Abstract
Intrinsic antisite defects pose a major challenge to understanding and predicting the exotic properties of the layered topological magnetic insulator MnBi2Te4 (MBT). In this work, we study the origin of the abundance of intrinsic defects in MBT, including many-body defect–defect interactions and many-body electronic correlations. Until now, ab initio methods have struggled to explain thermodynamic stability and properties influenced by defect behavior in MBT. We model native Mn–Bi antisite defects in MBT at finite temperatures using a cluster expansion that includes defect–defect interactions. To overcome the limitations of conventional density functional theory (DFT), we introduce a hybrid approach that incorporates high-accuracy quantum Monte Carlo (QMC) calculations, introducing missing correlations. This strategy allows for accurate estimation of defect energetics and finite-temperature properties. We compute the configurational free energy, defect concentration, and configurational heat capacity, revealing a second-order order–disorder phase transition near the experimental synthesis temperature. Our study provides the first theoretical insight into the thermodynamics of intrinsic defects in MBT. The negative free energy relative to pristine MBT at synthesis temperatures indicates that Mn–Bi antisite formation is thermodynamically spontaneous. We also present a broadly applicable general framework for correcting low-level theoretical theories using highly accurate many-body corrections from QMC.
Similar content being viewed by others
Introduction
Understanding materials that compete with other phases or are stable only in a narrow range of experimental conditions requires significantly accurate theory because subtle contributions to the free energy resulting from minor effects can decide their stability. For instance, the phonon entropy component of free energy has been found to contribute significantly to the phase stability of BN1, TiO22, and hydrogen at high pressures3. Furthermore, all these cases have required highly accurate quantum Monte Carlo (QMC) calculations to determine the internal energy. The influence of configurational entropy to free energy has been studied in less detail. One case in which configurational entropy could play a relevant role is the magnetic topological insulator MnBi2Te4 (MBT).
Magnetic topological insulators (TIs) have emerged as promising materials platforms for realizing a variety of exotic quantum phenomena, including the quantum anomalous Hall effect (QAHE)4,5, axion insulator states6, Weyl semimetal behavior7,8, and even potential realizations of Majorana fermions9. These phenomena originate from the interplay between nontrivial band topology and intrinsic magnetic order, which breaks time-reversal symmetry. As a consequence, magnetic TIs exhibit robust edge states and dissipationless transport that render them highly relevant for both fundamental studies and emerging quantum technologies. These characteristics position magnetic TIs as promising candidates for applications spanning quantum computing10,11, low-power spintronics12,13, and magneto-, opto-14, and thermoelectric15 devices.
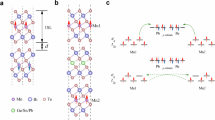
Among various magnetic TIs, MBT has drawn considerable attention as the first intrinsic A-type antiferromagnetic (AFM) topological insulator16,17. MBT crystallizes in a trigonal \(R\bar{3}m\) space group, adopting a septuple-layer (SL) stacking (Te-Bi-Te-Mn-Te-Bi-Te) separated by van der Waals gaps. Below its Néel temperature of ~25 K18,19, MBT exhibits A-type AFM order. In its AFM configuration, Mn moments are ferromagnetically aligned within each SL and antiferromagnetically coupled across adjacent layers. This unique magnetic and structural architecture makes MBT a model system for exploring the delicate interplay between magnetism and band topology.
Despite its promise, a persistent challenge in MBT lies in the prevalence of native defects, particularly Mn–Bi antisite defects20,21,22,23,24. Consequently, defect-free MBT crystals—which are essential for unambiguous characterization of the intrinsic properties of the material—are difficult to obtain. These defects disrupt the topological character of MBT by breaking long-range AFM order. Furthermore, such defects can alter the Dirac surface states by shifting the Fermi level out of the Dirac point (DP) gap to the conduction band. This shift may hinder the emergence of topological surface states18,22,23,24,25,26,27,28,29. Bulk conductivity increases with the concentration of intrinsic defects, undermining the insulating bulk and metallic surface state, which is crucial for topological properties20,30.
Scanning tunneling microscopy and spectroscopy have consistently revealed high concentrations of MnBi (Mn at Bi sites) antisites. These antisites show a strong correlation with the density of states near the Fermi level20. Low-temperature scanning tunneling spectroscopy and μ-laser ARPES measurements have shown that cation intermixing significantly modulates the DP gap22. The observations also revealed that short-range interplane magnetic order persists in MBT up to ~60 K, directly affecting the DP gap. Beyond 60 K, the MBT system transitions into a uncorrelated paramagnetic state19,31. Experimental reports estimate BiMn (Bi at Mn lattice site) antisite concentrations ranging from 4% to 40%20,22,32,33,34,35,36. In addition, BiTe defects have also been observed, though less frequently, and reported to have minimal effect on the band structure20. Furthermore, the Mn–Bi antisite defects play a key role in magnetic phase transitions from ferromagnetic to ferrimagnetic order, driven by Mn migration22,37,38. Magnetic disorder introduced by such defects increases electron scattering, undermining the robustness of edge states required for the QAHE. Therefore, suppressing Mn–Bi antisites is essential to reduce scattering, stabilize the magnetic configuration, and ensure the reproducibility of topological behavior in MBT39. This result clearly underscores the urgent need to mitigate these defects for achieving structural quality akin to its parent binary compound Bi2Te3.
To date, synthesizing Mn–Bi antisite-free MBT remains experimentally challenging. The antisite defect concentration in MBT is highly sensitive to synthesis conditions, particularly the growth temperature22,31,32,40. Notably, Mn–Bi intermixing remains prominent even in carefully annealed samples20,34,35,41.
By contrast, theoretical studies based on density functional theory (DFT) and high-accuracy diffusion Monte Carlo (DMC) predict Mn–Bi antisite pairs to be thermodynamically unfavorable at 0 K. These calculations yield positive formation energies: 0.28 eV from DMC and between 0.32 and 0.42 eV from DFT21,33,39. The discrepancy between theory and experiment suggests investigating the critical limitations of conventional ab initio methods, which typically neglect defect–defect interactions and finite-temperature effects. This need motivates going beyond zero temperature calculations of isolated defects. We believe that advancing beyond simplified approaches—such as DFT ground-state calculations at 0 K using idealized MBT crystals—is essential for improving theoretical accuracy. Incorporating finite-temperature effects and realistic defect configurations is key to a more reliable description.
Accurate characterization of defect thermodynamics in MBT, however, requires models that incorporate temperature-dependent configurational entropy and include accurate defect interaction energies. Although traditional ab initio techniques are well-suited for investigating the crystals at 0 K, they fall short of capturing the finite-temperature behavior of defects. We believe that configurational entropy could be important for stabilizing Mn–Bi antisite disorder in MBT, especially near the synthesis temperature (~850 K34,38,42,43). Defect energetics alone cannot fully explain the experimentally observed antisite concentrations. By calculating the configurational entropy, we aim to capture its contribution to free energy and better understand the thermodynamic origin of defect formation. The configurational entropy is obtained via the Boltzmann’s entropy formula, \({S}_{{\rm{config}}}={k}_{B}ln\Omega\) for idealized or non-interacting systems. Here, kB is the Boltzmann’s constant, and Ω denotes the number of distinct atomic configurations. This approach makes several oversimplifying assumptions. First, it treats defect configurations as randomly distributed and uncorrelated. Moreover, it ignores the local interactions and clustering effects that could be relevant in real materials. Furthermore, improving upon DFT calculations requires an approach that can adequately account for many-body effects, such as QMC44,45. However, direct use of these approaches is often not cost-effective because individual QMC calculations require significant computational resources.
In this work, we investigate the finite-temperature thermodynamic behavior of defects in MBT using a first-principles many-body-informed cluster expansion (CE) method46,47,48 combined with advanced sampling techniques. By employing both Metropolis Monte Carlo (MMC)49 and Wang–Landau (WL)50 algorithms, we capture configurational statistics across a wide range of temperature, enabling direct evaluation of temperature-dependent defect concentrations. Unlike conventional approaches that neglect defect–defect interactions, our CE framework incorporates up to three-body terms, revealing the complex interplay governing Mn–Bi antisite formation. Furthermore, we introduce a novel residual correction scheme that systematically refines the CE model by embedding many-body electronic effects, thus enhancing its final accuracy. Our results highlight the dominant role of defect correlations at finite temperatures. We provide a lower bound value (at thermodynamic equilibrium) of defect concentration in MBT using an accurate description of the defect formation and interactions energies.
The rest of this paper is organized as follows. In the Results section, we begin by demonstrating how many-body defect–defect interactions critically influence configurational heat capacity and antisite concentrations. Building on this knowledge, we assess the role of electronic correlations via QMC corrections, quantify the configurational entropy contribution to free energy, and analyze direct defect–defect interactions to establish the thermodynamic origins of antisite formation in MBT. The Discussion section includes the key findings of the study. Finally, the Methods section describes the overall methodological approach and provides technical details to ensure reproducibility.
Results
Heat capacity and many-body defect–defect interactions
We study the configurational heat capacity of MBT under Mn–Bi exchange using models of increasing complexity to highlight the role of higher-order interactions. This analysis also provides insight into the necessity of employing CE models and the QMC-based residual correction scheme. Unless otherwise specified, all calculations are performed under fixed MBT stoichiometry within the canonical ensemble.
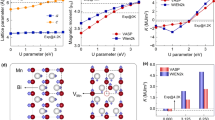
We begin by noting that the excellent agreement between the analytical solution (using only the noninteracting one-body formation energy) and the numerical simulation using the CEPBE+U [1B] model (as detailed in the SI section “Analytical limit”) validates our sampling algorithms, allowing us to proceed with higher-order models. Figure 1a presents the temperature-dependent configurational heat capacity of MBT. These calculations were performed on a 32–formula unit supercell with a cell shape (\(1\times 2\sqrt{14}\times 2\sqrt{14}\)) using MMC and WL sampling algorithms within the canonical ensemble using CE models. The primary reason for using both algorithms was to validate our sampling procedure through two independent methods, which show excellent consistency, as demonstrated in the Supplementary Fig. S5. The models include interactions up to 1B, 2B, and 3B terms. The figure contains five distinct curves. The dotted green curve corresponds to the analytical solution for the 1B term. The continuous blue curve on top is obtained from the sampling algorithm using the CEPBE+U [1B] model. Both of these curves represent a simplified model without any interatomic interactions and are superimposed on each other. Their agreement confirms the validity and accuracy of our sampling implementation. The solid green curve corresponds to a CEPBE+U [2B] model that includes up to 2B interactions. Importantly, this model exhibits a second-order configurational order–disorder phase transition. The ordered state corresponds to the pristine MBT structure, where Mn and Bi atoms occupy their ideal crystallographic sites in the \(R\bar{3}m\) space group without any antisite disorder. Both PBE+U and QMC calculations confirm that pristine MBT is the lowest-energy configuration (Fig. 2) at 0 K, exhibiting a positive formation energy for Mn–Bi antisite pair creation. This indicates that additional energy is necessary to drive the system from the ordered to the disordered phase. The transition to the disordered state is entropy driven at elevated temperatures. This transition deviates from the black curve, which corresponds to the CEPBE+U [3B] model, including up to 3B interactions. At lower temperatures, approximately up to 600 K, both the [2B] and [3B] models yield nearly identical values of Cv. Beyond this point, their behavior diverges: between ~600 K and the synthesis temperature, the [3B] model predicts a higher Cv than the [2B] model, but at higher temperatures, this trend reverses, and the [3B] model shows a lower Cv. These deviations highlight that the omission of three-body interaction energies in the [2B] model can lead to significant discrepancies in thermodynamic predictions, demonstrating the crucial role of many-body defect–defect interactions in determining the thermodynamic behavior of MBT. Although higher-order interactions can in principle be included, we found that extending the CE model to include up to four-body (4B) interactions does not lead to any substantial difference compared with the 3B model, as shown in Supplementary Fig. S4.
a Configurational heat capacity of MBT in the canonical ensemble as a function of temperature, computed using Metropolis sampling with CE models incorporating up to 1B, 2B, and 3B interactions. b Corresponding BiMn antisite concentrations predicted by each model. The synthesis temperature of MBT is indicated for reference.
Comparison of formation energies computed using PBE+U and QMC for a set of MnBi2Te4 configurations (e.g., MBT1, MBT2) containing different numbers of Mn–Bi antisite pairs and geometrically distinct arrangements. The number of antisite pairs in each configuration is indicated in parentheses.
The red curve in Fig. 1a corresponds to the \({{\rm{CE}}}_{Q}^{corr}\) model. It displays a bump around 300 K corresponding to order–disorder phase transition, which reaches a plateau beyond 500 K. Including QMC corrections yields a noticeable shift of the heat capacity curve toward lower temperatures, reflecting a reduced energy barrier for Mn–Bi antisite formation. This shift indicates that antisite pairs begin to form at lower temperatures. This shift toward lower temperatures with the \({{\rm{CE}}}_{Q}^{corr}\) model in Fig. 1a is consistent with Supplementary Fig. S5, where QMC predicts significantly lower defect formation energies than PBE+U for configurations with fewer antisites, but the trend reverses when more antisites are present. Therefore, with increasing temperature, more antisite pairs form with higher energy cost under the \({{\rm{CE}}}_{Q}^{corr}\) model, which explains the lower Cv compared to the PBE+U model.
At very high temperatures, the configurational heat capacity values from different models begin to converge because thermal energy becomes dominant. As a result, the system is driven into a highly disordered state. In this regime, the inclusion of higher-order interactions has a diminished effect on the specific heat. However, near the synthesis temperature—the thermal energy is relatively low compared to the defect–defect interaction energies in MBT. Only a limited set of low-energy configurations are accessible at this temperature. Therefore, many-body defect–defect interactions play a significant role in shaping the energy landscape because their energies are comparable to thermal energy.
As shown in Fig. 1a and listed in Table 1, the configurational heat capacity at low temperatures varies markedly across different models: simple noninteracting, CEPBE+U models with increasing interaction order (1B, 2B, and 3B), and the \({{\rm{CE}}}_{Q}^{corr}\) model. Deviations in the DMC-based models arise from differences in the treatment of electronic correlations, which influence the low-energy excitations that dominate the thermodynamics in this regime. This analysis underscores the importance of incorporating both quantum electronic corrections (via QMC) and many-body ionic interactions (via CE) to accurately capture Mn–Bi antisite formation in MBT.
Importance of electronic correlations
To understand the importance of many-body electronic correlations from QMC in MBT, we first compared the formation energies of several antisite-containing crystals using both QMC and PBE+U methods. Figure 2 presents a comparison of formation energies for several MBT configurations containing different numbers of Mn–Bi antisite pairs, computed using both PBE+U and QMC methods. The dataset includes one pristine MBT crystal (no antisites), two configurations with a single antisite pair (corresponding to the first- and second-nearest neighbors), two with two antisite pairs (corresponding to different combinations of the nearest neighbors), and one with four antisite pairs (all four Mn atoms were swapped). These specific configurations were selected to sample diverse local Mn–Bi swap environments within an MBT cell with four formula units, demonstrating the need for many-body electronic corrections. Although both methods capture general trends, the predicted formation energies differ notably between them. For instance, in the single antisite cases, QMC yields nearly identical formation energies of ~0.28 eV for both configurations. By contrast, PBE+U produces significantly different values, with one nearly twice as large as the other. This result highlights a key distinction: the discrepancy between PBE+U and QMC is not systematic and varies across different configurations. Furthermore, the formation energy does not scale linearly with the number of antisites, indicating that a simple additive model or one-body energy model is insufficient. These observations underscore the importance of many-body electronic corrections for modeling of defect energetics in MBT.
Antisite concentrations at thermodynamic equilibrium
An important result of our work is the prediction of the concentration of antisites at equilibrium at synthesis temperatures.
Figure 1b displays the BiMn antisite concentration as a function of temperature using different models. The calculations were performed in the canonical ensemble while preserving the stoichiometry of MBT. As a result, defect formation is limited to temperature-dependent native Mn–Bi antisite pairs. Due to the stoichiometry of MBT, the concentration of MnBi is expected to be half that of BiMn.
The number and energy of antisite pairs can differ between configurations, and the chance that a configuration is accepted during Metropolis sampling is determined by the Boltzmann probability: \(p(x)\propto \exp \left(-\beta \Delta E\right),\,\beta =\frac{1}{{k}_{B}T}\). Here, ΔE is the energy difference between the new (proposed) configuration and the current one. This probability means that lower-energy configurations are more likely to be accepted, especially at lower temperatures. This framework helps us understand the temperature at which the first antisite defects begin to appear. In MBT, second-nearest-neighbor Mn–Bi antisite pairs, which possess the lowest formation energy, tend to form first39. Once an initial antisite is created, the energy cost for forming additional antisites is reduced, thereby facilitating further defect generation as temperature increases (Supplementary Fig. S5).
In the simple, non-interacting analytical solution (dashed green curve) in Fig. 1b, the first antisite pair forms around 400 K, and the defect concentration increases linearly in BiMn concentration thereafter. This result can be explained from the following expression for analytical calculation of the antisite pairs: \(\langle n\rangle =\frac{1}{Z(T)}{\sum }_{n}n\cdot {g}_{n}\cdot {{\rm{e}}}^{-{E}_{n}/({k}_{B}T)}\). Once a few antisite pairs become thermally accessible, the system starts favoring configurations with more antisites as the temperature increases because the energy cost increases linearly with the number of swaps (nΔE), but the degeneracy of the configurations increases much faster. As a result, the average number of swaps rises steadily with temperature, appearing almost linear over a broad range.
When 2B and 3B interactions are included (as in the CEPBE+U model, shown by the black curve), the energy landscape becomes more complex. In this case, although antisite defects begin to form near 400 K, the concentration curve exhibits a nonlinear trend with distinct features. This trend suggests that higher-order interactions more accurately capture atomic correlations. As the temperature exceeds 400 K, a broader range of low-energy defect configurations becomes accessible, resulting in a rapid increase in antisite formation around 600 K. However, as the temperature approaches the synthesis point, most favorable configurations have already been explored, causing the defect concentration to increase slowly thereafter.
The \({{\rm{CE}}}_{Q}^{corr}\) model displays a similar qualitative trend, but the onset of antisite formation is noticeably shifted to lower temperatures. The initial antisites appear around 200 K, followed by a steep increase, reaching over 20% BiMn concentration by 300 K. Interestingly, around 400 K, the BiMn concentration predicted by the \({{\rm{CE}}}_{Q}^{corr}\) model begins to increase more slowly compared to the CEPBE+U model. From the synthesis temperature onward, although the defect concentration predicted by the \({{\rm{CE}}}_{Q}^{corr}\) model increases gradually, but it remains consistently lower than that predicted by the CEPBE+U model. These observations can be explained by examining Supplementary Fig. S5 in SI. The \({{\rm{CE}}}_{Q}^{corr}\) model predicts lower defect formation energies compared to the CEPBE+U model at low defect concentrations. However, this trend appears to reverse at higher defect densities. At elevated temperatures, the \({{\rm{CE}}}_{Q}^{corr}\) model predicts relatively higher incremental formation energies for creating additional defects. This result indicates that forming extra antisite pairs becomes less favorable as defect density increases. As a result, the overall defect concentration predicted by the \({{\rm{CE}}}_{Q}^{corr}\) model remains lower at high temperatures. This behavior reflects the increasing energetic cost of antisite formation in the high-defect regime under the \({{\rm{CE}}}_{Q}^{corr}\) model.
Limitations of simplified models
As demonstrated by the defect concentration and heat capacity trends in Fig. 1, conventional theoretical models, excluding defect–defect interactions, fail to capture key thermodynamic features of defects in MBT. Correcting only for configurational entropy using statistical microstate counting—such as Boltzmann’s entropy formula—is not sufficient to capture the full complexity of the system. The differences between models arise not only from entropy estimation but also from how energy landscapes and correlated defect energetics are represented. A deeper understanding of these variations is essential for accurate modeling of the behavior of antisite defects in MBT during synthesis. It is also crucial for reliable prediction of the material’s thermodynamic stability and phase transitions. Furthermore, it demonstrates that quantitatively reliable predictions of Mn–Bi antisite concentrations in MBT require the inclusion of many-body interaction effects and quantum-level corrections to defect energetics. This requirement highlights the inadequacy of simplified or purely entropic models in capturing the correlated disorder present in solids.
Configurational entropy contribution to free energy
Having established the significance of many-body corrections from QMC and higher-order interactions for accurate prediction of Mn–Bi antisite concentrations, we now turn our attention to the thermodynamic stability of MBT at finite temperatures. In particular, we examine how configurational entropy contributes to the system’s free energy landscape. This analysis offers deeper insight into the role of disorder and defect interactions in shaping the temperature-dependent behavior of MBT. This subsection presents the computed free energy profiles derived from statistical sampling over configurational micro-states.
Figure 3 presents the evolution of free energy as a function of temperature, computed using different energy models under MBT stoichiometry. The corresponding defect internal energy is shown in the inset. The free energy calculations include only the defect formation energy and the configurational entropy as defined by Supplementary Eq. (S6) in the SI, and all relative values are referenced to their respective zero-kelvin energies. The inset shows that the defect formation internal energy increases with temperature, while the main panel illustrates how this increase is progressively offset by the negative contribution from configurational entropy. This entropic stabilization effectively lowers the free energy, showing that configurational disorder can stabilize the system at elevated temperatures, despite the ~280 meV (in QMC) formation cost of a single Mn-Bi antisite pair at 0 K. In this plot, the gray solid curve represents the CEPBE+U [1B] sampling results using the MMC sampling method described in the Antisite concentrations at thermodynamic equilibrium section, and the dotted curve corresponds to the analytical calculation from the simple model based on the partition function described in Supplementary Eq. (S4) in the SI. The excellent agreement between the two approaches once again validates the application of the sampling algorithms (MMC and WL). Three more curves are shown, corresponding to sampling results from CE models with increasing interaction complexity: the PBE+U model, including up to 3B interactions (blue curve), and the \({{\rm{CE}}}_{Q}^{corr}\) model (red curve). For the simple 1B model without any interactions, the free energy and defect concentration remain constant at their baseline values up to ~400 K. Around this temperature, the initial Mn–Bi antisites appear, resulting in an increase in internal energy. However, as shown in Fig. 1b, the onset of antisite formation moves to progressively lower temperatures when higher-order interactions (up to 3B terms), and many-body electronic correlation (QMC correction) are incorporated into the model. Importantly, we observe a systematic downward shift in free energy as the model complexity improves: starting from the CEPBE+U [1B] model, moving to the CEPBE+U [3B] model and the corrected \({{\rm{CE}}}_{Q}^{corr}\) [3B] models. This trend indicates that the contribution of configurational entropy increases systematically with the inclusion of higher-order interactions. For example, the relative free energy contributions at the synthesis temperature range from ~2 to 4 meV/atom across the CEPBE+U [1B], CEPBE+U [3B], and \({{\rm{CE}}}_{Q}^{corr}\) [3B] models, with the \({{\rm{CE}}}_{Q}^{corr}\) model exhibiting the largest entropic contribution. This trend highlights the critical role of higher-order interactions in capturing the true thermodynamics of MBT at the synthesis temperatures. Furthermore, it is evident that the inclusion of configurational entropy significantly offsets the positive defect formation internal energy, leading to an overall negative free energy at high temperatures. The configurational heat capacity and defect concentration trends shown in Fig. 1 are consistent with this free energy behavior. More importantly, the emergence of negative free energy—even within the CEPBE+U [1B] model, albeit less pronounced than in the CEPBE+U [3B] model—relative to pristine MBT at synthesis temperatures strongly suggests that Mn–Bi antisite formation is thermodynamically spontaneous. This trend aligns well with experimental observations reporting the persistent difficulty in synthesizing antisite-free MBT samples20,22,32,33,34,35,36.
The inset shows the corresponding relative defect internal energy. All relative quantities are computed with respect to the bulk contribution to highlight variations between the models.
The significant configurational free energy contribution of ~4 meV/atom at synthesis temperature with the \({{\rm{CE}}}_{Q}^{corr}\) model could stabilize MBT against competitive phases.
Many-body defect–defect interactions
The Mn–Bi antisite pair formation energies and the analytical models based on these energies can help us understand defect formations at the non-interacting limit. However, Figs. 1 and 3 show that defect–defect interactions significantly contribute to the heat capacity, defect concentrations, and free energy. Therefore, we investigate defect–defect interactions in Fig. 4, using the CEPBE+U [3B] model to understand the role of higher-order interactions. To isolate these interactions, we compute the bare interaction energy by removing the interpolated energy (ϵD as defined in Eq. (6)) and individual defect contributions, defined as follows:
Here, Ed is the total energy of the defective MBT configuration. The terms NMn and NBi are the numbers of MnBi and BiMn antisite defects, respectively. The terms \({E}_{{{\rm{Mn}}}_{{\rm{Bi}}}}\) and \({E}_{{{\rm{Bi}}}_{{\rm{Mn}}}}\) represent the formation energies of isolated Mn atoms substituted at Bi sites and Bi atoms substituted at Mn sites, respectively, computed using CEPBE+U [3B] model. The horizontal dashed line in Fig. 4 effectively represents the reference baseline for the bare defect–defect interaction energy, excluding all contributions from point defect formation energies. Here, EInt denotes the bare interaction energy, rather than the raw CE effective cluster interactions parameters.
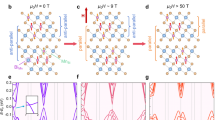
Two-antisite interaction configurations: a MnBi–MnBi, b MnBi–BiMn, c BiMn–BiMn. Three-antisite interaction configurations: d MnBi–MnBi–MnBi, e MnBi–MnBi–BiMn, f MnBi–BiMn–BiMn, g BiMn–BiMn–BiMn. The horizontal dashed lines represent the reference baseline constructed by subtracting the interpolated energy and the formation energy of isolated antisites. These lines indicate where data points would lie if higher-order defect–defect interactions were negligible.
We use sufficiently large supercells (32 formula units, corresponding to a more isotropic expansion of the primitive cell, expressed as \((1\times 2\sqrt{14}\times 2\sqrt{14})\)) to eliminate image interactions between defects and investigate symmetrically distinct defect configurations. To this end, we analyze how Mn–Bi antisites interact under various atomic environments by generating thousands of random antisite configurations within MBT crystals. These configurations are categorized into two groups based on the number of interacting antisites. The two-antisite interaction configurations are MnBi–MnBi, MnBi–BiMn, and BiMn–BiMn. The three-antisite interaction configurations are MnBi–MnBi–MnBi, MnBi–MnBi–BiMn, MnBi–BiMn–BiMn, and BiMn–BiMn–BiMn. The analyzed MBT structures may be stoichiometric, Bi-rich, or Mn-rich, depending on the specific arrangement and number of antisite defects.
Figure 4a–c, d–g shows the interaction energies plotted against two different geometric descriptors: (1) defect–defect distance and (2) the area of the triangle formed by three antisite points. The plots (a), (d), and (e) in Fig. 4 correspond to Mn-rich cases, whereas (c), (f), and (g) represent Bi-rich environments. In general, defect interactions are stronger in Mn-rich cases, and such motifs occur more frequently compared to those in Bi-rich cases. This result suggests that Bi-rich configurations are energetically more favorable, corroborating the experimental observation of Bi-rich MBT samples during synthesis32.
The plot in Fig. 4b corresponds to the stoichiometric case. It shows the interaction between a MnBi and a BiMn antisite pair. Notably, this pair exhibits an attractive interaction energy when separated by 6.1 Å, corresponding to the second-nearest-neighbor distance. As a result, Mn–Bi antisites forming at this separation are energetically favorable. Capturing this interaction requires at least 2B terms in the CE model. This requirement is further supported by the results in Fig. 1b, where the 1B model fails to reproduce the defect concentration trends obtained from models including up to 3B interactions. The sharp increase in antisite concentration with the CEPBE+U[3B] model near 600 K can be attributed to this favorable interaction energy, which is captured only when higher-order interactions are included. Furthermore, the plot illustrates that a model limited to calculating only a single nearest-neighbor antisite pair significantly overestimates the formation energy, highlighting the importance of many-body effects.
Evidently, according to Eq. (1), if one-body energies alone were sufficient to model Mn–Bi antisite defects in MBT, all data points in Fig. 4 would lie on the horizontal dashed line. This would imply that higher-order defect–defect interactions are negligible. By contrast, the observed spread of data points with finite interaction energies clearly indicates the presence and importance of such interactions.
In several cases, the three-body interaction terms are negative, indicating that certain correlated defect configurations are energetically favored. This promotes defect formation at lower temperatures because the presence of one antisite pair facilitates the formation of an additional antisite in its vicinity.
Discussion
Antisite formation is thermodynamically spontaneous in MBT near the synthesis temperature because an order–disorder transition occurs within that temperature range. Our work highlights the critical role of higher-order interactions in capturing this behavior.
Our study provides a detailed thermodynamic framework for understanding Mn–Bi antisite defect formation in MBT, bridging the gap between base-level single-defect DFT+U methods and a high-accuracy many-body QMC approach. We use a CE model that is improved using a residual learning approach trained on QMC data. This method allows us to more reliably predict defect energies and concentrations in MBT compared with DFT+U. Our results show that both many-body ionic interactions and quantum many-body electronic effects must be considered together for accurate modeling.
We show that simplified models that neglect higher-order cluster interactions fail to capture the correlated disorder underlying antisite behavior, particularly near the synthesis temperature. The inclusion of higher-order defect interactions, such as two-body and three-body interaction terms, in a CE model leads to systematic improvements in predictive accuracy. These interactions improve the modeling of key thermodynamic quantities, including configurational heat capacity, antisite concentration, and the configurational entropy contribution to free energy. Using QMC corrections further improves accuracy. Notably, we show that a residual correction model could be sufficient to significantly reduce energy prediction errors with respect to more accurate methods.
Our free energy analysis reveals a substantial configurational free-energy contribution—up to about 4 meV/atom at synthesis temperature with a QMC-corrected CE model. This energy contribution facilitates the Mn–Bi antisite defect formation and underpins the experimentally observed difficulty in synthesizing antisite-free MBT. The emergence of antisite defects at progressively lower temperatures with increasing model accuracy further emphasizes the thermodynamically induced Mn–Bi antisite disorder in this system. Ultimately, our findings establish a generalizable methodology for including localized many-body quantum corrections into a CE model trained on DFT. We believe this approach to be a robust path forward for defect thermodynamics in other correlated topological materials as well.
Methods
General approach
To capture the temperature-dependent configurational landscape of MBT, we employ a CE model trained on high-fidelity energy data derived from DMC calculations and DFT calculations. A CE expresses the total energy as a sum over interactions among atomic clusters, providing an efficient and scalable framework to model complex configurational spaces. When coupled with Monte Carlo sampling, CE enables the exploration of equilibrium defect distributions, phase stability, and thermodynamic observables over a broad temperature range. By leveraging symmetry constraints and many-body defect–defect interactions, CE has become a cornerstone method in computational materials science51,52,53,54,55. We assume here that elastic interactions are well accounted for in DFT methods, whereas exchange-correlation errors have shorter range. Accordingly, we use a hybrid model trained on the residual difference between Perdew–Burke–Ernzerhof (PBE)+U and QMC energies that accounts for correlation errors at shorter distances. This residual model builds upon the PBE+U-based CE model (CEPBE+U) by including the missing correlation energy, improving the transferability and accuracy of predictions across diverse configurations. We assume that this hybrid strategy enables more accurate description of the configurational space, especially in systems like MBT where correlation and exchange errors are nontrivial due to the presence of the transition metal Mn.
A CE model is linear-fit model that solves for effective cluster interactions (ECIs), as defined in Eqs. (3) and (4). It is the decomposition of the energy of a system into contributions from recurring atomic motifs. Once trained on a representative set of configurations, a CE model extrapolates the total energy of the sampled data as the linear sum of the cluster’s ECIs weighted by their corresponding multiplicities. The accuracy of a CE model therefore hinges on the accuracy of the ECIs, which in turn depends on the reliability of the energy data used during training. To enhance this reliability, a more accurate method (e.g., QMC) can be leveraged to correct the ECIs derived from a base-level method (e.g., PBE+U). The short-range interactions are typically dominant in crystalline materials56,57. Applying many-body corrections only to motifs within short cutoff radii can substantially improve model performance without significantly increasing cost for the many-body training data. As presented in Supplementary Table S1, both PBE+U and SCAN+U predict that interactions between Mn–Bi antisite pairs at large separations are weak, supporting the validity of the dilute limit approximation. Additionally, our recent study39 showed that the elastic contribution from neutral antisite defects becomes negligible at larger distances. These results justify the use of corrections limited to short-range interactions.
We use statistical methods such as MMC, which samples configurations based on Boltzmann weights, and WL, which estimates the density of states via flat-histogram sampling independent of temperature. This method enables a more realistic estimation of entropy by preferentially sampling energetically favorable states, including defect–defect interactions.
In general, phonon contributions are neglected in the formation energies of defects and are not included in this work. The goal of this study is to estimate the contribution of the configurational entropy. Here, the contribution of the phonon entropy is assumed to be independent of the antisite concentration. This assumption is consistent with previous work58, which found phonon contributions to be on the order of ~10 meV/atom near the synthesis temperature for the Bi2Te3 + MnTe → MnBi2Te4 reaction. Given that vibrational effects from neutral antisite defects tend to decay rapidly with distance in layered chalcogenides59, the phonon contribution in the present case is expected to be even smaller. Consequently, our framework captures the essential thermodynamic physics by focusing on configurational entropy as the leading contribution governing defect stability in MBT.
The interplay between magnetic and configurational entropy may be the subject of further studies, but it is beyond the scope of this work. We believe that its impact will not change our results qualitatively. The interlayer AFM magnetic ordering has a Néel temperature (~25 K ≃ 2 meV)18,19 that is one order of magnitude lower than that of the interlayer defect couplings we found. The intralayer magnetic ordering persists up to ~60 K, above which the system transitions to a disordered paramagnetic state19,31.
Technical details
To generate training data for CE modeling, DFT calculations were performed using the Vienna Ab Initio Simulation Package (VASP)60,61 with the PBE functional62 and Hubbard63U parameter (PBE+U) to account for on-site Coulomb interactions. We used 133 DFT configurations to fit the PBE+U model. These configurations were selected using simulated annealing, as implemented in ICET64, from a pool of thousands of candidate structures with supercells containing 1–10 formula units of MBT primitive cell. The ICET-selected configurations represent a diverse set of Mn–Bi atomic arrangements, including non-stoichiometric compositions, and were chosen to capture a broad range of relevant cluster environments. For validation of the PBE+U model, a 10-fold cross-validation technique was used on supercells similar to those in the training data, as discussed in the SI section “CE order analysis.” However, for residual model validation—ensuring the model accuracy and robustness of the cluster expansion—supercells with 16 formula units were randomly generated in the form (MnxBi3−xTe4)16, with x ranging from 0 to 1. Based on our previous calculations on MBT39, we used a kinetic cutoff of 350 eV, k-point sampling with ~1500 per atom, and Hubbard parameter U of 3.5 eV, which accurately captures the defect formation energies and forces. The magnetic structure was initialized with a spin moment of 5.0 μB per Mn atom, corresponding to a ferromagnetic configuration. Structural optimizations were conducted with forces converged to less than 0.01 eV/Å, and the energy convergence criterion was set to 10−5 eV. The van der Waals interactions were included using the Grimme’s DFT-D3 method with zero-damping function65.
We used ICET64, a python library for constructing the CE model and performing WL50 sampling. Our final goal was to construct a CE model with corrections derived from QMC. However, due to the demanding nature of QMC—as discussed in the Residual cluster expansion model subsection—even generating a single data point is computationally intensive. Therefore, we constructed a residual CE model trained on a small, carefully selected dataset. To validate the residual model, we used the strongly constrained and appropriately normed (SCAN)+U meta-Generalized Gradient Approximation functional as a proxy for the expensive QMC calculations. Although less computationally expensive than QMC, SCAN+U offers a more accurate description of weakly correlated materials compared with PBE+U. This step ensured that the energy corrections derived from the residual CE model were robust.
For the QMC calculations, the fixed-node DMC variant as implemented in the QMCPACK package66,67 was used in combination with the Nexus workflow automation package68. The trial wavefunctions for the DMC calculations were of the Slater–Jastrow type69,70, generated from spin-polarized DFT calculations performed using the Quantum Espresso package71. To produce the trial wavefunctions, we used DFT calculations with the PBE+U functional and a U value of 3.5 eV for the Mn atoms, consistent with prior studies39,72. The trial wavefunctions incorporated one-body (electron–ion) and two-body (electron–electron) correlations, optimized using the linear method. To reduce finite-size effects, all DMC calculations employed twist-averaged boundary conditions using 72 twists, corresponding to a 6 × 6 × 4 Monkhorst–Pack k-point grid. These calculations were performed on a supercell with four formula units constructed with an expansion of \((2\sqrt{2}\times 2\sqrt{2}\times 1)\). The correlation-consistent effective core potentials (ccECP)73,74 were employed for Mn, Bi, and Te atoms, ensuring computational efficiency (by replacing core electrons) while maintaining accuracy. To minimize the imaginary time step error in DMC projections, a time step of 0.005 Ha−1 was chosen. This time step has been found to yield accurate antisite defect pair formation energies in MBT39, and accurate magnetic ordering72. Time step errors depend on the pseudopotentials; we used the same pseudopotentials as in studies by Saritas et al.39 and Ahn et al.72. Furthermore, time step errors largely cancel in total energy differences.
In the fixed-node DMC approach, the total energy is estimated by projecting the ground-state component of a trial wavefunction ΨT(R) through imaginary time evolution while enforcing a nodal constraint. The projected energy is evaluated using a mixed estimator as an integral over the 3N-dimensional configuration space of all electron coordinates,
where f(R) ∝ ΨT(R)Ψ0(R) is the mixed distribution sampled by the DMC algorithm, and EL(R) is termed the local energy, defined as \({E}_{L}({\bf{R}})=\frac{\widehat{H}{\Psi }_{T}({\bf{R}})}{{\Psi }_{T}({\bf{R}})}\). Equation (2) requires a large number of Monte Carlo samples for statistical convergence. To achieve a statistical error of ϵ, the number of samples must scale as M ~ 1/ϵ2, significantly increasing the computational cost for higher accuracy. Because the square of variance of the statistical noise increases linearly with the number of electrons in the system, the statistics required for the same error bar also increase, which limits the practical size of the systems that can be studied in DMC.
Cluster expansion formalism
The CE formalism provides an approximated method to express the configurational energy of a crystalline solid as a function of the occupation variables of its lattice sites. This approach is especially effective for systems with compositional disorder, such as alloys or defect structures. For a configuration σ, where each lattice site is occupied by a specific atomic species or defect type, where σi are the site occupation variables, and the total energy E(σ) can be expanded as
where J0 is the reference energy of a chosen configuration (often the empty lattice or host material), Ji, Jij, Jijk, … are the ECI coefficients corresponding to point, pair, triplet, and higher-order interactions. The occupation variables σi can take values of −1 or +1 for Ising-type systems, or they can be generalized for multicomponent systems. Equation (3) can be rewritten using a compact and symmetry-aware formalism:
where α indexes the symmetrically distinct clusters (e.g., point, pair, triplet), mα is the multiplicity of cluster α, Jα is the corresponding ECI, and 〈Πα(σ)〉 is the averaged cluster correlation function for configuration σ. Once trained on a finite set of first-principles data, the CE model enables fast approximation to configurational energies across a large combinatorial space, making it ideal for Monte Carlo simulations of finite-temperature thermodynamic properties.
Residual cluster expansion model
The construction of the residual model for many-body corrections begins by expressing the total energy of each training configuration in terms of its interaction energy, obtained by subtracting the interpolated energies. The difference in interaction energy between the QMC (subscript Q) and the DFT (subscript D) is defined as follows:
Here and throughout this work, we use the superscript R to denote residual parameters. The interpolated energies (ϵ) account for the passive role of Te atoms in the CE modeling and is defined as follows:
where NMn, NBi, and nfu are the number of Mn atoms, Bi atoms, and formula units in the given MBT crystal, respectively. The ED/Q(MBT) and ED/Q(BBT) terms denote the energies per formula unit of the pristine MBT and a hypothetical structure where all Mn atoms are replaced by Bi atoms (BiBi2Te4), respectively, calculated with DFT (D) or QMC (Q).
To construct a residual correction model, we begin with a reduced CE, as defined in Eq. (4), but restricted to a smaller set of clusters—up to index \(\alpha {\prime}\), where \(\alpha {\prime} \subset \alpha\) and \({r}_{\alpha {\prime} } < {r}_{\alpha }\). This truncated CE model can be written as follows:
Here, ΔER(σ) corresponds to the residual energy defined in Eq. (5), and the superscript R is used to distinguish the residual model from the full PBE+U-based CE model (CEPBE+U) including 3B correction. Because both Eqs. (4) and (7) are linear in form, they can be combined cluster-wise to produce a corrected model (\({E}_{Q}^{corr}{(\sigma )}_{\alpha }\)):
For further details on the construction of the residual correction scheme, a comprehensive description is provided in the Supporting Information (SI) section, “Residual correction.” Fig. 5 provides a schematic illustration of the interaction ranges captured by the CEPBE+U model and the residual model. The interaction range of CEPBE+U is defined by the cutoff radius r, extending from the center to the larger circle, and the residual model is defined by a smaller cutoff radius \(r{\prime}\), extending to the inner circle. The cutoff radius r for the CEPBE+U model was 10 Å for pairs and 8 Å for triplets, and a smaller cutoff \(r{\prime} =6.5\) Å (for pairs only) was used for the residual correction. These cutoffs are sufficient to capture both intra- and interseptuple layer interactions because the minimum interseptuple layer distance at the active sites (Mn, Bi) is 6.2 Å. The selected cutoff radii for CEPBE+U were based on a convergence test, as presented in the Supplementary Fig. S7. All clusters within these cutoffs were included, totaling 52 for CEPBE+U and 8 for the residual model. The CEPBE+U model includes 1 zeroth-order, 2 one-body, 17 two-body, and 32 three-body clusters, and the residual correction model uses 1 zeroth-order, 2 one-body, and 5 two-body clusters.
The full PBE-based (cyan) cluster model and residual model (red) with their cutoff radius projected on the MBT crystal. The reddish disc represents the range up to which QMC electronic many-body correction is applied.
In principle, solving the corresponding set of linear equations (Eq. (4)) requires finding at least as many independent reference configurations as clusters. Finding eight structures with linearly independent cluster vectors is readily possible for training the residual model. However, for the CEPBE+U model, it is challenging to find 52 structures with 52 linearly independent cluster vectors. To navigate this issue, we used simulated structure annealing on several thousands of random MnxBi3−xTe4 (x = [0, 1]) structures with different cell sizes (1–10 formula units). In this way, we obtain an optimized set of 133 structures for the training by minimizing the condition number while keeping the number of structures large enough to generate a well-conditioned fit matrix.
The narrow bandgap of MBT (~150 meV28) constrains total energy shifts for charged defects: the Fermi-level shifts provide an upper bound for the Moss–Burstein75,76 correction. Thus only charge-neutral configurations are studied. Although the systems included in the present study are stoichiometric, local departures from stoichiometry occur due to disorder. Thus the training set must include non-stoichometric configurations. In fact, our CEPBE+U [3B] model accurately reproduces the energetics of stoichiometric, charge-neutral configurations, achieving an overall R2 of 0.96 for total formation energy and a root-mean-square error (RMSE) of 0.023 eV per antisite. The mean absolute error (MAE) of 0.019 eV per antisite is nearly an order of magnitude lower than the average formation energy of 0.181 eV per antisite (see Supplementary Fig. S1a, b), corresponding to a relative error of ~10.8% for predicting a single antisite. This evaluation highlights the model’s robust predictive capability and reliability in capturing defect energetics. It further suggests that global Fermi-level variations likely have a negligible influence on the model-predicted energies of the stoichiometric samples.
To test the residual correction scheme, we obtained SCAN+U-corrected model \({E}_{SCAN+U}^{corr}(\sigma )\) from EPBE+U(σ), as detailed in the SI section, “Validation of the residual model.” Our results show that even a small residual model, characterized by a shorter cutoff radius and fewer parameters, can be effective. Despite being trained on a relatively limited dataset, it significantly improves the accuracy of the underlying CE model. Motivated by the success of the SCAN-based validation, we constructed a QMC-based residual correction model (\({{\rm{CE}}}_{Q}^{corr}\)) using a similar approach. For this purpose, we employed the fixed-node DMC variant of QMC and applied it to the same set of training structures used for the SCAN-based model. Based on the rigorous validation with SCAN, we are confident in the transferability and reliability of the QMC-based residual corrections. Because the high computational cost of QMC precluded its use for full model validation, SCAN+U served as a robust and computationally feasible proxy to justify the QMC residual correction scheme.
Data availability
The data and analysis scripts in this study are openly available in the OLCF Constellation repository at https://doi.org/10.13139/OLCF/2574033.
References
Nikaido, Y. et al. Diffusion Monte Carlo study on relative stabilities of boron nitride polymorphs. J. Phys. Chem. C. 126, 6000–6007 (2022).
Luo, Y. et al. Phase stability of TiO2 polymorphs from diffusion quantum Monte Carlo. N. J. Phys. 18, 113049 (2016).
Ichibha, T., Zhang, Y., Hongo, K., Maezono, R. & Reboredo, F. A. Candidate structure for the H2-PRE phase of solid hydrogen. Phys. Rev. B 104, 214111 (2021).
Yu, R. et al. Quantized anomalous Hall effect in magnetic topological insulators. Science 329, 61–64 (2010).
Chang, C.-Z. et al. Experimental observation of the quantum anomalous Hall effect in a magnetic topological insulator. Science 340, 167–170 (2013).
Xiao, D. et al. Realization of the axion insulator state in quantum anomalous Hall sandwich heterostructures. Phys. Rev. Lett. 120, 056801 (2018).
Liu, E. et al. Giant anomalous Hall effect in a ferromagnetic kagome-lattice semimetal. Nat. Phys. 14, 1125–1131 (2018).
Wan, X., Turner, A. M., Vishwanath, A. & Savrasov, S. Y. Topological semimetal and fermi-arc surface states in the electronic structure of pyrochlore iridates. Phys. Rev. B 83, 205101 (2011).
He, Q. L. et al. Retracted: Chiral Majorana fermion modes in a quantum anomalous Hall insulator–superconductor structure. Science 357, 294–299 (2017).
Hasan, M. Z. & Kane, C. L. Colloquium: topological insulators. Rev. Mod. Phys. 82, 3045–3067 (2010).
He, M., Sun, H. & He, Q. L. Topological insulator: spintronics and quantum computations. Front. Phys. 14, 43401 (2019).
Zhang, Y. et al. Crossover of the three-dimensional topological insulator Bi2Se3 to the two-dimensional limit. Nat. Phys. 6, 584–588 (2010).
Lin, X., Yang, W., Wang, K. L. & Zhao, W. Two-dimensional spintronics for low-power electronics. Nat. Electron. 2, 274–283 (2019).
Tse, W.-K. & MacDonald, A. H. Giant magneto-optical Kerr effect and universal Faraday effect in thin-film topological insulators. Phys. Rev. Lett. 105, 057401 (2010).
Tian, W., Yu, W., Shi, J. & Wang, Y. The property, preparation and application of topological insulators: a review. Materials 10, 814 (2017).
Lee, S. H. et al. Spin scattering and noncollinear spin structure-induced intrinsic anomalous Hall effect in antiferromagnetic topological insulator MnBi2Te4. Phys. Rev. Res. 1, 012011(R) (2019).
Vidal, R. C. et al. Surface states and Rashba-type spin polarization in antiferromagnetic MnBi2Te4(0001). Phys. Re. B 100, 121104(R) (2019).
Chen, Y. et al. Topological electronic structure and its temperature evolution in antiferromagnetic topological insulator MnBi2Te4. Phys. Rev. X 9, 041040 (2019).
Otrokov, M. M. et al. Prediction and observation of an antiferromagnetic topological insulator. Nature 576, 416–422 (2019).
Huang, Z., Du, M.-H., Yan, J. & Wu, W. Native defects in antiferromagnetic topological insulator MnBi2Te4. Phys. Rev. Mater. 4, 121202(R) (2020).
Du, M., Yan, J., Cooper, V. R. & Eisenbach, M. Tuning Fermi levels in intrinsic antiferromagnetic topological insulators MnBi2Te4 and MnBi4Te7 by defect engineering and chemical doping. Adv. Functional Mater. 31, 2006516 (2020).
Garnica, M. et al. Native point defects and their implications for the Dirac point gap at MnBi2Te4(0001). npj Quantum Mater. 7, 7 (2022).
Tan, H. & Yan, B. Distinct magnetic gaps between antiferromagnetic and ferromagnetic orders driven by surface defects in the topological magnet MnBi2Te4. Phys. Rev. Lett. 130, 126702 (2023).
Lüpke, F. et al. Anti-site defect-induced disorder in compensated topological magnet MnBi2-xSbxTe4. Commun. Mater. 4, 82 (2023).
Liu, M. et al. Visualizing the interplay of Dirac mass gap and magnetism at nanoscale in intrinsic magnetic topological insulators. Proc. Natl Acad. Sci. 119, e2207681119 (2022).
Nevola, D. et al. Coexistence of surface ferromagnetism and a gapless topological state in MnBi2Te4. Phys. Rev. Lett. 125, 117205 (2020).
Swatek, P. et al. Gapless Dirac surface states in the antiferromagnetic topological insulator MnBi2Te4. Phys. Rev. B 101, 161109(R) (2020).
Hao, Y.-J. et al. Gapless surface Dirac cone in antiferromagnetic topological insulator MnBi2Te4. Phys. Rev. X 9, 041038 (2019).
Li, H. et al. Dirac surface states in intrinsic magnetic topological insulators EuSn2As2 and MnBi2nTe3n+1. Phys. Rev. X 9, 041039 (2019).
Hu, C., Qian, T. & Ni, N. Recent progress in MnBi2nTe3n+1 intrinsic magnetic topological insulators: crystal growth, magnetism and chemical disorder. Natl Sci. Rev. 11, nwad282 (2023).
Alfonsov, A. et al. Strongly anisotropic spin dynamics in magnetic topological insulators. Phys. Rev. B 103, L180403 (2021).
Yan, J.-Q. Perspective–the elusive quantum anomalous hall effect in MnBi2Te4: materials. ECS J. Solid State Sci. Technol. 11, 063007 (2022).
Hou, F. et al. Te-vacancy-induced surface collapse and reconstruction in antiferromagnetic topological insulator MnBi2Te4. ACS Nano 14, 11262–11272 (2020).
Yan, J.-Q. et al. Crystal growth and magnetic structure of MnBi2Te4. Phys. Rev. Mater. 3, 064202 (2019).
Yuan, Y. et al. Electronic states and magnetic response of MnBi2Te4 by scanning tunneling microscopy and spectroscopy. Nano Lett. 20, 3271–3277 (2020).
Liang, Z. et al. Mapping Dirac fermions in the intrinsic antiferromagnetic topological insulators (MnBi2Te4)(Bi2Te3)n (n = 0, 1). Phys. Rev. B 102, 161115(R) (2020).
Lai, Y., Ke, L., Yan, J., McDonald, R. D. & McQueeney, R. J. Defect-driven ferrimagnetism and hidden magnetization in MnBi2Te4. Phys. Rev. B 103, 184429 (2021).
Sahoo, M. et al. Ubiquitous order-disorder transition in the Mn antisite sublattice of the (MnBi2Te4)(Bi2Te3)n magnetic topological insulators. Adv. Sci. 11, 2402753 (2024).
Saritas, K. et al. Enhancing mnbi2te4 stability by doping. J. Phys. Chem. C. 128, 10108–10119 (2024).
Alfonsov, A. et al. Magnetic-field tuning of the spin dynamics in the magnetic topological insulators (MnBi2Te4)(Bi2Te3)n. Phys. Rev. B 104, 195139 (2021).
Hu, C. et al. Growth, characterization, and Chern insulator state in MnBi2Te4 via the chemical vapor transport method. Phys. Rev. Mater. 5, 124206 (2021).
Jiao, F. et al. The layer-inserting growth of antiferromagnetic topological insulator MnBi2Te4 based on symmetry and its X-ray photoelectron spectroscopy. J. Superconductivity Nov. Magn. 34, 1485–1493 (2021).
Li, Y. et al. Fabrication-induced even-odd discrepancy of magnetotransport in few-layer MnBi2Te4. Nat. Commun. 15, 3399 (2024).
Foulkes, W. M. C., Mitas, L., Needs, R. J. & Rajagopal, G. Quantum Monte Carlo simulations of solids. Rev. Mod. Phys. 73, 33–83 (2001).
Austin, B. M., Zubarev, D. Y. & Lester, W. A. Quantum Monte Carlo and related approaches. Chem. Rev. 112, 263–288 (2011).
Kikuchi, R. A theory of cooperative phenomena. Phys. Rev. 81, 988–1003 (1951).
Sanchez, J. M. & de Fontaine, D. The fee ising model in the cluster variation approximation. Phys. Rev. B 17, 2926–2936 (1978).
Sanchez, J., Ducastelle, F. & Gratias, D. Generalized cluster description of multicomponent systems. Phys. A Stat. Mech. Appl. 128, 334–350 (1984).
Metropolis, N., Rosenbluth, A. W., Rosenbluth, M. N., Teller, A. H. & Teller, E. Equation of state calculations by fast computing machines. J. Chem. Phys. 21, 1087–1092 (1953).
Wang, F. & Landau, D. P. Efficient, multiple-range random walk algorithm to calculate the density of states. Phys. Rev. Lett. 86, 2050–2053 (2001).
Van der Ven, A. & Ceder, G. Vacancies in ordered and disordered binary alloys treated with the cluster expansion. Phys. Rev. B 71, 054102 (2005).
Barroso-Luque, L. & Ceder, G. The cluster decomposition of the configurational energy of multicomponent alloys. npj Comput. Mater. 10, 158 (2024).
Lavrentiev, M. Y., Drautz, R., Nguyen-Manh, D., Klaver, T. P. C. & Dudarev, S. L. Monte Carlo study of thermodynamic properties and clustering in the bcc Fe-Cr system. Phys. Rev. B 75, 014208 (2007).
Hülsen, B., Scheffler, M. & Kratzer, P. Thermodynamics of the Heusler alloy Co2−xMn1+xSi: a combined density functional theory and cluster expansion study. Phys. Rev. B 79 (2009).
Kadkhodaei, S. & Muñoz, J. A. Cluster expansion of alloy theory: a review of historical development and modern innovations. JOM 73, 3326–3346 (2021).
Benedek, G. & Nardelli, G. F. Lattice response functions of imperfect crystals: effects due to a local change of mass and short-range interaction. Phys. Rev. 155, 1004–1019 (1967).
French, R. H. et al. Long range interactions in nanoscale science. Rev. Mod. Phys. 82, 1887–1944 (2010).
Ning, J. et al. Subtle metastability of the layered magnetic topological insulator MnBi2Te4 from weak interactions. npj Comput. Mater. 6, 157 (2020).
Tian, Y., Osterhoudt, G. B., Jia, S., Cava, R. J. & Burch, K. S. Local phonon mode in thermoelectric Bi2Te2Se from charge neutral antisites. Appl. Phys. Lett. 108, 041911 (2016).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Dudarev, S. L., Botton, G. A., Savrasov, S. Y., Humphreys, C. J. & Sutton, A. P. Electron-energy-loss spectra and the structural stability of nickel oxide:–An LSDA+U study. Phys. Rev. B 57, 1505–1509 (1998).
Angqvist, M. et al. ICET – a Python library for constructing and sampling alloy cluster expansions. Adv. Theory Simul. 2, 1900015 (2019).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Kim, J. et al. QMCPACK: an open source ab initio quantum Monte Carlo package for the electronic structure of atoms, molecules and solids. J. Phys. Condens. Matter 30, 195901 (2018).
Kent, P. R. C. et al. QMCPACK: advances in the development, efficiency, and application of auxiliary field and real-space variational and diffusion quantum Monte Carlo. J. Chem. Phys. 152, 174105 (2020).
Krogel, J. T. Nexus: a modular workflow management system for quantum simulation codes. Comput. Phys. Commun. 198, 154–168 (2016).
Slater, J. C. The theory of complex spectra. Phys. Rev. 34, 1293–1322 (1929).
Jastrow, R. Many-body problem with strong forces. Phys. Rev. 98, 1479–1484 (1955).
Giannozzi, P. et al. Quantum espresso: a modular and open-source software project for quantum simulations of materials. J. Phys. Condens. Matter 21, 395502 (2009).
Ahn, J. et al. Diffusion quantum Monte Carlo benchmarking of magnetic moments in MnBi2Te4. J. Phys. Chem. C 129, 7063–7072 (2025).
Annaberdiyev, A. et al. A new generation of effective core potentials from correlated calculations: 3d transition metal series. J. Chem. Phys. 149, 134108 (2018).
Bennett, M. C. et al. A new generation of effective core potentials for correlated calculations. J. Chem. Phys. 147, 224106 (2017).
Moss, T. S. The interpretation of the properties of indium antimonide. Proc. Phys. Soc. Sect. B 67, 775–782 (1954).
Burstein, E. Anomalous optical absorption limit in InSb. Phys. Rev. 93, 632–633 (1954).
Acknowledgements
This work was supported by the Materials Science and Engineering Division of the U.S. Department of Energy Office of Basic Energy Sciences. Computational resources were provided by the Oak Ridge Leadership Computing Facility at the Oak Ridge National Laboratory, supported by the Office of Science of the DOE under Contract no. DE-AC05-00OR22725.This manuscript has been authored by UT-Battelle, LLC, under contract DE-AC05-00OR22725 with the US Department of Energy (DOE). The US government retains and the publisher, by accepting the article for publication, acknowledges that the US government retains a nonexclusive, paid-up, irrevocable, worldwide license to publish or reproduce the published form of this manuscript, or allow others to do so, for US government purposes. DOE will provide public access to these results of federally sponsored research in accordance with the DOE Public Access Plan (https://www.energy.gov/doe-public-access-plan).We acknowledge Erica Heinrich for her editorial review.
Author information
Authors and Affiliations
Contributions
A.G. developed the computational methodology, performed all simulations and data analysis, generated all figures, contributed to scientific discussions, and wrote the initial draft of the manuscript. A.G. also led the manuscript revision process through multiple stages. K.S. contributed to scientific discussions, interpretation of the results, and participated in writing and editing the manuscript. F.R. conceived the original idea, supervised the research, developed key approximations used in the study, and contributed significantly to the manuscript writing. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ghaffar, A., Saritas, K. & Reboredo, F.A. The critical role of intrinsic defects and many-body interactions on the stability of MnBi2Te4. npj Comput Mater 12, 119 (2026). https://doi.org/10.1038/s41524-026-02019-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41524-026-02019-8