Abstract
The Davos Alzheimer’s Collaborative (DAC) Egypt Cohort (DAC-Egypt) is a newly established longitudinal study of cognitive aging in a community-based convenience sample of older Egyptian adults. The cohort’s purpose is to characterize trajectories of cognitive decline and dementia risk factors in an understudied population, filling a critical gap in aging research in the Middle East. Participants (n = 1,530) aged 55 and above were recruited via regionally diverse convenience sampling, with detailed baseline data collected on demographics, health status, lifestyle, and cognitive function. Cognitive assessments included both traditional neuropsychological testing and innovative digital tools (digital voice/speech & olfactory-sensory assessments) to enable comprehensive monitoring. Key preliminary findings indicated a high prevalence of chronic diseases and notable socioeconomic disparities in cognitive performance among older Egyptians. Blood samples were collected from 98% of participants, and dried blood spot (DBS) cards were obtained for 88% of participants to facilitate future biomarker and genetic research. This study seeks to enrich the scientific field of dementia and Alzheimer’s disease and related disorders (ADRD) for early detection and intervention strategies for cognitive health in aging populations.
Similar content being viewed by others
Introduction
Progress against Alzheimer’s disease (AD) requires a broad and nuanced understanding of the syndrome. Yet nearly all research to date has focused on white populations of Western European origin, excluding 90% of the world. This lack of diversity limits our ability to fully understand AD and identify new treatment targets and biomarkers. The goal of DAC’s Global Cohorts Program is to generate data and insights relevant to Alzheimer’s disease and brain aging from a global interlinked ecosystem of cohorts that are representative of the heterogeneity of the disease, patients’ environments, and lived experiences. The DAC-Egypt cohort is a key component of DAC’s Global Cohorts Program given high load of known dementia risk factors and tremendous genetic, cultural and socioeconomic diversity in the region.
Egypt is the most populous country MENA region (over 110 million people) and while its population remains relatively young, the proportion of older adults is rapidly growing, creating an urgent need to understand determinants of cognitive decline in this context. Currently, around 8% of Egyptians are aged 60 years or above, a share expected to nearly double by 20501. This demographic shift is accompanied by rising incidence of age-related conditions, including dementia and cognitive impairment, which pose significant public health challenges in the region. Alzheimer’s disease and related dementias are a growing concern in Egypt, yet there is a paucity of epidemiological data on cognitive aging in the country. Rough estimates suggest that about 1% of Egyptians over 50 years old have dementia, increasing to ~10% among those over 802.
The uniqueness of the Egyptian older adult population necessitates a structured, local context for study design. Egyptians face a “Triple Burden” of risk, a high prevalence of metabolic disorders (diabetes and hypertension), unique environmental hazards, and significant socioeconomic disparities. Egypt faces tremendous challenges with the burden of non-communicable diseases that account for 82% of mortality, driven by cardiovascular diseases, diabetes, cancer, and chronic respiratory illnesses3,4. The prevalence of hypertension, overweight/obesity, tobacco use, and insufficient physical activity is among the highest in the Middle East and North Africa region5. Egypt has the largest rate of diabetes in the region6,7 and faces high levels of air pollution8.
Beyond metabolic risk, cognitive aging in Egypt is deeply embedded in family-based, multigenerational households; nearly 90% of our cohort co-resides with immediate family, which serves as the primary site for informal caregiving and social support. Depression affects 64.6% of older adults9. Educational attainment remains a critical determinant, as only 25% of adults have education beyond the primary level, with just 18% of these being women10. This significant educational disparity—compounded by the fact that 53.4% of our cohort is illiterate—necessitated the use of non-literacy-dependent assessment tools to distinguish true cognitive impairment from lifelong low education. Consequently, a higher prevalence of dementia has been consistently found in illiterates11,12,13. Finally, the study’s intentional focus on the historically underserved rural population (comprising 88.2% of the sample) ensures the capture of region-specific environmental and occupational risks often invisible in urban-centric research.
Understanding how these factors, along with Egypt’s distinct social norms and socioeconomic conditions, affect cognitive aging will enrich the comparative literature on dementia. Furthermore, older Egyptian women and men experience different health outcomes shaped by gender norms—for example, women have longer life expectancy but may suffer poorer health and lower cognitive test scores than men in later life14,15. Comprehensive data on these patterns have been scarce. In this context, the Davos Alzheimer’s Collaborative (DAC) Egypt Cohort was established as part of an unprecedented global effort to study Alzheimer’s disease across diverse populations. By following a cohort of aging Egyptians, this study addresses a critical gap by examining the prevalence and predictors of cognitive decline in an Arab middle-income country. This cohort profile describes the study’s design and methodology, the characteristics of the participants, and key early findings, and it situates the implications of those findings for research and policy on cognitive health in Egypt and similar settings.
Results
Demographic characteristics
The definitive Wave 1 baseline successfully enrolled 1530 older adults after excluding those with missing informant data, demonstrating the feasibility of the recruitment strategy on a large scale (Table 1). The cohort includes a broad age range (55–98 years; Mean = 66.8, SD = 8.1), reflecting the strategic inclusion of the younger-older adult group to capture early aging and mid-life risk factors. The gender distribution consists of 46.0% males (n = 704) and 54.0% females (n = 826), a slight predominance of women that is consistent with the national demographic profile for older age groups in Egypt (CAPMAS, 2024).
Educational attainment within the cohort varies significantly, highlighting the socio-demographic diversity and the specific challenges of the Egyptian context. Over half of the participants (53.4%; n = 817) reported being illiterate with no formal education, a group primarily composed of older women from rural areas. In contrast, only 4.1% (n = 63) attained a college education, and a mere 0.8% (n = 12) achieved higher education degrees.
Residential distribution confirms the study’s intentional overrepresentation of rural populations; 88.2% (n = 1349) of participants reside in rural villages, while 11.8% (n = 181) live in urban governorates. This distribution ensures the cohort captures the unique health profiles and lifestyle patterns of the rural population, who are historically underrepresented in clinical dementia research.
Educational attainment among participants varied significantly, with a notable skew toward lower levels of formal education. 53.4% of the cohort reported being uneducated, many of whom were likely older women from rural areas. In contrast, only 4.1% of participants attained a college education, and a mere 0.8% achieved higher education. This heterogeneity in educational backgrounds will enable analysis of how literacy and educational attainment influence late-life cognitive outcomes.
The residential distribution highlights that the vast majority of participants (88.2%) reside in rural areas, reflecting an intentional overrepresentation of rural populations in the study. This ensures that the study captures the unique lifestyle patterns and health profiles of rural Egyptians, who often differ from their urban counterparts. While future sampling will include all regions, rural residents are expected to remain a substantial segment of the full cohort, given that approximately half of Egypt’s population lives outside major cities.
Household composition and living arrangements
Analysis of living arrangements revealed that the cohort is predominantly embedded in family-based households. The most common arrangement was living with a spouse and children (53.6%), followed by living with children only (20.6%), and with a spouse only (15.9%). In total, nearly 90% of participants co-resided with immediate family members. Only 2.5% lived alone, while the remaining 7.4% resided in extended family households that included siblings or parents. This pattern highlights the multigenerational family structure as a key context for social support and caregiving in this population.
Health conditions and lifestyle factors
Several health conditions and lifestyle risk factors were identified among the cohort, as summarized in Table 2.
Chronic health conditions are highly prevalent in the cohort. Approximately 70.0% (95% CI: 67.7–72.3%) of participants reported having at least one diagnosed chronic illness. Hypertension was the most frequent condition at 48.2% (95% CI: 45.7–50.7%), followed by Vision Impairment at 36.6% (95% CI: 34.2–39.0%) and Diabetes at 28.4% (95% CI: 26.1–30.7%).
Lifestyle risk factors were also assessed, revealing important areas for intervention. Smoking was more prevalent among men, with roughly 40% of male participants being current smokers, while it was rare among women, consistent with cultural norms that discourage female smoking. Obesity and overweight were highly prevalent in the cohort, with a mean BMI of 27.6 kg/m². Overall, over 60% of participants were overweight (BMI ≥ 25 kg/m²), and approximately 30% were obese (BMI ≥ 30 kg/m²). This burden was disproportionately higher among women; over 70% were classified as overweight, with an obesity rate approaching 40%. Physical inactivity was another notable issue, particularly among older women in rural areas, who often have limited opportunities for formal exercise.
The combination of widespread hypertension, diabetes, smoking (among men), and obesity indicates a substantial cardiovascular and metabolic risk profile for cognitive decline in this population. These findings underscore the need for targeted interventions to address these risk factors and improve health outcomes among older adults in Egypt.
Cognitive assessments and digital tools
Data and biospecimens were collected from the total cohort (N = 1530) using the following methods:
-
Mili app: A digital vocal biomarker assessment was administered to 1184 participants (77.4% of the cohort).
-
SensifyAware test: An olfactory-based cognitive function test was completed by 900 participants (58.8% of the cohort).
-
Blood samples: Venous blood samples were successfully collected from 1499 participants (98.0% of the cohort) for core biomarker analysis.
-
Dried blood spot (DBS) cards: DBS cards were additionally obtained from 1346 participants (88% of the cohort) for future molecular and genetic research.
Note on digital assessment completion: Participants who did not complete the Mili App (346 participants, 22.6%) or SensifyAware Test (630 participants, 41.2%) were unable to finish due to technical difficulties, user challenges, or partial completion depicted in Table 3.
Discussion
Although the DAC Egypt Cohort is in its early stages, several significant findings have already emerged from this baseline study. These results offer initial insights into the cognitive health of older Egyptians and highlight patterns for future longitudinal exploration. Regarding digital assessments, while preliminary feasibility is high, specific validation analyses are ongoing. Initial analysis also highlights variations in cognitive performance across governorates, potentially linked to socioeconomic and educational disparities.
One prominent observation is the extensive burden of chronic health conditions. Approximately 70% of participants (n = 1071) reported at least one chronic illness, with 48.2% (n = 737) being hypertensive and 28.4% (n = 435) having diabetes. These vascular and metabolic conditions are critical risk factors for cognitive decline. This high prevalence confirms that a large fraction of the Egyptian older population faces combined health risks, such as the compounded effect of diabetes and low education (53.4% of the cohort), which will be a primary focus of our interaction analyses.
Cognitive performance at baseline varies markedly across socio-demographic subgroups, particularly by education and residency. Participants with higher educational attainment performed better on memory and thinking tests than those with little or no formal education. An important nuance is that while urban residents initially appeared to perform better, the rural-urban gap in cognitive performance narrows significantly after adjusting for education level. This suggests that schooling and economic resources, rather than geography alone, drive cognitive inequalities.
The baseline data reveal gender differences shaped by social context. While overall cognitive scores did not differ drastically by sex after accounting for education, specific domains showed variation. Men performed better on numeracy-related tasks, whereas women demonstrated equal or slightly better performance on verbal memory when education levels were similar. These findings will be probed further, considering societal factors such as widowhood or lower social support that disproportionately affect older Egyptian women.
A key quantitative finding is the high engagement with digital tools despite the cohort’s low education levels. The Mili speech app achieved a 77.4% completion rate (n = 1184), and SensifyAware reached 58.8% (n = 900). These completion rates indicate that digital health tools can be successfully utilized by older individuals in Egypt with proper support, opening the door for technology-enhanced remote monitoring.
The early results from the DAC Egypt Cohort carry several important implications for research and policy on aging and cognitive decline in low- and middle-income countries. This cohort profile not only establishes the feasibility of conducting a large-scale study of cognitive aging in Egypt but also situates the findings in a broader context.
First and foremost, this cohort addresses a major knowledge gap regarding aging in the (MENA) region. Prior to this project, data on dementia and cognitive impairment in Egypt were extremely limited. By systematically collecting longitudinal data on cognitive function and its determinants, the DAC Egypt Cohort provides an infrastructure to generate evidence on questions that were previously unanswerable in the region4,10. For example, what is the incidence of Alzheimer’s disease in an Arab population, and how do risk factors like diabetes or illiteracy contribute? The cohort’s design, harmonized with Health and Retirement studies and other DAC global cohorts, means that the findings will be directly comparable with those from Europe, Asia, and the Americas. Early indications of strong education effects on cognition reinforce global theories of cognitive reserve, but the cohort will also investigate factors less studied elsewhere, such as the potential effects of long-term exposure to regional environmental toxins (e.g., pesticides in rural farming communities) and the impact of the Triple Burden of metabolic, environmental, and infectious risk on cognitive health. In the coming years, analyses from this study can help generalize findings across diverse populations and ensure that global models of dementia risk are not solely based on Western data.
The findings have significant public health implications for Egypt. The high prevalence of risk factors like hypertension (48.2%) and diabetes (28.4%) in those over 55 signals a need for integrating cognitive health into chronic disease management. Furthermore, policy interventions must account for the unique Egyptian household structure; with 90% of participants residing in multigenerational homes, caregiving strategies must be community- and family-centered rather than solely institution-based. Another critical insight is the socio-economic disparity in cognitive aging, which points policymakers toward the importance of improving lifelong learning opportunities. Additionally, the fact that poorer individuals may under-report health issues implies that strengthening primary healthcare systems in disadvantaged areas (rural clinics, outreach programs) could lead to earlier detection. Scientific Contributions On the research front, the DAC Egypt Cohort offers an opportunity to integrate digital tools like Mili and SensifyAware. While these tools are currently in the feasibility phase, if they prove predictive, they could revolutionize screening in low-resource settings by allowing for efficient, large-scale remote monitoring. The cohort also sets the stage for intervention studies, such as randomized trials for blood pressure control with cognitive outcomes.
The DAC Egypt Cohort is part of a global network under the Davos Alzheimer’s Collaborative. Comparisons with China’s aging cohorts or Europe’s SHARE study can highlight universal versus context-specific traits. Egypt’s older adults share high vascular risk factors with peers in other middle-income countries but possess unique characteristics—such as lower education levels (53.4% illiterate) and distinctive gendered life-course exposures—that may modulate dementia risk. Regionally, the success of this cohort can catalyze similar research in neighboring countries, with the North African Dementia Registry (NADR) serving as a direct offshoot to amplify regional impact that will help put Egypt’s data in a regional context and amplify its impact.
The DAC Egypt Cohort is built upon a strong framework of collaborations, both within Egypt and internationally. These partnerships enhance the study’s capacity, ensure high-quality implementation, and facilitate the broader use of the data for scientific discovery. Internationally, the study is a flagship project under the Davos Alzheimer’s Collaborative (DAC), connecting the Egypt cohort team with a global network of researchers and resources. In return, the cohort contributes de-identified data to DAC’s global data partner, the ADDI platform (https://www.alzheimersdata.org/), enabling meta-analyses and cross-national comparisons. The DAC Egypt cohort has been intentionally aligned in terms of data and bio-sample collection protocols with other DAC Global Cohorts in Kenya, India, and Chile.
A key research collaboration with University College London (UCL) has led to the development of the North African Dementia Registry (NADR). The NADR aims to compile regional data to support research into genetic and environmental risk factors distinctive to North Africa, such as specific regional environmental toxins or the impact of the Triple Burden of metabolic diseases. The Egyptian cohort serves as a feeder into this registry by identifying and characterizing dementia cases. Furthermore, select Egyptian researchers are receiving training at UCL to enhance protocol developments, such as future amyloid/tau biomarker collection.
A cornerstone of the project’s ethos is open data. Approximately one year after each wave’s completion, clean and anonymized data will be made available free of charge to bona fide researchers. The cohort has also spurred industry partnerships, such as with the developers of the Mili App and SensifyAware, to validate digital health solutions and olfactory sensing in an Arabic-speaking population.
In interpreting the cohort’s findings, some caveats are acknowledged. The challenge of attrition in longitudinal studies—driven by internal migration and regional instability—requires intensive monitoring. While we have implemented retention strategies (e.g., medical follow-ups and “return of value” health metrics), we will address future loss-to-follow-up using analytic strategies such as Inverse Probability Weighting (IPW) or Multiple Imputation to minimize selection bias.
Additionally, while our cognitive measures are culturally adapted, subtle bias may remain for 53.4% of the cohort with no formal education. To address this, we do not use raw cut-offs but define cognitive impairment based on education-stratified norms derived from our local validation study, ensuring illiterate participants are compared only against their peers. Furthermore, we acknowledge that self-reported variables, such as the “Polluted Environment” metric (3.7% prevalence), may underestimate objective exposure compared to sensor data.
Methods
Cohort establishment
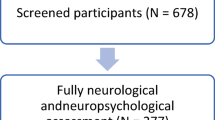
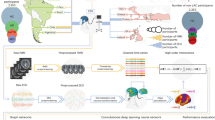
The DAC-Egypt baseline data collection began in 2024 as a collaborative effort between the American University in Cairo and international partners under the Davos Alzheimer’s Collaborative. This study was approved by the American University in Cairo’s Institutional Review Board (Case# 2023-2024-181). All participants provided written or oral informed consent in accordance with the World Medical Association Declaration of Helsinki. Participants recruitment used community-based convenience sampling approach, ensuring coverage of both urban (11.8%) and rural (88.2%) areas with initial enrollment response rate 79.7%. We have provided a Participant Flow Diagram (Fig. 1) to track the recruitment pipeline, including reasons for non-participation to assist in assessing potential selection. Recruitment was conducted across eight governorates to ensure geographic diversity, with a deliberate focus on enrolling participants from rural health centers to address a critical gap in research inclusion. and spanned across eight governorates: Cairo, Beni Suef, Dakahlia, Sohag, Alexandria, Ismailia, Menoufia, and Al-Sharqia to ensure coverage of Egypt’s three major ecological zones: the Urban Governorates, Lower Egypt (Delta), and Upper Egypt (Fig. 2).
Of 1920 individuals approached, 390 were excluded (primary reasons: lack of time [68%], refusal of blood draw [25%]). The final enrolled cohort comprised 1530 individuals. All with complete core data were analyzed. Voluntary sub-study participation rates are shown (venous blood: 98%; DBS: 88%; Mili digital app: 77.4%; Sensify olfactory test: 58.8%) for the Egyptian population.
Data collection sites in Egyptian governorates and tools used for cognitive assessment.
All individuals aged 55 years or older were eligible for participation. The lower age cutoff of 55 was chosen to study the transition into older age and to examine midlife risk factors 5 years before the typical retirement age in Egypt. There was no strict exclusion criteria related to health status at baseline; cognitively impaired individuals were included, with caregiver or informant assistance provided during assessments when necessary. This approach allowed for the study of the full spectrum of cognitive function, from normal aging to dementia.
The cohort is designed for biennial (2-year) follow-up waves. To minimize bias from attrition, we will monitor loss-to-follow-up and utilize analytic strategies such as IPW or multiple imputation in longitudinal models. Our retention strategy relies on local team members and their established relationship with the participants, with follow-up calls 3–5 months after the study to check up on participants and offer medical assistance in the form of medication or free physician consult. We also leveraged the “return of value” strategy by providing participants with basic health metrics (blood pressure, glucose check) at each visit, which incentivizes retention in areas with limited healthcare access. Reasons for non-participation primarily included lack of time (68%), refusal of blood draw (25%) and lack of interest in digital tools (8%).
Data collection
Participants underwent a comprehensive cognitive assessment using the validated Egyptian Harmonized Cognitive Assessment Protocol (HCAP) battery16 along with digital assessments (Mili app and SensifyAware). Additionally, biological samples were collected to support biomarker research, including whole blood samples and DBS cards. Figure 2 provides an overview of data collection sites and modalities used.
The HCAP is a 30-min in-depth cognitive test module covering multiple domains (memory, executive function, language, orientation, and visuospatial ability) administered by specially trained interviewers. Given the high proportion of illiterate participants (53.4%), the protocol prioritizes non-literacy-dependent tasks, such as auditory attention and visual memory, to minimize educational bias. Instructions were provided in the standard Arabic dialect to ensure comprehension across different governorates. It includes another part dedicated to an informant, who can be a family member, a friend, colleague, nurse, or anyone who has known the participant well over the past 5 years. Cognitive outcomes were operationalized as follows: Cognitive Impairment is defined by education-stratified HCAP norms (score >35 in the local validation study), while Dementia is defined by the convergence of a low cognitive score AND informant-reported functional decline. The cognitive testing framework was designed to assess baseline cognitive function and monitor changes over time, while ensuring cultural and linguistic appropriateness.
A distinctive feature of the DAC Egypt Cohort is the incorporation of digital health tools to complement traditional assessments. Participants (and their caregivers, when applicable) were introduced to two mobile applications designed to monitor cognitive and sensory health remotely. The Mili App (developed by ki elements GmbH) is a smartphone-based platform that administers speech-based cognitive tasks. Through short verbal exercises—for example, verbalizing words starting with a certain letter or repeating sentences—the app collected speech samples to be analyzed for linguistic and acoustic markers of cognitive change. Participants could use the Mili app periodically at home, allowing for more frequent, low-burden cognitive monitoring between formal study visits. The audio recordings are then analyzed using artificial intelligence algorithms to detect features sensitive to cognitive changes, such as speech fluency, vocabulary richness, syntactic complexity, and the presence of hesitations or errors. These subtle changes in spontaneous speech may serve as early biomarkers of cognitive decline, potentially preceding detectable changes on standard tests.
SensifyAware is an olfactory digital health app that focuses on sensory and cognitive monitoring through smell identification tasks. It is a self-administered digital tool provided to cohort members who have access to a smartphone or tablet. Participants used a provided scent kit to complete these tasks. The app is based on the premise that sensory deficits, particularly in olfaction, can be early indicators of neurodegenerative changes. By combining olfactory testing with cognitive exercises, SensifyAware provides a more comprehensive assessment of brain health. The completion rate for SensifyAware was 58.8% (n = 900), as it required access to specific technology and a scent kit. Validation of this tool in the Egyptian population is an ongoing objective for future study waves.
Both apps were translated to Arabic and tested for usability with a small group of older adults before wider rollout. During the baseline, participants with the requisite technology and interest were enrolled in the digital tools sub-study; ongoing technical support was provided to maintain engagement. The integration of these digital assessments aims to enrich the cohort data with high-frequency observations and to evaluate the feasibility of remote cognitive monitoring in an older Middle Eastern population.
To further enhance the cohort’s capacity for biomarker and genetic research, blood samples were collected from 98% participants to support investigations into potential molecular indicators of cognitive decline. In addition, DBS cards were obtained from 88% of participants, offering a convenient and stable means of storing blood samples for long-term analysis. The collection of these biological specimens will enable future research on inflammatory markers, metabolic pathways, and genetic predispositions linked to dementia and neurodegenerative diseases. These samples provide a valuable resource for exploring the interplay between cognitive health, systemic health conditions, and genetic risk factors in an Egyptian population.
All participants underwent a comprehensive cognitive evaluation using the HCAP, which was adapted to include both traditional neurocognitive tests and innovative digital assessment tools. While the Egyptian HCAP has already been validated16. The digital components remain in the feasibility phase for this specific population. In alignment with the innovation goals of the Davos Alzheimer’s Collaborative (DAC), both the Mili and SensifyAware apps were rigorously adapted for the Egyptian context. This involved professional Arabic translations and the selection of culturally relevant content to ensure ease of use for local participants. This collaborative effort between DAC and technology providers underscores a shared commitment to validating digital health solutions in diverse, middle-income settings.
Data were collected by trained field teams through face-to-face interviews. To minimize missing data, a dedicated digital application for the HCAP was developed for use on tablets. Interviewers received specialized training to ensure standardized administration. Supervisors randomly observed interviews and re-contacted a subset of respondents with short validation questionnaires to verify response integrity.
Data availability
We will provide data upon reasonable request.
References
United Nations, Department of Economic and Social Affairs, Population Division. (2022). World Population Prospects 2022: Data Sources (UN DESA/POP/2022/DC/NO. 9). https://population.un.org/wpp/publications/Files/WPP2022_Data_Sources.pdf.
Central Agency for Publica Mobilization and Statistics 2024 (CAPMASS, 2024).
World Health Organization. Global Health Observatory data repository. Retrieved September 30, 2024, from https://www.who.int/data/gho.
Moustafa, S. A., Boersch-Supan, A. & Salama, M. Aging in an Arab country: knowledge gaps in Egypt. Nat. Aging 3, 1042–1044 (2023).
World Health Organization. (n.d.). Egypt: Noncommunicable diseases. Retrieved March 9, 2025, from https://www.emro.who.int/egy/programmes/noncommunicable-diseases.html#:~:text=Noncommunicable%20diseases%20(NCDs)%2C%20including,and%2067%25%20of%20premature%20deaths.
Jayaraj, R. L., Azimullah, S. & Beiram, R. Diabetes as a risk factor for Alzheimer’s disease in the Middle East and its shared pathological mediators. Saudi J. Biol. Sci. 27, 736–750 (2020).
Kalan Farmanfarma, K. H., Ansari-Moghaddam, A., Zareban, I. & Adineh, H. A. Prevalence of type 2 diabetes in Middle-East: systematic review & meta-analysis. Prim. Care Diab. 14, 297–304 (2020).
Isaifan, R. J. Air pollution burden of disease over highly populated states in the Middle East. Front. Public Health 10, 1002707 (2023).
Ahmed, H. et al. Depression in geriatrics: a systematic review and meta-analysis of prevalence and risk factors in Egypt. BMC Geriatr. 24, 930 (2024).
Khadr, Z. Growing old in an Egyptian family: beyond the bare statistics. In Aging Across Cultures, Science Across Cultures: The History of Non-Western Science (ed. Selin, H.) Vol. 10, pp. 35–50 (Springer, 2021).
Elshahidi, M. H., Elhadidi, M. A., Sharaqi, A. A., Mostafa, A. & Elzhery, M. A. Prevalence of dementia in Egypt: a systematic review. Neuropsychiatr. Dis. Treat. 13, 715–720 (2017).
El Tallawy, H. N. et al. Prevalence of dementia in Al-Quseir city, Red Sea Governorate, Egypt. Clin. Interv. Aging 9, 9–14 (2014).
El Tallawy, H. N. et al. Prevalence of dementia in Al Kharga District, New Valley Governorate, Egypt. Neuroepidemiology 38, 130–137 (2012).
Sabry, N., Tadros, G. & Odejimi, O. A systematic review of the prevalence of mental disorders, cognitive impairment and dementia amongst older adults populace in Egypt. BJPsych Open 7, S286 (2021).
Yount, K. M. & Agree, E. M. The power of older women and men in Egyptian and Tunisian families. J. Marriage Fam. 66, 126–146 (2004).
Moustafa, S. A. et al. Validation of harmonized cognitive assessment protocol within the Egyptian context. Soc. Psychiatry Psychiatric Epidemiol. https://doi.org/10.1007/s00127-024-02783-2.
Acknowledgements
This work was supported by the Davos Alzheimer’s Collaborative (DAC) as part of its global cohort program for advancing research in dementia.
Author information
Authors and Affiliations
Contributions
S.A.M.: Data curation, formal analysis, writing—original draft, writing—review and editing, participant recruitment, data collection. S.M.: Participant recruitment and data collection. G.F.: Participant recruitment and data collection. M.O., S.H., A.S.M., H.H., O.A.H., F.M., A.S.Y., E.M.A., R.M., M.M., M.Mo., J.S., M.W., S.E., N.S.K., K.K., L.T., F.M., Z.G., F.Z., E.Q., M.E., S.S., M.El., I.S., S.E.M., S.S., E.G., M.S.: Participant recruitment and data collection. M.R.: Writing—Review and editing. I.M.: Writing—review and editing. V.N.: Writing—review and editing. M.S.: Conceptualization, funding acquisition, project administration, supervision, writing—review and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Moustafa, S.A., Mowafi, S., Fawi, G. et al. Cohort profile Davos Alzheimer’s Collaborative DAC Egypt Cohort. npj Aging 12, 58 (2026). https://doi.org/10.1038/s41514-026-00378-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41514-026-00378-6