Abstract
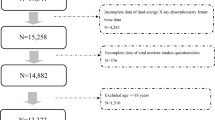
Chronic obstructive pulmonary disease (COPD) and osteoporosis are significant public health concerns, often co-occurring due to shared risk factors such as ageing, smoking, and systemic inflammation, as well as treatment-related factors such as long-term glucocorticoid use. However, large-scale studies exploring these associations, their sex-specific effects, and mediating factors remain limited. A total of 8,274 participants aged ≥50 years from NHANES cycles 2005–2018 were included. COPD and osteoporosis were identified based on self-reported diagnoses, with Bone Mineral Density (BMD), measured by Dual-energy X-ray Absorptiometry (DXA), used as a sensitivity outcome. Weighted logistic regression analyzed the association between COPD and osteoporosis. Interaction and stratified analyses explored effect modification by sex, BMI, prednisone use, vitamin D, and race. Exploratory mediation analysis examined the indirect effects of prednisone, sleep problems, and vitamin D. COPD was significantly associated with osteoporosis risk (OR = 2.24, P < 0.001). A nominal sex interaction was observed (unadjusted P = 0.03), with a stronger association in males (adjusted OR = 4.85, 95% CI: 2.49–9.42, P < 0.001) than females (adjusted OR = 1.86, 95% CI: 1.30–2.65, P < 0.001). Exploratory mediation analyses suggested that prednisone use (mediated 5.1%) and sleep problems (mediated 9.3%) accounted for portions of the association, while vitamin D level did not show meaningful mediation. Sensitivity analyses confirmed an association between COPD and lower BMD (β = −0.032, P < 0.001), with significant mediation by prednisone (2.2%, P = 0.034). COPD is significantly associated with osteoporosis, with a stronger relative effect observed in males. Exploratory findings suggest potential mediation by prednisone use and sleep disorders. These results highlight the importance of integrated bone health management in COPD patients, with particular attention to sex-specific risks and modifiable factors such as glucocorticoid exposure and sleep quality.

Similar content being viewed by others
Data availability
This study used publicly available data from the National Health and Nutrition Examination Survey (NHANES). The datasets analysed are available from the NHANES website (https://www.cdc.gov/nchs/nhanes/) following the survey’s data-use guidelines.
References
Organization, W. H. Chronic obstructive pulmonary disease (COPD), <https://www.who.int/news-room/fact-sheets/detail/chronic-obstructive-pulmonary-disease-(copd)> (2024).
Adeloye, D. et al. Global, regional, and national prevalence of, and risk factors for, chronic obstructive pulmonary disease (COPD) in 2019: a systematic review and modelling analysis. Lancet Respir. Med. 10, 447–458 (2022).
Compston, J. E., McClung, M. R. & Leslie, W. D. Osteoporosis. Lancet 393, 364–376 (2019).
Johnell, O. & Kanis, J. A. An estimate of the worldwide prevalence and disability associated with osteoporotic fractures. Osteoporos. Int. 17, 1726–1733 (2006).
Zhao, G. et al. Potential drug targets for osteoporosis identified: A Mendelian randomization study. Heliyon 10, e36566. https://doi.org/10.1016/j.heliyon.2024.e36566 (2024).
Du, X. et al. Research progress in the mechanism of calcium ion on contraction and relaxation of airway smooth muscle cells. J. Recept. Signal. Transduct. Res. 41, 117–122 (2021).
Inoue, D., Watanabe, R. & Okazaki, R. COPD and osteoporosis: links, risks, and treatment challenges. Int. J. Chron. Obstruct Pulmon Dis. 11, 637–648 (2016).
Chan, Y. H., Teo, C. B., Tay, J. K. & Cheong, C. S. The association between obstructive sleep apnea and osteoporosis: A systematic review and meta-analysis. Sleep. Med. Rev. 78, 102006. https://doi.org/10.1016/j.smrv.2024.102006 (2024).
Kalyanaraman, H., Pal China, S., Casteel, D. E. & Pilz, R. B. Crosstalk between androgen receptor and protein kinase G signaling in bone: Implications for osteoporosis therapy. Trends Pharmacol. Sci. 46, 279–294 (2025).
Mohanty, S. et al. Molecular mechanisms and treatment strategies for estrogen deficiency-related and glucocorticoid-induced osteoporosis: a comprehensive review. Inflammopharmacology, https://doi.org/10.1007/s10787-025-01749-3 (2025).
MacGregor, K. A., Gallagher, I. J. & Moran, C. N. Relationship between insulin sensitivity and menstrual cycle is modified by BMI, fitness, and physical activity in NHANES. J. Clin. Endocrinol. Metab. 106, 2979–2990 (2021).
Benowitz, N. L., Bernert, J. T., Caraballo, R. S., Holiday, D. B. & Wang, J. Optimal serum cotinine levels for distinguishing cigarette smokers and nonsmokers within different racial/ethnic groups in the United States between 1999 and 2004. Am. J. Epidemiol. 169, 236–248 (2009).
Wang, D. & Yang, Y. The relationship between Serum 25-Hydroxyvitamin D levels and osteoporosis in postmenopausal women. Clin. Interv. Aging 18, 619–627 (2023).
Weinstein, R. S. Glucocorticoids, osteocytes, and skeletal fragility: The role of bone vascularity. Bone 46, 564–570 (2010).
Aguirre-Quezada, M. A., Aranda-Ramirez, M. P., Del Carmen-Garcia, M. & Reivan-Ortiz, G. Association of blood pressure with metabolic factors, stress levels, physical activity, and nutrient intake in overweight or obese ecuadorian university students: A study based on mediation analysis. Nutrients 18, 201 (2026).
Tang, D., Yang, J., Zhang, Y. & He, J. Q. Risk factors for sputum smear-positive in COPD patients first diagnosed with active tuberculosis. Microb. Pathog. 213, 108319 (2026).
Li, J., Zhao, H., Wang, F., Wang, C. & Pan, J. Socioeconomic status influences the relationship between residential green space and the risk of osteoporosis among rural adults: a large-scale population-based study. Front. Public. Health 13, 1695153 (2025).
Kurabayash, T. et al. Importance of changes in body mass index from adolescence to middle age as a risk factor for osteoporosis: the Japan Nurses’ Health Study. Menopause, https://doi.org/10.1097/GME.0000000000002740 (2026).
Graumam, R. Q., Pinheiro, M. M., Nery, L. E. & Castro, C. H. M. Increased rate of osteoporosis, low lean mass, and fragility fractures in COPD patients: association with disease severity. Osteoporos. Int. 29, 1457–1468 (2018).
Wang, T. & He, C. TNF-alpha and IL-6: The Link between Immune and Bone System. Curr. Drug. Targets 21, 213–227 (2020).
Chen, T., Jin, L., Li, J. & Liu, Y. Pyroptosis mediates osteoporosis via the inflammation immune microenvironment. Front. Immunol. 15, 1371463 (2024).
Kaneki, H. et al. Tumor necrosis factor promotes Runx2 degradation through up-regulation of Smurf1 and Smurf2 in osteoblasts. J. Biol. Chem. 281, 4326–4333 (2006).
Azuma, Y., Kaji, K., Katogi, R., Takeshita, S. & Kudo, A. Tumor necrosis factor-alpha induces differentiation of and bone resorption by osteoclasts. J. Biol. Chem. 275, 4858–4864 (2000).
Zhang, H. et al. NOTCH inhibits osteoblast formation in inflammatory arthritis via noncanonical NF-kappaB. J. Clin. Invest. 124, 3200–3214 (2014).
Diarra, D. et al. Dickkopf-1 is a master regulator of joint remodeling. Nat. Med. 13, 156–163 (2007).
Wang, X. et al. N‑acetyl cysteine inhibits the lipopolysaccharide‑induced inflammatory response in bone marrow mesenchymal stem cells by suppressing the TXNIP/NLRP3/IL‑1beta signaling pathway. Mol. Med. Rep. 22, 3299–3306 (2020).
Wang, L. et al. NLRP3 inflammasome activation in mesenchymal stem cells inhibits osteogenic differentiation and enhances adipogenic differentiation. Biochem. Biophys. Res. Commun. 484, 871–877 (2017).
Kaneshiro, S. et al. IL-6 negatively regulates osteoblast differentiation through the SHP2/MEK2 and SHP2/Akt2 pathways in vitro. J. Bone Miner. Metab. 32, 378–392 (2014).
Franchimont, N., Gangji, V., Durant, D. & Canalis, E. Interleukin-6 with its soluble receptor enhances the expression of insulin-like growth factor-I in osteoblasts. Endocrinology 138, 5248–5255 (1997).
Yeh, L. C., Zavala, M. C. & Lee, J. C. Osteogenic protein-1 and interleukin-6 with its soluble receptor synergistically stimulate rat osteoblastic cell differentiation. J. Cell Physiol. 190, 322–331 (2002).
Ross, F. P. & Teitelbaum, S. L. alphavbeta3 and macrophage colony-stimulating factor: partners in osteoclast biology. Immunol. Rev. 208, 88–105 (2005).
Srivastava, R. K., Dar, H. Y. & Mishra, P. K. Immunoporosis: Immunology of osteoporosis-role of T cells. Front. Immunol. 9, 657 (2018).
Hodge, J. M., Collier, F. M., Pavlos, N. J., Kirkland, M. A. & Nicholson, G. C. M-CSF potently augments RANKL-induced resorption activation in mature human osteoclasts. PLoS one 6, e21462 (2011).
Theill, L. E., Boyle, W. J. & Penninger, J. M. RANK-L and RANK: T cells, bone loss, and mammalian evolution. Annu. Rev. Immunol. 20, 795–823 (2002).
Lam, J. et al. TNF-alpha induces osteoclastogenesis by direct stimulation of macrophages exposed to permissive levels of RANK ligand. J. Clin. Invest. 106, 1481–1488 (2000).
Takayanagi, H., Kim, S. & Taniguchi, T. Signaling crosstalk between RANKL and interferons in osteoclast differentiation. Arthritis Res. 4, S227–S232 (2002). Suppl 3.
Wong, B. R. et al. The TRAF family of signal transducers mediates NF-kappaB activation by the TRANCE receptor. J. Biol. Chem. 273, 28355–28359 (1998).
Takayanagi, H. et al. T-cell-mediated regulation of osteoclastogenesis by signalling cross-talk between RANKL and IFN-gamma. Nature 408, 600–605 (2000).
Cheung, W. Y., Liu, C., Tonelli-Zasarsky, R. M., Simmons, C. A. & You, L. Osteocyte apoptosis is mechanically regulated and induces angiogenesis in vitro. J. Orthop. Res. 29, 523–530 (2011).
Tang, M., Lu, L. & Yu, X. Interleukin-17A Interweaves the Skeletal and Immune Systems. Front. Immunol. 11, 625034 (2020).
Mansoori, M. N. et al. IL-18BP is decreased in osteoporotic women: Prevents Inflammasome mediated IL-18 activation and reduces Th17 differentiation. Sci. Rep. 6, 33680 (2016).
Palmqvist, P., Persson, E., Conaway, H. H. & Lerner, U. H. IL-6, leukemia inhibitory factor, and oncostatin M stimulate bone resorption and regulate the expression of receptor activator of NF-kappa B ligand, osteoprotegerin, and receptor activator of NF-kappa B in mouse calvariae. J. Immunol. 169, 3353–3362 (2002).
O’Brien, C. A., Lin, S. C., Bellido, T. & Manolagas, S. C. Expression levels of gp130 in bone marrow stromal cells determine the magnitude of osteoclastogenic signals generated by IL-6-type cytokines. J. Cell Biochem. 79, 532–541 (2000).
Kudo, O. et al. Interleukin-6 and interleukin-11 support human osteoclast formation by a RANKL-independent mechanism. Bone 32, 1–7 (2003).
Yoshitake, F., Itoh, S., Narita, H., Ishihara, K. & Ebisu, S. Interleukin-6 directly inhibits osteoclast differentiation by suppressing receptor activator of NF-kappaB signaling pathways. J. Biol. Chem. 283, 11535–11540 (2008).
Duplomb, L. et al. Interleukin-6 inhibits receptor activator of nuclear factor kappaB ligand-induced osteoclastogenesis by diverting cells into the macrophage lineage: key role of Serine727 phosphorylation of signal transducer and activator of transcription 3. Endocrinology 149, 3688–3697 (2008).
Alam, M. I. et al. NLRP3 inflammasome negatively regulates RANKL-Induced osteoclastogenesis of mouse bone marrow macrophages but positively regulates it in the presence of lipopolysaccharides. Int. J. Mol. Sci. 23, 6096 (2022).
Ding, J. et al. Pore-forming activity and structural autoinhibition of the gasdermin family. Nature 535, 111–116 (2016).
Wu, Y. L. et al. Propionate and butyrate attenuate macrophage pyroptosis and osteoclastogenesis induced by CoCrMo alloy particles. Mil. Med. Res. 9, 46 (2022).
Place, D. E. & Kanneganti, T. D. Recent advances in inflammasome biology. Curr. Opin. Immunol. 50, 32–38 (2018).
Coll, R. C. et al. A small-molecule inhibitor of the NLRP3 inflammasome for the treatment of inflammatory diseases. Nat. Med. 21, 248–255 (2015).
Ni, B. et al. MCC950, the NLRP3 inhibitor, protects against cartilage degradation in a mouse model of osteoarthritis. Oxid. Med. Cell Longev. 2021, 4139048 (2021).
Jilka, R. L. et al. Increased osteoclast development after estrogen loss: mediation by interleukin-6. Science 257, 88–91 (1992).
Roggia, C. et al. Up-regulation of TNF-producing T cells in the bone marrow: a key mechanism by which estrogen deficiency induces bone loss in vivo. Proc. Natl Acad. Sci. USA 98, 13960–13965 (2001).
Wang, D., Ruan, W., Peng, Y. & Li, W. Sleep duration and the risk of osteoporosis among middle-aged and elderly adults: a dose-response meta-analysis. Osteoporos. Int. 29, 1689–1695 (2018).
Irwin, M. R., Olmstead, R. & Carroll, J. E. Sleep disturbance, sleep duration, and inflammation: A systematic review and meta-analysis of cohort studies and experimental sleep deprivation. Biol. Psychiatry 80, 40–52 (2016).
D’Cruz, R. F., Murphy, P. B. & Kaltsakas, G. Sleep disordered breathing and chronic obstructive pulmonary disease: A narrative review on classification, pathophysiology and clinical outcomes. J. Thorac. Dis. 12, S202–S216 (2020).
Li, S. Q. et al. Impact of insomnia and obstructive sleep apnea on the risk of acute exacerbation of chronic obstructive pulmonary disease. Sleep. Med. Rev. 58, 101444 (2021).
Humphrey, M. B. et al. 2022 American college of rheumatology guideline for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Care Res. 75, 2405–2419 (2023).
Balasubramanian, A. et al. Glucocorticoid exposure and fracture risk in a cohort of US patients with selected conditions. J. Bone Min. Res. 33, 1881–1888 (2018).
Tanaka, Y. et al. The 2023 Guidelines for the management and treatment of glucocorticoid-induced osteoporosis. J. bone Miner. Metab. 42, 143–154 (2024).
Ru, J. Y. & Wang, Y. F. Osteocyte apoptosis: the roles and key molecular mechanisms in resorption-related bone diseases. Cell Death Dis. 11, 846 (2020).
Hu, L. et al. Mesenchymal stem cells: Cell fate decision to osteoblast or adipocyte and application in osteoporosis treatment. Int. J. Mol. Sci. 19, 360 (2018).
Lee, J. E., Park, S., Kim, Y., Wi, S. & Kim, Y. T. Novel evidence in vivo: Berberine ameliorated glucocorticoid-induced post-natal growth retardation by regulating the GH/IGF-1 axis through KMT1A downregulation. Toxicol. Appl. Pharmacol. 500, 117362 (2025).
Locatelli, V. & Bianchi, V. E. Effect of GH/IGF-1 on Bone Metabolism and Osteoporsosis. Int. J. Endocrinol. 2014, 235060 (2014).
Chen, Y. C., Greenbaum, J., Shen, H. & Deng, H. W. Association between gut microbiota and bone health: Potential mechanisms and prospective. J. Clin. Endocrinol. Metab. 102, 3635–3646 (2017).
Dong, Y. et al. Modulation of the gut-bone axis: Lacticaseibacillus paracasei LC86 improves bone health via anti-inflammatory metabolic pathways in zebrafish models of osteoporosis and cartilage damage. Front. Immunol. 16, 1493560 (2025).
Paccou, J. et al. Prevention and treatment of glucocorticoid-induced osteoporosis in adults: recommendations from the European Calcified Tissue Society. Eur. J. Endocrinol. 191, G1–G17 (2024).
Khan, M., Jose, A. & Sharma, S. in StatPearls (2025).
Aryal, S., Diaz-Guzman, E. & Mannino, D. M. COPD and gender differences: an update. Transl. Res. 162, 208–218 (2013).
Su, L. et al. Association between systemic immune inflammation index and serum testosterone and free testosterone in middle-aged and elderly men. Andrology, https://doi.org/10.1111/andr.70036 (2025).
Cover, P. O., Baanah-Jones, F., John, C. D. & Buckingham, J. C. Annexin 1 (lipocortin 1) mimics inhibitory effects of glucocorticoids on testosterone secretion and enhances effects of interleukin-1beta. Endocrine 18, 33–39 (2002).
O’Brien, C. J. O. Macrophage regulation of hypothalamic-pituitary-adrenal and gonadal axis homeostasis and hormonal output. Biomed J, 100866, https://doi.org/10.1016/j.bj.2025.100866 (2025).
Acknowledgements
We would like to express our sincere thanks to all participants in the NHANES survey. This research was supported by the 2024 Shantou Science and Technology Plan Medical and Health Project (Grant No: 240506186498668, 240922169602002).
Author information
Authors and Affiliations
Contributions
YG: Conceptualization, Data curation, Formal Analysis, Visualization, Software, Writing - original draft, Writing - review & editing. JW: Data curation, Software, Visualization, Writing - review & editing. ZL: Data curation, Writing - review & editing. LD: Data curation, Conceptualization, Writing - review & editing. MM: Methodology, Supervision, Writing - review & editing. LL: Conceptualization, Methodology, Writing - review & editing. XC:Methodology, Writing - review & editing. ZZ: Conceptualization, Methodology, Project administration, Supervision, Writing - review & editing. SC: Project administration, Methodology, Supervision, Funding acquisition, Writing - review & editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Gao, Y., Li, Z., Wu, J. et al. The risk of osteoporosis in COPD: An analysis of sex differences and mediating effects based on NHANES. npj Prim. Care Respir. Med. (2026). https://doi.org/10.1038/s41533-026-00490-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41533-026-00490-w