Abstract
Human skin is repaired by scar formation, lacking hair follicles, arrector pili muscles, and targeted innervation. Scarring leads to significant losses in skin functionality. Contrary to humans, spiny mice (Acomys spp.) repair skin via scar-free regeneration, regrowing hair follicles and muscles. However, skin across the body is diverse, and whether Acomys can regenerate specialized skin remains unclear. Here, we report that Acomys regenerated whisker pad skin with whisker follicles (i.e., vibrissae), blood sinuses, sebaceous glands, skeletal muscles, and targeted innervation. In contrast, CD-1 mice (Mus) healed via scarring and poor innervation of the scar. While whisker pad skin regeneration in Acomys was remarkable, only 20% of whiskers regenerated on average, ranging from 0% to 75%. Regenerated axons were bundled in epineurial sheaths, targeting the regenerated whisker, with an average of 75% of the uninjured innervation. This expands our understanding of Acomys skin regeneration and provides novel models for skin regeneration and sensorimotor recovery.
Similar content being viewed by others
Introduction
Spiny mice (Acomys) are the first known mammals to regenerate injured skin without scarring1. The regenerated skin includes hair follicles, sebaceous glands, arrector pili muscles, and panniculus carnosus muscle1,2,3. In contrast, mice (Mus) and humans replace injured skin with fibrotic scar tissue. This scar tissue lacks hair follicles and other tissues but is often innervated with fewer fibers than the original tissue4,5,6. In addition to skin regeneration, Acomys can regenerate ear cartilage1,7, skeletal muscle8, spinal cord9,10, and kidney tissue11, and they have remarkable resistance to scarring after myocardial infarction12,13,14. This natural regenerative ability makes Acomys a novel animal model to help us understand how scarless tissue regeneration is possible in mammals15.
The whisker pad is an area of specialized skin on the upper lip of most mammals comprised of whisker (i.e., vibrissae) follicles used for touch sensation16. Each whisker follicle is six times larger than a hair follicle and contains a blood-filled sinus, a sebaceous gland, and innervation17,18,19 (Fig. 1A). The whisker follicle is also surrounded by intrinsic follicular skeletal muscles, supporting whisking behavior for tactile investigation (Fig. 1A, B). Whiskers are arranged in a stereotypical pattern with five rows (A, B, C, D, and E) and a straddler column between the rows (α, β, ɣ, δ). Rows A and B have four or five whiskers, row C has seven, and rows D and E have eight or nine20. Typically, the whisker hair shafts undergo turnover, or physiological regeneration, where they are maintained and replaced, similar to the replacement of hairs21. However, when the entire whisker follicle is removed (i.e., hair shaft, blood sinus, sebaceous gland, and dermal papilla) via surgery, the whisker follicle is permanently missing, and a scar is formed22,23.
A Side-view transection of the whisker pad skin, 1: ordinary pelage hair follicles, 2: shaft of the whisker hair follicle, 3: sebaceous gland, 4: blood sinus, 5: follicular muscles, 6: deep follicular (i.e., vibrissal) nerve, branching from the infraorbital nerve of the trigeminal nerve, 7: dermal papilla of the whisker follicle. B Horizontal diagram parallel with skin, denoting the arrangement of the whisker follicles and their name assignments depending on the position on the maxilla. The follicular muscles (in pink) wrap around each follicle (in brown). Note that this is a simple representation of relevant structures and is not exhaustive of all structures within the whisker pad skin. This figure was created in Inkscape.
It remains unknown whether Acomys can reparatively regenerate whisker pad skin with whisker follicles, similar to dorsal skin with hair follicles. The current study investigated this question, comparing the healing response of CD-1 mice (Mus) and Acomys after full-thickness removal of whisker pad skin tissue. We followed the methods of previous studies that removed whisker follicles in mice and rats, with a slight modification of leaving the wound open without a suture. This modification was done because previous studies on Acomys skin regeneration had left open wounds to heal without suturing. Following the injuries, histology was done several days post-injury (dpi) to observe the healing response. We show that Acomys regenerated whisker pad skin tissue while Mus healed via scarring. Condensations of whisker follicles were present by 10 dpi, and appeared fully regenerated at 44 dpi with blood sinuses, surrounded by regenerated skeletal muscle, and significantly innervated by axons within epineurial sheaths.
Results
Mus scar after full-thickness removal of whisker pad skin
On the day of injury, several whisker follicles were removed from the whisker pad of CD-1 mice, Mus, by making a freeform full-thickness wound around the whisker pad (Fig. 2A, B and Supplementary Fig. 1). The wound was not sutured. This method removed the whisker follicles, transected the deep nerves innervating the follicles, and removed or transected the muscles surrounding the whiskers. Mus healed the area with a scar, significant contraction, and a 90° rotation of the uninjured or remaining whiskers compared to an uninjured whisker pad (n = 13, see Supplementary Table 1 for more details) (Fig. 2C–F).
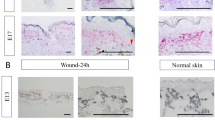
A An external image of Mus whisker pad, B external image of Mus whisker pad after wounding, C external image of Mus whisker pad 21 dpi, D trichrome stain of uninjured whisker pad, notice the arrangement of whisker follicles, large circles surrounded by C-shaped muscles, in five rows and four to eight columns. Scale bar represents 1 mm, E trichrome stain of injured whisker pad 35 dpi, notice the new arrangement of follicles (annotated with white dotted arrows) and the scarred area in deep blue (circled with yellow dotted line). Scale bar represents 1 mm. F trichrome stain of scarred skin section with epidermis on top. No hair follicles or whisker follicles are present. Scale bar represents 250 µM.
Acomys regenerates some whisker follicles and muscle after full-thickness removal of whisker pad skin
Whisker follicles were removed from Acomys using the same method (Fig. 3A, B and Supplementary Fig. 1). Within 24 dpi, whiskers were emerging and apparent from the wound bed (n = 3) (Fig. 3C). Histology at 10 dpi showed that the induction of the follicle was apparent from the healed epidermis of the injured area (n = 4) (Fig. 3D), and larger than the induction of pelage hair follicles at 14 dpi (n = 5) (Fig. 3E, F). The whisker follicle continued to grow into the dermis by 14 dpi (Fig. 3F). By 21 dpi, the regenerating whisker follicle grew larger, and ordinary pelage hair follicles continued to regenerate in the epidermis (n = 4) (Fig. 3G). By 44 dpi, whisker follicles were surrounded by follicular muscles (n = 3) (Figs. 3H and 4B). Notably, some whiskers failed to regenerate, with 0% to 75% regenerating across injured whisker pads (Table 1 and Supplementary Table 1), and some whiskers only partially regenerated (Supplementary Figs. 3 and 4). In the cases of partial whisker regeneration, other tissues like pelage hairs and muscles regenerated, and there were no signs of scarring in Acomys.
A An external image of Acomys whisker pad, they only have 4 whiskers in row A and 9 whiskers in rows D and E (see Supplementary Fig. 2) B external image of Acomys whisker pad after wounding the 2nd column of whiskers to the most anterior whiskers in each row (see Supplementary Fig. 1), C external image of Acomys whisker pad 24 dpi. Regenerating whisker follicles are marked with an asterisk, emerging from the wound bed, while a cluster of regenerating pelage hair follicles is marked with an arrow. D Trichrome stain of whisker pad skin 10 dpi, with whisker follicle induction in the skin. E Trichrome stain of whisker pad skin 14 dpi, with pelage hair follicle induction in the skin. F Trichrome stain of whisker pad skin 14 dpi, with follicular condensations in the skin, outlined in dotted line, an uninjured follicle is to the right labeled with “uf,” and yellow arrows point to regenerating pelage hair follicles. G trichrome stain of whisker pad skin 21 dpi, again the regenerating whisker follicle is outlined, the uninjured whisker follicle is labeled with “uf”, and yellow arrows point to regenerating hair follicles. G trichrome stain of horizontal section parallel with skin 44 dpi with three regenerated follicles in a row, showing the regenerated muscle with disorganized matrix on either side of the regenerated row. Scale bars in (D–H) represent 250 µM.
A Trichrome stain of uninjured whisker pad skin, with hair follicles and two whisker follicles surrounded by muscle with a sebaceous gland, blood sinuses, innervation, and dermal papillae. B Comparable section of regenerated whisker pad skin 70 dpi with the same structures. There appear to be more adipose cells and fewer hair follicles in the regenerated skin, just left of the whisker follicle. Scale bars represent 250 µM.
Acomys regenerate bundles of axons within epineurial sheaths targeting regenerated whisker follicles, while Mus regenerate single axon fibers into scar tissue
To understand innervation after healing, whisker pad skin tissue was stained with RT-97 antibody, a marker of heavy-weight neurofilament. Deep follicular nerves innervating the regenerated whisker follicles of Acomys were similar in general appearance to uninjured whiskers 44 dpi (n = 12 whiskers in 1 animal) and 90 dpi (n = 8 whiskers across 4 animals) (Fig. 5A, B). Hematoxylin and Masson’s trichrome stains suggested that the nerves resided within peripheral nerve connective tissue and epineurial sheaths. Complementary skin sections of Mus tissue 47 dpi showed dense scarring via trichrome stain (n = 4 animals) (Fig. 5C). Axon regeneration was significantly more robust in Acomys (Fig. 5D, E) than in Mus (Fig. 5F). In summary, the regenerated axons in Acomys varied in diameter and density, while only single fibers randomly regenerated into scar tissue in Mus.
A Horizontal section of an uninjured whisker follicle from Acomys with RT-97 antibody stain (brown) with hematoxylin counterstain, showing three bundles of nerves. B A complementary section of a regenerated follicle 90 dpi from Acomys with similar morphology and anatomy. C A complementary section of scarred whisker pad skin from Mus, no whisker follicles were present in the injured zone. D A section of regenerated nerve 44 dpi from Acomys with RT-97 antibody stain (brown) depicting the density of axons. E Another regenerated nerve from the same Acomys, 44 dpi, depicting a lower density of axons. F A subsequent serial section of (C), depicting the unguided and sporadic innervation of scar tissue with single axon fibers in Mus. Scale bars represent 50 µM.
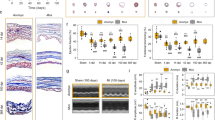
The numbers of regenerated axons per regenerated whisker follicle were counted to understand the variability observed in Acomys. Regenerated Mus axons were not included in this analysis since they did not regenerate whisker follicles and thus had no targeted innervation. First, comparisons of axon numbers at each whisker position in uninjured whisker pads indicated differences across the whisker pad (Fig. 6A). Axon numbers significantly decreased across the columns (F(8, 78) = 6.158, p < 0.001), where the straddler column (i.e., whisker column 0) had a mean count of 157.2 ± 36.63 and the eighth column had a mean count of 85.6 ± 15.82 axons (Supplementary Fig. 5). Axon numbers also slightly decreased across rows (F(4, 77) = 0.689, p = 0.602), where row A had a mean count of 145.50 ± 41.96 and row E had a mean count of 102.73 ± 31.80 (Supplementary Fig. 6). There were no statistical differences between the left and right whisker pads of Acomys when controlling for whisker position (F(1, 29) = 0.385, p = 0.540). There were also no statistical differences between Acomys and Mus (F(1, 11) = 1.829, p = 0.204) (Fig. 6B). A simple analysis between the same animals’ uninjured and regenerated whisker follicles, controlling for whisker position, revealed that Acomys regenerated around 75% of the uninjured innervation, ranging from 22.5% to 132.82%. Formal statistical analysis indicated that while 75% innervation was remarkable, it was significantly less (F(1, 23) = 11.37, p = 0.003; t = -3.372, p = 0.003) (Fig. 6C). Notably, there appeared to be three subsets of axon regeneration: most had fewer axons, some had similar numbers, and one whisker had more axons. Also, most regenerated whisker follicles (17/21) were within the “normal range” of axon counts across the entire whisker pad (47–245 axons per whisker follicle; Fig. 6A, B).
A A heatmap of axon counts for each whisker of an Acomys whisker pad (n = 2–3 whiskers per position). B Bar chart of axon counts between Acomys and Mus; there was no significant difference in species. C Bar chart of differences in axon counts between uninjured and regenerated whiskers; there were significantly fewer axons in the regenerated whiskers, *p < 0.05. This figure was created using ggplot2.
Discussion
Acomys can regenerate dorsal skin and hair follicles without scarring1,2,3, and here we demonstrate that this regenerative ability extends to the whisker pad skin with regeneration of some whisker follicles, follicular muscles, and deep follicular innervation. A full-thickness wound to the whisker pad skin removed the whisker follicles and intrinsic follicular muscles surrounding the follicles and transected the deep follicular nerves innervating the whisker follicles. The wound was left open without suturing. Mus healed the wound with scarring and significant skin contraction, rearranging the uninjured whisker follicles. In contrast, Acomys regenerated the whisker pad skin without scarring or rearranging, regrowing new whisker follicles surrounded by follicular skeletal muscles and innervated by deep follicular nerves, complete with epineurial sheaths. While this ability to regenerate whisker pad skin, whisker follicles, muscle, and transected nerve is remarkable for a mammal, whisker pad skin regeneration was imperfect. On average, approximately 20% of the removed whisker follicles regenerated in Acomys. However, our injuries were unsystematic in the number and position of whiskers removed, and more systematic studies will need to be completed to parse whether the whisker quantity or position contributes to this variability. Also, the regenerated whisker follicles had approximately 75% of their uninjured innervation, with variable axon density. The rest of the discussion will place this regenerative ability in context with other animals, compare the imperfect regeneration with other Acomys studies, and expand on future directions concerning underlying mechanisms and sensorimotor recovery.
The ability of Acomys to regenerate whisker pad skin is remarkable and extends our understanding of their ability to regenerate skin. Skin is an important mechanosensory organ system, and whisker follicle regeneration can be compared to the regeneration of other sensory organs in highly regenerative animals. Compared to dorsal skin, the whisker pad skin is strikingly complex, with whisker follicles six times larger than ordinary hair follicles, dense innervation, and powerful musculature. This system is the primary sensory system for most rodents, occupying approximately 70% of the primary somatosensory cortex. Thus, Acomys whisker pad skin is specialized for sensation and can be compared to other major sensory systems that regenerate in other animals. For example, catfish and zebrafish are known to regenerate their barbels; maxillary appendages used for taste and mechanosensation24,25. While amphibians (i.e., caecilians and some larval frogs and salamanders) and reptiles (i.e., tentacled snakes) have barbels, several studies suggest they cannot regenerate or the regenerative capacity remains unknown25. Another example is lateral line regeneration in axolotls after tail amputation26. The lateral line is a sensory system comprised of modified epithelial hair cells that sense changes in water displacement. Many studies have determined that fish can regenerate lateral line hair cells after chemical or laser ablation27. However, we are unaware of any studies on lateral line regeneration following mechanical-induced wounds in fish28, which would be more similar to the skin wounds reported in this study. Thus, whisker pad skin regeneration in Acomys can be compared to barbel regeneration in some fishes and lateral line regeneration in axolotls.
While whisker pad skin regeneration is remarkable, the regeneration was imperfect and variable. On average, 20% of the whisker follicles with associated musculature and innervation regenerated after full-thickness skin wounding. Some animals healed the skin without scarring but never regenerated any whisker follicles, while others regenerated 75% of the removed follicles (n = 1, 4 follicles removed). The mechanism underlying this variability is unclear. A recent study from our lab showed that ear wound regeneration is imperfect and variable in Acomys and linked to delayed regeneration and poor regeneration quality in two-year-old animals29. However, the animals in the current study ranged in age from 2 to 12 months. Further mechanisms beyond age remain unclear. Notably, the observed variability in regeneration quality suggests that the system is plastic and capable of better regeneration. Studying the underlying mechanisms of regeneration and manipulating factors to increase or decrease regeneration quality must be uncovered to improve our understanding of Acomys regeneration, which would also be a boon for improving regenerative therapies in the clinic.
Since regeneration often uses developmental signals, studies on the embryonic development of whiskers in mice and rats may provide insight into the underlying mechanisms of whisker pad skin regeneration. For example, whisker follicle development is known to be more rapid than pelage hair follicle development29. The current study has some evidence to suggest this difference also occurs in regeneration because whisker follicular induction was clear at 10 dpi, with smaller pelage hair follicle induction occurring at 14 dpi (Fig. 3D, E). Developmental studies also show that a nerve plexus forms under the epithelium with fine fibers penetrating the mesenchyme before follicle induction30. This nerve plexus becomes larger with thicker fibers as the follicle grows. Innervation was present in the follicular condensations observed at 14 dpi, but earlier time points are necessary to understand whether deep follicular nerve regeneration precedes follicle induction. Developmental studies could also be used to examine transcriptional signals in follicle regeneration. Studies show that Prdm1 (i.e., Blimp1) is expressed at the earliest stage of follicle induction, and associated with other signals like Sox2, Lef1, Bmp2, Shh, Bmp4, Krt17, Edar, and Gli131,32. However, these signals are also shared with pelage hair follicle induction, and whisker follicle-specific signals remain unclear32. Also, whisker follicles contain pluripotent neural crest stem cells33. Whether these stem cells are present in the regenerated tissue remains unknown. Thus, many follow-up experiments derived from the developmental biology literature may provide insight into mechanisms underlying whisker pad skin regeneration.
The robust peripheral nerve regeneration observed in Acomys whisker pad skin establishes a novel model for studying peripheral nerve regeneration. The current study transected the peripheral nerves innervating the whisker pad skin and removed all subsequent target tissue (e.g., whisker follicles and muscles). Acomys then regenerated whisker follicles with muscles and targeted innervation, complete with regenerated epineurial sheaths. Mus, in contrast, healed via scarring with non-targeted innervation of the scar. This establishes a novel model system to compare mechanisms underlying peripheral nerve regeneration and target innervation. The trigeminal ganglia containing the neuronal cell bodies of the transected nerves can be studied to determine signals for axon regrowth and neuronal preservation. Cellular signals involved in the regeneration of the epineural sheath can be studied, focusing on the role of Schwann cells and peripheral-nerve-derived fibroblasts. Signals involved in targeted innervation could also be studied, presumably deriving from the epithelium. Comparisons between Acomys and Mus in each area (i.e., ganglia, peripheral nerve injury site, and regenerating skin tissue) would likely reveal novel signaling molecules for peripheral nerve regeneration.
The sensorimotor capacity of the whisker pad skin also makes Acomys a novel model for understanding sensorimotor recovery following injury and regeneration. Whisker follicles are central to rodent touch sensation and densely filled with multiple nerve receptors, including Merkel cells, Pacinian-like corpuscles, lanceolate receptors, and unmyelinated fibers33. These nerve endings send afferent signals down the deep follicular nerves with cell bodies in the trigeminal ganglia. The signals are then sent to the brainstem, whose cell bodies are organized in a topographic map representing the arrangement of whiskers in a point-to-point correspondence34. Each receptive field is an individual cluster or “barrel” of neurons, which exist in the brainstem as barrelettes, the thalamus as barreloids, and the cortex as barrels. These receptive fields are plastic and alter in response to whisker removal22,35,36,37. Mouse experiments that crushed the infraorbital nerve, which branches into each deep follicular nerve, showed that the nerve crush led to unresponsiveness in the whisker cortical area38. This whisker cortical area was then invaded by an expansion of the forepaw cortical area. Ten days after the nerves had regenerated, the whisker and forepaw cortical areas overlapped. By 60 days post nerve crush, the forepaw areas withdrew from the whisker cortical area. Whether similar phenomena occur in Acomys remains unclear. Research on this question and studies on sensory behavior are essential to understanding sensory recovery with skin regeneration.
In summary, Acomys regenerated whisker pad skin tissue with whisker follicles, muscles, and innervation, while Mus scarred the wounded area with significant skin contraction. This regenerative ability can be compared to barbel regeneration in some fish and lateral line regeneration in axolotls. While this regenerative ability is remarkable, regeneration was imperfect and varied in quality from 0% to 75% follicle regeneration. The regenerated whisker follicles also had targeted innervation with regenerated deep follicular nerves ranging in axon density from 22.5% to 132.82% of the uninjured follicles at the same whisker position on the opposite uninjured whisker pad. Future studies should consider using findings from the embryonic development of whiskers to help determine the mechanisms underlying whisker pad skin regeneration. Also, regenerating whisker pad skin is a novel model system to understand sensorimotor recovery following injury and regeneration.
Methods
Subjects
Male and female Acomys cahirinus used in this study were from our breeding colony at the University of Florida and studied in accordance with the IACUC protocol (201807707 and 202400000403), while male and female Mus musculus were from the CD-1 strain and ordered from Charles River before being studied following the IACUC protocol (2018030505). All histology on the basic anatomy of the Acomys whisker pad and whisker follicles was performed with extra tissue from previous studies. All Acomys and Mus were 55 days to 1 year old. After removing the section of the whisker pad, the following time points were taken: 10 dpi (Acomys n = 4), 14 dpi (Acomys n = 5), 21 dpi (Acomys n = 4), 34–35 dpi (Acomys n = 4; Mus n = 6), 44–49 dpi (Acomys n = 8; Mus n = 4), 65–70 dpi (Acomys n = 1; Mus n = 3), and 90 dpi (Acomys n = 3) (See Supplementary Table 1 for more details). All animals were euthanized via carbon dioxide asphyxiation with 30–70% displacement of the chamber volume per minute for at least 1 min after respiration had ceased and secondary cervical dislocation.
Full-thickness mystacial pad injury
On the day of injury, the animals were placed under deep anesthesia via 4% (v/v) isoflurane inhalation and maintained at 2.5%. Due to the location of the injury, on the snout, both a hind limb pinch and a whisker pad pinch with tweezers were done to ensure the animal was adequately anesthetized. After trimming the whisker follicles and pelage hairs with scissors, the skin was pinched and lifted with tweezers and an incision was made caudal to the whisker pad. Continual scissor cuts along the outside of the whisker pad and then under the full-thickness skin were made to remove the desired area (see Supplementary Fig 1 for more details). Any follicular stumps were further excised with scissors planar to the surface. Excessive bleeding of the whisker pad was dabbed with sterile gauze. Following subcutaneous analgesic treatment with meloxicam (15 mg/kg), the animal was allowed to wake up and placed back in their cage. Several animals were randomly selected to be euthanized 24 h after injury to ensure that entire follicles were removed during the injury process (Fig. 7) .
This is a vertical histological section through the muzzle of an Acomys 24 h after full-thickness mystacial pad injury. The number “1” refers to the nose of the animal in the center. To the left, “2” refers to the injured area (dotted line) with missing tissue on the left side of the muzzle. This injured area should mirror the opposite side, “3”, where an uninjured whisker follicle on the right side of the snout is marked.
Histology
Several histological methods were used including Harrison’s hematoxylin and eosin (H&E), Masson’s trichrome, and antibody staining with anti-RT97, a neurofilament-heavy-weight antigen (mouse monoclonal, Developmental Studies Hybridoma Bank, RT97, AB 528399). All tissue was excised immediately after euthanasia and fixed in 10% neutral buffered formalin overnight and processed for paraffin embedding following increasing serial dilution with ethanol and 100% xylene. Excised tissue that included the nasal bone and muzzle was decalcified using daily changes of 14% EDTA solution for 10 days prior to paraffin embedding. Paraffin-embedded whisker pads were then sliced on the microtome at 10 µM for sagittal sections and 7 µM for tangential sections parallel to the skin surface. All slides were then rehydrated with decreasing serial dilution of ethanol before subsequent staining. The manufacturer’s protocol was followed for H&E and Masson’s trichrome staining. For antibody staining, the slides underwent an immersion in 2% hydrogen peroxide to remove peroxidases and were immersed in 0.1 M citrate buffer at pH 6.0 and microwaved for 2 min at 50% power and left at room temperature for 23 min for antigen retrieval. The blocking solution with the primary antibody was then soaked on the slide overnight at 4 °C, and a biotinylated anti-mouse secondary antibody was applied the following day for 1 h at room temperature after several washes. Samples were then prepped with an avidin/biotin-based peroxidase step (Vectastain ABC kit) prior to 3,3’-diaminobenzidine (DAB) staining (Vectastain DAB kit). All samples were mounted with cytoseal and a coverslip before viewing under a light microscope. Each batch of antibody staining included a no-primary-antibody control.
Axon counting of the deep follicular nerve
The number of axons in the deep follicular nerve was counted for individual follicles in Acomys or Mus following anti-RT97 of tangential histological sections. For comparisons, samples were blinded before counting between Acomys and Mus for basic anatomy or Acomys injured and uninjured whisker pads for regeneration. Individual follicles were located and their spatial position was noted at ×10 with a light microscope prior to images being taken at ×40 or ×100 for axon counting. Individual axons were counted for each blinded image using ImageJ software39 and the “Cell Counter” plugin. Axons were operationally defined as small circles of dark brown coloration (i.e., DAB positive) residing within the deep vibrissal nerve of the follicular complex. Counting was reliable with an intra-class correlation coefficient of 0.922 for 10% inter-rater reliability and 0.995 for 5% intra-rater reliability.
Statistical analyses
All statistical analyses were performed in R (v4.4.0), and used a p < 0.05 as the critical threshold. The normality and homogeneity of variance in each dataset was examined graphically, and no data transformations were performed. For all axon count analyses, linear mixed-effects models were used to account for whisker position and individual animal. It was not appropriate to run simple paired t tests because there were two levels of multiple or repeated comparisons (e.g., 30+ whiskers nested within individual animals). All analyses used “animal id” as a random effect. Analyses on axon counts across the whisker pad within Acomys used “whisker number” and “whisker letter” as fixed effects. Comparisons between Acomys and Mus used the same model, adding “species” as a fixed effect. Left and right comparisons used “whisker side” and “whisker position” as fixed effects. Comparisons between regenerated and uninjured whiskers used “injury type” and “whisker position” as fixed effects.
Data availability
Data is provided within the Supplementary Information files and the following github repository: https://github.com/javarhol/AcomysWhiskerRegeneration.
References
Seifert, A. W. et al. Skin shedding and tissue regeneration in African spiny mice (Acomys). Nature 489, 561–565 (2012).
Maden, M. Optimal skin regeneration after full thickness thermal burn injury in the spiny mouse, Acomys cahirinus. Burns 44, 1509–1520 (2018).
Jiang, T.-X., Harn, H. I.-C., Ou, K.-L., Lei, M. & Chuong, C.-M. Comparative regenerative biology of spiny (Acomys cahirinus) and laboratory (Mus musculus) mouse skin. Exp. Dermatol. 28, 442–449 (2019).
Mehta, A. S. et al. Quantifying innervation facilitated by deep learning in wound healing. Sci. Rep. 13, 16885 (2023).
Altun, V., Hakvoort, T. E., van Zuijlen, P. P., van der Kwast, T. H. & Prens, E. P. Nerve outgrowth and neuropeptide expression during the remodeling of human burn wound scars. A 7-month follow-up study of 22 patients. Burns 27, 717–722 (2001).
Waris, T. Innervation of scar tissue in the skin of the rat. Scand. J. Plast. Reconstr. Surg. 12, 173–180 (1978).
Matias Santos, D. et al. Ear wound regeneration in the African spiny mouse Acomys cahirinus. Regeneration 3, 52–61 (2016).
Maden, M. et al. Perfect chronic skeletal muscle regeneration in adult spiny mice, Acomys cahirinus. Sci. Rep. 8, 8920 (2018).
Streeter, K. A. et al. Molecular and histologic outcomes following spinal cord injury in spiny mice, Acomys cahirinus. J. Comp. Neurol. 528, 1535–1547 (2020).
Nogueira-Rodrigues, J. et al. Rewired glycosylation activity promotes scarless regeneration and functional recovery in spiny mice after complete spinal cord transection. Dev. Cell 57, 440–450.e7 (2022).
Okamura, D. M. et al. Spiny mice activate unique transcriptional programs after severe kidney injury regenerating organ function without fibrosis. iScience 24, 103269 (2021).
Qi, Y. et al. Functional heart recovery in an adult mammal, the spiny mouse. Int. J. Cardiol. 338, 196–203 (2021).
Koopmans, T. et al. Ischemic tolerance and cardiac repair in the spiny mouse (Acomys). npj Regen. Med. 6, 1–16 (2021).
Peng, H. et al. Adult spiny mice (Acomys) exhibit endogenous cardiac recovery in response to myocardial infarction. npj Regen. Med. 6, 1–15 (2021).
Gaire, J. et al. Spiny mouse (Acomys): an emerging research organism for regenerative medicine with applications beyond the skin. npj Regen. Med. 6, 1 (2021).
Muchlinski, M. N., Wible, J. R., Corfe, I., Sullivan, M. & Grant, R. A. Good vibrations: the evolution of whisking in small mammals. Anat. Rec. 303, 89–99 (2020).
Rice, F. L., Mance, A. & Munger, B. L. A comparative light microscopic analysis of the sensory innervation of the mystacial pad. I. Innervation of vibrissal follicle-sinus complexes. J. Comp. Neurol. 252, 154–174 (1986).
Ahl, A. S. Relationship of vibrissal length and habits in the Sciuridae. J. Mammal. 68, 848–853 (1987).
Vincent, S. B. The tactile hair of the white rat. J. Comp. Neurol. 23, 1–34 (1913).
Welker, E. & Van der Loos, H. Quantitative correlation between barrel-field size and the sensory innervation of the whiskerpad: a comparative study in six strains of mice bred for different patterns of mystacial vibrissae. J. Neurosci. 6, 3355–3373 (1986).
Davidson, P. & Hardy, M. H. The development of mouse vibrissae in vivo and in vitro. J. Anat. 86, 342–356 (1952).
Melzer, P. & Smith, C. B. Whisker follicle removal affects somatotopy and innervation of other follicles in adult mice. Cereb. Cortex 5, 301–306 (1995).
Van der Loos, H. & Woolsey, T. A. Somatosensory cortex: structural alterations following early injury to sense organs. Science 179, 395–398 (1973).
LeClair, E. E. & Topczewski, J. Development and regeneration of the zebrafish maxillary barbel: a novel study system for vertebrate tissue growth and repair. PLoS ONE 5, e8737 (2010).
Fox, H. Barbels and barbel-like tentacular structures in sub-mammalian vertebrates: a review. Hydrobiologia 403, 153–193 (1999).
Jones, J. E. & Corwin, J. T. Replacement of lateral line sensory organs during tail regeneration in salamanders: identification of progenitor cells and analysis of leukocyte activity. J. Neurosci. 13, 1022–1034 (1993).
Rubel, E. W., Furrer, S. A. & Stone, J. S. A brief history of hair cell regeneration research and speculations on the future. Hear. Res. 297, 42–51 (2013).
Sveen, L., Karlsen, C. & Ytteborg, E. Mechanical induced wounds in fish – a review on models and healing mechanisms. Rev. Aquac. 12, 2446–2465 (2020).
Varholick, J. A. et al. Older 6-9-month-old spiny mice (Acomys cahirinus) have delayed and spatially heterogenous ear wound regeneration. Biol. Open 13, bio060565 (2024).
Van Exan, R. J. & Hardy, M. H. A spatial relationship between innervation and the early differentiation of vibrissa follicles in the embryonic mouse. J. Anat. 131, 643–656 (1980).
Manti, P. G. et al. The transcriptional regulator Prdm1 is essential for the early development of the sensory whisker follicle and is linked to the beta-catenin first dermal signal. Biomedicines 10, 2647 (2022).
Robertson, E. J. et al. Blimp1 regulates development of the posterior forelimb, caudal pharyngeal arches, heart and sensory vibrissae in mice. Development 134, 4335–4345 (2007).
Sieber-Blum, M., Grim, M., Hu, Y. F. & Szeder, V. Pluripotent neural crest stem cells in the adult hair follicle. Dev. Dyn. 231, 258–269 (2004).
Petersen, C. C. H. Sensorimotor processing in the rodent barrel cortex. Nat. Rev. Neurosci. 20, 533–546 (2019).
Melzer, P. & Smith, C. B. Plasticity of cerebral metabolic whisker maps in adult mice after whisker follicle removal–II. Modifications in the subcortical somatosensory system. Neuroscience 83, 43–61 (1998).
Kossut, M. & Juliano, S. L. Anatomical correlates of representational map reorganization induced by partial vibrissectomy in the barrel cortex of adult mice. Neuroscience 92, 807–817 (1999).
Waite, P. M. E. & Cragg, B. G. The peripheral and central changes resulting from cutting or crushing the afferent nerve supply to the whiskers. Proc. R. Soc. Lond. Ser. B Biol. Sci. 214, 191–211 (1982).
Kis, Z. et al. Comparative study of the neuronal plasticity along the neuraxis of the vibrissal sensory system of adult rat following unilateral infraorbital nerve damage and subsequent regeneration. Exp. Brain Res. 126, 259–269 (1999).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012).
Acknowledgements
The authors gratefully acknowledge the animal care staff and veterinarians at the University of Florida. We also acknowledge the technical assistance of Jazmine Thermolice and Vanessa Dos Santos. This research was funded by a National Institutes of Health T32 training fellowship on regenerative medicine from the National Institutes of Diabetes and Digestive and Kidney Diseases T32DK074367-15 [J.A.V.] and a grant from the National Science Foundation (NSF) - 2052459 [M.M.].
Author information
Authors and Affiliations
Contributions
This research was conceptualized by J.A.V. and M.M.; software, validation, formal analysis, data curation, original draft, visualization, and project administration by J.A.V.; funding and supervision by M.M.; methodology, investigation, reviewing, and editing by J.A.V., M.M., and R.K.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Varholick, J.A., Kondapaneni, R. & Maden, M. Spiny mice (Acomys) regenerate wounded whisker pad skin with whisker follicles, muscles, and targeted innervation. npj Regen Med 10, 28 (2025). https://doi.org/10.1038/s41536-025-00415-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41536-025-00415-0