Abstract
Pressurised metered dose inhalers (pMDIs) contain propellant gases with high global warming potential yet remain a cornerstone of management for asthma and chronic obstructive pulmonary disease (COPD). The aim of this study was to determine whether non-propellant alternatives of dry powder inhalers (DPIs) and soft mist inhalers (SMIs) had similar efficacy and safety. A systematic review was performed finding 44 randomised trials (24,710 participants) and moderate certainty evidence for most outcomes. No statistically significant or clinically important differences were found between inhaler types for any assessed measure. For asthma maintenance, the mean difference in peak expiratory flow rate between groups was 1.07 L/min (95% confidence interval [CI] -0.93 to 3.06). For COPD, the mean difference in FEV1 between groups was 0.01 L (95% CI -0.01 to 0.02). While the choice of optimal inhaler for an individual patient is a multifaceted decision, this review provides reassurance that non-pMDI devices can perform equally well.
Similar content being viewed by others
Introduction
Asthma and chronic obstructive pulmonary disease (COPD) are highly prevalent globally, affecting hundreds of millions of individuals1,2. A cornerstone of therapy for these conditions is the directed delivery of effective medications via inhalers3,4. Pressurised metered dose inhalers (pMDIs) are one of the most frequently used inhaler devices, with almost one billion manufactured every year5. Current pMDIs contain fluorinated propellant gases (F-gases) which are potent greenhouse gases – they exert an effect thousands of times greater than the equivalent volume of carbon dioxide (CO2). For example, a single inhaler of a common short-acting beta-agonist product, using the propellant HFA-134a, has the same global warming potential as 25.2 kg CO2 (equivalent to driving over 120 kilometres in a petrol car)6. The healthcare sector contributes 4-5% of global greenhouse emissions7,8, and the proportionally large contribution of inhalers (particularly pMDIs) is increasingly recognised. In the United Kingdom, pMDIs alone are responsible for around 3% of the entire National Health Service carbon footprint9.
A key component of this issue is the marked variation between countries in the proportion of prescribed inhalers that are pMDIs, due to both cultural and cost reasons. For example, there is a seven-fold difference in pMDI prescribing rates across northern European countries10. These observations in the face of the climate crisis have led to a call for much greater use of propellant-free options such as dry powder inhalers (DPIs) and soft mist inhalers (SMIs), especially in countries that predominantly use pMDIs11. A conscious consideration of the environmental impact is now included in inhaler guidelines in the UK12.
However, some uncertainty remains as to whether there is equipoise between the benefit of pMDIs and propellant-free inhalers for management of asthma and COPD. Previous systematic reviews with a narrow focus have considered this question for different drug classes across both conditions; however, these reviews are now 20 or more years old and did not include many drugs, formulations (e.g. extrafine particles) and devices (e.g. SMIs) that are now used routinely13,14,15,16. Some countries have made calls to move away from pMDIs when clinically safe to do so17, however strong contemporary data are needed to support such decisions. We therefore undertook a comprehensive systematic review and meta-analysis to establish if there was any evidence of a difference in clinical effectiveness or safety of treating individuals with asthma or COPD acutely or for maintenance therapy with pMDIs versus propellant-free devices, when the drugs and doses administered were broadly equivalent.
Methods
This review was not registered, but we developed a protocol before commencing the review, containing additional detail on the methods used and a complete list of changes made to the planned methods18. This review is reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement19.
Search strategy
We searched PubMed, Embase and the Cochrane Central Register of Controlled Trials to 25 September 2025, as well as reference lists of systematic reviews identified in the search. Full search strategies are provided in the Supplementary Information (Section A, Tables S1-S3).
Eligibility criteria
Study designs
Only randomised controlled trials (RCTs) were eligible for inclusion. In accordance with the protocol, observational studies were initially identified in the search but not included in the review as sufficient RCTs were available. We excluded studies not written in English and crossover studies (due to insufficient data regarding washout periods in many studies).
Population
We included studies of patients of any age with confirmed asthma or COPD. Asthma studies were categorised as management of acute asthma episodes (e.g. presenting for emergency care) or asthma maintenance therapy. Results were analysed separately for these three conditions. There were no studies comparing relevant devices in acute COPD.
Intervention
We included studies comparing the delivery of equivalent inhaled medication(s) by either DPI or SMI (grouped together as non-pMDI inhalers) versus pMDI. Studies of nebulised medication delivery were not included. Medications were considered ‘equivalent’ if they were from the same drug class (e.g. short-acting beta-agonist (SABA), or inhaled corticosteroid (ICS)) and given at approximately equipotent doses. ICS doses were categorised into ‘high,’ ‘medium’ and ‘low’ dose according to National Institute for Health and Care Excellence (NICE) guidelines20. Non-ICS medication doses were classified as ‘standard’ based on comparative clinical therapeutic equivalence, and ‘high’ if a multiple of that standard dose was used3,4. Studies assessing different combinations of drug classes (e.g. ICS-alone versus ICS/SABA combination) were excluded. We permitted slight variations in the frequency of medication administration (e.g. one versus two doses per day) but excluded larger discrepancies (e.g. once versus three or four doses per day) due possible differences in adherence, pharmacokinetic profiles and therefore effect. There were no restrictions on which drugs participants were taking prior to the trial, or on lead-in or washout periods. Results were analysed together for all drugs and doses.
The inclusion criteria for interventions and the definition of equipotency were intentionally broad to include the most complete picture possible of the available evidence comparing treatment regimens with and without propellant gases. Differences in effects (heterogeneity) observed in the results could then be fully investigated through subgroup analysis.
Outcomes
Our primary outcomes were physiological lung function measurements (forced expiratory volume in 1 s (FEV1) and peak expiratory flow rate (PEFR)), symptom control (any scale), quality of life (any scale), exacerbations (as defined by each study) and use of additional reliever medication (any measure). Secondary outcomes focused on safety, including mortality, overall or treatment-related adverse events (AEs) and serious adverse events (SAEs) as defined by each study. Studies were eligible if they reported one or more primary or safety outcomes.
We had initially planned to report hospital admissions and emergency room attendance separately, but these were frequently incorporated by our included studies into composite measures of adverse events or exacerbations, and so we have reported these composite outcomes in this review. We had also planned to include additional secondary measures of satisfaction, adherence to therapy and inhaler technique, but found that many studies measuring these factors did not address effectiveness or safety outcomes and were therefore ineligible for inclusion in the review. To avoid presenting misleading estimates of these outcomes from a subset of the available literature, we decided to exclude these outcomes.
Studies were excluded if the duration of follow-up after commencement of treatment was less than 48 h for COPD and asthma maintenance, but there was no minimum duration of treatment or follow-up for studies of acute asthma exacerbations.
Minimal clinically important differences (MCIDs) for various outcomes in asthma and COPD were identified from the published literature where possible or otherwise determined by consensus of our Respiratory Experts; these are summarised in the Supplementary Information (Section B, Table S4).
Study selection and data extraction
Title and abstract screening and full-text review were conducted using Covidence software21. All identified titles were initially screened by one of MJL, LP or HW, before all abstracts of papers deemed potentially relevant were independently screened by two authors (MJL and LP). These two authors then performed independent full-text reviews of all remaining papers. Any disputes were resolved by consensus, or by consultation with the review content experts (AG, JB, MR) where needed.
We extracted data including the study design, clinical condition of participants, inclusion of children and/or adults, inhaler type, drugs and doses used, particle size (normal or extra fine), use of spacers, and outcome data. Using a standardised extraction form, one researcher (SB, MJL, LP or RS) extracted and a second researcher verified the data, with inconsistencies discussed until consensus was reached. To improve consistency of data extracted from figures, a web-based plot digitising software was used22. No additional data were sought from authors of included studies. Where studies reported results at more than one time point, the latest time point was used. Within each analysis, only one measure from each study was included. Different measures of the same outcome and measures taken at different time points from different studies were combined in the analysis where possible.
Risk of bias
Studies were assessed for risk of bias using the Cochrane Risk of Bias tool23. Assessment was completed independently for all included studies by two of SB, LP and RS, and reviewed by a third author (MC). Studies assessed as high risk for any domain were assessed overall as being at high risk of bias. Studies assessed as unclear risk for three or more domains, with no high-risk domains, were assessed overall as being at unclear risk of bias. Studies assessed as unclear risk for two or fewer domains, with no high-risk domains, were assessed overall as being at low risk of bias.
Data analysis
Data synthesis
Included studies were grouped by condition (asthma maintenance, asthma acute exacerbations and COPD) for analysis and the available outcome measures were tabulated for selection as described above. Where two or more studies within a group reported results for an outcome of interest, we performed meta-analysis using a random-effects model and inverse variance statistical methods using RevMan Web24. Where possible, we reported dichotomous outcomes as risk ratios (RRs) with 95% confidence intervals, and continuous outcomes as mean differences (where all studies used the same outcome measure), or standardised mean differences (where studies used a variety of outcome measures), with 95% confidence intervals.
Where additional studies reported outcome measures or data that could not be included in the meta-analysis, effect estimates were calculated where possible and all results were reported in table format. No synthesis was performed on these results.
Imputation and standardisation
Where results were reported with no measure of variance, we imputed standard deviations (SDs) for group-level data from other studies that used the same outcome measure, using the average of SDs from the intervention and control groups of the study with the highest available SD values for the same measure, as a conservative approach. Sensitivity analysis was conducted excluding studies with imputed data from the analysis, and in all cases no meaningful change on the overall meta-analysis results was observed. For standardised analyses, where studies used a range of outcome measures with opposing directions of effect (e.g. scales for which a lower score indicated improvement, and scales for which a higher score indicated improvement), results for the least frequent direction were multiplied by -1 in the analysis. Also, for standardised analyses, either change or endpoint scores were used for all included studies, based on which was the most frequently reported for that outcome. Where outcome data was converted from endpoint to change scores, appropriate measures of variance were also calculated where possible, or otherwise imputed as described above.
Heterogeneity and subgroup analyses
Heterogeneity was assessed using the I2 statistic. Exploration of the causes of heterogeneity was planned using subgroup analyses, including adults and adolescents (13+ years) vs children (up to 12 years), and SMI vs DPI inhalers. An additional subgroup analysis was conducted to explore the influence of study funding by the manufacturer of either the pMDI or non-pMDI inhalers, or neither. These subgroup analyses were conducted using the primary outcome of FEV1, which was commonly measured across the greatest number of studies.
Certainty in the evidence
The possibility of reporting bias was considered in the context of the available studies. The presence of small study effects that may indicate reporting bias was assessed using funnel plots where 10 or more studies contributed to a meta-analysis.
We assessed certainty in the evidence using the Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) approach25, incorporating the risk of bias, imprecision, inconsistency, indirectness and reporting bias. We summarised assessments for main outcomes in ‘summary of findings’ tables and used standardised language to report the certainty of results26.
Results
Description of studies
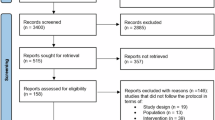
Of 3287 records identified in the search and 51 records identified from references of systematic reviews, 44 RCTs (reported in 46 papers) were included (32 on asthma maintenance therapy, five on acute asthma exacerbations, and seven on COPD) (Fig. 1 and Table 1). All studies of acute asthma exacerbations were in children. The majority of studies (38/44, 86.4%) were funded by device manufacturers, including all seven studies assessing patients with COPD (Table 1). Among these 38 studies, in 23 (60.5%) the study received funding from the manufacturer of the DPI/SMI device, in 10 (26.3%) the study received funding from the manufacturer of the pMDI device, and in 5 (13.2%) the same manufacturer produced the devices used in both study arms and funded the study (Table S5 in the Supplementary Information).
Flow diagram of the systematic review following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines.
Risk of bias assessments are summarised in Figure S1 in the Supplementary Information. Across all 44 studies, 21 were considered high-risk for bias, 6 were low-risk and the remaining 17 were unclear. Of those studies rated at high risk, the most frequent domain assessed as high risk of bias related to blinding of participants and personnel.
Analyses
Therapy via either a pMDI or non-pMDI device had similar effects on all outcomes of interest. Most evidence was considered moderate certainty, with certainty downgraded due to the proportion of studies at high or unclear risk of bias, but the observed results were highly robust to all sensitivity analyses, and imprecision and heterogeneity were both very low across most outcomes. No indication of reporting bias was identified, and small study effects were not detected in funnel plots for any meta-analyses. Summary of findings tables including GRADE assessments of the certainty of the evidence are presented in the Supplementary Information (Section C, Tables S6-S8).
Forced expiratory volume in 1 s (FEV1)
There was moderate certainty evidence of little or no difference in FEV1 between pMDI and non-pMDI devices for both asthma maintenance (standardised mean difference (SMD) 0.05, 95% CI 0 to 0.10; 29 studies; n = 9958) and COPD (SMD 0.03, 95% CI -0.03 to 0.09; seven studies; n = 3946) (Fig. 2). In asthma maintenance, this equates to a mean difference in percent predicted FEV1 of 0.71% (95% CI 0% to 1.42%), well below the MCID of 12%27 (see minimal clinically important differences in Table S4 in the Supplementary Information). The effect estimate among COPD patients is equivalent to a mean difference of 0.01 L (95% CI -0.01 L to 0.02 L), well below the MCID of 0.1 L28.
Meta-analysis of the association between device type and forced expiratory volume in 1 s (FEV1) in (A) asthma maintenance and (B) chronic obstructive pulmonary disease. The centre of the squares or diamonds indicates the point estimate and the width is the 95% confidence interval (CI). SE = standard error. SMD = standard mean difference. IV = inverse variance.
Only one study assessed FEV1 in acute asthma, which showed low certainty evidence of little to no difference in this population (mean difference 2% predicted, 95% CI -2.9% to 6.9%). One additional study reported FEV1 in asthma maintenance that could not be included in the meta-analysis (Table S9 in the Supplementary Information).
Peak expiratory flow rate (PEFR)
There was moderate certainty evidence of little or no difference in PEFR between pMDI and non-pMDI devices for asthma maintenance (mean difference 1.07 L/min, 95% CI -0.93 to 3.06; 26 studies; n = 8860) (Fig. 3), with these values well short of the MCID of 18.8 L/min for asthma maintenance studies29.
The centre of the squares or diamond indicates the point estimate and the width is the 95% confidence interval (CI). SE = standard error. MD = mean difference. IV = inverse variance.
There was low certainty evidence of little or no difference in PEFR between device types for acute asthma exacerbations (mean difference 1.38 L/min, 95% CI -17.97 to 20.73; two studies; n = 259). This difference is smaller than the MCID used of 12% predicted among adults with acute asthma exacerbations30 (which equates to approximately 25 L/min in paediatric populations, such as those in the two acute asthma studies included in our analysis). There was moderate certainty evidence of little or no difference among COPD patients (mean difference -2.56 L/min, 95% CI -9.17 to 4.05; two studies; n = 644) (Figure S3 in the Supplementary Information), acknowledging that there is no published MCID for PEFR in COPD.
There was one additional study that measured PEFR in asthma maintenance that could not be included in the meta-analysis (Table S10 in the Supplementary Information).
Use of reliever medication
There was moderate certainty evidence of little or no difference in use of reliever medication between pMDI and non-pMDI devices for both asthma maintenance (SMD 0.02, 95% CI -0.06 to 0.09; 13 studies; n = 4308) and COPD (mean difference -0.21 puffs/day, 95% CI = -0.51 to 0.10; three studies; n = 1265) (Figure S4 in the Supplementary Information). The asthma maintenance SMD of 0.02 equates to approximately 0.05 puffs/day (95% CI -0.16 to 0.23), well below the minimal patient perceivable improvement value of 0.81 puffs/day29.
There were seven additional studies that reported results on reliever use for asthma maintenance that could not be included in the meta-analysis (Table S11 in the Supplementary Information). This was not a relevant outcome for people with acute asthma exacerbations.
Symptom control
Regarding symptom control scores, for asthma maintenance there was moderate certainty evidence of little or no difference between pMDI and non-pMDI devices (SMD -0.04, 95% CI -0.11 to 0.02; eight studies; n = 3836). This SMD of -0.04 equates to approximately -0.02 points (95% CI -0.05 to 0.01) on the Asthma Control Questionnaire (ACQ-7), well below the MCID of 0.5 points. For COPD, using the COPD Assessment Test (CAT) score, there was moderate certainty evidence of little or no difference between pMDI and non-pMDI devices (mean difference -0.59 points, 95% CI = -1.19 to 0.01, one study; n = 1006). The observed mean difference was below the MCID of 2 points (Figure S5 in the Supplementary Information).
For acute asthma exacerbations, using the Modified Wood Clinical Asthma Score there was low certainty evidence of little or no difference between pMDI and non-pMDI devices (mean difference -0.1 points, 95% CI -0.72 to 0.52, one study; n = 32) (Figure S5 in the Supplementary Information).
There were eight additional studies for asthma maintenance and one additional study for acute asthma exacerbations that reported symptom control results that could not be included in the meta-analysis (Table S12 in the Supplementary Information).
Quality of life
Comparisons between studies were challenging due to the variety of quality of life scores used across studies. Meta-analysis was only possible for two asthma maintenance studies assessing the proportion of participants with an Asthma Quality of Life Questionnaire score improving ≥0.5 points. There was very low certainty evidence on the effect of device type on quality of life (relative risk (RR) 1.02, 95% CI 0.91 to 1.14; two studies; n = 871) (Figure S6 in the Supplementary Information). Seven further studies reported on quality of life measures (five for asthma maintenance, two for COPD) (Table S13 in the Supplementary Information).
Exacerbations
There was moderate certainty evidence of little or no difference between pMDI and non-pMDI devices in the risk of experiencing ≥1 exacerbation for both asthma maintenance (RR 0.87, 95% CI 0.72 to 1.05; 19 studies; n = 7532) and COPD (RR 1.08, 95% CI 0.94 to 1.24; seven studies; n = 4101) (Figure S7 in the Supplementary Information). This was not a relevant outcome for people with acute asthma exacerbations.
Adverse events
Results around AEs and SAEs – including those that were deemed to be treatment-related – are summarised in the Supplementary Information (Figures S8-S11). No clinically important difference was demonstrated between pMDI and non-pMDI devices for any condition. It should be noted that for severe and treatment-related adverse events, fewer studies were included in the analysis and fewer events reported, increasing the imprecision of the estimates and reducing the certainty in the estimate.
Mortality
No deaths were reported in any studies of asthma maintenance or acute asthma. For COPD, there was low certainty evidence of little or no difference between pMDI and non-pMDI devices (RR 1.40, 95% CI 0.59 to 3.32; six studies; n = 3657) (Figure S12 in the Supplementary Information).
Subgroup analyses
Subgroup analyses did not identify any factors that significantly modified the results, as little or no heterogeneity was observed. Studies in either children or adults were available for subgroup analysis by age group in asthma maintenance (Figure S13 in the Supplementary Information, test for subgroup difference P = 0.68). Studies using either SMI or DPI were available for subgroup analysis by non-pMDI device type in COPD (Figure S14 in the Supplementary Information, test for subgroup difference P = 0.48). Studies funded by the manufacturer of either the pMDI or non-pMDI device were available for subgrouping in asthma maintenance (Figure S15 in the Supplementary Information, test for subgroup difference P = 0.22).
Discussion
In this systematic review of 44 studies, we have found consistent evidence of no clinically meaningful difference between pMDIs and non-pMDI devices (DPIs and SMIs) in the management of asthma and COPD. The certainty of evidence was mainly rated as moderate rather than high due to the presence of some studies with high or unclear risk of bias and the possibility that biases could be operating in the direction of demonstrating equivalence. However, the overwhelming consistency of our findings and the robustness of these results to sensitivity analyses provides a clear conclusion. Results were uniform across a range of different outcomes, not just FEV1 and PEFR measurements but also markers of disease control (exacerbations and reliever use) – the latter being important given the greater sensitivity of symptom-based endpoints to detect meaningful differences31. Our results are consistent with more focused previous systematic reviews around two decades ago that also showed no differences between devices regarding clinical effectiveness and adverse events13,14,15,16. Importantly, our study contains updated data from newer studies, new drugs (or drug combinations), as well as a new device type (soft mist inhaler) that were not included in the previous reviews.
Climate change is the greatest global health threat of the twenty-first century32, and patients with respiratory diseases are among those projected to be affected disproportionately33. Globally, both governments and industry have made commitments to develop more sustainable healthcare systems, with a strong focus on decarbonisation. Increasing the proportion of inhalers without hydrofluorocarbons is an immediate step that can be taken to begin realising these ambitions. Our review supports that such a change may be possible without compromising patient care, though evidently the device class is only one of several important aspects of successful therapeutic changes. We acknowledge that there may be further issues relating to device selection and/or switching at an individual level, which are beyond the scope of this review and cannot be directly answered by our research findings. Furthermore, additional strategies may be employed to reduce inhaler-associated emissions – such as promoting uptake of propellants with a lower carbon footprint, improving inhaler disposal and recycling, reducing reliance on reliever treatments and improving disease control34,35,36.
Selecting the most appropriate inhaler device for an individual patient is a complicated and multifaceted decision. This choice will be influenced by factors including the available agents (e.g. extrafine therapies are only widely available in pMDIs), patient familiarity and preference, patient ability and dexterity, as well as cost and accessibility37. Increasingly, clinicians and patients are also being encouraged to consider the environmental impact of inhaler selection in position statements from expert bodies38 and decision aids39. There are important ethical issues around potentially trading patient preference for population health40, and avoiding any shaming of individuals who continue with pMDIs for strong user or disease reasons41. Nevertheless, multiple surveys have shown that for most respiratory patients the environmental impact of their inhaler is an important consideration and one that could lead them to seek a device change42,43,44.
When contemplating a widespread move away from pMDIs to other devices (primarily DPIs), two potential concerns should be considered. The first concern is the suitability of DPIs for subgroups with limited lung function (e.g. the very young, the very old, or those experiencing an exacerbation), due to the need to generate sufficient inspiratory flow for adequate drug delivery45. However, studies have shown that the majority of patients, including hospitalised patients nearing discharge, can achieve sufficient inspiratory flow to use an appropriately selected DPI46,47,48. A recent systematic review focusing of DPIs for primary school aged children with asthma found that the majority of children could use DPIs with adequate training and support49. Nevertheless, there are likely subpopulations for whom a pMDI will be the most appropriate device based on considerations of flow or other technical factors. The second concern is the potential negative impact of changing device type on disease control. Although the CRITIKAL study reported no clear difference between MDIs and DPIs in critical errors rates50, interpretation of findings across the wider inhaler-error literature remains challenging due to inconsistent definitions of errors and of what qualifies as ‘critical.’51 Any switching of individuals between devices requires investment of time in education and training. Outcomes may be worse if this is not undertaken, but large-scale real-world evidence suggests it can be undertaken successfully52,53,54,55. Changing inhaler devices does not have a predictable effect on outcomes56, and large-scale switching for non-clinical reasons may lead to worsened disease control57. Avoiding a loss of disease control is important not only for individual patient benefit, but also for sustainability. The greatest environmental impact of respiratory care comes from poor disease control leading to frequent use of reliever medications, unscheduled healthcare attendance and hospital admissions36. A preventer inhaler that an individual can and will use is therefore preferable to one which is “greener” but remains in the cupboard.
Some strengths of our research are the large number of included studies, and the robust methodologies that were employed. Our research does have some limitations. First, to ensure that solely the inhaler device was being compared, we only included studies with equivalent drugs and doses in each arm – this limited the pool of available studies but does increase the confidence in our findings. Second, we acknowledge that participants in clinical trials are not wholly representative of the wider population with asthma or COPD, which potentially casts some doubt on the ability to generalise these findings. However, limiting our assessment to RCTs helped to reduce unmeasured confounders and other influences on results. Additionally, “real world” trials and analyses of primary care databases have also shown non-inferiority of dry powder inhalers in broader populations “treated as” asthma or COPD, giving greater support to our review’s conclusions52,53,58. Third, the review contained a number of equivalence or non-inferiority trials, which may potentially lack assay sensitivity and the ability to detect a true difference if one exists59. However, the consistency of results across all conditions and subgroups assessed was notable, and the use of meta-analysis increases the combined power of the set of included studies to counter this limitation. Fourth, a number of pharmaceutical companies are presently developing new propellants with lower global warming potential34. While these propellants will not alter the validity of our systematic review’s findings, they may reduce the impetus to move patients away from pMDIs for environmental reasons. Finally, for our assessment of acute asthma management we were only able to identify studies focusing on children.
The safest and most productive way to implement greater use of inhalers that do not contain propellants with a high global warming potential is uncertain. This review provides reassurance to patients, clinicians, and researchers that – at least based on RCT data – DPIs and SMIs are equivalent to pMDIs when administering corresponding drugs and doses among subjects trained to satisfactorily use their device. It lays a strong basis upon which to undertake future studies assessing optimal strategies to reduce the overall environmental impact of inhaler therapy.
Data availability
Our research protocol is publicly available on the Monash University research repository (https://doi.org/10.26180/26065789.v2). The datasets generated and/or analysed during the current study are also available on the Monash University research repository (https://doi.org/10.26180/28916777.v2).
References
Safiri, S. et al. Burden of chronic obstructive pulmonary disease and its attributable risk factors in 204 countries and territories, 1990-2019: results from the Global Burden of Disease Study 2019. Bmj 378, e069679 (2022).
Wang, Z. et al. Global, regional, and national burden of asthma and its attributable risk factors from 1990 to 2019: a systematic analysis for the Global Burden of Disease Study 2019. Respir. Res. 24, 169 (2023).
Global Initiative for Asthma. Global Strategy for Asthma Management and Prevention, www.ginasthma.org (2023).
Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global Strategy for Diagnosis, Management and Prevention of COPD, www.goldcopd.org (2023).
MCTOC: Medical and Chemical Technical Options Committee. 2018 Assessment Report, https://ozone.unep.org/sites/default/files/2019-04/MCTOC-Assessment-Report-2018.pdf (2018).
Wilkinson, A. J. K., Braggins, R., Steinbach, I. & Smith, J. Costs of switching to low global warming potential inhalers. An economic and carbon footprint analysis of NHS prescription data in England. BMJ open. 9, e028763 (2019).
Lenzen, M. et al. The environmental footprint of health care: a global assessment. Lancet Planet. Health 4, e271–e279 (2020).
Pichler, P.-P., Jaccard, I. S., Weisz, U. & Weisz, H. International comparison of health care carbon footprints. Environ. Res. Lett. 14, 064004 (2019).
NHS England. Delivering a Net Zero National Health Service, https://www.england.nhs.uk/greenernhs/a-net-zero-nhs/ (2021).
Lavorini, F. et al. Retail sales of inhalation devices in European countries: so much for a global policy. Respir. Med. 105, 1099–1103 (2011).
Wilkinson, A. & Woodcock, A. The environmental impact of inhalers for asthma: A green challenge and a golden opportunity. Br. J. Clin. Pharmacol. 88, 3016–3022 (2022).
British Thoracic Society and Scottish Intercollegiate Guideline Network. BTS/SIGN British guideline on the management of asthma: A national clinical guideline, https://www.brit-thoracic.org.uk/quality-improvement/guidelines/asthma/ (2019).
Brocklebank, D., Wright, J. & Cates, C. Systematic review of clinical effectiveness of pressurised metered dose inhalers versus other hand held inhaler devices for delivering corticosteroids in asthma. Bmj 323, 896–900 (2001).
Dolovich, M. B. et al. Device selection and outcomes of aerosol therapy: Evidence-based guidelines: American College of Chest Physicians/American College of Asthma, Allergy, and Immunology. Chest 127, 335–371 (2005).
Ram, F. S., Brocklebank, D. M., Muers, M., Wright, J. & Jones, P. W. Pressurised metered-dose inhalers versus all other hand-held inhalers devices to deliver bronchodilators for chronic obstructive pulmonary disease. Cochrane Database Syst. Rev. 2002, Cd002170 (2002).
Ram, F. S., Wright, J., Brocklebank, D. & White, J. E. Systematic review of clinical effectiveness of pressurised metered dose inhalers versus other hand held inhaler devices for delivering beta (2)agonists bronchodilators in asthma. Bmj 323, 901–905 (2001).
Department of Health and Aged Care. National Health and Climate Strategy, https://www.health.gov.au/our-work/national-health-and-climate-strategy (2023).
Loftus M. J. et al. Systematic Review Protocol - Metered Dose inhalers versus Dry Powder Inhalers and Soft Mist Inhalers for Asthma and COPD, https://doi.org/10.26180/26065789.v2 (2024).
Page, M. J. et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Bmj 372, n71 (2021).
National Institute for Health and Care Excellence (NICE). Inhaled corticosteroid doses for NICE’s asthma guideline, https://www.nice.org.uk/guidance/ng80/resources/inhaled-corticosteroid-doses-pdf-4731528781 (2023).
Covidence systematic review software (Veritas Health Innovation, Melbourne, Australia, 2024).
Rohatgi A. WebPlotDigitizer version 5.1, https://automeris.io (2024).
Higgins J. P. T. & Altman D. G. in Cochrane Handbook for Systematic Reviews of Interventions (eds Higgins J. P. T. & Green S.) (John Wiley & Sons, 2008).
The Cochrane Collaboration. Review Manager (RevMan). Version 8.5.2, www.revman.cochrane.org (2024).
Schünemann HJ, H. J. et al. in Cochrane Handbook for Systematic Reviews of Interventions version 6.4 (ed Thomas J. Higgins J. P. T., Chandler J., Cumpston M., Li T., Page M. J., Welch V. A.) (Cochrane, 2023).
Santesso, N. et al. GRADE guidelines 26: informative statements to communicate the findings of systematic reviews of interventions. J. Clin. Epidemiol. 119, 126–135 (2020).
Pellegrino, R. et al. Interpretative strategies for lung function tests. Eur. Respir. J. 26, 948–968 (2005).
Donohue, J. F. Minimal clinically important differences in COPD lung function. Copd 2, 111–124 (2005).
Santanello, N. C., Zhang, J., Seidenberg, B., Reiss, T. F. & Barber, B. L. What are minimal important changes for asthma measures in a clinical trial?. Eur. Respir. J. 14, 23–27 (1999).
Karras, D. J. et al. Clinically meaningful changes in quantitative measures of asthma severity. Acad. Emerg. Med. 7, 327–334 (2000).
Dissanayake, S., Jain, M., Grothe, B., McIver, T. & Papi, A. An evaluation of comparative treatment effects with high and low dose fluticasone propionate/formoterol combination in asthma. Pulmonary Pharmacology Therapeutics 35, 19–27 (2015).
Costello, A. et al. Managing the health effects of climate change: lancet and university college london institute for global health commission. Lancet 373, 1693–1733 (2009).
Vicedo-Cabrera, A. M. et al. Climate change and respiratory health: a european respiratory society position statement. Eur. Respir. J. 62, 2201960 (2023).
Pernigotti, D. et al. Reducing carbon footprint of inhalers: analysis of climate and clinical implications of different scenarios in five European countries. BMJ Open. Respir. Res. 8, e001071 (2021).
Murphy, A., Howlett, D., Gowson, A. & Lewis, H. Understanding the feasibility and environmental effectiveness of a pilot postal inhaler recovery and recycling scheme. NPJ Prim. Care Respir. Med. 33, 5 (2023).
Wilkinson, A. J. K. et al. Greenhouse gas emissions associated with suboptimal asthma care in the UK: the SABINA healthCARe-Based envirONmental cost of treatment (CARBON) study. Thorax 79, 412–421 (2024).
Lavorini, F., Janson, C., Braido, F., Stratelis, G. & Løkke, A. What to consider before prescribing inhaled medications: a pragmatic approach for evaluating the current inhaler landscape. Ther. Adv. Respir. Dis. 13, 1753466619884532 (2019).
British Thoracic Society. Position Statement Sustainability and the Environment: Climate Change & Lung Health, https://www.brit-thoracic.org.uk/about-us/position-statements/ (2024).
National Institute for Health and Care Excellence (NICE). Patient Decision Aid: Inhalers for Asthma, https://www.nice.org.uk/guidance/ng80/resources/inhalers-for-asthma-patient-decision-aid-pdf-6727144573/ (2020).
Parker, J. Barriers to green inhaler prescribing: ethical issues in environmentally sustainable clinical practice. J. Med. Ethics 49, 92–98 (2023).
Keeley, D., Scullion, J. E. & Usmani, O. S. Minimising the environmental impact of inhaled therapies: problems with policy on low carbon inhalers. Eur. Respir. J. 55, 2001122 (2020).
D’Ancona, G., Cumella, A., Renwick, C. & Walker, S. The sustainability agenda and inhaled therapy: what do patients want?. European Respiratory Journal 58, PA3399 (2021).
Liew, K. & Wilkinson, A. P280 How do we choose inhalers? patient and physician perspectives on environmental, financial and ease-of-use factors. Thorax 72, A235–A237 (2017).
Metting, E. I., Dijk, L. V., Messlaki, H. E., Luers, J. & Kock, J. Development of a shared decision-making tool to support patients and their healthcare provider in choosing the best inhaler device. Eur. Respiratory J. 52, OA1643 (2018).
Woodcock, A. et al. The environmental impact of inhaled therapy: making informed treatment choices. Eur. Respir. J. 60, 2102106 (2022).
Clark, A. R., Weers, J. G. & Dhand, R. The confusing world of dry powder inhalers: it is all about inspiratory pressures, not inspiratory flow rates. J. Aerosol Med. Pulm. Drug. Deliv. 33, 1–11 (2020).
Mahler, D. A., Waterman, L. A., Ward, J. & Gifford, A. H. Comparison of dry powder versus nebulized beta-agonist in patients with COPD who have suboptimal peak inspiratory flow rate. J. Aerosol Med. Pulm. Drug. Deliv. 27, 103–109 (2014).
Sharma, G. et al. Prevalence of low peak inspiratory flow rate at discharge in patients hospitalized for COPD exacerbation. Chronic Obstr. Pulm. Dis. 4, 217–224 (2017).
Kuek, S. L. et al. Dry powder inhaler use in primary school aged children with asthma: a systematic review. ERJ Open. Res. 10, 00455–02024 (2024).
Price, D. B. et al. Inhaler errors in the CRITIKAL study: type, frequency, and association with asthma outcomes. J. Allergy Clin. Immunol. Pract. 5, 1071–1081.e1079 (2017).
Usmani, O. S. et al. Critical inhaler errors in asthma and COPD: a systematic review of impact on health outcomes. Respir. Res. 19, 10 (2018).
Vestbo, J. et al. Effectiveness of fluticasone furoate-vilanterol for COPD in clinical practice. N. Engl. J. Med. 375, 1253–1260 (2016).
Woodcock, A. et al. Effectiveness of fluticasone furoate plus vilanterol on asthma control in clinical practice: an open-label, parallel group, randomised controlled trial. Lancet 390, 2247–2255 (2017).
Jones, R. et al. The comparative effectiveness of initiating fluticasone/salmeterol combination therapy via pMDI versus DPI in reducing exacerbations and treatment escalation in COPD: a UK database study. Int. J. Chron. Obstruct Pulmon Dis. 12, 2445–2454 (2017).
Price, D. et al. Device type and real-world effectiveness of asthma combination therapy: an observational study. Respir. Med. 105, 1457–1466 (2011).
Usmani, O. S. et al. Real-world impact of nonclinical inhaler regimen switches on asthma or COPD: a systematic review. J. Allergy Clin. Immunol. Pract. 10, 2624–2637 (2022).
Thomas, M. et al. Inhaled corticosteroids for asthma: impact of practice level device switching on asthma control. BMC Pulm. Med. 9, 1 (2009).
Price, D. et al. Switching patients from other inhaled corticosteroid devices to the Easyhaler(®): historical, matched-cohort study of real-life asthma patients. J. Asthma Allergy 7, 31–51 (2014).
International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use ICH Harmonised Tripartite Guideline: Choice of Control Group and Related Issues in Clinical Trials E10. ICH: Geneva, 2000.
Amar, N. J., Shekar, T., Varnell, T. A., Mehta, A. & Philip, G. Mometasone furoate (MF) improves lung function in pediatric asthma: A double-blind, randomized controlled dose-ranging trial of MF metered-dose inhaler. Pediatr. Pulmonol. 52, 310–318 (2017).
Corrigendum. Pediatr Pulmonol. 54: 655–656. https://doi.org/10.1002/ppul.24291.
Barnes, N. et al. Stepping-across controlled asthmatic patients to extrafine beclometasone/formoterol combination. Pulm. Pharmacol. Ther. 26, 555–561 (2013).
Bateman, E. D., Silins, V. & Bogolubov, M. Clinical equivalence of salmeterol/fluticasone propionate in combination (50/100 microg twice daily) when administered via a chlorofluorocarbon-free metered dose inhaler or dry powder inhaler to patients with mild-to-moderate asthma. Respir. Med. 95, 136–146 (2001).
Bernstein, D. I. et al. Efficacy and onset of action of mometasone furoate/formoterol and fluticasone propionate/salmeterol combination treatment in subjects with persistent asthma. Allergy Asthma Clin. Immunol. 7, 21 (2011).
Bodzenta-Lukaszyk, A. et al. Fluticasone/formoterol combination therapy versus budesonide/formoterol for the treatment of asthma: a randomized, controlled, non-inferiority trial of efficacy and safety. J. Asthma 49, 1060–1070 (2012).
Bracamonte, T., Schauer, U., Emeryk, A., Godwood, A. & Balsara, S. Efficacy and safety of salmeterol/fluticasone propionate combination delivered by the diskustrade mark or pressurised metered-dose inhaler in children with asthma. Clin. Drug. Investig. 25, 1–11 (2005).
Bronsky, E. et al. Comparison of inhaled albuterol powder and aerosol in asthma. J. Allergy Clin. Immunol. 79, 741–747 (1987).
Busse, W. W. et al. Comparison of adjustable- and fixed-dose budesonide/formoterol pressurized metered-dose inhaler and fixed-dose fluticasone propionate/salmeterol dry powder inhaler in asthma patients. J. Allergy Clin. Immunol. 121, 1407–1414 (2008). 1414.e1401-1406.
O’Connor, R. D., Patrick, D. L., Parasuraman, B., Martin, P. & Goldman, M. Comparison of patient-reported outcomes during treatment with adjustable- and fixed-dose budesonide/formoterol pressurized metered-dose inhaler versus fixed-dose fluticasone propionate/salmeterol dry powder inhaler in patients with asthma. J. Asthma 47, 217–223 (2010).
Dusser, D., Vicaut, E. & Lefrançois, G. Double-blind, double-dummy, multinational, multicenter, parallel-group design clinical trial of clinical non-inferiority of formoterol 12 microg/unit dose in a b.i.d. regimen administered via an HFA-propellant-pMDI or a dry powder inhaler in a 12-week treatment period of moderate to severe stable persistent asthma in adult patients. Respiration 72, 20–27 (2005).
Kanniess, F., Scuri, M., Vezzoli, S., Francisco, C. & Petruzzelli, S. Extrafine beclomethasone/formoterol combination via a dry powder inhaler (NEXThaler(®)) or pMDI and beclomethasone monotherapy for maintenance of asthma control in adult patients: A randomised, double-blind trial. Pulm. Pharmacol. Ther. 30, 121–127 (2015).
Kemp, J. P. et al. Albuterol treatment for children with asthma: a comparison of inhaled powder and aerosol. J. Allergy Clin. Immunol. 83, 697–702 (1989).
Koskela, T. et al. Equivalence of two steroid-containing inhalers: easyhaler multidose powder inhaler compared with conventional aerosol with large-volume spacer. Respiration 67, 194–202 (2000).
Lundback, B. et al. Evaluation of fluticasone propionate (500 micrograms day-1) administered either as dry powder via a Diskhaler inhaler or pressurized inhaler and compared with beclomethasone dipropionate (1000 micrograms day-1) administered by pressurized inhaler. Respir. Med. 87, 609–620 (1993).
Lundback, B. et al. A comparison of fluticasone propionate when delivered by either the metered-dose inhaler or the Diskhaler inhaler in the treatment of mild-to-moderate asthma. Eur. J. Clin. Res. 5, 11–19 (1994).
Morice, A. H., Peterson, S., Beckman, O. & Osmanliev, D. Therapeutic comparison of a new budesonide/formoterol pMDI with budesonide pMDI and budesonide/formoterol DPI in asthma. Int. J. Clin. Pract. 61, 1874–1883 (2007).
Morice, A. H., Hochmuth, L., Ekelund, J., Thorén, A. & Puterman, A. S. Comparable long-term safety and efficacy of a novel budesonide/formoterol pressurized metered-dose inhaler versus budesonide/formoterol Turbuhaler in adolescents and adults with asthma. Pulm. Pharmacol. Ther. 21, 32–39 (2008).
Nelson, H., Kemp, J. P., Bieler, S., Vaughan, L. M. & Hill, M. R. Comparative efficacy and safety of albuterol sulfate Spiros inhaler and albuterol metered-dose inhaler in asthma. Chest 115, 329–335 (1999).
Papi, A., Paggiaro, P. L., Nicolini, G., Vignola, A. M. & Fabbri, L. M. Beclomethasone/formoterol versus budesonide/formoterol combination therapy in asthma. Eur. Respir. J. 29, 682–689 (2007).
Papi, A. et al. Step-down from high dose fixed combination therapy in asthma patients: a randomized controlled trial. Respir. Res. 13, 54 (2012).
Pauwels, R. A., Hargreave, F. E., Camus, P., Bukoski, M. & Ståhl, E. A 1-year comparison of turbuhaler vs pressurized metered-dose inhaler in asthmatic patients. Chest 110, 53–57 (1996).
Poukkula, A. et al. Comparison of a multidose powder inhaler containing beclomethasone dipropionate (BDP) with a BDP metered dose inhaler with spacer in the treatment of asthmatic patients. Clin. Drug. Investig. 16, 101–110 (1998).
Reichel, W. et al. Extrafine beclomethasone dipropionate breath-actuated inhaler (400 micrograms/day) versus budesonide dry powder inhaler (800 micrograms/day) in asthma. Int. J. Clin. Pract. 55, 100–106 (2001).
Srichana, T., Juthong, S., Thawithong, E., Supaiboonpipat, S. & Soorapan, S. Clinical equivalence of budesonide dry powder inhaler and pressurized metered dose inhaler. Clin. Respir. J. 10, 74–82 (2016).
Stradling, J. R., Pearson, M. G., Morice, A. H., Peake, M. D. & Barnes, N. C. Efficacy and safety of a novel beclomethasone dipropionate dry powder inhaler (Clickhaler) for the treatment of adult asthma. Amsterdam Clinical Study Group. J. Asthma 37, 183–190 (2000).
van Noord, J. A., Lill, H., Diaz, T. C., Greefhorst, A. P. & Davies, P. Clinical equivalence of a salmeterol/fluticasone propionate combination product (50/500μg) delivered via a chlorofluorocarbon-free metered-dose inhaler with the diskus™ in patients with moderate to severe asthma. Clin. Drug. Investigation 21, 243–255 (2001).
Vincken, W., Bantje, T., Middle, M. V., Gerken, F. & Moonen, D. Long-term efficacy and safety of ipratropium bromide plus fenoterol via respimat((R)) soft misttrade mark inhaler (SMI) versus a pressurised metered-dose inhaler in asthma. Clin. Drug. Investig. 24, 17–28 (2004).
von Berg, A. et al. Efficacy and safety of ipratropium bromide plus fenoterol inhaled via respimat soft mist inhaler vs. a conventional metered dose inhaler plus spacer in children with asthma. Pediatr. Pulmonol. 37, 264–272 (2004).
von Berg, A. et al. Comparison of the efficacy and safety of ciclesonide 160 microg once daily vs. budesonide 400 microg once daily in children with asthma. Pediatr. Allergy Immunol. 18, 391–400 (2007).
Wardlaw, A. et al. Efficacy and safety of mometasone furoate dry powder inhaler vs fluticasone propionate metered-dose inhaler in asthma subjects previously using fluticasone propionate. Ann. Allergy Asthma Immunol. 93, 49–55 (2004).
Wolfe, J. et al. Comparison of powder and aerosol formulations of salmeterol in the treatment of asthma. Ann. Allergy Asthma Immunol. 84, 334–340 (2000).
Zheng, J. et al. Comparison of extrafine beclomethasone dipropionate/formoterol fumarate dry powder inhaler and pressurized metered-dose inhaler in Chinese patients with asthma: the FORTUNE study. J. Asthma 61, 360–367 (2024).
Zhou, Y. et al. Efficacy of inhaler devices in new-onset mild asthma: a randomized blinded multicenter trial. Adv. Ther. 42, 3861–3881 (2025).
Direkwatanachai, C. et al. Comparison of salbutamol efficacy in children-via the metered-dose inhaler (MDI) with Volumatic spacer and via the dry powder inhaler, Easyhaler, with the nebulizer-in mild to moderate asthma exacerbation: a multicenter, randomized study. Asian Pac. J. Allergy Immunol. 29, 25–33 (2011).
Drblik, S. et al. Comparative efficacy of terbutaline sulphate delivered by Turbuhaler dry powder inhaler or pressurised metered dose inhaler with Nebuhaler spacer in children during an acute asthmatic episode. Arch. Dis. Child. 88, 319–323 (2003).
Khaled, M. S., Akter, F., Rahman, K., Ullah, M. S. & Rahman, M. H. Bronchodilator response to salbutamol delivered by metered dose inhaler with spacer and dry powder inhaler in acute asthma in children: a comparative study. Bangladesh J. Child. Health 38, 68–73 (2014).
Lodha, R., Gupta, G., Baruah, B. P., Nagpal, R. & Kabra, S. K. Metered dose inhaler with spacer versus dry powder inhaler for delivery of salbutamol in acute exacerbations of asthma: a randomized controlled trial. Indian. Pediatr. 41, 15–20 (2004).
Vangveeravong, M. A comparative study of efficacy of salbutamol via metered dose inhaler with volumatic spacer and via dry powder inhaler, easyhaler, to nebulization in mild to moderate severity acute asthma exacerbation in childhood. J. Med. Assoc. Thai 91(Suppl 3), S115–S123 (2008).
Ferguson, G. T., Ghafouri, M., Dai, L. & Dunn, L. J. COPD patient satisfaction with ipratropium bromide/albuterol delivered via Respimat: a randomized, controlled study. Int. J. Chron. Obstruct Pulmon Dis. 8, 139–150 (2013).
Ferguson, G. T. et al. Budesonide/formoterol MDI with co-suspension delivery technology in COPD: the TELOS study. Eur. Respir. J. 52, 1801334 (2018).
Kilfeather, S. A. et al. Improved delivery of ipratropium bromide/fenoterol from Respimat Soft Mist Inhaler in patients with COPD. Respir. Med. 98, 387–397 (2004).
Koser, A., Westerman, J., Sharma, S., Emmett, A. & Crater, G. D. Safety and efficacy of fluticasone propionate/salmeterol hydrofluoroalkane 134a metered-dose-inhaler compared with fluticasone propionate/salmeterol diskus in patients with chronic obstructive pulmonary disease. Open. Respir. Med. J. 4, 86–91 (2010).
Maltais, F. et al. A randomized, double-blind, double-dummy study of glycopyrrolate/formoterol fumarate metered dose inhaler relative to umeclidinium/vilanterol dry powder inhaler in COPD. Adv. Ther. 36, 2434–2449 (2019).
Wang, C. et al. Efficacy and safety of budesonide/glycopyrrolate/formoterol fumarate metered dose inhaler in chinese patients with COPD: a subgroup analysis of KRONOS. Adv. Ther. 37, 1591–1607 (2020).
Zuwallack, R. et al. Efficacy and safety of ipratropium bromide/albuterol delivered via respimat inhaler versus MDI. Respir. Med. 104, 1179–1188 (2010).
Acknowledgements
Thanks to Dr Kim Jachno of the Australian Living Evidence Collaboration in the School of Public Health and Preventive Medicine, Monash University, for statistical advice. Michael Loftus is supported by a Royal Australasian College of Physicians (RACP) Research Establishment Fellowship. Miranda Cumpston is supported by philanthropic research funding from the Walter Thomas Cottman Charitable Trust and The Phyllis Connor Memorial Trust, managed by Equity Trustees. The funders played no role in study design, data collection, analysis and interpretation of data, or the writing of this manuscript.
Author information
Authors and Affiliations
Contributions
Author Contribution Statement : MJL: conceptualization, methodology, investigation, formal analysis, writing – original draft, writing – review and editing. MSC: methodology, investigation, visualisation, formal analysis, writing – original draft, writing – review and editing. SB: investigation, writing – review and editing. JB: conceptualization, methodology, supervision, writing – original draft, writing – original draft, writing – review and editing. AG: conceptualization, methodology, supervision, writing – original draft, writing – original draft, writing – review and editing. SM: methodology, investigation, writing – review and editing. LP: investigation, writing – review and editing. MR: conceptualization, methodology, supervision, writing – original draft, writing – original draft, writing – review and editing. RS: investigation, writing – review and editing. HW: methodology, investigation, writing – review and editing. TT: conceptualization, methodology, supervision, writing – review and editing. KL: conceptualization, methodology, supervision, writing – original draft, writing – original draft, writing – review and editing. All authors (except AG, who died in December 2024) read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
Author JB declares personal and institutional fees from Chiesi, Boehringer Ingelheim, GSK, AstraZeneca and Sanofi for educational presentations or speaking engagements, as well as support for travelling to conferences from GSK and AstraZeneca. Author JB has participated in a study steering committee for GSK, unrelated to this topic. All other authors declare no financial or non-financial competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Loftus, M.J., Cumpston, M.S., Barnes, S. et al. Efficacy and safety of different inhaler types for asthma and chronic obstructive pulmonary disease. a systematic review and meta-analysis. npj Prim. Care Respir. Med. 36, 18 (2026). https://doi.org/10.1038/s41533-026-00488-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41533-026-00488-4