Abstract
This study integrates first-principles calculations, computational chemistry, system simulations, experiments, and machine learning to identify redox perovskite oxides for solar thermochemical hydrogen production. Using two random forest regressions and one classification model, the approach predicts materials’ stability and the enthalpy of oxygen vacancy formation (\(\Delta {h}_{o}\)), a critical property for selecting materials for thermochemical hydrogen production. B-site composition significantly influences \(\Delta {h}_{o}\) predictions. The methodology led to the discovery of Ba0.875Ca0.125Zr0.875Mn0.125O3 (BCZM), which reduces at temperatures up to 250 °C lower than CeO2 and is expected to outperform other perovskites in water splitting. However, CeO2 remains the benchmark for solar thermochemical hydrogen production. The combined use of machine learning and DFT calculations refined \(\triangle {h}_{o}\) predictions and provided insights into experimental results. This framework not only enhances database creation for material screening but also establishes a novel approach for perovskite discovery for hydrogen production applications.
Similar content being viewed by others
Introduction
Green hydrogen, which involves water splitting using renewable energy sources, is gaining attention for its potential to replace liquid fossil fuels in the transport sector. Water splitting is an energy-intensive process that, in practice, requires more energy to drive the chemical reactions than the theoretical minimum: 237 kJ mol−1 or 13.16 MJ per liter of water1. To enable large-scale hydrogen deployment, it is essential to produce it at an affordable cost. Nowadays, commercial green hydrogen is linked to renewable electricity, and it is still unclear how much of it can be effectively generated by electrolyzers. Solar thermochemical hydrogen production (STCH) offers an alternative route that can convert solar energy into heat and lately into hydrogen without the need for electricity production. Even though it is a technology with great potential to achieve high solar-to-hydrogen efficiency, there are still uncertainties regarding its commercialization. Among the main barriers to overcome are the development of suitable metal oxides with long durability and higher hydrogen productivity2,3,4,5, more efficient solar reactors6,7,8 and efficient plant configurations allowing heat recovery9,10.
Finding novel redox materials has gathered a great deal of attention since early 1970´s with non-volatile metal oxides being the most studied since 1990s, including iron oxides and ferrites11,12, manganese oxides13,14,15,16, ceria and doped-ceria materials17,18,19,20, hercynite21 and perovskites2,22,23. In 2010, ceria was first tested in a solar reactor showing stability in multiple cycles24 and this work has led to the first demonstration of the production of solar kerosene in 202225. In 2013, perovskites were first proposed as redox material26 and more than 50 different compositions have been tested for hydrogen and carbon monoxide production (from CO2) splitting in the last decade27,28. However, only a few perovskites have been deeply studied, including the description of their thermodynamic properties required to establish the performance of the materials for water or carbon dioxide splitting. In addition, many computational methods have been employed to explore perovskites and other metal oxides for STCH from different angles: high-throughput DFT analysis29,30,31, DFT perturbation theory32,33,34, molecular dynamics2, and lately machine learning (ML). These theoretical approaches were used to predict certain properties of materials, including: heat capacity, band gap, and vibrational entropy, among other physicochemical characteristics35.
Appropriate thermodynamic properties are the first cut-off criteria to determine the ability of a material to thermodynamically split water and carbon dioxide via STCH process6,36. However, obtaining these thermodynamic properties demands weeks of experimental work to evaluate a single material26,37,38. Experimental methods alone for evaluating materials could possibly test up to 100 possible oxides in dedicated labs. If 1000 or even 10,000 materials are aimed, obtaining thermodynamic properties via computational methods would be appropriate. This work also requires months or years and, therefore, if the purpose is to cover 100,000 or even 1,000,000 possible structures, this will entail the allocation of a huge number of research hours and researchers performing daily calculations for decades39,40. Currently, databases do not have all possible perovskite compositions, not even reaching 300,000 perovskite oxides in total2, covering only a relatively small compositional space, especially for doped perovskites.
In this work, we developed a full methodology to predict the ability of materials to produce hydrogen via STCH combining machine learning, first-principles thermodynamics, computational chemistry simulations, experiments and system modeling. Recent works have focused on predicting the enthalpy of oxygen vacancy formation (\(\Delta {h}_{o}\)) using defect graph neural networks and random forest models41,42,43. Other studies also focused on finding linear models for predicting oxygen vacancy formation energies. Deml et al. combined the oxide enthalpy of formation, the midgap energy relative to the O 2p states, and atomic electronegativities within a range of ∼0.2 eV, offering insights into key influencing properties44. On the other hand, Wexler et al. developed a compact linear model for predicting neutral vacancy formation energy in perovskites, achieving a mean absolute error of 0.45 eV across diverse perovskites. Incorporating intuitive metrics like bond dissociation energies and reduction potentials, the model enables efficient, high-throughput screening and identifies new candidate materials with promising thermochemical water-splitting properties, such as (Bi,Y)(Fe,Co)O345.
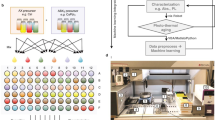
The novelty of the current work extends into experimentally demonstrating the machine learning predictions on unknown and known materials from the literature, performing computational calculations of the predicted enthalpy, and connecting them into a simulation platform to obtain the system performance of the materials. The complete methodology is shown in Fig. 1. First, we used a different set of features as compared to previously published works41,42. Our database helps to explore materials never registered in computational repositories, enabling the discovery of novel compositions. To achieve this, we required a database that relies solely on elemental composition without including any DFT or structurally related data as the materials list for discovery are gathered only by element combination. Even with limitations, our approach ensures a wide discovery of materials. The machine learning methods allowed for an explanation of the physical significance of the predictors. The variable importance analysis combined with Pearson correlation enables to retraining of the models with a reduced number of features from more than 250 to 33 or less. The retrained models were then applied to a dataset of combined elements, and among the predicted compositions, four new perovskites were experimentally tested. Predictions were also performed for selected materials from the literature. Results were compared with predictions finding mean average errors below 16%, for 80% of the experimental samples.
An iterative frame-work combining machine learning, experimental and DFT validation, along with process simulations and first-principles thermodynamics enables the selection and assessment of candidate perovskites under relevant conditions.
The ML modeling method also introduces novelty to the current work compared to previous approaches. Three ML models are combined for the prediction of the target property (\(\Delta {h}_{o}\)) and stability. Two numerical predictions were incorporated for different values of δ, specifically 0.5 and 0.25, using datasets derived from perovskite compounds reported in Vieten et al.46 and Baldassarri et al.41, respectively. These two models (based on random forest, RF, regression) allowed for the generation of predictions at multiple δ values, to enable a better representation of a range of \(\triangle {h}_{o}\) with δ. The methodology also applied a feature reduction method using variable importance and Pearson correlation. The ML procedure was then used in a large set of 6264 perovskites generated through elemental combinations ensuring charge neutrality. A third machine learning model was developed for stability prediction, ensuring that all experimental perovskites were stable prior to experimental demonstration. The stability dataset was obtained from Talapatra et al.47 (using other sets of features) and encoded into a RF classification approach. Predictions from ML models were rationalized and explained using Density Functional Theory (DFT) calculations.
To finish, a full description of the thermodynamic performance of the most promising material was used in a thermodynamic model mimicking their performance in a solar thermochemical hydrogen plant9,48,49,50,51, allowing to obtain the working conditions that can maximize the solar conversion into hydrogen. The full methodology showed in this work, allowed us to discover, a perovskite that shows operational temperatures more adequate for thermochemical water splitting as compared to other perovskites in the literature.
Results
Exploratory analysis of the databases
An initial overview of the Vieten and Baldassarri DFT databases reveals distinct patterns in composition and \(\Delta {h}_{o}\). In the Vieten database, the element distribution in the A, A’, B, and B’ sites of the AA’BB’O3 perovskite structure is uneven. The A site is dominated by La, while the most common elements in the A’ and B’ (dopant) sites are non-doped (“N.E.”), followed by Sm and Mn, respectively (Supplementary Fig. 1). The B site is primarily occupied by Mn and Ti. The \(\Delta {h}_{o}\) in this database ranges from 188–517 kJ/mol, with a notable concentration around 350 kJ mol−1, a value favorable for water splitting applications. Approximately 57% of the data is above this average, with peaks at 260 kJ mol−1 and 500 kJ mol−1. The Baldassarri database displays a broader elemental composition compared to Vieten but still exhibits uneven distribution (Supplementary Fig. 2). In the A site, Sr, Ca, and Ba are most prevalent, while the A’ site is again dominated by non-doped (“N.E.”) entries. Ti is the primary element in the B site, while Fe and Pb are prominent dopants in the B’ site, followed by Ru, Rh, Mn, and Ir. Predictions for perovskites containing commonly represented elements in both databases (e.g., La, Sm, Mn, Ti in Vieten; Sr, Ca, Ba, Ti, Fe in Baldassarri) are expected to be more accurate due to their higher representation in the training datasets. The \(\triangle {h}_{o}\) is delimited within 189 and 482 kJ mol−1. The average is 327 kJ mol−1 and 52% of the data is above this value.
Random forest regression models train and test results
Initially, RF models are trained to identify the best predictors from a set of 258 features generated through feature engineering. The acronyms used for the list of features are included in the Supplementary Table 12. The predicted values versus the original data, using all 258 features, are shown in Fig. 2, along with R2, RMSE and MAE for both databases. Using the Vieten database, the model is able to explain with 84% certainty the variability of the target property, \(\triangle {h}_{o}\). In the context of \(\triangle {h}_{o}\) prediction, an R2 of 84% is considered satisfactory until a larger dataset can be generated. Additionally, RMSE and MAE values are reasonable and similar to previous publications on the prediction of the same property (see works from Witman et al. 42 and Baldassarri et al. 41). Using the Baldassarri database, the RF model achieves an R2 of 83%, similar to the model with Vieten database, with slightly lower MAE and RMSE, also in line with previous publications.
Regression results using Vieten (a, b) and Baldassarri (c, d) databases for the training (a, c) and the test sets (b, d).
Variable importance analysis and Pearson correlation were performed for 258 predictors on RF model using the Vieten database (see Supplementary Figs. 3, 4) narrowing them to 39 key features. Variable importance is assessed using %IncMSE (impact on prediction error when a variable is permuted) and IncNodePurity (enhancement of tree node homogeneity), with %IncMSE serving as the primary cutoff criterion. The melting point of AB average (MP_AB_Avg) is identified as the most impactful feature, followed by the B site heat of fusion (B_site_heat_fus) and boiling point of AB average (BP_AB_avg). These thermal properties are highly correlated due to their shared dependence on intermolecular forces. The dimensionality reduction process selects features with at least 25% of the maximum importance, streamlining the model for Pearson correlation analysis to eliminate redundant features. Using a correlation cut-off of 0.7, variables with correlations of 0.7 or higher (positive or negative) are discarded, reducing the predictors to 10 final variables. Among these, 5 predictors show positive correlation with \(\triangle {h}_{o}\), while the other 5 show negative correlation (see Supplementary Fig. 4 in Supplementary Information). On the other hand, variable importance and Pearson correlation differs significantly for the RF model using Baldassarri database (Supplementary Fig. 5).
The RF model using the Vieten database was retrained to consider only the 10 selected predictors while the model using Baldassarri database was retrained using 33 predictors. Note how Baldassarri database have a larger number of samples so the feature reduction gives larger number of features too. The prediction versus original data of both retrained models are shown in Fig. 3. It is possible to observe by comparing Figs. 2, 3 (using 10/33 and 258 predictors, respectively) that values of R2, RMSE and MAE are very similar with less than 10% decrement in performance, while a faster training and smaller model is used in the case of the retrained RFs. This corroborates the feature reduction procedure was appropriate for databases. As compared to previous works on \(\triangle {h}_{o}\) predictions, our MAEs (31.8 kJ mol−1 and 24.7 kJ mol−1 for RF models using Vieten and Baldassarri database respectively) are similar to the best performing MAE of Witman et al. (0.34 eV - 32.80 kJ mol−1)42 and Baldassarri et al. (0.3 eV – 28.87 kJ mol−1)41. Indeed, our MAE of the retrained model, 24.7 kJ mol−1, is smaller to the work of Baldassarri et al.41, 28.87 kJ mol−1. While these results are promising, note that different regression techniques, training datasets and testing conditions were used across the various experiments presented, including those conducted in this study and prior works. Consequently, direct comparisons of final performance metrics between studies may lack generalizability and should be interpreted cautiously.
Regression results for Δho prediction using Vieten (a, b) and Baldassarri (c, d) databases for the training (a, c) and the test sets (b, d).
Although it is recognized that in most ML works, increasing the number of samples improves model accuracy and generalization, this should be approached with caution. Increasing the amount of data could lead to a more generalized model but may make it less specific for targeting certain predictions52. In our case, the model uses fewer samples (506, limited to double perovskites) than the original Baldassarri et al. model (>1000 samples across diverse oxide chemistries) and is trained exclusively on compositional features. As such, it inherently lacks the ability to distinguish between non-equivalent oxygen sites. Furthermore, the dataset used from Baldassarri et al. includes only the formation energy of the lowest-energy vacancy site per material. This limits the model’s ability to capture site-specific behavior, but aligns with the scope of composition-based screening.
To verify consistency, a standard cross-validation procedure was also performed on both retrained RF models, as is common in ML workflows53. Although RF models are based on bootstrapping and this validation should show redundant information, it is included for verification. They obtained a R2 values close those of the retrained RFs (Supplementary Table 1) and slightly larger RMSE and MAE values.
A second variable importance analysis of the retrained models exhibits minor changes compared to the first. For the RF model using the Vieten database, 70% of the top 10 final features are related to the A and B sites (weighted averages), while the remaining features are specific to B site elements. This highlights the strong influence of B site characteristics—either alone or in combination with A site features—on \(\triangle {h}_{o}\) values (Supplementary Fig. 6). The most important features by %IncMSE are MP_AB_avg, B_site_SIP, and IE_AB_avg, with the second to fifth variables showing similar importance, while the importance decreases significantly from the fifth to tenth features. From the second to the fifth variables, the %IncMSE are very close to each other meaning that they have similar importance. Then from fifth to tenth variables the importance is relatively lower as compared to the previous ones. The model based on Baldassarri database shows more than 90% of features related to B site elements with a smaller number of A and B site influence (Supplementary Fig. 6). Previous studies support the influence of B-site cation in oxygen vacancy formation linking them to the electronic structure of the B elements29,54.
Analyzing how MP_AB_avg interacts with B_site_SIP and IE_AB_avg to influence \(\triangle {h}_{o}\) in the RF model retrained with the Vieten database reveals two distinct trends. Higher B_site_SIP values tend to lower \(\triangle {h}_{o}\), by facilitating oxygen vacancy formation (Supplementary Fig. 7a). In contrast, a simultaneous increase in MP_AB_avg and IE_AB_avg is associated with higher \(\triangle {h}_{o}\), relating lower MP/IE values to easier oxygen vacancy formation and higher values to greater resistance.
A more detailed analysis of the ML models used in this study is provided in the Supplementary Information. This includes: the selection of hyperparameters for RF predictions (Supplementary Fig. 8), the effect of adding structural features (Supplementary Figs. 9, 10, 11, 12 and Supplementary Table 2), and the confusion matrix along with additional analyses of perovskite stability (Supplementary Fig. 13). Furthermore, the comparison of the use of a larger dataset or a delimited dataset is presented (Supplementary Fig. 14), and the results of using of a linear model instead of random forest for predictions are shown (Supplementary Figs. 15, 16, 17, Supplementary Tables 3, 4, 5, and Supplementary Notes1, 2).
Experimental validation with selected materials
After the machine learning models are trained, tested and retrained the next step is to validate the prediction \({\triangle h}_{o}\) of new materials using experimental methods. For this demonstration, four experimental materials were selected: BCZM, LSFM, LBFC and LNC, as these represent diversity in regards to their composition and predicted \({\triangle h}_{o}\) (see chemical formulas in Table 1). The XRD results (Fig. 4a–d) show that the target perovskite structures were achieved as the main phase present in all samples, with not significative contributions of impurity phases. As the materials are essentially new, the XRD patterns do not match perfectly any reference from the PDF database. However, they have similarities with close structures. BCZM have a partial match with BaZrO3 cubic structure (ref. JCPDS 01-074-1299) while LSFM matches La0.8Sr0.2FeO3 with orthorhombic symmetry (ref. JCPDS file No 00-035-1480). On the other hand, LCN matches with LaCoO3 with cubic symmetry (ref. JCPDS 01-075-0279) and LBFC with also cubic LaFeO3 (ref JCPDS 01-075-0541). The samples show mostly a single phase without segregation of metal oxide species (Fig. 4a–d). The Goldschmidt tolerance factor analysis is also used to predict the stability of the crystal structure. BCZM and LCN should stabilize in cubic structure as their τ values are close to unity (τ = 1.005 and 1.003 respectively) although they could be slightly distorted to tetragonal. Indeed, the peak splitting shown in LCN suggests a distortion from the ideal cubic crystal. LSFM should display an orthorhombic symmetry (τ = 0.98) while LBFC should show rhombohedral distortion (τ = 0.97). As these values are also close to unity, it is possible that these perovskites could stabilize in slightly distorted cubic structures. Rietveld refinement for all materials, with the mentioned symmetries, was performed (see Fig. 4a–d to observe the relative difference). For all materials the Rwp was around 5, indicating a good quality fit and an accurate model refinement55. SEM/EDS was conducted before and after thermochemical testing (Fig. 4e–l). All materials showed evidence of sintering, as revealed by the increased particle size after cycling, except BCZM, which showed relatively lower signs of particle size change comparatively.
XRD and SEM for (a) BCZM, (b) LSFM, (c) LBFC and (d) LNC. The dashed blue lines represent the XRD diffraction data, while orange is the best fit and the gray lines are the residual difference between observed and Rietveld calculated. SEM images are shown for BCZM (e, f), LSFM (g, h), LBFC (i, j) and LNC (k, l).
Figure 5 shows the equilibrium data (points) experimentally obtained from TGA experiments and the predicted equilibrium values from the Bulfin model (lines)56. The model exhibits reasonable fitting for all the samples. Van’t Hoff analysis was conducted to evaluate \({\triangle h}_{o}\) and \({\triangle s}_{o}\) using the model predictions, those results are presented in Table 1 and Supplementary Fig. 18, respectively. Table 1 also includes other experimentally measured values for literature materials that were predicted by the machine learning model. Considering all experimental vs predicted values for \({\triangle h}_{o}\)(in the δ range) the model predicted 9 out of 11 experimental samples accurately (with errors using either one of the predictions below 22%). When removing the 2 conflictive samples the statistical analysis shows a MAE of 28 kJ mol−1 which is acceptable. However, when all of the samples are considered, the MAE increases to 50 kJ mol−1, being 56% higher than the test of the reduced model (32 kJ mol−1 of MAE).
Experimental data (dots) and Bulfin model adjustment (lines) of δ, oxygen partial pressure and temperature for (a) LNC, (b) LSFM, (c) LBFC, and (d) BCZM. The calculated values of \({\triangle h}_{o}\) for the corresponding van´t Hoff analysis are shown on Table 1. Bulfin model was applied from ref. 56.
We must consider that predictions of \(\triangle {h}_{o}\) are accounted at 0 K as the DFT databases contain calculations performed without temperature corrections. As pointed by Lany in57, there is a difference of around 0.3 eV between experimental and DFT calculated values at the expected reduction conditions of 1500 oC and \({p}_{{O}_{2}}\) = 10−5. This difference is within the error of the ML predictions. Additionally, the use of the van´t Hoff method helps integrating this dependency by approximating the experimental \(\triangle {h}_{o}\) to be temperature independent. This helps comparing predictions and experimental values fairly although it is recognized that temperature corrections in both DFT and experimental fitting methods would enhance predictions.
Using two predictions at different oxygen vacancy concentration (δ = 0.25 and 0.5) helps selecting novel materials for STCH exploration as, a priori, it is not possible to know the range of oxygen vacancies a material will have without experimental testing. By using two predictions, we can observe if a material will potentially have a large or small difference in \(\triangle {h}_{o}\) and its potential for performing STCH. As expected, when predicting \(\triangle {h}_{o}\) using the two RF models with the databases of Vieten and Baldassarri, different values are obtained. Most materials show a rather constant enthalpy of oxygen vacancy formation in the range 0.25 to 0.5. This is also observed experimentally for most perovskites extracted from literature (see examples of CTM, LSM20, LSM50 and LCMA experimental values gathered in their respective references in Table 1). The difference with predictions for these materials range between 15−68 kJ mol−1. In most cases, the experimental values are close to one of the two predictions, with most of them being closer to the RF prediction using Vieten database than to that using the Baldassarri database (Supplementary Table 1). However, differences between predictions are not large for most perovskites. In addition, as discussed, inherent differences exist between experimentally-obtained and computed DFT \(\triangle {h}_{o}.\) Besides, using a range of predicted \(\triangle {h}_{o}\) values help to outline the expected behavior of the materials without previous knowledge of their maximum possible δ.
Notice that CCTM material has been studied only by DFT and a single value was reported58. The main difference found in CCTM materials and predictors can be associated to the amount of oxygen vacancies (which are highly diluted in CCTM reference) as well as the different DFT procedure use in that work. Additionally, CCTM accounts for a complex reduction mechanism involving the simultaneous reduction of Ce⁴⁺ and Mn⁴⁺, which is not accounted for in the database that assumes single-element redox behavior. However, the largest discrepancy in predictions is observed for BCZM, with a difference of 130 kJ mol−1 (Table 1). To clarify the origin of this variation, a further DFT analysis was performed for BCZM.
DFT analysis of BCZM
Following the observed differences in the predicted \(\triangle {h}_{o}\), further DFT analysis was performed for BCZM, a newly discovered perovskite with potential for water splitting due to its relatively high \(\triangle {h}_{o}\). It is important to note that all calculations assumed a ferromagnetic alignment for unpaired spins, unless stated otherwise, based on test calculations indicating weak magnetic coupling. The analysis involved constructing structures with different crystal symmetries and varying supercell sizes (see Supplementary Table 7 and Supplementary Fig. 19 and 20 for more details of the structures construction and optimization). As mentioned previously, the calculated Goldschmidt tolerance factor for fully oxidized BCZM suggests a slightly distorted cubic crystal. However, DFT simulations at 0 K often favor orthorhombic over cubic symmetry in ABO₃-type perovskites59.
The Vieten database comprises single and double perovskite structures with various dopants in 1/8 increments at both A and B sites, all featuring cubic unit cells in space group 221 (Pm3̅m). For the reduced structure, brownmillerite-type orthorhombic cells in space group 46 (Ima2) were used, unless a more stable phase was available in the Materials Project. The Baldassarri database contains only double perovskite structures with the general formula AxA'1-xB0.5B'0.5O3, where x is 0.5 or 1.0, with ten structural distortions considered across various crystal systems and space groups: monoclinic P2₁/c (14) and C2/m (12); triclinic P1 (1) and P1̅ (2); tetragonal I4/m (87) and I4/mmm (139); rhombohedral R3 (146), R3̅ (148), and R3̅m (166); and cubic Fm3̅m (225). Among these, monoclinic P2₁/c (space group 14) is the most frequently observed41,60. No phase transitions were considered for the reduced A2A’2B2B’2O11 and A4B2B’2O11 double perovskite structures.
Several factors in DFT calculations can influence \(\triangle {h}_{o}\), including the choice of crystalline structures for the oxidized and reduced phases (orthorhombic vs. cubic), oxygen vacancy concentrations, phase transition upon reduction, the selected Hubbard U parameter, and the value used for \(\frac{1}{2}{E}_{{O}_{2}}\). Tables 2 and 3 summarize these findings, revealing significant variations in \(\triangle {h}_{o}.\)
One of the primary sources of variation in \(\triangle {h}_{o,{\rm{pred}}}\) is the oxygen vacancy concentration. As previously noted, the numerical predictions correspond to two distinct δ values, 0.5 and 0.25, derived from perovskite datasets reported by Vieten et al.46 and Baldassarri et al. 41, respectively. For example, removing a single oxygen vacancy from Ba7CaZr7MnO24 (δ = 0.125) versus sixteen vacancies from Ba28Ca4Zr28Mn4O96 (equivalent to 4 × Ba7Ca7Zr7Mn4O24, δ = 0.5) results in a DFT-calculated enthalpy difference of up to ~190 kJ mol−1 (see \(\Delta {h}_{o,{\rm{DFT}},{\rm{o}}}\) for δ = 0.125 and 0.5 in Table 4 for U = 2 eV). This trend aligns with ML model predictions, which show a \(\triangle {h}_{o,{\rm{pred}}}\) difference of ~130 kJ mol−1 between the Vieten (δ = 0.5) and Baldassarri (δ = 0.25) datasets (see \(\triangle {h}_{o,{\rm{pred}}}\) in Table 2). These findings underscore oxygen vacancy concentration as the dominant factor influencing \(\triangle {h}_{o}\), in agreement with experimental studies—for instance, CeO₂ demonstrates variations exceeding 100 kJ mol−1 with changes in vacancy concentration23.
Structural changes also contribute to the observed differences. Preserving the same symmetry (orthorhombic or cubic) for both oxidized and reduced phases versus including a structural transition to an orthorhombic brownmillerite phase (space group 46, Ima2), phase with a larger supercell leads to a 31.5 kJ mol−1, independent of the choice of \(\frac{1}{2}{E}_{{O}_{2}}\,\)(see \(\Delta {h}_{o,{\rm{DFT}},{\rm{o}}-{\rm{t}}}\) vs. \(\Delta {h}_{o,{\rm{DFT}},{\rm{o}}}\) for δ = 0.5 in Table 2, U = 2 eV). Another important factor is the value used for \(\frac{1}{2}{E}_{{O}_{2}}\). Using −4.523 eV (\(\frac{1}{2}{E}_{{O}_{2}}\)corr) as suggested by Baldassarri et al. vs. −4.929 eV (\(\frac{1}{2}{E}_{{O}_{2}}\)PBE) from Vieten et al. results in an overall 39 kJ mol−1 difference (Supplementary Table 3, where all other parameters remain constant). Finally, the choice of the Hubbard U value also impacts \(\Delta {h}_{o,{\rm{DFT}}}\,\) calculations. Using U = 2 eV (Baldassarri approach) versus 3.9 eV (Vieten approach) for Mn4+ results in a difference of approximately 19.4 kJ mol−1 (see \(\Delta {h}_{o,{\rm{DFT}},{\rm{o}}-{\rm{t}}}\) for δ = 0.5 in Table 2).
For BCZM, the discrepancy between enthalpy predictions from different RF models trained on different databases primarily arises from differences in vacancy concentration (δ), rather than structural considerations, the choice of U, or the reference energy of \(\frac{1}{2}{E}_{{O}_{2}}\), although all these factors contribute to the final \(\Delta {h}_{o}\) value. A final set of DFT calculations—using the original computational protocols specific to each database—successfully reproduced the ML predictions. For example, the value of 478.554 kJ mol⁻¹ obtained using the Vieten DFT protocol (Table 3) closely matches the ML-predicted value of 457 kJ mol−1 (Table 2). Similarly, for BCZM, using the Baldassarri calculation setup (applied to both cubic and orthorhombic phases, with no phase transition observed upon reduction), the DFT-derived values of 314.592 and 315.168 kJ mol−1 (Table 2) are in good agreement with the ML prediction of 327 kJ mol−1 and even closer to the experimental measurement of 305 kJ mol−1.
In the reduced BCZM calculations, simulation cells with compositions Ba₇Ca₁Zr₇Mn₁O₂₃ and Ba₂₈Ca₄Zr₂₈Mn₄O₈₀ (bronwmillerite phase with 144 atoms)—corresponding to oxygen vacancy concentrations of δ = 0.125 and 0.5, respectively—contained either one or four Mn cations. The DFT results showed that Mn⁴⁺ was present in the corresponding fully oxidized phases, while Mn²⁺ emerged in the reduced phases in both cases. The projected Density of States (DOS) analysis presented in Supplementary Fig. 21 provides clear evidence of the reduction of Mn4+ to Mn2+ in both cubic and orthorhombic BCZM perovskite structures, as the excess charge resulting from the formation of a neutral oxygen vacancy Mn4+ localizes on the Mn site. This reduction is reflected in the total magnetic moment, which increases from +3 μB to +5 μB, due to the higher number of unpaired electrons in the Mn d orbitals. In the pristine BCZM, the projected DOS for Mn (green line) shows a lower contribution near the Fermi level, consistent with the electronic configuration of Mn4+, where fewer electrons occupy the valence band. Upon reduction to Mn2+, a significant increase in the density of Mn states is observed near the Fermi level, indicating additional electron localization associated with the Mn2+ oxidation state following the creation of an oxygen vacancy near the Mn site.
To investigate alternative redox pathways, we carried out a convergence test of oxygen vacancy (see Supplementary Table 11) formation energy with respect to supercell size, following oxygen removal—specifically the reduction of 2 × Mn⁴⁺ to either 2 × Mn³⁺ (in accordance with the experimental observations in other Mn-based perovskites61,62) or a mixed-valence Mn²⁺/Mn⁴⁺ configuration. We employed a larger 2 × 1 × 1 cubic supercell of Ba₇CaZr₇MnO₂₄, resulting in a Ba₁₄Ca₂Zr₁₄Mn₂O₄₈ composition. Upon removing the most labile oxygen atom (δ = 0.0625), we evaluated the magnetic and energetic stability of various Mn valence configurations. For both the Mn⁴⁺/Mn⁴⁺ and Mn³⁺/Mn³⁺ configurations, the energy difference between ferromagnetic (FM) and antiferromagnetic (AFM) states was negligible ( ~ 2 meV), indicating weak magnetic coupling. In contrast, the mixed Mn²⁺/Mn⁴⁺ AFM configuration, specifically designed to enforce a redox-asymmetric state, was approximately 0.5 eV higher in energy than the Mn³⁺/Mn³⁺ AFM configuration. This result indicates that the 2 × Mn⁴⁺ → 2 × Mn³⁺ redox pathway is energetically preferred over the Mn²⁺ + Mn⁴⁺ alternative at lower vacancy concentrations. The corresponding DFT-calculated enthalpy change, \(\triangle {h}_{o,{DFT},c}\), is 261.984 kJ mol−1 (δ = 0.0625). Notably, the DFT enthalpy values for BCZM bracket the experimental measurement of 305 kJ mol−1, with 315.168 kJ mol−1 at δ = 0.125 slightly above, and 261.984 kJ mol⁻¹ at δ = 0.0625 somewhat below. Supplementary Fig. 22 presents the DOS of the reduced 80-atom cubic BZCM perovskite supercell across the different magnetic configurations considered.
Thermodynamic performance of BCZM against state-of-art materials
BCZM is further studied in a comparative thermodynamic analysis to CeO2 and LCMA6464 as reference state-of-art materials. The comparison against CeO2 is shown in Fig. 6 for conditions of maximum water-to-hydrogen conversion (θmax) and degree of reduction and oxidation (δred and δox). There are common parameters selected to study both materials under similar performance which is the rate of carrier N2 used during reduction per mol of H2 produced (nN2/nH2 = 3), the minimum water-to-hydrogen conversion (θmin = 0.001). First, it can be observed that for any variable condition, CeO2 achieve higher operational temperatures than BCZM (higher reduction and oxidation equilibrium temperatures and a smaller temperature difference). This is associated to the higher enthalpy and entropy of CeO2 as compared to BCZM for the same δ (Supplementary Table 4 of comparative values). These high enthalpy and entropy also make CeO2 a unique material able to achieve, so far, record high oxidation temperatures with fast kinetics. When compare to a state-of-art perovskite such as LCMA6464, the performance of BCZM improves specially in the oxidation reaction. Due to its higher enthalpy and lower entropy compared to LCMA6464, the difference between reactions is reduced, as highlighted in the work of Bayon et al. 6. Further experiments should still be made to ensure long-term performance of BCZM to ensure it can be used in solar plants.
Results of thermodynamic performance where the operating temperatures are compared for BCZM (dashed lines), CeO2 (solid lines) and LCMA6464 (dashed-dot lines) with reduction temperatures shown in red and oxidation temperatures in blue. Conditions explored: (a) δred = 0.04, δox = 0.005, θmax = 0.1, (b) δred = 0.045, δox = 0.01, θmax = 0.05, (c) δred = 0.04, δox = 0.005, θmax = 0.01, and (d) δred = 0.04, δox = 0.005, θmax = 0.05 (including LCMA in dot-dashed lines83 for comparison). Common parameters: nN2/nH2 = 3, pO2, min = 10 Pa (0.00001 bar) and θmin = 10-3.
Specifically, from Fig. 6 BCZM can optimize its operational conditions when fixing a θmax of 5% or lower (Fig. 6b), achieving the lower temperature difference between both steps, as compared to LCMA, and, at the same time, a relatively low thermal reduction temperature as compared to CeO2 (maximum of 1523 °C for BCZM as compared to 1734 °C for CeO2). Lower conversion values are favorable for water-splitting temperature (Fig. 6c), however, it will be detrimental to the system efficiency9,51. If higher values of conversion are fixed (Fig. 6d), then the minimum oxidation temperature is too low (179 °C) implying very slow kinetics6,21. It is also noticeable that such small changes in \(\Delta {h}_{o}\) and \(\Delta {s}_{o}\) values make materials to have disparate performance in operational conditions (Fig. 6). This is associated with a large variation of δ at different oxygen partial pressures (especially for LCMA6464 and BCZM) that result in very different operational temperatures. Therefore, even if the \(\Delta {h}_{o}\) and \(\Delta {s}_{o}\) are the first cut-off criteria for selecting STCH materials, a full description of their thermodynamic performance (δ, T and \({{po}}_{2}\)) is required in order to compare materials by water splitting capability and operational conditions.
Discussion
Combining machine learning (ML) models with experimental demonstration, computational chemistry calculations (DFT), and system simulations is a promising and effective approach for discovering new redox materials for solar thermochemical hydrogen production. In this study, the target property selected was the enthalpy of oxygen vacancy formation (\(\left.\Delta {h}_{o}\right)\), as it serves as the primary screening criterion for identifying suitable materials for thermochemical water splitting. RF regression and classification were chosen as the ML models due to its ability to provide reliable predictions even when the sample size is limited. The training and test results showed accurate predictions with a small number of selected features (only 10 and 33 for both databases use in this work), yielding relatively small errors (R² = 81 and 84%, MAE = 24.7 and 31.78 kJ mol−1, and RMSE = 36.8 and 43.44 kJ mol−1). The use of variable importance and Pearson correlation to reduce the dimensionality of the models are essential to avoid noise and reduce dimensionality. The retrained models were used to predict the \(\Delta {h}_{o}\) of experimentally tested materials while stability was also checked using a classification method. Since the databases are composed of \(\Delta {h}_{o}\) values at different vacancy concentrations (δ) the use of two ML models helped searching materials for STCH. Based on the prediction of \(\Delta {h}_{o}\) values within the range of interest, a series of previously unexplored perovskites were selected for a more in detail investigation. In 9 out of 11 materials the predictions have reasonable errors as compared to the experimental data. A large variation of \(\Delta {h}_{o}\) values was found in BCZM which was further explored by DFT analysis. DFT analysis showed that primarily differences between RF predictions arises from differences in δ, rather than structural considerations, the choice of the U value, or the reference energy of \(\frac{1}{2}{E}_{{O}_{2}}\), although all the parameters have some influence in the final value of \(\Delta {h}_{o,{\rm{DFT}}}\). The specific example of the BCZM was further studied by system modeling as it is a promising perovskite discovered in this work. BCZM was not previously synthesized or computationally explored, showing improved performance against previously published perovskites. The material exhibits a 250 °C lower reduction temperature as compared to CeO2 and a nearly 500 °C higher oxidation temperature as compared to LCMA6464 material. This is associated with its relatively high enthalpy than other perovskites, although not as high as CeO2, requiring less reduction temperature. However, it is also noticeable that a full description of the material performance is required to finally asses its performance in STCH and not only the thermodynamic properties (enthalpy and entropy) can be used as water-splitting performance descriptors. The material BCZM is not proved to outperform CeO2 although it could offer a greater performance among perovskites. Future work on extending the database for improved accuracy of the machine learning model to explore other compositions, using DFT combined with experiments, is currently ongoing.
Methods
Thermodynamic principles of solar thermochemical hydrogen
The first-principles thermodynamic model is essential to initially evaluate whether a material will produce hydrogen or not. Current research in thermochemical cycles primarily focuses on two-step redox-active, off-stoichiometric metal oxide cycles, which are the subject of this study. These cycles involve two reactions (one for each step, shown in Eqs. (1) and (2), transitioning between a (partially) reduced state and a re-oxidized state of a metal oxide (MOx), where the metal cation exhibits multiple oxidation states. In these reactions, water is the only reactant consumed:
In Eqs. (1) and (2), \({\delta }_{{red}}\) and \({\delta }_{{ox}}\) represent the extents of reduction reaction (off-stoichiometry) and re-oxidation processes, respectively. The difference in off-stoichiometry between the reduced and re-oxidized states of MOx-δ is given by \(\varDelta \delta \,=\,{\delta }_{{red}}\,-{\delta }_{{ox}}\). Typically, the reduction process is highly endothermic and occurs under conditions of high temperatures and low partial pressure of oxygen (\({p}_{{O}_{2}}\)). In contrast, the reoxidation process is mildly exothermic and occurs under conditions of low temperatures and high-water vapor concentrations. The full model description can be consulted in previous works6,63. Essentially, the description of the thermodynamic equilibrium of a metal oxide performing water (or carbon dioxide) splitting can be expressed by the following two Eqs. (3) and (4):
where \({T}_{{red}}\) and \({T}_{{ox}}\) are the temperatures of reduction and oxidation reactions; \(\frac{{p}_{{O}_{2},{red}}}{{p}^{o}}\) is the oxygen partial pressure at the inlet of the reduction reactor divided by the reference pressure (\({p}^{o}\) = 1 bar); \({x}_{{H}_{2},{out}}\) represents the molar fraction of H2 at the outlet of the oxidation reactor; \({K}_{{WS}}\) are the equilibrium constant of the water splitting reaction (for carbon dioxide, the constant and subscripts should be modified); \(\Delta {s}_{o}\) and \(\Delta {h}_{o}\) are the entropy and enthalpy of oxygen vacancy formation. When the material is performing both, reduction and oxidation reactions in counter-current operation, then \(\Delta {s}_{o,{red}}\,\)= \(\Delta {s}_{o,{ox}}\), and \(\Delta {h}_{o,{red}}\) = \(\Delta {h}_{o,{ox}}\). Resolving both equations for the entropy given the enthalpy in a wide range of temperatures, it is possible to evaluate the thermodynamic properties required to split water to hydrogen at a certain \({x}_{{H}_{2},{out}}\) and \(\frac{{p}_{{O}_{2},{red}}}{{p}^{o}}\). [6]
Data architecture and feature engineering
In this work, the \(\triangle {h}_{o}\) is selected as the target variable. This selection is based on the fact that, from a thermodynamic perspective, enthalpy plays a crucial role in a material’s ability to split water (o CO2). While entropy is also an important property and is directly related to enthalpy through the heat capacity relationship29,33,34, the available data on materials’ entropy is more limited. Additionally, a near-linear correlation between entropy and enthalpy has been observed in redox metal oxides6,64, making it practical to focus on one of these properties.
Materials databases are essential to run machine learning models. The acquisition and processing of this data is known as data architecture. Two databases were created using the \(\triangle {h}_{o}\) published in Vieten et al.46 and Baldassarri et al.41 accounting for δ = 0.5 and 0.25 respectively. These predictions of \(\triangle {h}_{o}\) will help us covering different values of δ as well as accounting for a phase transition used in Vieten et al. DFT calculations46. Both databases are completed using the same features to be able to predict two values of \(\triangle {h}_{o}\) for the same list of perovskites for discovery. Although ideally, we should cover more values of \(\triangle {h}_{o}\,\) with δ variation, we are currently working on expanding the databases shown here to a wider range of data and future works are expected to cover it.
Github repositories were used for feature engineering39,46,53. First, for the \(\triangle {h}_{o}\) data, the perovskite formulas found in Vieten et al.46 were checked against Materials Project to find if they were optimized. All formulas that were not found in materials project were discarded from the database. After that, the list is reduced to the number of possible perovskites that are in the range of 188 – 517 kJ mol−1, removing all perovskites with enthalpies outside this range. The specific values of 188 and 517 kJ mol−1 corresponds to the exact minimum and maximum \(\triangle {h}_{o}\) values found in the database for the compounds Sr0.625Ca0.375Ti0.375Mn0.625O3 and RbWO3. This particular range is chosen for training and testing as, perovskites showing lower values of enthalpy will not split water while higher values of enthalpy will not show realistic reduction temperatures65. Although the minimum value of enthalpy would be around 246 kJ mol−1, 188 kJ mol−1 is chosen to increase the diversity of data for training. Additionally, the maximum value of the enthalpy should be similar to the \(\triangle {h}_{o}\,\) for CeO2 (around 490 kJ mol−1)23, and accordingly 517 kJ mol−1 can be considered a reasonable limit. Widening the range of data excessively can be counterproductive as adding too much data outside the range of interest could increase the variance error with the risk of overfitting the model, not increasing the predictability in the range of interest66. For Baldassarri et al.41 database, we selected formulas containing perovskites (excluding others like pyrochlores) and a set of 506 compounds were used for training and test. The range of values in Baldassarri et al. is already within the selected range 188–517 kJ mol−1 and no additional data was discarded.
RF models require both the target variable data and predictors, which are generated through feature engineering using features from literature data for both databases. This is a critical step to produce an appropriate database. For this work, a number of features were created using the methodology from Li et al.53 as well as the Github repository “Materials Simulation Toolkit for Machine Learning” (MAST-ML)67 adapted to this current work. In this way, data are able to generate predictors for unknown materials (never explored computationally or experimentally) and serve for a quick screening of compositions which is the purpose of this work. This strategy was followed in previous machine learning works and it is similarly applied here41,42. with some differences. Thus, Witman et al.42 used DFT data of pristine materials as predictors in graph neural networks, while the work of Baldassarri et al.41 used formation energy of oxides and band gaps as predictors.
For the purpose of materials discovery, it is essential to create a database that contains properties that do not rely on experimental or computational results. In our current work, we aimed to explore unknown materials (where DFT or formation energy data are not available) using a wider screening of compositions and not limiting it to materials already gathered in computational repositories. Including structural parameters could limit materials discovery to compounds with known crystal structures, space groups, or lattice parameters, which are typically available only through experimental data or DFT-relaxed geometries. In contrast, our list of perovskite candidates was generated combinatorially based solely on chemical composition, ensuring charge neutrality and applying specific selection criteria—without requiring prior structural knowledge. This approach differs from previous machine learning efforts41,42 that incorporate geometric descriptors derived from known structures. Our main limitation lies in the absence of structure-dependent features, which prevents the model from resolving local geometric effects or distinguishing among non-equivalent atomic sites. While accurate predictions using structure-dependent models require access to the “real” (i.e., experimentally resolved or DFT-relaxed) crystal structure, alternative strategies are possible in the absence of such information. One approach employed in this work is the complete exclusion of structure-dependent features. An alternative strategy, not pursued here due to the need for structure-independent features, involves substituting target elements into a prototypical perovskite structure—such as the ideal cubic phase—without conducting a full DFT relaxation. Although this introduces approximations, as the real structure may involve different distortions, lattice parameters, or atomic environments, it remains a feasible route to generate approximate geometries. These approximate structures can then serve as inputs to existing structure-based models such as ‘Feature set I’ and ‘Element-only’ in41,42.
The final databases for training and testing are composed of 79 and 506 perovskites, respectively, sourced from the Vieten and Baldassarri databases, each with 258 features. Following autocorrelation and variable importance analyses, the number of features was reduced to 10 for the Vieten dataset and 33 for the Baldassarri dataset. Another dataset is constructed to predict the stability of the materials using Talapatra et al.47 stability data with the 258 initial predictors. This dataset is composed of 3469 perovskites with varying stoichiometry. Details of this database can be found in the Supplementary Information document and in Talapatra et al. 47. Table 5 summarizes the main characteristics of the four databases used in this work. Details of databases such as methods of calculations, crystalline structures, space groups among others can be consulted in the respective references41,46.
Machine learning model
Once the databases are prepared, it is possible to start searching for materials with adequate \(\triangle {h}_{o}\) using machine learning. Supervised machine learning is used in this work as we pretend to perform predictions over tabulated data. From the supervised machine learning algorithms, a RF regression is selected. RF are a combination of tree predictors such that each tree depends on the values of a random vector sampled independently and with the same distribution for all trees in the forest68. RF are also adequate for ranking the importance of predictors, helping to explain the physical significance of the model. Additionally, considering the small datasets used in this work (79 and 506 samples), RF models have also been demonstrated to be able to provide accurate results, while mitigating overfitting, which can be a common issue found in more complex models (like deep learning) used on reduced datasets69. An exploratory analysis is performed before training to know the variety of elements and the distribution of the target property (\(\triangle {h}_{o})\). After that, the machine learning training and test are performed. After initial training, an exploratory analysis of the predictors is carried out, searching for the autocorrelated variables with the Pearson coefficient (ρ), in order to reduce the complexity of the model and the noise, maintaining the same accuracy. In addition, a variable importance analysis together with heat maps is included in this work to clarify the predictions of the model and give a physical meaning to the results. These steps are different from previous approaches shown in41,42.
The RF workflow used in this work is described as follows (and use to train both ML models with the two DFT databases):
-
1.
Sets of databases are formed by bootstrapping the initial database (using a split of 70-30% for training and testing)68 using a number of trees (ntrees) of 500. The RF model is stabilized, and the lowest error is achieved. RF hyperparameters are optimized during the training procedure. We used 500 trees to ensure stable predictions, as the validation error plateaued after ~200 trees. Increasing the number of trees further reduces variance through averaging, without increasing bias or causing overfitting68.
-
2.
Each subset of data (train and test) is used independently to give a set of parameters for the RF regression with its corresponding R2 (goodness-of-fit), MAE (Mean absolute error) and RMSE (Root square mean error) to predict the \(\triangle {h}_{o}\).
-
3.
The final regression model is built by averaging each set of parameters in the 500 regression trees.
-
4.
The prediction results over the testing and training datasets are given along with the R2, MAE and RMSE. The values of R2 for the train set are higher than for the test set, and lower MAE and RMSE are also obtained as commonly found in machine learning models70.
-
5.
A cross-validation is performed using a 5-fold method.
For the construction of the database and training and testing of the ML model, we used the open-source R-language and the R Studio environment71. The source code and database are available at SmartSolFuel GitHub repository.
In order to test if RF is a good predictive model against other simpler alternatives (as proposed by works from Wexler et al.45 and Deml et al.72, we have tested several linear regression models and compared them with RF. Linear models could be an alternative specially to datasets using small number of samples such as the Vieten database. However, across all relevant metrics, the RF model consistently outperformed the linear models. Specifically, RF achieved higher R² values (see Supplementary Table 5 and Supplementary Fig. 17), indicating its superior ability to capture the variability and complexity of the dataset, while also exhibiting reduced bias.
List of perovskites for discovery and predictions
A list of perovskites for discovery was created for prediction purposes before selecting materials for experimental demonstration. This process, as described in Fig. 7, involves a preliminary selection of elements to form compositions. This step was necessary because selecting all possible combinations of elements would create an unmanageable problem, with most of the resulting compounds expected to be unstable or even not forming a perovskite structure. To address this, elements were selected based on those commonly found to form stable perovskites for STCH applications, following these criteria: non-metals such as F, Cl, Br, I, and noble gases (He, Ne, Ar, Kr, Xe, Rn) are incapable of forming the necessary oxide structures, and other non-metals like H, C, N, O, P, S, and Se are similarly unsuitable for perovskite formation. Alkali metals, including Li, Na, K, Rb, Cs, and Fr, often introduce volatility and instability, preventing the formation of robust perovskite oxides. Among alkaline earth metals, Be, Mg, and Ra are excluded as their oxides are unstable, unlike Ca, Sr, and Ba, which form stable perovskite structures. Post-transition metals such as Ga, In, Tl, Sn, Pb, and Bi have limited stability in perovskites, while metalloids like B, Si, Ge, As, Sb, and Te rarely form perovskite oxides and are scarcely studied. Certain transition metals, particularly Ag, Au, Zn, Cd, and Hg, are excluded due to volatility or inability to adopt the required oxidation states. In the lanthanide and actinide series, most elements (e.g., Pm, Eu, Tb, Dy, Ho, Er, Tm, Yb, and Lu) do not consistently form stable perovskite oxides, though exceptions include La, Ce, Sm, and Y. Actinides are generally excluded due to their radioactivity and limited practical applications. As a result, the elements that do not form perovskite oxides adequate for STCH include H, Li, Be, B, C, N, O, F, Na, Mg, Si, P, S, Cl, K, Sc, V, Cr, Zn, Ga, Ge, As, Se, Br, Rb, Zr, Nb, Mo, Ag, Cd, In, Sn, Sb, Te, I, Cs, Ba, Hf, Ta, W, Re, Os, Ir, Pt, Au, Hg, Tl, Pb, Bi, Po, At, Ra, Ac, and all actinides.
Overview of the compositional space, down-selection strategy, and representative candidate materials identified through the selection process.
This approach reduces uncertainty regarding the synthesizability of the materials. According to the literature, most materials that demonstrate effective water splitting with suitable thermodynamic properties contain Mn, Ni or Fe as redox-active elements. Other elements, such as Co, Al, Ti, Cu or Zr, are used as redox-inactive dopants73. On the other hand, the A-site elements were selected based on their frequent occurrence in perovskites, with La being the most common in the literature, followed by Ba, Ca, Sr, Ce, Y, and Sm73. The variability in elements was introduced by adding dopants from 0 to 1 in increments of 0.125 in the A and B sites, providing a wide range of compositions. Despite this relatively limited set of elements considered, a combinatorial analysis revealed 34300 possible combinations, of which 6264 were charge neutral. In order to verify the stability of the materials in the discovery database, a machine-learning model is used. Here, the stability database was constructed using the same methodology as per the \(\triangle {h}_{o}\,\) prediction database using elemental information as in the materials discovery database. The machine learning model utilized was a random forest classification (RFC) as the stability value is a binary distribution (e.g. 0,1) that contains information of 3469 perovskites (classified as stable and unstable). For those materials, the elemental features are again 258. In this case, the whole set of features can be used for training and test as the number of samples is larger than 10 times the number of features ensuring avoiding overfitting; however, random forest models are well known to not produce overfitting even with larger number of features. The RFC model splits the dataset into training and test with 70% of data for training and the remaining for test.
The model results are shown in the Supplementary Information document along with details of the database for stability prediction. In summary, from 6264 formulas over 97% are predicted as stable while less than 3% are predicted as unstable. From the stable list, a final selection of materials for the experimental demonstration was performed according to their representative predicted \(\triangle {h}_{o}\) given the following list: Ba0.875Ca0.125Zr0.875Mn0.125O3, La0.125Sr0.875Fe0.875Mn0.125O3, La0.875Ba0.125Fe0.875Co0.125O3 and LaCo0.875Ni0.125O3.
DFT calculations
Spin-polarized DFT + U calculations for computing \(\triangle {h}_{o}\) (Eqs. (5) and (6)) were performed using the Vienna Ab Initio Simulation Package (VASP)74. The Generalized Gradient Approximation with the Perdew–Burke–Ernzerhof (GGA-PBE) exchange-correlation functional and projector augmented wave (PAW) pseudo-potentials were employed. DFT calculations were performed for both the oxidized (Ba0.875Ca0.125Zr0.875Mn0.125O₃) and the reduced (Ba0.875Ca0.125Zr0.875Mn0.125O₃ −δ) forms of the BCZM perovskite. To ensure reliable results, full structural optimizations were carried out using an increased energy cutoff (ENCUT = 1.3 × max(ENMAX)). Based on the highest ENMAX value from the pseudopotential files (400 eV, corresponding to the O species), ENCUT was set to 520 eV. At least two consecutive optimizations were performed, allowing adjustments to ionic positions, cell volume, and cell shape. The global break condition for the electronic SC-loop was set to 10-4 eV, while the ionic relaxation stopping condition was 10⁻3 eV.
The computed \(\triangle\)ho is obtained from DFT data using the Eq. (5), along with the conversion of eV to kJ mol−1, with the \(\frac{1}{2}{E}_{{O}_{2}}\) value of −4.523 eV, as recommended by Baldassarri et al.41, to address the well-known tendency of the PBE functional to overestimate binding energies:
To analyze the effect of different off-stochiometry (δ) values on the predicted oxygen vacancy formation enthalpy, \({\triangle h}_{o}\), reduced BCZM structures were constructed with δ = 0.125 and δ = 0.5. Here, δ is defined relative to the ABO₃ formula unit, and compositions are expressed as ABO₃₋δ. For the δ = 0.125 case, both cubic and orthorhombic structures were generated from Ba₇Ca₁Zr₇Mn₁O₂₄ supercells obtained from the Materials Project database. The orthorhombic BCZM structure was constructed from the cubic structure by applying small lattice parameter modifications, followed by full structural optimization (see Supplementary Table 7). These structures were fully relaxed using a Γ-centered 5 × 5 × 5 k-point mesh (to see the k-point effect, see Supplementary Table 6 for more details). To study the effect of structural change in obtaining ∆ho, we constructed orthorhombic brownmillerite-phase cells in space group 46 (Ima2), based on Materials Project entry mp-1077688. The structure has lattice parameters of 12.024 \(\dot{{\rm{A}}}\) × 12.020 \(\dot{{\rm{A}}}\) × 18.747 \(\dot{{\rm{A}}}\). This supercell contains 144 atoms, corresponding to the Ba28Ca4Zr28Mn4O80 (4 × Ba7CaZr7MnO20) composition with a non-stoichiometry coefficient of δ = 0.5. The structure was fully optimized using a 3 × 3 × 2 k-point grid, representing the reduced phase considered by Vieten et al., which assumes a structural phase transition from the orthorhombic to the triclinic phase upon reduction (see Supplementary Fig. 19a). In this case, only the FM configuration was considered, based on the weak magnetic coupling observed in the cubic phase with δ = 0.125 (see Results). For the construction of the reduced orthorhombic phase (Ba28Ca4Zr28Mn4O80) and the oxidized orthorhombic Ba28Ca4Zr28Mn4O96 supercell (See Supplementary Fig. 19, b), we started with the Ba7Ca1Zr7Mn1O24 orthorhombic structure (Supplementary Table 7) and doubled the a and b lattice parameters, followed by full structural optimization using a 5 × 3 × 3 k-point grid. The optimized lattice parameters are 8.382 \(\dot{{\rm{A}}}\) × 16.796 \(\dot{{\rm{A}}}\) × 16.796 \(\dot{{\rm{A}}}\). We then removed 16 equivalent oxygen atoms, corresponding to a vacancy concentration of 16/96 = 1/6 (δ = 0.5). A final structural optimization was performed to ensure stability and accommodate adjustments to the ionic positions, cell volume, and shape.
To form the oxygen vacancies, a single oxygen atom was removed per supercell of 40 atoms, corresponding to an oxygen vacancy concentration of θ = 1/24. When normalized to the ABO₃ formula unit, this yields δ = (1/24) × 3 = 0.125, consistent with typical experimental conditions. The removed oxygen atoms corresponded to those identified as the most labile in the smaller unit cell using energy-based criteria, as discussed below. As with the brownmillerite phase, only the ferromagnetic (FM) configuration was considered, justified by the weak magnetic coupling observed in the cubic structure at δ = 0.0625.
Because the cubic and orthorhombic BCZM phases contain multiple inequivalent oxygen atoms with distinct coordination environments, each type of oxygen was systematically removed to generate reduced structures (see Supplementary Table 8, 9, 10 for more details removing the different types of oxygen atoms). All resulting configurations were then fully relaxed, and the most stable configuration for each symmetry was identified and used for \({\triangle h}_{o}\) calculations. For δ = 0.125, no structural phase transitions were considered upon reduction; both oxidized and reduced structures retained either cubic or orthorhombic symmetry, depending on the starting phase
For structures with δ = 0.5, the reduced brownmillerite phase was modeled using a triclinic supercell with the composition Ba₂₈Ca₄Zr₂₈Mn₄O₈₀, consistent with structures reported in the Vieten et al. database. This structure was fully optimized using a 3 × 3 × 2 k-point grid. Additionally, an orthorhombic phase was constructed from a 2 × 2 × 1 supercell of Ba₇CaZr₇MnO₂₄, resulting in a Ba₂₈Ca₄Zr₂₈Mn₄O₉₆ composition. In this case, 16 equivalent oxygen atoms were removed, corresponding to a vacancy concentration of θ = 1/6 (δ = 0.5). Oxygen removal was guided by energy-based criteria, prioritizing the most labile oxygen sites as identified from calculations on the smaller unit cell. A final full structural relaxation was then performed on the reduced orthorhombic structure using a 5 × 3 × 3 k-point mesh.
For δ = 0.5, the effect of a structural phase transition upon reduction was explicitly considered by comparing two pathways: one in which both the oxidized and reduced phases remain orthorhombic, and another in which the oxidized structure is orthorhombic but the reduced phase adopts an orthorhombic brownmillerite phase (space group 46, Ima2).
Spin-polarized DFT + U calculations were performed on the double perovskite BCZM (Ba₀.₈₇₅Ca₀.₁₂₅Zr₀.₈₇₅Mn₀.₁₂₅O₃) in both cubic and orthorhombic structures for oxidized and reduced phases. These simulations used cells with compositions Ba₇Ca₁Zr₇Mn₁O₂₄ (oxidized) and Ba₇Ca₁Zr₇Mn₁O₂₃ (reduced), corresponding to a non-stoichiometry δ = 0.125. Magnetic configurations were initialized by assigning initial magnetic moments of +3, +4, and +5 μB to the Mn cations, corresponding to their expected oxidation states of Mn⁴⁺, Mn³⁺, and Mn²⁺. For structures with δ = 0.125, the simulation cells contained only a single Mn cation—Mn⁴⁺ in the oxidized phases and Mn²⁺ in the reduced phases —so magnetic ordering was not a relevant degree of freedom in those cases (see Supplementary Information Table 11). To investigate alternative redox pathways following oxygen removal, specifically the reduction of 2 × Mn⁴⁺ to either 2 × Mn³⁺ or to a mixed-valence state of 1 × Mn⁴⁺ and 1 × Mn²⁺, we employed a larger 2 × 1 × 1 cubic supercell of Ba₇CaZr₇MnO₂₄, corresponding to a Ba₁₄Ca₂Zr₁₄Mn₂O₄₈ composition (80-atom supercell). Ferromagnetic (FM) and antiferromagnetic (AFM) spin configurations were generated by aligning the Mn spins in parallel or antiparallel orientations. After removing the most labile oxygen atom (δ = 0.0625), we evaluated the magnetic and energetic stability of various reduced states. For the Mn³⁺/Mn³⁺ configuration, both FM and AFM alignments were considered. In the Mn²⁺/Mn⁴⁺, only an AFM configuration was tested, specifically constructed to enforce a redox-asymmetric state. Here, the Mn cations were initialized with magnetic moments of +5 μB and −3 μB, and the total magnetization was constrained to 2 μB to enforce the desired mixed-valence redox state. Total energies of the relaxed FM and AFM structures were then compared to assess the stability of each magnetic configuration. For the orthorhombic structure with Ba₂₈Ca₄Zr₂₈Mn₄O₉₆ composition and the brownmillerite structure with Ba₂₈Ca₄Zr₂₈Mn₄O₈₀ composition (δ = 0.5), the simulation cell contained four Mn cations—Mn⁴⁺ in the oxidized state and Mn²⁺ in the reduced state. In this case, only the FM configuration was considered, based on the weak magnetic coupling observed in the cubic phase.
Experimental Demonstration
Experimental demonstration is performed on selected materials from the list of perovskites for prediction, prepared by a modified sol-gel synthesis method. These are: Ba0.875Ca0.125Zr0.825Mn0.125O3, La0.125Sr0.875Fe0.875Mn0.125O3, La0.125Ba0.875Fe0.875Co0.125O3 and LaCo0.875Ni0.125O3. The samples are identified by cation composition as: BCZM, LSFM, LBFC and LCN respectively. The compositions were chosen due to its variability on the B-site cation and the B-site oxidation state. Besides, we are continuously testing new materials from the 6264 list of discovery in search for novel perovskites. Sigma-Aldrich metal nitrate precursors, ACS reagents 97.0 – 99.9% trace metals, were used in the Pechini method to produce perovskite oxides75. Stoichiometric quantities of metal nitrates were dissolved in 75 ml of milliQ water with citric acid added in a proportion of 5 times the molar content of metals. The solution was heated to 70 °C for 3 h under continuous agitation. Ethylene glycol was then added at a mol ratio of 3:2 citric acid to ethylene glycol. The solution was further stirred at 90 °C for 3 h to obtain a polymeric resin. Combustion of the resin was achieved by slowly heating using a muffle furnace to 400 °C in air (0.1 °C min−1, holding for 4 hours isothermal), to obtain metal oxide. This solid was ground in a mortar and pestle before calcination at 1400 °C for 6 h.
The predicted thermodynamic properties were experimentally validated using a thermogravimetric analyzer (SETARAM SETSYS Evolution 16) and the van´t Hoff principle where that \(\triangle {h}_{o}\) and \(\triangle {s}_{o}\) are obtained by a linearization of the \(\triangle {g}_{o}\) equation assuming independency from the temperature76. The selected sample for further study (BCZM) was also replicated in a 449 F5 Jupiter (NETSZCH). In these devices, a series of T and \({p}_{{O}_{2}}\) steps were performed, and the δ at chemical equilibrium was calculated, producing a dataset of T, \({p}_{{O}_{2}}\), and δ. Typically, a wide dataset is obtained, however, to accelerate data acquisition, we have limited the dataset to 4 temperatures and 4 \({p}_{{O}_{2}}\) to each perovskite, with the exception of BCZM, which was evaluated at 5 temperatures. Figure 8 illustrates an example of this experiment conducted on LCN material.
Thermogravimetric experiments conducted over a range of temperatures and \(p{o}_{2}\) to evaluate δ.
The most common method to obtain \(\triangle {h}_{o}\) and \(\triangle {s}_{o}\) from these datasets is using the van’t Hoff approximation23,37,77, which assumes both variables are independent of the temperature78. With a large dataset, the interpolation of δ as a function of \({p}_{{O}_{2}}\) for a fixed T allows for easy calculation of \(\triangle {h}_{o}\). However, if the dataset is not sufficiently large, the error in enthalpy becomes magnified. To reduce this error and accelerate the material discovery, a chemical equilibrium model that fits the dataset can be used to capture the material trend. One of the simplest is the one described by Bulfin et al.56, which relies only on four fitting parameters:
Where \({\delta }_{m}\), \(\frac{{A}_{{red}}}{{A}_{{ox}}}\), \(n\) and \({E}_{a}\) are parameters. Appling van’t Hoff approximation on the Bulfin model, \(\triangle {h}_{o}\) and \(\triangle {s}_{o}\) are obtained. Note that this model assumes this variable independent of T and δ. Although it is recognized that these assumptions have limitations79, it has been adopted in many previous works37,80,81,82.
Other alternative methods to the van’t Hoff approximation for obtaining \(\triangle {h}_{o}\) and \(\triangle {s}_{o}\) are reported. The “chemical potential method” described by Lany57, addresses the limitations of van’t Hoff by using oxygen chemical potential instead of \({p}_{{O}_{2}}\), decoupling gas-phase and solid-state contributions. There are also other equilibrium models, such as the one described by Takacs et al.83, and the compound energy formalism84,85,86, which has demonstrated high accuracy when combined with thermogravimetric data. These methods and models require fitting a larger number of parameters and a deeper understanding of the material being studied57,84, as well as longer datasets to avoid overfitting and there is no guarantee that more accurate results will be obtained compared to the Bulfin model87.
Incorporating those approaches could enhance the understanding of the material’s behavior under varying experimental conditions, offering a more detailed analysis of performance. While we employed the van’t Hoff method in this study to align with established experimental materials used for predictions in this work23,37,77, future investigations could benefit from integrating advanced methodologies to assess a material’s water-splitting capability and operational equilibrium temperatures more comprehensively.
Powder X-ray diffraction (XRD) measurements were performed using a PANalytical Aeris diffractometer at room temperature. Data were collected over a 2θ range of 5° to 90°, with a step size of 0.006° and a total acquisition time of one hour. The data were analyzed using PANalytical HighScore software, which included background correction for low-angle effects and peak identification. Subsequently, Rietveld refinement was carried out, utilizing a parent structure as the initial model. The parent structure is assumed to have similar space group and crystalline structure to the target crystalline material. Modifications to the parent structure were applied to reflect the stoichiometry of the perovskite phase under investigation prior to refinement. The Rwp value, or weighted profile R-factor (a statistical parameter used to assess the goodness of fit in Rietveld), is given for all the refined structures. Rwp between 5–10 indicates an acceptable goodness of fit.
A Zeiss Sigma VP scanning electron microscope coupled with a Bruker energy dispersive X-ray system, SEM/EDS, was used to image particle surfaces. Acceleration voltages of 5 or 15 keV were used with BSE, SE and In-lenses detectors, while the magnification of 5-50 k was used.
System performance of materials
A computational tool has been developed to compare materials under equal operating conditions. This approach assumes reactions in counter-current flow reactors, which maximize the utilization of the chemical potential of the redox-active material. This tool is based on the findings by de la Calle et al. in thermochemical water splitting counter-current operation48,51. This system model helps to compare materials under the same critical parameters such as oxygen partial pressure and water-to-hydrogen conversion ratios were fixed to ensure a fair and independent comparison with other perovskites. This offers reliability as compared to complex system modeling platforms that require assumptions of system performance parameters [9]. The tool, developed in Matlab, calculates the behavior of both reactors under specific operating conditions (extent of reduction and re-oxidation, inlet partial pressure of oxygen, reduction sweep ratio, inlet molar fraction of hydrogen and hydrogen yield). Note that mass balance lines are depicted in Fig. 9 as straight lines under \(\delta -{p}^{* }\) and \(\delta -{x}_{{{\rm{H}}}_{2}}\) coordinates in counter-current reduction and re-oxidation reactions respectively (\({p}^{* }\) is the ratio between the partial pressure of oxygen and inert gas and \({x}_{{{\rm{H}}}_{2}}\) is the molar fraction of H2)48,51. As a result, temperature profiles at chemical equilibrium for both reactions are obtained. Figure 9 shows a schematic of the system modeling tool. It was used to obtain a comparative assessment against CeO2 and a selected perovskite representing state-of-art materials.
Data availability
The raw data used in this work were obtained from Vieten et al.46, Baldassarri et al.41 and Talapatra et al.47. The curated dataset: Data architecture and feature engineering, is available at SmartSolFuel GitHub repository: https://github.com/ABayonSandoval/SmartSolFuel.
Code availability
Custom code used for the findings described: Results available at SmartSolFuel GitHub repository: https://github.com/ABayonSandoval/SmartSolFuel.
References
Lide, D. R. CRC Handbook of Chemistry and Physics 88th edn. (CRC Press, 2007).
Bayon, A., de la Calle, A., Ghose, K. K., Page, A. & McNaughton, R. Experimental, computational and thermodynamic studies in perovskites metal oxides for thermochemical fuel production: a review. Int. J. Hydrogen Energy 45, 12653–12679 (2020).
Bhosale, R. R. et al. A decade of ceria based solar thermochemical H2O/CO2 splitting cycle. Int. J. Hydrogen Energy 44, 34–60 (2019).
Bader, R. & Lipiński, W. In Advances in Concentrating Solar Thermal Research and Technology (ed. Blanco, M. J.) 403–459 (Elsevier Inc., 2017).
Bayon, A., Hashimoto, J. & Muhich, C. In Advances in Chemical Engineering Vol. 58 (ed. Lipiński, W.) 55–90 (Academic Press Inc., 2021).
Bayon, A., de la Calle, A., Stechel, E. B. & Muhich, C. L. Operational limits of redox metal oxides performing thermochemical water splitting. Energy Technol 10, 2100222 (2022).
Siegel, N. P., Miller, J. E., Ermanoski, I., Diver, R. B. & Stechel, E. B. Factors affecting the efficiency of solar driven metal oxide thermochemical cycles. Ind. Eng. Chem. Res. 52, 3276–3286 (2013).
Ermanoski, I., Siegel, N. P. & Stechel, E. B. A new reactor concept for efficient solar-thermochemical fuel production. J. Sol. Energy Eng. 135, 31002 (2013).
de la Calle, A. & Bayon, A. Annual performance of a thermochemical solar syngas production plant based on non-stoichiometric CeO2. Int. J. Hydrogen Energy 44, 1409–1424 (2019).
Ermanoski, I., Miller, J. E. & Allendorf, M. D. Efficiency maximization in solar-thermochemical fuel production: challenging the concept of isothermal water splitting. Phys. Chem. Chem. Phys. 16, 8418–8427 (2014).
Roeb, M. et al. Test operation of a 100 kW pilot plant for solar hydrogen production from water on a solar tower. Sol. Energy 85, 634–644 (2011).
Abanades, S. Metal Oxides Applied to Thermochemical Water-Splitting for Hydrogen Production Using Concentrated Solar Energy. ChemEngineering 1–28 (MDPI AG, 2019).
Gao, X. et al. Earth-abundant transition metal oxides with extraordinary reversible oxygen exchange capacity for efficient thermochemical synthesis of solar fuels. Nano Energy 50, 347–358 (2018).
Bayón, A., De La Penã O’Shea, V. A., Serrano, D. P. & Coronado, J. M. J. Exploring the alternative MnO-Na2CO3 thermochemical cycle for water splitting. J. CO2 Util. 42, 101264 (2020).
Bayón, A., De La Peña O’Shea, V. A., Coronado, J. M. & Serrano, D. P. Role of the physicochemical properties of hausmannite on the hydrogen production via the Mn3O4–NaOH thermochemical cycle. Int. J. Hydrogen Energy 41, 113–122 (2016).
Bayón, A., De La Peña O’Shea, V. A., Serrano, D. P. & Coronado, J. M. Int. J. Hydrogen Energy https://doi.org/10.1016/j.ijhydene.2013.07.101 (2013).
Gao, X. et al. J. Mater. Chem. A https://doi.org/10.1039/c6ta02187e (2016).
Jiang, Q., Zhou, G., Jiang, Z. & Li, C. Thermochemical CO2 splitting reaction with CexM1−xO2−δ (M=Ti4+, Sn4+, Hf4+, Zr4+, La3+, Y3+ and Sm3+) solid solutions. Sol. Energy 99, 55–66 (2014).
Lu, Y. et al. Solar Fuels Production: Two-Step Thermochemical Cycles with Cerium-Based Oxides. Progress in Energy and Combustion Science 100785 (Elsevier Ltd., 2019).
Kaneko, H. et al. Reactive ceramics of CeO2–MOx (M=Mn, Fe, Ni, Cu) for H2 generation by two-step water splitting using concentrated solar thermal energy. Energy 32, 656–663 (2007).
Millican, S. L. et al. Oxidation kinetics of hercynite spinels for solar thermochemical fuel production. Chem. Eng. J. 401, 126015 (2020).
McDaniel, A. H. et al. Sr- and Mn-doped LaAlO3-δ for solar thermochemical H2 and CO production. Energy Environ. Sci. 6, 2424–2428 (2013).
Carrillo, R.J. & Scheffe, J. R. Advances and trends in redox materials for solar thermochemical fuel production. Sol. Energy 156, 3–20 (2017).
Chueh, W. C. et al. High-flux solar-driven thermochemical dissociation of CO2 and H2O using nonstoichiometric ceria. Science 330, 1797–1801 (2010).
Schäppi, R. et al. Drop-in fuels from sunlight and air. Nature 601, 63–68 (2022).
Scheffe, J. R. & Weibel, D. & Steinfeld, A. Lanthanum-strontium-manganese perovskites as redox materials for solar thermochemical splitting of H2O and CO2. Energy Fuels 27, 4250–4257 (2013).
Sastre, D., Carrillo, A. J., Serrano, D. P., Pizarro, P. & Coronado, J. M. Exploring the redox behavior of La0.6Sr0.4Mn1−xAlxO3 perovskites for CO2-splitting in thermochemical cycles. Top. Catal. 60, 1108–1118 (2017).
Haeussler, A., Abanades, S., Julbe, A. Jouannaux, J. & Cartoixa, B. Two-Step CO2 and H2O splitting using perovskite-coated ceria foam for enhanced green fuel production in a porous volumetric solar reactor. J. CO2 Util. 41, 101257 (2020).
Ghose, K. K., Brown, J. J., Frankcombe, T. J., Page, A. & Bayon, A. Density functional theory modeling of critical properties of perovskite oxides for water splitting applications. Wiley Interdiscip. Rev. Energy Environ. 14, e476 (2023).
Deml, A. M. et al. Tunable oxygen vacancy formation energetics in the complex perovskite oxide SrxLa1−xMnyAl1−yO3. Chem. Mater. 26, 6595–6602 (2014).
Emery, A. A., Saal, J. E., Kirklin, S., Hegde, V. I. & Wolverton, C. High-throughput computational screening of perovskites for thermochemical water splitting applications Chem. Mater. 28, 5621–5634 (2016).
Ghose, K. K., Bayon, A., Hinkley, J. & Page, A. J. Electronic structure and high-temperature thermochemistry of BaZrO(3-δ) perovskite from first-principles calculations. Phys. Chem. Chem. Phys. 21, 12468–12476 (2019).
Ghose, K. K., Bayon, A. & Page, A. J. First-principles thermochemical properties of hexagonal and cubic phase BaMnO3. Mater. Today Commun. 31, 103453 (2022).
Ghose, K. K., Bayon, A. & Page, A. J. J. Phys. Chem. C 124, 27055–27063 (2020).
Tao, Q. Xu, P., Li, M. & Lu, W. Machine learning for perovskite materials. npj Comput. Mater. 7, 23 (2021).
Li, S. et al. Thermodynamic guiding principles for designing nonstoichiometric redox materials for solar thermochemical fuel production: ceria, perovskites, and beyond. Energy Technol. 10, 2000925 (2021).
Qian, X. et al. Outstanding properties and performance of CaTi0.5Mn0.5O3–δ for solar-driven thermochemical hydrogen production. Matter 4, 688–708 (2021).
Takacs, M., Scheffe, J. R. & Steinfeld, A. Oxygen nonstoichiometry and thermodynamic characterization of Zr doped ceria in the 1573-1773 K temperature range. Phys. Chem. Chem. Phys. 17, 7813–7822 (2015).
Jain, A. et al. Commentary: The Materials Project: a materials genome approach to accelerating materials innovation. APL Mater 1, 11002 (2013).
Saal, J. E., Kirklin, S., Aykol, M., Meredig, B. & Wolverton, C. Materials design and discovery with high-throughput density functional theory: the Open Quantum Materials Database (OQMD). J. Miner. Met. Mater. Soc. 65, 1501–1509 (2013).
Baldassarri, B. et al. Oxygen vacancy formation energy in metal oxides: high-throughput computational studies and machine-learning predictions. Chem. Mater. 35, 10619–10634 (2023).
Witman, M. D., Goyal, A., Ogitsu, T., McDaniel, A. H. & Lany, S. Defect graph neural networks for materials discovery in high-temperature clean-energy applications. Nat. Comput. Sci. 3, 675–686 (2023).
Choudhary, K. & Sumpter, B. G. Can a deep-learning model make fast predictions of vacancy formation in diverse materials. AIP Adv 13, 0–6 (2023).
Deml, A. M., Holder, A. M., O’Hayre, R. P., Musgrave, C. B. & Stevanović, V. Intrinsic material properties dictating oxygen vacancy formation energetics in metal oxides. J. Phys. Chem. Lett. 6, 1948–1953 (2015).
Wexler, R. B., Gautam, G. S., Stechel, E. B. & Carter, E. A. Factors governing oxygen vacancy formation in oxide perovskites. J. Am. Chem. Soc. 143, 13212–13227 (2021).
Vieten, J. et al. Materials design of perovskite solid solutions for thermochemical applications. Energy Environ. Sci. 12, 1369–1384 (2019).
Talapatra, A., Uberuaga, B. P., Stanek, C. R. & Pilania, G. A machine learning approach for the prediction of formability and thermodynamic stability of single and double perovskite oxides. Chem. Mater. 33, 845–858 (2021).
de la Calle, A., Ermanoski, I. & Stechel, E. B. Towards chemical equilibrium in thermochemical water splitting. Part 1: thermal reduction. Int. J. Hydrogen Energy 47, 10474–10482 (2022).
de la Calle Alonso, A. & Bayon, A. Annual performance of a solar-thermochemical hydrogen production plant based on CeO2 redox cycle. https://doi.org/10.3384/ecp17132857 (2017).
Bayon, A. & De La Calle, A. Preface: Proceedings of the 24th SolarPACES International Conference (SolarPACES 2018). AIP Conf. Proc. 2126, 010001 (2019).
de la Calle, A., Ermanoski, I., Miller, J. E. & Stechel, E. B. Towards chemical equilibrium in thermochemical water splitting. Part 2: Re-oxidation. Int. J. Hydrogen Energy 72, 1159–1168 (2024).
Fu, Y. et al. Exploring the effects of training samples on the accuracy of crop mapping with machine learning algorithm. Sci. Remote Sens. 7, 100081 (2023).
Li, W., Jacobs, R. & Morgan, D. Predicting the thermodynamic stability of perovskite oxides using machine learning models. Comput. Mater. Sci. 150, 454–463 (2018).
Su, H. Y. & Sun, K. DFT study of the stability of oxygen vacancy in cubic ABO3 perovskites. J. Mater. Sci. 50, 1701–1709 (2015).
Toby, B. H. https://doi.org/10.1154/1.2179804 (2016).
Bulfin, B. et al. Analytical model of CeO2 oxidation and reduction. J. Mater. Chem. C 117, 24129−24137 (2013).
Lany, S. Chemical potential analysis as an alternative to the van't Hoff method: hypothetical limits of solar thermochemical hydrogen. J. Am. Chem. Soc. 146, 14114–14127 (2024).
Wexler, R. B. et al. Multiple and nonlocal cation redox in Ca–Ce–Ti–Mn oxide perovskites for solar thermochemical applications. Energy Environ. Sci. 16, 2550–2560 (2023).
Baldassarri, B. et al. Phys. Rev. Mater. 7, 1–13 (2023).
Wang, Y., Baldassarri, B., Shen, J., He, J. & Wolverton, C. Chem. Mater. 36, 6816–6830, https://doi.org/10.1021/acs.chemmater.4c00576 (2024).
Ding, J., Zhao, L., Yang, Y. & Liu, J. Unraveling the role of a Cu dopant in formaldehyde catalytic oxidation over a La₀.₈Sr₀.₂Mn₁₋ₓCuₓO₃ perovskite: an experimental and theoretical study. J. Mater. Chem. A 13, 7371–7380 (2025).
Jin, F. et al. CaCo₀.₀₅Mn₀.₉₅O₃−δ: a promising perovskite solid solution for solar thermochemical energy storage. ACS Appl. Mater. Interfaces 13, 3856–3866 (2021).
Perry, J., Jones, T. W., Coronado, J. M., Donne, S. W. & Bayon, A. Thermodynamic analysis of a novel two-step high temperature thermo-electrochemical water splitting cycle. Energy 276, 127412 (2023).
Lenarduzzi, G. & Cooper, T. A. The role of entropy in the success of nonstoichiometric oxides for two-step thermochemical water and CO2 splitting. Appl. Phys. Lett. 119, 263902 (2021).
Bayon, A., de la Calle, A., Stechel, E. B. & Muhich, C. Operational limits of redox metal oxides performing thermochemical water splitting. Energy Technol. 10, 2100222 (2022).
Ying, X. An overview of overfitting and its solutions. J. Phys.: Conf. Ser. 1168, 22022 (2019).
Jacobs, R. et al. The Materials Simulation Toolkit for Machine learning (MAST-ML): an automated open source toolkit to accelerate data-driven materials researchComput. Mater. Sci. 176. (2020).
Breiman, L. Random forests Mach. Learn. 45, 5–32 (2001).
Li, L. et al. Studies on the regularity of perovskite formation via machine learning. Comput. Mater. Sci. 199, 110712 (2021).
Toyao, T. et al. Machine Learning for Catalysis Informatics: Recent Applications and Prospects 2260–2297 (ACS Catalysis American Chemical Society, 2020).
Rs. Team. RStudio: Integrated Development Environment for R. Posit Software, PBC, Boston, MA. http://www.posit.co/ (2025).
Deml, A. M., O’Hayre, R., Wolverton, C. & Stevanovic, V. Predicting density functional theory total energies and enthalpies of formation of metal-nonmetal compounds by linear regression. Phys. Rev. B 93, 85142 (2016).
Bayon, A., de la Calle, A., Ghose, K. K., Page, A. J. & McNaughton, R. Experimental, computational and thermodynamic studies in perovskites metal oxides for thermochemical fuel production: a review. Int. J. Hydrogen Energy 45, 12653–12679 (2020).
Dudarev, S. & Botton, G. Electron-energy-loss spectra and the structural stability of nickel oxide: an LSDA+U study. Phys. Rev. B 57, 1505–1509 (1998).
Jana, P., De La Peña O’Shea, V. A., Coronado, J. M. & Serrano, D. P. Cobalt based catalysts prepared by Pechini method for CO2-free hydrogen production by methane decomposition. Int. J. Hydrogen Energy 35, 10285–10294 (2010).
Panlener, R. J., Blumenthal, R. N. & Garnier Medley, J. E. A Thermodynamic Study of Nonstoichiometric Cerium Dioxide Vol. M (Puganwa Press, 1975).
Qian, X. et al. Favorable redox thermodynamics of SrTi0.5Mn0.5O3−δ in solar thermochemical water splitting. Chem. Mater. 32, 9335–9346 (2020).
Mastronardo, E., Sánchez, M., González-Aguilar, J., Haile, S. M. & Coronado, J. M. Proc.—ISES Sol. World Congr. 2021, Vol. 9, 875–880 (2021).
Cooper, T. et al. Lanthanum manganite perovskites with Ca/Sr A‐site and Al B‐site doping as effective oxygen exchange materials for solar thermochemical fuel production. Energy Technol 3, 1130–1142 (2015).
Panlener, R. J., Garnier, J. E. & Blumenthal, R. N. A thermodynamic study of non-stoichiometric cerium dioxide. Solid State Commun. 17, iv--v (1975).
Mastronardo, E., Sánchez, M., González-Aguilar, J. & Coronado, J. M. Experimental assessment of Fe-doped CaMnO3 porous pellets in a bench-scale packed-bed reactor for thermochemical energy storage. J. Energy Storage 57, 106226 (2023).
Mastronardo, E., Qian, X., Coronado, J. M. & Haile, S. M. 2020, https://doi.org/10.1039/d0ta02031a.
Takacs, M. et al. Oxygen nonstoichiometry, defect equilibria, and thermodynamic characterization of LaMnO3 perovskites with Ca/Sr A-site and Al B-site doping. Acta Mater. 103, 700–710 (2016).
Wilson, S. A., Stechel, E. B. & Muhich, C. L. Overcoming significant challenges in extracting off-stoichiometric thermodynamics using the compound energy formalism through complementary use of experimental and first principles data: a case study of Ba1−xSrxFeO3−δ. Solid State Ionics 390, 116115 (2023).
Zinkevich, M., Djurovic, D. & Aldinger, F. Thermodynamic modelling of the cerium–oxygen system☆. Solid State Ionics 177, 989–1001 (2006).
Hillert, M. The compound energy formalism. J. Alloys Compd. 320, 161–176 (2001).
Miller, J. E. et al. Modified calcium manganites for thermochemical energy storage applications. Front. Energy Res. 10, 774099 (2022).
Qian, X., Haile, S. M., Davenport, T. C. & Mastronardo, E. Experimental protocols for the assessment of redox thermodynamics of nonstoichiometric oxides: a case study of YMnO3‐δ. J. Am. Ceram. Soc. 105, 4375–4386 (2022).
Acknowledgements
J.P. and T.J. acknowledge that this activity received funding from ARENA as part of ARENA’s Research and Development Program – Renewable Hydrogen for Export. AC and RP gratefully acknowledge the financial support by Programa Atracción de Talento Fellowship of “Comunidad de Madrid” (2022-T1/AMB-23875). J.C., A.B., and A.C. thank Comunidad de Madrid” for the financial support to ACES4NET0-CM project (TEC-2024/ECO-116), through the R&D activities programme “Tecnologías 2024”. SD acknowledges support from the University of Newcastle Priority Research Centre for Frontier Energy Technologies and Utilization. M.V.G.P. thanks for the Grant PID2021-128915NB-I00 funded by MCIN/AEI/10.13039/501100011033 and by ERDF, UE. A.B. and L.M. gratefully acknowledge the financial support by Programa de Atracción de Talento Fellowship of “Comunidad de Madrid” (2020-T1/AMB-19884). A.B. also acknowledge the funding from Consolidacion Investigadora Fellowship (CNS2023-144887). We thank B. Baldassarri for helpful discussions. Computer time provided by the RES (Red Española de Supercomputación) resources at MareNostrum (BSC,Barcelona) node is acknowledged.
Author information
Authors and Affiliations
Contributions
Jonathan Perry: Conceptualization, Methodology, Investigation, Writing—Original Draft Preparation. Laura Molina: Conceptualization, Methodology, Data curation, Software, Formal Analysis, Writing—Original Draft Preparation. Alberto de la Calle: Conceptualization, Investigation, Software, Formal Analysis, Supervision, Writing—Original Draft Preparation. Raul Peño: Methodology, Investigation, Writing—review & editing. Timothy W. Jones: Methodology, Supervision, Writing—review & editing, M. Verónica Ganduglia-Pirovano: Methodology, Supervision, Writing—review & editing. Silvia Jimenez-Fernandez: Methodology, Supervision, Writing—review & editing. Scott W. Donne: Methodology, Supervision, Writing—review & editing. Juan M. Coronado: Methodology, Supervision, Writing—review & editing. Alicia Bayon: Conceptualization, Investigation, Data curation, Software, Formal Analysis, Supervision, Writing—Original Draft Preparation, Funding acquisition, Project administration.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Perry, J., Molina, L., de la Calle, A. et al. Discovery of materials for solar thermochemical hydrogen combining machine learning, computational chemistry, experiments and system simulations. npj Comput Mater 11, 247 (2025). https://doi.org/10.1038/s41524-025-01726-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41524-025-01726-y