Abstract
Space travel could be accelerating ageing. This could be because life is being exposed to conditions too far outside its evolved Goldilocks zone. For optimal health, this suggests that humans may need to develop spacecraft that mimic Earth’s conditions exactly. In this perspective we suggest that origins of life theories, quantum biology and thermodynamics may provide insight into whether humans, and their companion organisms, are “trapped on Earth”.
Similar content being viewed by others
Introduction
Due to the large number of negative physiological changes that occur when humans go into space, it has been suggested that this could represent an accelerated ageing phenotype (AAP)1,2,3. This has been raised as a concern in the recently published “Thriving in Space: Ensuring the Future of Biological and Physical Sciences Research: A Decadal Survey for 2023-2032”, which is a consensus study report supported by contracts with NASA (the National Aeronautical and Space Administration)4. This report also raises questions about whether or not humans, or the species they may take with them, can even adapt to space, and whether biology may need help in order to do this, for instance, via some kind of genetic manipulation5.
It is becoming clear that “biological age” can be different from “chronological age”. As an organism ages, there is a gradual loss of physiological function and accumulation of damage, which enables a reasonable statistical prediction of mortality, with disease increasing with age. Based on this, an organism can have a biological age that can be higher or lower than their chronological age6. In short, once in space, is life pushed outside the limits of an evolved “adaptive metabolic envelope” (AME—akin to the well described flight envelope of aircraft), or Goldilocks zone, which results in chronic unresolving pathology, indicated as a biological age greater than their predicted chronological age?
Data suggest that excessive stress can result in an AAP, but if not too severe or long term, this can be reversed7. An example of a chronic stressor that leads to an AAP is a sedentary modern lifestyle8. In contrast, physical activity is an effective medicine leading to a longer and healthier life9 and is perhaps the best described inducer of an adaptive response as it triggers oxidative stress10, including in the brain11. Exercise is well known to be a cornerstone approach in trying to maintain health in space. It also highlights the point that zero physical activity is as bad as too much, although the latter is more difficult to achieve. In biology this biphasic effect is true for many “stressors”, describable by a “U” shaped curve, and is often called “hormesis”12. In short, not only can biology be stressed by too much of something, but also by its absence – and this could be key in space travel.
In this perspective, we embrace some less mainstream approaches that could help in understanding the origins of the space-induced AAP, such as thermodynamics, bioelectricity, magnetic fields, photobiomodulation (PBM, which is the practice of using light to treat various medical conditions) and hormesis. Key in this is that life is electrical, as it relies on the movement of charged particles. This suggests that quantum biology may provide helpful insight. For instance, although there are obvious boundaries of a human AME, for instance, temperature, oxygen, carbon dioxide, gravity, water, food, radiation, pressure, and altered circadian rhythms, it may also include light spectra and electromagnetic fields. With regards the latter, as redox and reactive oxygen species (ROS) are central to how life works13, the phrase from a Barnes and Greenebaum paper discussing the effects of weak magnetic fields on radical pairs in biology14, is particularly relevant:
“Finally, we suggest that observable magnetic field effects might generally happen under situations where an organism is stressed by other conditions that lead to changes in the radical concentrations so the combination of these stresses and the magnetic field changes take the radical concentrations outside the range of values where they are compensated for by radical scavengers. If this is so, it may explain why it is hard to get reproducible results, as the initial conditions in biological systems are hard to reproduce.”
In effect, it may well be that quantum spin is important in everyday redox15, suggesting that several environmental factors may affect it, and in combination, could push redox control beyond its limits to compensate, in other words, outside the AME. Given that mitochondria are central to eukaryotic cellular redox, the observation that they rapidly become dysfunctional in space16, suggests we should focus on them as the “metabolic canary in the coalmine”. So, a key question is will living a healthy lifestyle in space be enough to maintain optimal mitochondrial health, especially for longer trips, and if so, how do we achieve it?
The life electric and quantum biology
Szent-Györgyi reputedly once said; “life is nothing but an electron looking for a place to rest”17. The idea that “life is electric” has been recognised for 100s of years, although whether the phenomenon should be considered from the biological or the physics point of view was often debated, particularly by Volta and Galvani in the 18th Century. However, whichever angle one views it from, evidence continues to confirm its importance in life18,19, as it is based on the movement of fundamental charged particles and the fields they generate being pivotal in homoeostasis20.
That life is electrical of course underlies a long-standing debate about whether it has harnessed quantum effects beyond what might be predicted by the generally accepted limits of quantum mechanics, as biology has long been thought of as being too “warm and wet”. However, ever since quantum mechanics was developed, many have suggested that biology is “enhancing” quantum mechanical properties21,22,23. Indeed, quantum tunnelling of electrons in biological systems was championed by Britton Chance24, and it was thus suggested that it was important in the origins and evolution of life25, for instance, being selected for in electron transport chains (ETCs)26, or proton transfer in enzymatic reactions27,28.
Another property, quantum spin, is perhaps one of the easiest to describe in biological terms because of its relationship to magnetic fields and triplet born radical pairs and can potentially be predicted to occur from theory alone29. This is of course particularly relevant to space travel beyond Earth’s magnetic embrace, as due to the solar wind, the magnetosphere only extends to about ten Earth Radii on the dayside, but more than a 100 nightside. The Moon orbits at around 60 Earth radii. So, although the magnetic field is pivotal in protecting life from radiation and high energy particles, it has been suggested that a hypomagnetic field in space could have direct negative effects on metabolism30,31.
The Proterozoic “flux capacitor” canary in the metabolic coalmine
As indicated above, life is electrical, but did it start this way? Certainly, it is likely the underlying chemistry of life is based on the early geochemistry of the Earth32. A key component of all life is the reliance on the ETC, which drives the formation of a proton gradient. This led to theories involving alkaline thermal vents where hydrogen upwelling from the Earth’s crust met acidic sea water33. In turn, these conditions enabled the development of autocatalytic molecular networks, in effect, chemical reactions where the product catalysed its own production. These reactions would have relied on the flow of electrons, but at some point, this reversed when life evolved to use other sources of energy to create its own proton gradient. It also makes the case for why mitochondria are so central to all eukaryotic life today, and why, despite life’s apparent immense complexity, at its core is something much simpler34. This simplicity is apparent in the relationship between the mitochondrial membrane potential and the Kreb’s cycle—turning in one direction it works to create ATP, but reversing it, which is perhaps the original direction, it is key in biosynthesis. Membrane potential also controls the production of ROS. Mitochondria can thus be described as “flux capacitors” because of how the metabolic flux is linked to the charge across their membranes and the existence of electrical fields, which are likely to be part of metabolic flux control35.
A charge-based metabolism also ties in well with the bioelectric field concept, which is not only important in development, but in regeneration and the evolution of multicellularity. In effect, information about morphology and cooperation is encoded in an electric field and is largely generated by modulation of ion channels36. This suggests that an electromagnetic (EM)/bioelectric field may have been key in the origins of life itself as a kind of “ethereal skeleton”37. The concept of the “morphogenetic field” was first described by Alexander Gurwitsch in the early part of the 20th century as a kind of supra-cellular dynamic law that embraced biological organisation at the molecular, cellular and morphological level20. Although there is still a debate around the origins of life being metabolism or genetics first, it is possible that genes and proteins arose through direct biophysical interactions between amino acids and nucleotides in a protometabolic network38. Life started simply, but got more complex, and with multicellularity, became much smarter, with bioelectricity likely being part of the field based informational, sensing and cooperative glue that helps it all work. Dysfunction of this could well be key as to why pathologies, such as cancer, begin to occur more often with increasing age39. In short, complex organisms, such as humans, can be viewed as a giant flux capacitors built on cooperative smaller ones, with the brain perhaps being the most complex unit of them all40.
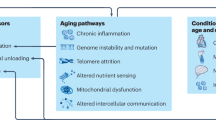
This suggests that ageing, and our ability to resist it, is determined by our ability to maintain a structure that enables metabolic flux that is intimately tied in with the movement of charge, and thus the creation of electric fields. The existence of the Kreb’s cycle and large electric potentials, especially in the mitochondrion, very likely not only reflects life’s origins, but also indicates that mitochondria are perhaps the best “canary in the metabolic mine” of life (Fig. 1). It is possible that mitochondria may also rely on quantum effects, especially if we view electrons from the wave-particle perspective and if they age, their ability to flow electrons will decrease, leading to oxidative stress41.
Life is clearly electrical, a principle that was likely engrained from the origins of life where the movement of charge may well have resulted in a kind of self-organising and dissipating flux capacitor. The earliest lifeforms were prokaryotes, and the strongest theory at the moment suggests that eukaryotes arose when an Archaean underwent symbiosis with a bacterium, which later became the mitochondrion. This would suggest that bioelectricity and EM fields are fundamental to life, hinting that quantum mechanics may help to understand how it works, especially if exposed to unusual environments. The role of gravity could be particularly important in structural organisation. Figure created in Microsoft PowerPoint.
Life as a global dissipating concept: space ageing in different organisms
“Life is a far from equilibrium self-maintaining chemical system capable of processing, transforming and accumulating information acquired from the environment”. A reworking of NASA’s definition of life by Vitas and Dobovišek, 201942.
In the previous sections we have highlighted the importance of life being electrical, but there is another concept that may also help to explain why various species age at different rates. This is that life can be viewed as a far from equilibrium self-organising and self-replicating structure that is an emergent property of the Earth’s geochemistry to dissipate energy, in effect, life is an emergent property of our planet32. As Schrödinger put it, life is simply a piece of negative entropy43. Indeed, it has been said that life is a good way to dissipate solar potential44. In effect, the organisation of matter into life, and its drive to replicate, can be explained by thermodynamics45. This thermodynamic approach not only helps describe the physical properties of life and its origins, but why it evolves towards higher states of entropy production46,47,48. Although life can be defined in the above thermodynamic terms, it displays homoeostasis, which is the ability to maintain a constant internal environment – despite changes in both the internal and external conditions. This can be further refined using the term “dynamic homoeostasis”, meaning it dynamically maintains biological and chemical equilibrium, and is thus not static, but constantly adjusting to keep a steady state. The origins of this can be traced back to biophysical theories such as self-organisation and Dynamic Systems Theory, which has led to the idea that rather than homoeostasis, the term homeodynamic is a better description of how life manages to maintain a steady state49.
A key observation is that inflammatory tone rises with age and has been coined “inflammaging”, which is associated with the gradual ageing and thus reduction in function of the immune system termed “immunosenescence”50. Key in this process is failing mitochondrial function51. If we combine thermodynamic self-organisation, inflammation and hormesis at a global level, we can make some interesting inferences – especially if we include natural selection. It can be argued that inflammation and hormesis are flipsides of the same adaptive process, where individual molecules, organelles, cells, organs, organisms and even entire species are disposable as part of an adaptive global dissipating system. In effect, if inefficient dissipating systems cannot adapt, they are removed in response to stress to enable rebuilding of functional ones, a kind of global “inflammatory/hormetic” approach to life. The key is that as stress is experienced, the damaged system is broken down into simpler and simpler components until it can rebuild. It could be viewed as a kind of “scale free” adaptive process. In a way, chronic inflammation can be viewed as a negative marker of fitness, and if it cannot be resolved, it may be accelerating the ageing process to make way for something that is better adapted. What this effectively suggests is that the more complex an organism, and the better it can use information to adapt to an environment, the longer lived it can be without having to resort to genetically-based Darwinian evolution52. Figure 2 outlines the concept.
Key here is that as life became more complex, an individual organism became better at adapting to stress by using retained information, rather than having to evolve through genetically based natural selection. This would imply that simple short-lived organisms, such as prokaryotes, could evolve to survive in space quickly, whereas long-lived and complex ones, such as humans, would simply develop inflammation and age faster. Figure created in Microsoft PowerPoint.
From these ideas, it could be inferred that a complex organism, if pushed outside its AME, will become inflamed which is likely linked to an AAP. In contrast, short lived prokaryotes, even if they do age faster, because of their short replication cycles will evolve through genetic selection, something that a long-lived complex human cannot do during a few months to years in space. So, from this we might predict that complex organisms in space may develop inflammaging if they cannot adapt, as they cannot evolve through genetic based natural selection, but smaller ones, such as prokaryotes, may well be able to.
Stress defines form
The above theories suggest that thermodynamics “organises” life, with stress having been a factor in determining an organism’s AME and identity. Indeed, a key concept in defining life is how it identifies self from non-self in relation to cognition and a computational spatio-temporal boundary. Bioelectricity provides one explanation of this and highlights how any organism will build its own internal model that embraces a primal homoeostatic drive to reduce stress when exposed to a changing environment, and explains how complex multicellular life emerged from something simpler, indicating that cognition is scale-free53. However, if it became too good at removing stressors, it could paradoxically remove the very stressors that shaped it, resulting in it becoming less fit, something humans seem to be good at. Indeed, humanity is today plagued by a plethora of self-inflicted conditions including obesity, type 2 diabetes and cardiovascular disease54.
In relation to this, quantum biology seems to become more relevant when it is considered as an interaction with the environment, for instance, rather than a noisy environment disrupting any significant quantum effects such as coherence and superposition, it actually enhances them55,56. This has led to the idea of environment-assisted quantum transport (ENAQT) in biological systems57. This all suggests that the general thermodynamic mechanism that explains dissipative adaptation and self-organisation and the generation of order out of disorder. In effect, how external work leads to the existence of the most adaptive dissipative system, and thus, longest lasting that can dissipate heat into the environment, which also involves the quantum realm58.
In short, several different scientific fields all converge to suggest that a challenging environment, in other words, stress, invoke limited adaptation and an organism’s identity that can be explained from different viewpoints, such as quantum mechanics, thermodynamics and bioelectricity. However, if the stress is too far outside of its AME, it invokes a programme that accelerates its demise, enabling other, fitter organisms to flourish.
Proof of quantum biology–a medical approach
A key test of any theory is the ability to apply it: pharmacology, biochemistry and genetics have been pivotal in medicine for many years, but many diseases are still not explained by this approach. What about quantum biology in medicine? There are perhaps two key approaches, one, as already mentioned involves spin, and the other is PBM, which may or not be described as quantum biology, but both likely affect the ETC. Although there are clear medical implications, they also tell us something fundamental about how life works, and what might happen in space. Critically, PBM and magnetic fields will likely interact controlling outcome, suggesting “mito-tuning” for health is possible using these modalities.
Quantum spin
Numerous studies have shown that altering magnetic fields can affect cell growth and death, which are often associated with changes in ROS and calcium, likely induced by a radical pair mechanism59. This is leading to the possibility that weak static magnetic, radiofrequency and electromagnetic fields (EMF) that modulate ROS could be important in both tissue regeneration and as an anti-cancer strategy60. Recently, one group reported an alternating current EMF technique to tune electron tunnelling and thus the redox state in cytochrome C in complex IV, which is part of the mitochondrial ETC, opening up the possibility of not only a quantum based sensor of metabolism, but also one that could manipulate it61. Another group has shown that very low frequency magnetic field pulses can induce mitophagy and rejuvenate mitochondria62, while rotating magnetic fields have been shown to inhibit mitochondrial respiration in cancer cells63. In short, there could be a correlation between spin, magnetic fields and ROS, in particular, involving mitohormesis and calcium flux, especially in relation to extremely low frequency electromagnetic field (ELF-EMF) exposure64.
A key point is that differing field strengths can have multi-phasic effects. For example, in one study it was found that using fields slightly stronger than that of Earth modulated proliferation via a process involving ROS and heat shock protein 70 (HSP 70) in a biphasic manner; compared to 45 µT (Earth standard), 200 µT inhibited planarian regeneration, while 500 µT stimulated it65. That a hormetic process is involved seems to be clear, as there is a correlation with ROS, and it appears, the mitochondrion. It is well described that “mitohormesis”, however it is induced, is a therapeutic approach66,67. It would seem that modulation of ETC using EMF can be both beneficial and harmful.
PBM
There is a long history of using light to accelerate healing, treat cancer and neuropathological diseases – with near infra-red light (NIR) being one of the most effective. It has been called both PBM and low-level light therapy (LLLT). One possible target of NIR is cytochrome C oxidase (complex IV) in the ETC68,69; it has also been shown that light at 808 nm enhanced the activity of complex IV, but suppressed complex III70. In a cellular model of inflammation, NIR at 730 nm has been shown to initially stimulate mitochondrial ROS, which then induces an anti-inflammatory response to counteract inflammation induced by the SARS-CoV-2 spike protein, which is associated with a longer term enhancement of mitochondrial function, essentially a hormetic response71. Interestingly, NASA has long held an interest in PBM, as it developed light-therapy based devices for potential use in space (NASA Research Illuminates Medical Uses of Light | NASA Spinoff).
In terms of a mode of action, it was originally thought that NIR stimulated reduction of the ETC, mainly by absorption at the copper centre in complex IV, enhancing electron transfer72, although it has also been suggested that it is not the cytochrome that is primary target, but mitochondrially bound water73. This is likely to depend on the absorption spectrum of water, which is quite varied depending on the wavelength. However, more recent data support the bigger picture that the ETC can be modulated by light of many different wavelengths, ranging from the UV to the NIR, depending on the chromophore, with a unique biphasic and thus therapeutic response at each, with the changes in the mitochondrial membrane potential (MMP) and thus level of ROS dictating outcome (growth, apoptosis, death) – again, supporting hormesis74.
Perhaps of relevance is that basic metabolism produces low number of photons of many different wavelengths, especially during oxidative stress. This was originally known as “mitogenetic radiation” (literally, radiation that stimulates mitosis), but later, biophotons or ultra-weak photon emission (UPE). Its existence was first observed by Alexander Gurwitsch in the 1920s and seems to have been largely confirmed in many organisms75,76. Indeed, it has been suggested that these metabolic photons could be part of a non-chemical cellular communication system77; it even appears that isolated mitochondria can communicate via photons78,79.
When one considers that life is based on chromophoric molecules that may well ensure dissipative stability, hinting at a possible mechanism for why many plant sunscreen compounds are key in stress resistance and could explain why they act as medicines80, then if life also produces photons, which could interact with other components in the cell, then it is possible that PBM is modulating an existing non-chemical photonic homoeostasis system.
Can we achieve optimal health in space?
A key question is whether humans, and their companion organisms, cannot just survive in space, but thrive in optimal health without either genetic modification and/or completely replicating Earth’s environment. The evidence suggests that even the fittest astronauts, living in space in the current spacecraft, seem to develop an AAP despite taking regular exercise3. Unfortunately, how much exercise is required is still unclear81, even though on Earth humans can get stronger and faster through physical exercise training, which is associated with not only a longer and healthier life82, but better cognition83. Although it has been said that endurance exercise can save the (Western) world84, one estimate suggests that only 6.8% of U.S. adults can be classified as being in “good” cardiometabolic health85. This of course raises an interesting question: in microgravity is it ever going to be possible to do enough exercise to provide protection, and what happens when less fit and/or older populations start living in space?
So, what other factors determine the boundaries of the AME? Clearly, as suggested this will vary depending on the species. Humans are long lived, with extended reproductive cycles, but we cannot fly in the air or breath underwater unaided, equally, we struggle if oxygen levels get too low, or we run out of food and water, or if there is too much sunlight, or too little, or if the temperature gets too high or too low. More subtly, too much food and too much oxygen can also be damaging, as can too much radiation, and it seems, a lack of gravity. There is also now evidence, in mice, that higher than normal carbon dioxide levels found on the ISS could accelerate the somatic mutation rate due to increased oxidative stress, which may be independent of radiation86. However, differences in light and magnetic fields could also play a role, in particular, around entraining of circadian rhythms. They are likely all interlinked, in particular, through the mitochondrion and redox. It is possible that with so many factors close to, or outside the adaptability envelope, the system cannot adapt. Figure 3 outlines these factors in relation to the AME.
All aircraft, and spacecraft, have a designed “flight envelope”, outside of which they either stop flying, or suffer severe damage. Life also has a metabolic envelope that is the result of its evolution in a particular niche. However, unlike aircraft and spacecraft, individual organisms have the advantage of a limited ability to expand the envelope to some degree when exposed to stress, known as hormesis, hence a better description is the “adaptative metabolic envelope” (AME). However the downside is without the stress, it shrinks – it seems to be very plastic. Figure created in Microsoft PowerPoint.
What is optimal health?
It could be said that this would be a phenotype that develops minimal disease, is aerobically fit with good strength and balance, adaptable, and metabolically flexible, which exhibits minimal inflammaging, good mitochondrial function for their age, and a healthspan that gets close to their species predicted realistic lifespan, in effect, shows morbidity compression. In short, the opposite of an AAP. Unfortunately for humans, this healthy phenotype is something that is not really well defined in medicine, as most data is based on actuarial data, which, certainly for modern Western cultures, includes a large percentage of the population suffering from diseases induced by a sedentary lifestyle with a poor diet. This phenotype can be described as the metabolic syndrome, which is associated with a plethora of other diseases, including an increased risk of developing severe SARs-CoV-2 due to reduced metabolic adaptability87. Although the maximum theoretical limit for human lifespan is thought to be between 115–120, with 105 being the limit for most people, suggesting an asymptomatic (healthy for age) of around 95, what we actually saw in the early part of the 21st century was an average absolute life expectancy of 80 or so, with a healthy lifespan around 70. The implication being that humans have made themselves too comfortable, with a lack of physical activity being a big component of this54. Figure 4 depicts a way of viewing this.
Multiple data suggest that the average human living a perfect lifestyle could certainly live into their 90s, only suffering severe frailty in the last few years of their life, so demonstrating morbidity compression. In comparison, a very poor lifestyle seems to be associated with a greatly reduced life expectancy, with many years of ill health. It is also possible that a transgenerational epigenetic inheritance (TGI) mechanism could affect future generations (see last point in conclusions). The question is where, if living in space unless very special measures are taken, would this put the average astronaut in relation to these curves. Figure created in Microsoft PowerPoint.
Gravity
In space, one of the biggest problems is the lack of gravity, which of course directly impacts the efficacy of exercise. The solution for this, which has long been proposed, is a large centrifuge, which could provide many benefits88. Indeed, calculations suggest that a hypothetical comfort zone could be achieved with a spacecraft rotating between 3–5 rpm, with a radius of around 10–100 m, achieving between 0.3 to 1 G89. Although an astronaut-sized centrifuge has yet to be launched/built in orbit, the ISS has had smaller laboratory scale devices in place for many years, which have enabled in situ comparison of microgravity and artificial gravity (AG) on a number of organisms. For instance, these have shown clear effects on a variety of plant parameters, such as upregulation of membrane associated proteins involved in the stress response under microgravity90. Further experiments, which compared plant transcriptional responses in two consecutive experiments under microgravity, when compared to centrifuge data, highlighted many changes, but somewhat surprisingly, an apparent downregulation of genes involved in the stress response91, hinting there is still a lot to learn. In terms of animal studies, early experiments using a similar system comparing C.elegans in microgravity and in a centrifuge, showed that they too lost muscle mass, but critically, showed evidence of down regulation of the TCA cycle, as well as changes in sirtuins, supporting alterations to mitochondrial function92. The Japan Aerospace Exploration Agency (JAXA) have also developed the “Multiple Artificial-gravity Research System” (MARS), which is effectively a rotating habitat for mice, which clearly showed that AG could protect the mice from bone and muscle loss when on the ISS93. Multiple studies have now shown that, for instance, AG in space could protect the optic nerve and retina of mice94, reduce CNS damage in fruit flies95, partially offset hepatic oxidative stress in mice96, and protect against thymus atrophy97. In terms of cell studies, osteoblastic cells, when kept in a centrifuge on the ISS were much healthier than the controls kept in microgravity. The control cells displayed reduced rates of cell proliferation, lysosomal function and DNA repair, and enhanced inflammation, ferroptosis and senescence, as well as enhanced microtubule acetylation, hinting at severe cytoskeleton disruption98.
The lack of gravity could be predicted to upset cell function due to the principle of tension induced integrity, or “tensegrity”99. Critically, mitochondrial function is integrated with the cytoskeleton100, which is connected to the deformation sensitive plasma membrane. Indeed, it has been suggested that cytoskeletal reorganisation under microgravity would induce mitochondrial dysfunction101. Moreover, as all membranes have an amphipathic structure and a large electric charge across them, changes in charge will exert a mechanical force on them102,103,104. In fact, most cells have very sensitive mechanotransduction systems (in effect, they can detect even the slightest change in their structure induced by both internal and external forces), which are integrated with multiple signalling systems and ion channels105, with redox shifts being pivotal106. Hence, decompressing cells by removing gravity is going to alter redox signalling and their bioelectric field. As it is possible that the radical pair mechanism can also affect microtubule reorganisation107, this might suggest an interaction between the Earth’s magnetic field and gravitational shifts and cell signalling.
Radiation protection from magnetic fields
The engineering of spacecraft has taken care of most of the lethal problems of simply surviving in space, but the radiation aspect, especially once outside the Earth’s magnetic field from Solar Energetic Particles and Galactic Cosmic Rays, is a serious potential problem4. It was originally thought that ionising radiation primarily accelerated ageing by inducing cancer, but data now suggest it affects multiple systems, including metabolism and individual cell survival, inducing oxidative stress and damaging mitochondria108. Certainly, evidence is that ionising radiation can induce mitochondrial ROS production109. No human has been exposed to a hypomagnetic field for any length of time, as the ISS orbits within the Earth’s magnetosphere, offering some protection. However, the establishment of lunar bases is more of an issue, as the Moon only spends about 7 days a month within this protective magnetic field, and even this varies depending on whereabouts it is on the Moon110.
Direct effects of hypomagnetic fields
As discussed, there is reasonable evidence that shifts in magnetic fields, both above, and below the strength of the Earth’s magnetic field can alter cell growth and this is related to changes in ROS65, supporting that fact the biology is sensitive to these fields and likely involves a radical pair mechanism and mitochondria in many cases59. So, in addition to the lack of protection against radiation, a hypomagnetic field may also directly induce metabolic perturbations.
Circadian rhythms
Ageing and the circadian rhythm are linked, especially via nutrient sensing; it is a two-way street, ageing worsens the inherent circadian clock, while damage to the circadian clock can accelerate ageing111. Critically, exercise and stress are also circadian entrainment factors112, and the circadian system controls mitochondrial function113.
Disruption of circadian clock in space is recognised as a concern for space flight. Accepted “zeitgebers” that entrain the clock are light, exercise and food114. But another “zeitgeber” may be added to this. In the 1950s, Frank Brown discovered that Atlantic oysters when removed from the sea and moved 100 s of miles and kept in a tank and shielded from most normal entrainment cues, including light, still seemed to synchronise to the lunar cycle. New data does support the idea that many organisms are also entrained to small changes in the Earth’s magnetic field due to the effects of the Moon and day/night cycles, which is also involves change in ROS115.
The best described zeitgeber is light, which controls melatonin, which is also an antioxidant and is produced in mitochondria in many tissues. Although mitochondrial dysfunction can result in a reduced capacity to produce melatonin, melatonin can also improve mitochondrial function. Blue light supresses pineal production of melatonin, while darkness enhances it. However, extra-pineal production of melatonin does not seem to be suppressed in the same way by blue light, while NIR may even stimulate it. As NIR can penetrate much further into many tissues, it has been suggested that a modern lifestyle of living indoors, and the increasing use of light sources that do not produce NIR, could be resulting in ill health. Given that NIR modulates mitochondrial function, it has been suggested that some of its action may involve melatonin production, triggered by the release of nitric oxide (NO), or even ROS. The key point here is that extra-pineal production of melatonin far exceeds that induced by retinal blue light exposure, and could well be triggered by exposure to NIR right throughout the body, including the brain (see review by Tan on melatonin116). Interestingly, recent data also show that NIR can promote bone regeneration, possibly via modulation of a sodium channel and cryptochrome 1117, while light at 670 nm has been shown to reduced blood glucose levels, possible via manipulation of mitochondria function118. It is generally accepted that 5–7% of the light reaching the Earth is UV (300–370 nm), about 38–43% visible (370–740 nm), of which about 25% is blue, and 52–54% is NIR (740–3000 nm)119,120,121. It has been suggested that early morning sunlight contains just the right amount of NIR to prepare the skin for later exposure to higher intensities of UV, hinting at an evolved mechanism as to why it is beneficial122.
In contrast, although cortisol production is also entrained to light cycles, in particular of shorter wavelengths, it tends to peak early morning and fall during the day. However, short high intensity exposure of blue light in the evening can induce it and alter circadian rhythms123. Cortisol is central to the allostatic stress response, which requires energy. Mitochondria both produce it, but are also controlled by it, critically, in a biphasic fashion – short duration exposure induces mitochondrial function, but too much for too long inhibits124.
This raises the possibility that another factor in space, which puts humans outside of their AME, is not only the potential for an unusual circadian rhythm, for instance, the ISS experiences 16 sun rises and sets per day, but a totally artificial light system. It is entirely possible that life has evolved to be dependent on the sun’s spectrum, in particular, NIR, with mitochondrial function again being pivotal. It could be envisioned that too little melatonin, and too much cortisol, could quickly result in mitochondrial dysfunction.
Conclusions
Being in space clearly stresses organisms, in particular, complex ones like humans, perhaps resulting in an AAP, which can be partially explained by conventional biological approaches. However, slightly less conventional approaches may also be providing some insight, which may include quantum biology and a focus on mitochondria. This could have implications not just for the astronauts themselves, but potentially their offspring in relation to epigenetics.
The life electric
Life is electric as it relies on the movement of fundamental particles, which suggests further insight could come from quantum mechanics. For instance, the role of quantum spin and tunnelling in relation to redox and ROS production. Data, and theory, do seem to suggest that travelling outside of the Earth’s magnetosphere could impact basic metabolism, and even within it but in orbit, could upset circadian rhythms. Furthermore, alterations in cell structure, and cell to cell communications, for instance, through the lack of gravity, would affect the bioelectric field.
The ancestral Proterozoic flux capacitor – the canary in the coalmine
Within most eukaryotic cells, especially those which are part of multicellular organism, such as a human, is the mitochondrion, which is central to cell function, both in sensing the environment and controlling homoeostasis and adaptation, and balances hormesis and inflammation. It is constantly flowing electrons and moving charge, creating large electric fields, and could well be dependent on some interesting quantum effects, ranging from tunnelling, to coherence, and spin41. That mitochondria appear to suffer in space suggests that in order to thrive, we need to find ways to restore their function.
Life as a global dissipating concept
Thermodynamics suggest that life is a far from equilibrium self-organising dissipating vortex of negative entropy that operates at a global scale to dissipate energy potentials, in effect, it is the flow of energy that results in the structures we call life. When viewed like this, each component, from the molecular upwards, is disposable. To maintain dissipation under stress, it effectively undergoes retrograde evolution to simpler forms that enable rebuilding, hence hormesis and inflammation are flipsides of the same adaptive process, which can be viewed at the global level. In effect, accelerated ageing, if an organism cannot adapt, is a marker of global adaptation, with the ultimate expression being death and its removal to enable fitter variants to survive. In terms of space travel, this means that simpler, short lived and fast reproducing organisms, such as prokaryotes, will evolve quickly through genetic selection, but multicellular long-lived ones, like humans, will not, and will therefore simply age faster. This raises an interesting scenario that an astronaut’s microbiome may be in a much healthier state, say, when they get to Mars than the astronaut themselves, unless we can find a way to keep the host healthy as well.
Learnings from hormesis
The main question, as posed in the “thriving in space document”, is whether or not humans can adapt to extended periods in space, in particular, on long journeys to other planets, or whether they need to adapt the space environment to make it more like home. Hormesis, and other terms, such as allostasis, describe the ability of individual organisms to adapt, or not, to stress; if the organism cannot adapt, it seems to develop chronic inflammation. As it ages, the ability to adapt decreases. All in all, whether we consider shifts in acceleration/gravity, magnetism, circadian rhythms or radiation, even alterations in gas levels, they are all inter-related due to the evolution of life on Earth. Life is both entrained with and can adapt to constant changes in this environment within a certain Goldilocks zone, whether if fails or grows stronger is determined by it hormetic adaptability. Figure 5 summarises these concepts.
Life has become canalised to its environment, which determines its AME; throughout evolution this has been constantly stimulated by environmental factors, such as physical activity, plant polyphenols, extremes of temperature, lack of food or water, and infection, but has been entrained to circadian rhythms. Without these stressors, the adaptive envelope likely narrows, which is highly reliant on a sufficient mitochondrial reserve, making it more susceptible to other factors, resulting in an inability to flow electrons correctly, leading to inflammation and an accelerated ageing phenotype. A marker for this could be the relationship between the mitochondrial membrane (Ψ) potential generated by the ETC and the amount of ROS, which if imbalanced, leads to increased oxidative stress. Figure created in Microsoft PowerPoint.
It seems that humans require a certain amount of the right kind of stress for optimal health, which raises the question, which is the most important Earth environment to maintain in space? The forerunner here has to be gravity, as exercise does offset some of the problems, but not all of them. Certainly, hypergravity seems to induce beneficial changes125, as increased gravity beyond Earth standard effectively stresses the system in a direction it can, to some degree, adapt to, as it mimics the effects of exercise. In effect, it is hormetic.
Are humans trapped on Earth?
The evidence is that even very fit astronauts develop health problems once in space, even if they are orbiting within the Earth’s magnetosphere, with mitochondrial dysfunction being a key marker of non-adaptation, despite eating a good diet and taking lots of exercise. Whether this translates, when they return to Earth, into a shortened life expectancy remains to be determined, as there is no readily available long-term data on the 600 plus people who have been in space. If astronauts are experiencing an AAP and do not adapt, then this does not bode well for extended periods of time in space, in particular, in transit to other planets and moons. Lunar or Mars bases may be slightly better due to some gravity, and the ability to reduce radiation exposure, but even here, the reduced magnetic fields, and different circadian cycles and light spectra might result in problems.
So are humans trapped on Earth? This depends on how we define it, clearly staying on the ISS for several months is entirely possible, although we don’t know what the longer-term effects might be. The data is simply not available yet. Even trips to Mars, which may be of similar time scales, suggest it is survivable, except there are other issues that could worsen health, in particular, a lack of a magnetic field. But if astronauts are developing an AAP, then there is a difference between survivable and thriving. It therefore seems that unless we re-engineer our own bodies, or completely replicate the Earth environment on a spacecraft, very long travel times could well be a problem, as we are likely to always develop an AAP. As to off-Earth bases, say on the Moon or Mars, the presence of some gravity, and the ability to build much more substantial structures, may help. As we still do not fully understand the ageing process, space travel, somewhat ironically, appears to be a valid model for studying it. Interestingly, exposing people to microgravity has raised ethical concerns about whether it is fair to send people into space, given the obvious problems it causes126. This of course raises an important question about how space companies adhere to the occupational safety and health acts of various countries for their employees, especially if they leave Earth for significant periods of time. Unless companies can guarantee health in space, a barrier to extended space travel could simply arise from legal considerations.
A role for quantum mechanics and other less traditional treatments?
In this perspective we ask the question, can thermodynamics, and quantum mechanics, provide any further insight into space health? Certainly, from the angle of quantum spin, we believe it can, as it raises the possibility that hypomagnetic fields could have a direct effect, for instance, via redox manipulation. This in turn raises the possibility that other factors that alter, or influence, electron movement, especially in the mitochondrion, could also be important, which then ties in with the bioelectricity, cellular structure and communication, and thus regeneration and growth, as well as senescence and death. This of course means both gravity and photonic involvement. Whether or not how they influence metabolism can be described as classical or quantum, is still up for debate, but once we view life as being electrical, and reliant on fields due to the movement of charge, which can be explained by the organising principles around thermodynamics, new ways of explaining what happens to life in space become possible, which potentially leads to ways of improving space health. In terms of research direction, differentiating and working out the actual contribution of gravity, magnetic fields, circadian zeitgebers and light spectra to health will be key, in particular, how they interact with each other to induce off-earth phenotypes is going to be pivotal. For example, measuring the differences in physiology between animals, prokaryotes and plants, in light controlled and magnetically shielded centrifuges, compared with appropriate controls, both on the ISS, and the soon to come Moon bases, could be really informative.
A transgenerational mitochondrial/epigenetic epilogue
It is well accepted that extended space travel could be harmful for human reproduction, for instance, radiation induced teratogenesis127. However, it is also clear that epigenetic imprinting can occur, for example when an organism is stressed, which can affect both the offspring, and potentially, their offspring too – and likely displays hormetic biphasic effects128. For instance a poor lifestyle can lead to epigenetic inheritance of obesity and diabetes129, as well as mental health issues130. This would strongly suggest that longevity of future generations might also be epigenetically controlled to some degree. Because of their central role in maintaining cell function and epigenetics, mitochondria play a key role in how cells respond to stress, in particular, radiation131. Might the observed space travel induced mitochondrial dysfunction be important here? Understanding the effects of space flight on epigenetic transgenerational inheritance may need to be considered (see Fig. 4). If astronauts are experiencing an AAP, could the propensity for this be passed on to their children and grandchildren? We simply do not know and will probably need to before we launch ourselves on long space journeys.
Data availability
No datasets were generated or analysed during the current study.
References
Vernikos, J. & Schneider, V. S. Space, gravity and the physiology of aging: parallel or convergent disciplines? A mini-review. Gerontology 56, 157–166 (2010).
Strollo, F., Gentile, S., Strollo, G., Mambro, A. & Vernikos, J. Recent progress in space physiology and aging. Front. Physiol. 9, 1551 (2018).
Ratushnyy, A. Y. & Buravkova, L. B. Microgravity effects and aging physiology: similar changes or common mechanisms? Biochem 88, 1763–1777 (2023).
National Academies of Sciences, E. & Medicine. Thriving in Space: Ensuring the Future of Biological and Physical Sciences Research: A Decadal Survey for 2023-2032, (The National Academies Press, 2023).
Mason, C. E. The Next 500 Years: Engineering Life to Reach New Worlds (The MIT Press, 2021).
Li, Z. et al. Progress in biological age research. Front. Public Health 11, 1074274 (2023).
Poganik, J. R. et al. Biological age is increased by stress and restored upon recovery. Cell Metab. 35, 807–820 (2023).
Nunn, A. V., Bell, J. D. & Guy, G. W. Lifestyle-induced metabolic inflexibility and accelerated ageing syndrome: insulin resistance, friend or foe? Nutr. Metab. 6, 16 (2009).
Katzmarzyk, P. T. & Jakicic, J. M. Physical activity for health-every minute counts. JAMA 330, 213–214 (2023).
Lesmana, R. et al. The role of exercise-induced reactive oxygen species (ROS) hormesis in aging: friend or foe. Cell Physiol. Biochem. 56, 692–706 (2022).
Gradari, S., Palle, A., McGreevy, K. R., Fontan-Lozano, A. & Trejo, J. L. Can exercise make you smarter, happier, and have more neurons? a hormetic perspective. Front. Neurosci. 10, 93 (2016).
Calabrese, E. J. & Baldwin, L. A. Hormesis as a biological hypothesis. Environ. Health Perspect. 106, 357–362 (1998).
Menon, S. G. & Goswami, P. C. A redox cycle within the cell cycle: ring in the old with the new. Oncogene 26, 1101–1109 (2007).
Barnes, F. S. & Greenebaum, B. The effects of weak magnetic fields on radical pairs. Bioelectromagnetics 36, 45–54 (2015).
Usselman, R. J. et al. The quantum biology of reactive oxygen species partitioning impacts cellular bioenergetics. Sci. Rep. 6, 38543 (2016).
da Silveira, W. A. et al. Comprehensive multi-omics analysis reveals mitochondrial stress as a central biological hub for spaceflight impact. Cell 183, 1185–1201 (2020).
Camprubí, E. et al. The emergence of life. Space Sci. Rev. 215, 56 (2019).
Becker, R. O. & Selden, G. The Body Electric; Electromagnetism and the Foundation of Life, (Morrow, 1985).
Adee, S. We Are Electric: The New Science of Our Body’s Electrome (Canogate Books, 2023).
Fels, D., Cifra, M. & Scholkmann, F. E. Fields of the Cell (Research Signpost, 2015).
Marais, A. et al. The future of quantum biology. J. R. Soc. Interface 15, 20180640 (2018).
Al-Khalili, J. & McFadden, J. Life on the Edge: the Coming of Age of Quantum Biology, (Transworld Publishers, 2014).
Aeillo, C. It’s time to take quantum biology seriously. Physics 16, 20180640 (2023).
Chance, B. Tunneling in Biological Systems (Academic Press, 1979).
Trixler, F. Quantum tunnelling to the origin and evolution of life. Curr. Org. Chem. 17, 1758–1770 (2013).
Moser, C. C., Page, C. C. & Dutton, P. L. Darwin at the molecular scale: selection and variance in electron tunnelling proteins including cytochrome c oxidase. Philos. Trans. R. Soc. Lond. B Biol. Sci. 361, 1295–1305 (2006).
Sutcliffe, M. J. et al. Hydrogen tunnelling in enzyme-catalysed H-transfer reactions: flavoprotein and quinoprotein systems. Philos. Trans. R. Soc. Lond. B Biol. Sci. 361, 1375–1386 (2006).
Layfield, J. P. & Hammes-Schiffer, S. Hydrogen tunneling in enzymes and biomimetic models. Chem. Rev. 114, 3466–3494 (2014).
Woodward, J. R. Triplet born radical pairs and the low field effect. Appl. Mag. Res. 54, 47–58 (2023).
Mo, W., Liu, Y. & He, R. Hypomagnetic field, an ignorable environmental factor in space? Sci. China Life Sci. 57, 726–728 (2014).
Sarimov, R. M., Serov, D. A. & Gudkov, S. V. Hypomagnetic conditions and their biological action (Review). Biology 12, 1513 (2023).
Morowitz., E.S.a.H.J. The Origin and Nature of Life on Earth. (Cambridge University Press, 2016).
Sousa, F. L. et al. Early bioenergetic evolution. Philos. Trans. R. Soc. Lond. B Biol. Sci. 368, 20130088 (2013).
Lane, N. The Vital Question: Why is Life the way it is?, (Profile Books Ltd, Great Britain, 2015).
Lane, N. Transformer: The Deep Chemistry of Life and Death, (W. W. Norton, 2022).
Levin, M. Bioelectric signaling: Reprogrammable circuits underlying embryogenesis, regeneration, and cancer. Cell 184, 1971–1989 (2021).
Nunn, A. V., Guy, G. W. & Bell, J. D. Bioelectric fields at the beginnings of life. Bioelectricity 4, 237–247 (2022).
Harrison, S. A. et al. Life as a guide to its own origins. Annu. Rev. Ecol. Evol. Syst. 54, 327–350 (2023).
Levin, M. Bioelectrical approaches to cancer as a problem of the scaling of the cellular self. Prog. Biophys. Mol. Biol. 165, 102–113 (2021).
McFadden, J. Integrating information in the brain’s EM field: the cemi field theory of consciousness. Neurosci. Conscious 2020, niaa016 (2020).
Nunn, A. V., Guy, G. W. & Bell, J. D. The quantum mitochondrion and optimal health. Biochem Soc. Trans. 44, 1101–1110 (2016).
Vitas, M. & Dobovišek, A. Towards a general definition of life. Orig. Life Evol. Biospheres 49, 77–88 (2019).
Schrodinger, E. What is Life? The Physical Aspect of the Living Cell. (Cambridge University Press, 1944).
Michaelian, K. Thermodynamic dissipation theory for the origin of life. Earth Syst., Dynam 2, 37–51 (2011).
England, J. Every Life is on Fire: How Thermodynamics Explains the Origins of Living Things, (Basic Books, 2020).
Pulselli, R. M., Simoncini, E. & Tiezzi, E. Self-organization in dissipative structures: a thermodynamic theory for the emergence of prebiotic cells and their epigenetic evolution. Biosystems 96, 237–241 (2009).
Kondepudi, D. K., De Bari, B. & Dixon, J. A. Dissipative structures, organisms and evolution. Entropy 22, 1305 (2020).
Prigogine, I. & George, C. The second law as a selection principle: the microscopic theory of dissipative processes in quantum systems. Proc. Natl. Acad. Sci. USA 80, 4590–4594 (1983).
Lloyd, D., Aon, M. A. & Cortassa, S. Why Homeodynamics, Not. Homeost.? Sci.World J. 1, 133–145 (2001).
Franceschi, C., Garagnani, P., Parini, P., Giuliani, C. & Santoro, A. Inflammaging: a new immune-metabolic viewpoint for age-related diseases. Nat. Rev. Endocrinol. 14, 576–590 (2018).
Picca, A. et al. Fueling inflamm-aging through mitochondrial dysfunction: mechanisms and molecular targets. Int J. Mol. Sci. 18, 933 (2017).
Nunn, A. V., Guy, G. W. & Bell, J. D. Thermodynamics and inflammation: insights into quantum biology and ageing. Quantum Rep. 4, 47–74 (2022).
Levin, M. The computational boundary of a “Self”: developmental bioelectricity drives multicellularity and scale-free cognition. Front., Psychol. 10, 2688 (2019).
Nunn, A. V., Guy, G. W. & Bell, J. D. The intelligence paradox; will ET get the metabolic syndrome? Lessons from and for Earth. Nutr. Metab. (Lond.) 11, 34 (2014).
Huelga, S. & Plenio, M. Vibrations, quanta and biology. Contemp. Phys. 54, 181–207 (2013).
Scholes, G. D. et al. Using coherence to enhance function in chemical and biophysical systems. Nature 543, 647–656 (2017).
Zerah-Harush, E. & Dubi, Y. Universal origin for environment-assisted quantum transport in exciton transfer networks. J. Phys. Chem. Lett. 9, 1689–1695 (2018).
Valente, D., Brito, F. & Werlang, T. Quantum dissipative adaptation. Commun. Phys. 4, 11 (2021).
Zadeh-Haghighi, H. & Simon, C. Magnetic field effects in biology from the perspective of the radical pair mechanism. J. R. Soc. Interface 19, 20220325 (2022).
Hack, S. J., Kinsey, L. J. & Beane, W. S. An open question: is non-ionizing radiation a tool for controlling apoptosis-induced proliferation? Int. J. Mol. Sci. 22, 11159 (2021).
Jain, A. et al. Wireless electrical-molecular quantum signalling for cancer cell apoptosis. Nat. Nanotechnol. 19, 106–114 (2024).
Toda, T. et al. Extremely low-frequency pulses of faint magnetic field induce mitophagy to rejuvenate mitochondria. Commun. Biol. 5, 453 (2022).
Sharpe, M. A., Baskin, D. S., Pichumani, K., Ijare, O. B. & Helekar, S. A. Rotating magnetic fields inhibit mitochondrial respiration, promote oxidative stress and produce loss of mitochondrial integrity in cancer cells. Front. Oncol. 11, 768758 (2021).
Franco-Obregon, A. Harmonizing magnetic mitohormetic regenerative strategies: developmental implications of a calcium-mitochondrial axis invoked by magnetic field exposure. Bioengineering 10, 1176 (2023).
Van Huizen, A. V. et al. Weak magnetic fields alter stem cell-mediated growth. Sci. Adv. 5, eaau7201 (2019).
Ristow, M. & Schmeisser, K. Mitohormesis: promoting health and lifespan by increased levels of reactive oxygen species (ROS). Dose Response 12, 288–341 (2014).
Tapia, P. C. Sublethal mitochondrial stress with an attendant stoichiometric augmentation of reactive oxygen species may precipitate many of the beneficial alterations in cellular physiology produced by caloric restriction, intermittent fasting, exercise and dietary phytonutrients: “Mitohormesis” for health and vitality. Med. Hypotheses 66, 832–843 (2006).
Hamblin, M. R., Nelson, S. T. & Strahan, J. R. Photobiomodulation and cancer: what is the truth? Photomed. Laser Surg. 36, 241–245 (2018).
Hamblin, M. R. & Salehpour, F. Photobiomodulation of the Brain: shining light on Alzheimer’s and other neuropathological diseases. J. Alzheimers Dis. 83, 1395–1397 (2021).
Pope, N. J. & Denton, M. L. Differential effects of 808-nm light on electron transport chain enzymes in isolated mitochondria: Implications for photobiomodulation initiation. Mitochondrion 68, 15–24 (2023).
Aguida, B. et al. Near-Infrared Light Exposure Triggers ROS to Downregulate Inflammatory Cytokines Induced by SARS-CoV-2 Spike Protein in Human Cell Culture. Antioxidants 12, 1824 (2023).
Karu, T. I., Pyatibrat, L. V., Kolyakov, S. F. & Afanasyeva, N. I. Absorption measurements of a cell monolayer relevant to phototherapy: reduction of cytochrome c oxidase under near IR radiation. J. Photochem. Photobio. B 81, 98–106 (2005).
Sommer, A. P. Mitochondrial cytochrome c oxidase is not the primary acceptor for near infrared light-it is mitochondrial bound water: the principles of low-level light therapy. Ann. Transl. Med. 7, S13 (2019).
Golovynska, I., Golovynskyi, S. & Qu, J. Comparing the impact of NIR, visible and UV light on ROS upregulation via photoacceptors of mitochondrial complexes in normal, immune and cancer cells. Photochem. Photobio. 99, 106–119 (2023).
Cifra, M. & Pospisil, P. Ultra-weak photon emission from biological samples: definition, mechanisms, properties, detection and applications. J. Photochem. Photobio. B 139, 2–10 (2014).
Volodyaev, I. & Beloussov, L. V. Revisiting the mitogenetic effect of ultra-weak photon emission. Front. Physiol. 6, 241 (2015).
Prasad, A., Rossi, C., Lamponi, S., Pospisil, P. & Foletti, A. New perspective in cell communication: potential role of ultra-weak photon emission. J. Photochem Photobio. B 139, 47–53 (2014).
Mould, R. R. et al. Non-chemical signalling between mitochondria. Front. Physiol. 14, 1268075 (2023).
Bat’ianov, A. P. Distant-optical interaction of mitochondria through quartz]. Biull Eksp. Biol. Med. 97, 675–677 (1984).
Nunn, A. V. W., Guy, G. W., Botchway, S. W. & Bell, J. D. From sunscreens to medicines: can a dissipation hypothesis explain the beneficial aspects of many plant compounds? Phytother. Res. 34, 1868–1888 (2020).
Hackney, K. J. et al. The Astronaut-athlete: optimizing human performance in space. J. Strength Cond. Res. 29, 3531–3545 (2015).
Garcia, L. et al. Non-occupational physical activity and risk of cardiovascular disease, cancer and mortality outcomes: a dose-response meta-analysis of large prospective studies. Br. J. Sports Med. 57, 979–989 (2023).
Fernandes, R. M. et al. The effects of moderate physical exercise on adult cognition: a systematic review. Front. Physiol. 9, 667 (2018).
Kolodziej, F. & O’Halloran, K. D. Re-evaluating the oxidative phenotype: can endurance exercise save the western world? Antioxidants 10, 609 (2021).
O’Hearn, M., Lauren, B. N., Wong, J. B., Kim, D. D. & Mozaffarian, D. Trends and disparities in cardiometabolic health among U.S. adults, 1999-2018. J. Am. Coll. Cardiol. 80, 138–151 (2022).
Stolc, V. et al. Metabolic stress in space: ROS-induced mutations in mice hint at a new path to cancer. Redox Biol. 78, 103398 (2024).
Nunn, A. V. W. et al. SARS-CoV-2 and mitochondrial health: implications of lifestyle and ageing. Immun. Ageing 17, 33 (2020).
van Loon, J. J. W. A. et al. Benefits of a rotating – Partial gravity – Spacecraft. Acta Astronautica 220, 403–415 (2024).
Clement, G. R., Bukley, A. P. & Paloski, W. H. Artificial gravity as a countermeasure for mitigating physiological deconditioning during long-duration space missions. Front. Syst. Neurosci. 9, 92 (2015).
Mazars, C. et al. Microgravity induces changes in microsome-associated proteins of Arabidopsis seedlings grown on board the international space station. PLoS One 9, e91814 (2014).
Land, E. S., Sheppard, J., Doherty, C. J. & Perera, I. Y. Conserved plant transcriptional responses to microgravity from two consecutive spaceflight experiments. Front. Plant Sci. 14, 1308713 (2023).
Higashibata, A. et al. Microgravity elicits reproducible alterations in cytoskeletal and metabolic gene and protein expression in space-flown Caenorhabditis elegans. npj Microgravity 2, 15022 (2016).
Shiba, D. et al. Development of new experimental platform ‘MARS’-multiple artificial-gravity research system-to elucidate the impacts of micro/partial gravity on mice. Sci. Rep. 7, 10837 (2017).
Kremsky, I. et al. Artificial gravity attenuates the transcriptomic response to spaceflight in the optic nerve and retina. Int J. Mol. Sci. 25, 12041 (2024).
Mhatre, S. D. et al. Artificial gravity partially protects space-induced neurological deficits in Drosophila melanogaster. Cell Rep. 40, 111279 (2022).
Kurosawa, R. et al. Impact of spaceflight and artificial gravity on sulfur metabolism in mouse liver: sulfur metabolomic and transcriptomic analysis. Sci. Rep. 11, 21786 (2021).
Horie, K. et al. Impact of spaceflight on the murine thymus and mitigation by exposure to artificial gravity during spaceflight. Sci. Rep. 9, 19866 (2019).
Garbacki, N. et al. Microgravity triggers ferroptosis and accelerates senescence in the MG-63 cell model of osteoblastic cells. npj Microgravity 9, 91 (2023).
Gardiner, J. Cytoskeletal tensegrity in microgravity. Life 11, 1091 (2021).
Alberti, P., Semperboni, S., Cavaletti, G. & Scuteri, A. Neurons: the interplay between cytoskeleton, ion channels/transporters and mitochondria. Cells 11, 2499 (2022).
Nguyen, H. P., Tran, P. H., Kim, K. S. & Yang, S. G. The effects of real and simulated microgravity on cellular mitochondrial function. npj Microgravity 7, 44 (2021).
Taghian, T., Narmoneva, D. A. & Kogan, A. B. Modulation of cell function by electric field: a high-resolution analysis. J R Soc. Interface 12, 20150153 (2015).
Escobar, J. F., Vaca-González, J. J., Guevara, J. M. & Garzón-Alvarado, D. A. Effect of magnetic and electric fields on plasma membrane of single cells: A computational approach. Eng. Rep. 2, e12125 (2020).
Brownell, W. E., Qian, F. & Anvari, B. Cell membrane tethers generate mechanical force in response to electrical stimulation. Biophys. J. 99, 845–852 (2010).
Uray, I. P. & Uray, K. Mechanotransduction at the plasma membrane-cytoskeleton interface. Int J. Mol. Sci. 22, 11566 (2021).
Balta, E., Kramer, J. & Samstag, Y. Redox regulation of the actin cytoskeleton in cell migration and adhesion: on the way to a spatiotemporal view. Front. Cell Dev. Biol. 8, 618261 (2020).
Zadeh-Haghighi, H. & Simon, C. Radical pairs may play a role in microtubule reorganization. Sci. Rep. 12, 6109 (2022).
Richardson, R. B. Ionizing radiation and aging: rejuvenating an old idea. Aging 1, 887–902 (2009).
Yamamori, T. et al. Ionizing radiation induces mitochondrial reactive oxygen species production accompanied by upregulation of mitochondrial electron transport chain function and mitochondrial content under control of the cell cycle checkpoint. Free Radic. Biol. Med. 53, 260–270 (2012).
Winglee, R. M. & Harnett, E. M. Radiation mitigation at the Moon by the terrestrial magnetosphere. Geophys. Res. Lett. 34, 30507 (2007).
Liu, F. & Chang, H. C. Physiological links of circadian clock and biological clock of aging. Protein Cell 8, 477–488 (2017).
Tahara, Y., Aoyama, S. & Shibata, S. The mammalian circadian clock and its entrainment by stress and exercise. J. Physiol. Sci. 67, 1–10 (2017).
de Goede, P., Wefers, J., Brombacher, E. C., Schrauwen, P. & Kalsbeek, A. Circadian rhythms in mitochondrial respiration. J. Mol. Endocrinol. 60, R115–R130 (2018).
Malhan, D., Schoenrock, B., Yalcin, M., Blottner, D. & Relogio, A. Circadian regulation in aging: implications for spaceflight and life on earth. Aging Cell 22, e13935 (2023).
Zadeh-Haghighi, H. & Simon, C. Radical pairs can explain magnetic field and lithium effects on the circadian clock. Sci. Rep. 12, 269 (2022).
Tan, D. X., Reiter, R. J., Zimmerman, S. & Hardeland, R. Melatonin: both a messenger of darkness and a participant in the cellular actions of non-visible solar radiation of near infrared light. Biology 12, 89 (2023).
Peng, J. et al. Low intensity near-infrared light promotes bone regeneration via circadian clock protein cryptochrome 1. Int J. Oral. Sci. 14, 53 (2022).
Powner, M. B. & Jeffery, G. Light stimulation of mitochondria reduces blood glucose levels. J. Biophotonics 17, e202300521 (2024).
de Galvez, E. N. et al. The potential role of UV and blue light from the sun, artificial lighting, and electronic devices in melanogenesis and oxidative stress. J. Photochem. Photobio. B 228, 112405 (2022).
Mayhew, K. W. New Thermodynamics: rethinking the science of climate change. Eur. J. Eng. Technol. Res. 5, 559–564 (2020).
Barolet, D., Christiaens, F. & Hamblin, M. R. Infrared and skin: friend or foe. J. Photochem. Photobio. B 155, 78–85 (2016).
Barolet, D. Near-infrared light and skin: why intensity matters. Curr. Probl. Dermatol. 55, 374–384 (2021).
Robertson-Dixon, I., Murphy, M. J., Crewther, S. G. & Riddell, N. The influence of light wavelength on human HPA axis rhythms: a systematic review. Life 13, 1968 (2023).
Picard, M., McEwen, B. S., Epel, E. S. & Sandi, C. An energetic view of stress: focus on mitochondria. Front. Neuroendocrinol. 49, 72–85 (2018).
Isasi, E., Isasi, M. E. & van Loon, J. The application of artificial gravity in medicine and space. Front. Physiol. 13, 952723 (2022).
van Loon, J., Cras, P., Bouwens, W., Roozendaal, W. & Vernikos, J. Gravity deprivation: is it ethical for optimal physiology? Front. Physiol. 11, 470 (2020).
Mathyk, B. et al. Understanding how space travel affects the female reproductive system to the Moon and beyond. npj Women’s. Health 2, 20 (2024).
Rothi, M. H. & Greer, E. L. From correlation to causation: the new frontier of transgenerational epigenetic inheritance. Bioessays 45, e2200118 (2023).
Kaspar, D., Hastreiter, S., Irmler, M., Hrabe de Angelis, M. & Beckers, J. Nutrition and its role in epigenetic inheritance of obesity and diabetes across generations. Mamm. Genome 31, 119–133 (2020).
Monaco, A. P. An epigenetic, transgenerational model of increased mental health disorders in children, adolescents and young adults. Eur. J. Hum. Genet 29, 387–395 (2021).
Averbeck, D. & Rodriguez-Lafrasse, C. Role of mitochondria in radiation responses: epigenetic, metabolic, and signaling impacts. Int J. Mol. Sci. 22, 11047 (2021).
Acknowledgements
To the founders of quantum mechanics, and subsequent multi-disciplinary scientists who had and have the foresight to consider its role in biology. This project had no specific source of funding.
Author information
Authors and Affiliations
Contributions
A.V.W.N. and G.W.G. discussed and developed the concept that humans could be trapped on Earth due to their evolved physiology, which might be partly explained by our systems being coupled quantum mechanically to the conditions we evolved in. J.D.B. reviewed the idea and provided feedback, in particular, from the ageing aspect. A.V.W.N. wrote and edited the manuscript, and J.D.B. and G.W.G. provided editorial support during the development of the drafts.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Nunn, A., Bell, J. & Guy, G. Are we trapped on Earth: space and accelerated ageing. npj Microgravity 12, 10 (2026). https://doi.org/10.1038/s41526-025-00551-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41526-025-00551-3