association with upper and lower respiratory tract infections. J. Infect. 29, 23–31 (1994).
Most, Z. M., Perl, T. M. & Sebert, M. Respiratory virus infections in symptomatic and asymptomatic children upon hospital admission: new insights. Antimicrob. Steward Health. Epidemiol. 4, e162 (2024).
Jansen, R. R. et al. Frequent detection of respiratory viruses without symptoms: toward defining clinically relevant cutoff values. J. Clin. Microbiol. 49, 2631–2636 (2011).
Self, W. H. et al. Respiratory viral detection in children and adults: comparing asymptomatic controls and patients with community-acquired pneumonia. J. Infect. Dis. 213, 584–591 (2016).
Dickson, R. P. The microbiome and critical illness. Lancet Respiratory Med. 4, 59–72 (2016).
Mourani, P. M. et al. Temporal airway microbiome changes related to ventilator-associated pneumonia in children. Eur. Respir. J. 57, 2001829 (2021).
Durairaj, L. et al. Patterns and density of early tracheal colonization in intensive care unit patients. J. Crit. Care 24, 114–121 (2009).
Ewig, S. et al. Bacterial colonization patterns in mechanically ventilated patients with traumatic and medical head injury. Incidence, risk factors, and association with ventilator-associated pneumonia. Am. J. Respir. Crit. Care Med. 159, 188–198 (1999).
Mick, E. et al. Integrated host/microbe metagenomics enables accurate lower respiratory tract infection diagnosis in critically ill children. J. Clin. Invest. 133, e165904 (2023).
Tsitsiklis, A. et al. Lower respiratory tract infections in children requiring mechanical ventilation: a multicentre prospective surveillance study incorporating airway metagenomics. Lancet Microbe 3, e284–e293 (2022).
Lydon, E. et al. Proteomic profiling of the local and systemic immune response to pediatric respiratory viral infections. mSystems 10, e0133524 (2025).
United States Centers for Disease Control and Prevention. CDC/NHSN Surveillance Definitions for Specific Types of Infections. https://www.cdc.gov/nhsn/pdfs/pscmanual/pcsmanual_current.pdf (2021).
Nitu, M. E. & Eigen, H. Respiratory failure. Pediatrics Rev. 30, 470–478 (2009).
Lin, H. & Peddada, S. D. Analysis of compositions of microbiomes with bias correction. Nat. Commun. 11, 3514 (2020).
Dong, W. et al. An expanded database and analytical toolkit for identifying bacterial virulence factors and their associations with chronic diseases. Nat. Commun. 15, 8084 (2024).
St Geme, J. W. & Yeo, H.-J. A prototype two-partner secretion pathway: the Haemophilus influenzae HMW1 and HMW2 adhesin systems. Trends Microbiol. 17, 355–360 (2009).
Akhtar, A. A. & Turner, D. P. J. The role of bacterial ATP-binding cassette (ABC) transporters in pathogenesis and virulence: therapeutic and vaccine potential. Microb. Pathogenesis 171, 105734 (2022).
Beghini, F. et al. Integrating taxonomic, functional, and strain-level profiling of diverse microbial communities with bioBakery 3. eLife 10, e65088 (2021).
Furuhashi, M., Saitoh, S., Shimamoto, K. & Miura, T. Fatty acid-binding protein 4 (FABP4): pathophysiological insights and potent clinical biomarker of metabolic and cardiovascular diseases. Clin. Med. Insights Cardiol. 8, 23–33 (2014).
Schneider, W. M., Chevillotte, M. D. & Rice, C. M. Interferon-stimulated genes: a complex web of host defenses. Annu. Rev. Immunol. 32, 513–545 (2014).
Hofer, U. Granzyme B’s roundhouse kick against bacteria. Nat. Rev. Microbiol 15, 707–707 (2017).
Icardi, M. et al. CD64 index provides simple and predictive testing for detection and monitoring of sepsis and bacterial infection in hospital patients. J. Clin. Microbiol. 47, 3914–3919 (2009).
Albiger, B., Dahlberg, S., Henriques-Normark, B. & Normark, S. Role of the innate immune system in host defence against bacterial infections: focus on the Toll-like receptors. J. Intern. Med. 261, 511–528 (2007).
Lydon, E. C. et al. Pulmonary FABP4 is an inverse biomarker of pneumonia in critically ill children and adults. Am. J. Respir. Crit. Care Med. 210, 1480–1483 (2024).
Phan, H. V. et al. Integrating a host biomarker with a large language model for diagnosis of lower respiratory tract infection. Nat. Commun. 16, 10882 (2025).
Langelier, C. et al. Integrating host response and unbiased microbe detection for lower respiratory tract infection diagnosis in critically ill adults. Proc. Natl. Acad. Sci. USA 115, E12353–E12362 (2018).
Flanagan, J. L. et al. Loss of bacterial diversity during antibiotic treatment of intubated patients colonized with Pseudomonas aeruginosa. J. Clin. Microbiol. 45, 1954–1962 (2007).
Bai, A. D. et al. Anaerobic antibiotic coverage in aspiration pneumonia and the associated benefits and harms: a retrospective cohort study. CHEST 166, 39–48 (2024).
Marik, P. E. & Careau, P. The role of anaerobes in patients with ventilator-associated pneumonia and aspiration pneumonia: a prospective study. Chest 115, 178–183 (1999).
Metlay, J. P. et al. Diagnosis and treatment of adults with community-acquired pneumonia. An official clinical practice guideline of the American Thoracic Society and Infectious Diseases Society of America. Am. J. Respir. Crit. Care Med. 200, e45–e67 (2019).
Wesolowska-Andersen, A. et al. Dual RNA-seq reveals viral infections in asthmatic children without respiratory illness which are associated with changes in the airway transcriptome. Genome Biol. 18, 12 (2017).
Wolsk, H. M. et al. Picornavirus-induced airway mucosa immune profile in asymptomatic neonates. J. Infect. Dis. 213, 1262–1270 (2016).
Følsgaard, N. V. et al. Pathogenic bacteria colonizing the airways in asymptomatic neonates stimulates topical inflammatory mediator release. Am. J. Respir. Crit. Care Med. 187, 589–595 (2013).
Bisgaard, H. et al. Childhood asthma after bacterial colonization of the airway in neonates. N. Engl. J. Med. 357, 1487–1495 (2007).
Cho, H.-J. et al. Differences and similarities between the upper and lower airway: focusing on innate immunity. Rhinology 59, 441–450 (2021).
Mick, E. et al. Upper airway gene expression reveals suppressed immune responses to SARS-CoV-2 compared with other respiratory viruses. Nat. Commun. 11, 5854 (2020).
Mick, E. et al. Upper airway gene expression shows a more robust adaptive immune response to SARS-CoV-2 in children. Nat. Commun. 13, 3937 (2022).
Ichinohe, T. et al. Microbiota regulates immune defense against respiratory tract influenza A virus infection. Proc. Natl. Acad. Sci. USA 108, 5354–5359 (2011).
Vanderweele, T. J. & Vansteelandt, S. Conceptual issues concerning mediation, interventions and composition. Stat. Interface 2, 457–468 (2009).
Gaston, D. C. Clinical metagenomics for infectious diseases: progress toward operational value. J. Clin. Microbiol. 61, e01267-22 (2023).
Benoit, P. et al. Seven-year performance of a clinical metagenomic next-generation sequencing test for diagnosis of central nervous system infections. Nat. Med. 30, 3522–3533 (2024).
National Human Genome Research Institute. DNA Sequencing Costs: Data. https://www.genome.gov/about-genomics/fact-sheets/DNA-Sequencing-Costs-Data (2024).
Self, W. H. et al. Procalcitonin as a marker of etiology in adults hospitalized with community-acquired pneumonia. Clin. Infect. Dis. 65, 183–190 (2017).
van der Meer, V., Neven, A. K., van den Broek, P. J. & Assendelft, W. J. J. Diagnostic value of C reactive protein in infections of the lower respiratory tract: systematic review. BMJ 331, 26 (2005).
Patel, R. et al. Clinically adjudicated reference standards for evaluation of infectious diseases diagnostics. Clin. Infect. Dis. 76, 938–943 (2023).
A’Court, C. H. et al. Microbiological lung surveillance in mechanically ventilated patients, using non-directed bronchial lavage and quantitative culture. Q J. Med. 86, 635–648 (1993).
Gu, W. et al. Depletion of Abundant Sequences by Hybridization (DASH): using Cas9 to remove unwanted high-abundance species in sequencing libraries and molecular counting applications. Genome Biol. 17, 1–13 (2016).
Gold, L. et al. Aptamer-based multiplexed proteomic technology for biomarker discovery. PLoS One 5, e15004 (2010).
Kim, C. H. et al. Stability and reproducibility of proteomic profiles measured with an aptamer-based platform. Sci. Rep. 8, 8382 (2018).
Candia, J. et al. Variability of 7K and 11K SomaScan Plasma Proteomics Assays. J. Proteome Res. 23, 5531–5539 (2024).
Kalantar, K. L. et al. IDseq-An open source cloud-based pipeline and analysis service for metagenomic pathogen detection and monitoring. Gigascience 9, giaa111 (2020).
Lu, D. et al. Simultaneous detection of pathogens and antimicrobial resistance genes with the open source, cloud-based, CZ ID platform. Genome Med. 17, 46 (2025).
Bray, N. L., Pimentel, H., Melsted, P. & Pachter, L. Near-optimal probabilistic RNA-seq quantification. Nat. Biotechnol. 34, 525–527 (2016).
Chen, S., Zhou, Y., Chen, Y. & Gu, J. fastp: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 34, i884–i890 (2018).
Ruby, J. G., Bellare, P. & Derisi, J. L. PRICE: software for the targeted assembly of components of (Meta) genomic sequence data. G3 Bethesda 3, 865–880 (2013).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat Methods. 9, 357–359 (2012).
Jain, S. et al. Community-acquired pneumonia requiring hospitalization among U.S. adults. N. Engl. J. Med. 373, 415–427 (2015).
Jain, S. et al. Community-acquired pneumonia requiring hospitalization among U.S. children. N. Engl. J. Med. 372, 835–845 (2015).
Iwai, S. et al. The lung microbiome of Ugandan HIV-infected pneumonia patients is compositionally and functionally distinct from that of San Franciscan Patients. PLOS One 9, e95726 (2014).
Magill, S. S. et al. Multistate point-prevalence survey of health care-associated infections. N. Engl. J. Med. 370, 1198–1208 (2014).
Soneson, C., Love, M. I. & Robinson, M. D. Differential analyses for RNA-seq: transcript-level estimates improve gene-level inferences. F1000Res. 4, 1521 (2015).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Ritchie, M. E. et al. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 43, e47 (2015).
Croft, D. et al. The Reactome pathway knowledgebase. Nucleic Acids Res 42, D472–D477 (2014).
Tingley, D., Yamamoto, T., Hirose, K., Keele, L. & Imai, K. mediation: R Package for causal mediation analysis. J. Stat. Softw. 59, 1–38 (2014).
Friedman, J. et al. glmnet: Lasso and Elastic-Net Regularized Generalized Linear Models CRAN (Comprehensive R Archive Network, 2023).
Robin, X. et al. pROC: an open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinforma. 12, 77 (2011).
Lydon, E. & Glascock, A. Code for: Host–Microbiome Archetypes Differentiate Infection from Pathogen Carriage in the Human Lower Airway. https://doi.org/10.5281/zenodo.19078486 (2026).
Acknowledgements
We thank all patients and their families for participating in this study. We acknowledge the contributions of the principal investigators, coinvestigators, and research coordinators at each of the study sites where patients were enrolled. This study was supported by the Collaborative Pediatric Critical Care Research Network, the Eunice Kennedy Shriver National Institute of Child Health and Human Development, the National Heart, Lung and Blood Institute UG1HD083171 (P.M.M.), 1R01HL124103 (P.M.M.), R01AI182308 (P.M.M. and C.R.L.), 5R01HL155418 (C.R.L.), and the Chan Zuckerberg Biohub (C.R.L.). The study sponsors were not involved in study design, in the collection, analysis, or interpretation of data, in writing of the manuscript, or in the decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Contributions
E.C.L., P.D., A.G. and C.R.L. conceived and designed the study. C.R.L. and P.M.M. supervised the study. C.R.L., P.M.M., C.M.M., M.K.L. and L.A. enrolled the cohort. C.R.L., P.M.M., C.M.M. and M.K.L. performed clinical adjudications. P.D., A.G., H.V.P., E.M. and B.D.W. generated the data. E.C.L., P.D. and A.G. analyzed the data and generated the figures. E.C.L. and C.R.L. wrote the manuscript. E.C.L., P.D., A.G., H.V.P., C.M.M., M.K.L., J.A., E.M., B.D.W., J.L.D., L.A., P.M.M. and C.R.L. edited the manuscript. C.R.L. and P.M.M. acquired funding.
Corresponding author
Ethics declarations
Competing interests
E.C.L., H.V.P., C.R.L., and P.M.M. are listed as inventors on a provisional patent application titled “Diagnostic & prognostic immune markers for critical infections” filed by the University of California San Francisco and currently pending. All other authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Vanya Gant and Leopoldo Segal for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lydon, E.C., Deosthale, P., Glascock, A. et al. Host–microbiome archetypes differentiate infection from pathogen carriage in the human lower airway. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71863-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71863-5