Abstract
Altered hippocampal morphology and metabolic pathology, but also hippocampal circuit dysfunction, are established phenomena seen in psychotic disorders. Thus, we tested whether hippocampal subfield volume deficits link with deviations in glucose metabolism commonly seen in early psychosis, and whether the glucose parameters or subfield volumes change during follow-up period using one-year longitudinal study design of 78 first-episode psychosis patients (FEP), 48 clinical high-risk patients (CHR) and 83 controls (CTR). We also tested whether hippocampal morphology and glucose metabolism relate to clinical outcome. Hippocampus subfields were segmented with Freesurfer from 3T MRI images and parameters of glucose metabolism were determined in fasting plasma samples. Hippocampal subfield volumes were consistently lower in FEPs, and findings were more robust in non-affective psychoses, with strongest decreases in CA1, molecular layer and hippocampal tail, and in hippocampal tail of CHRs, compared to CTRs. These morphometric differences remained stable at one-year follow-up. Both non-diabetic CHRs and FEPs had worse glucose parameters compared to CTRs at baseline. We found that, insulin levels and insulin resistance increased during the follow-up period only in CHR, effect being largest in the CHRs converting to psychosis, independent of exposure to antipsychotics. The worsening of insulin resistance was associated with deterioration of function and symptoms in CHR. The smaller volume of hippocampal tail was associated with higher plasma insulin and insulin resistance in FEPs, at the one-year follow-up. Our longitudinal study supports the view that temporospatial hippocampal subfield volume deficits are stable near the onset of first psychosis, being more robust in non-affective psychoses, but less prominent in the CHR group. Specific subfield defects were related to worsening glucose metabolism during the progression of psychosis, suggesting that hippocampus is part of the circuits regulating aberrant glucose metabolism in early psychosis. Worsening of glucose metabolism in CHR group was associated with worse clinical outcome measures indicating a need for heightened clinical attention to metabolic problems already in CHR.
Similar content being viewed by others
Introduction
Psychotic disorders have been consistently associated with altered brain morphology1,2 including the hippocampus3,4,5,6,7,8,9,10, as well as with glucose metabolism already in first episode of psychosis11,12. Also, hippocampal circuit dysfunction in psychotic disorders, such as schizophrenia, is supported by several studies on brain structure and function13,14,15. Meta-analyses16,17 have demonstrated that hippocampal volume reductions are present in first-episode psychosis, but it is less clear whether the same is true for the individuals with clinical high-risk for psychosis18,19,20,21. Based on recent longitudinal study, the subfield volumes remain stable during the early years after the onset of psychosis22. The data is inconsistent whether hippocampal morphology predicts the transition to psychosis in high-risk individuals18,20,23. Furthermore, it is not known whether these morphological changes are associated with glucose metabolism disturbances in FEP or CHR, or in relation to clinical outcomes.
Hippocampal circuits are traditionally linked to regulation of learning, memory, spatial processing, emotions and stress responses24. These behavioral domains are all afflicted to a varying degree in psychotic disorders14. In addition, several lines of research suggest that hippocampus regulates endocrine functions and glucose metabolism25,26. Also, diabetes associates with brain morphology4 including the volume of the hippocampus27,28,29,30,31. Further, an inverse association between insulin resistance and total hippocampal volume has been reported in healthy individuals32, but also between insulin resistance and specific subfield volumes in type 2 diabetes33,34. There is also abundant evidence of altered glucose homeostasis in psychoses35,36,37,38 which cannot be attributed solely to the effects of antipsychotic medications11,39,40,41. In fact, psychotic disorders show peripheral changes of both glucose and lipid metabolism12,39,42, and the regulation between hippocampal circuitries and peripheral metabolism seems to be bidirectional25.
Associations between fasting glucose or glycated hemoglobin, and clinical outcome have been studied in FEPs, but not insulin or insulin resistance43. Also, recent study44 showed that worsening of fasting glucose values was related to progression of psychotic illness in recent-onset psychosis. Only some longitudinal studies have been published on subfields and clinical outcome in CHR10,23 or first-episode schizophrenia45. However, these studies do not combine glucose metabolism parameters and subfield volumetry to investigate the clinical outcome trajectories, in contrast to our study. Given this evidence base it is important to study whether peripheral metabolic factors relate to alterations in brain morphology as well as to illness progression and clinical outcome, even as early as during the first episode of psychosis.
In this longitudinal study, we first tested whether patients with first-episode psychosis, clinical high-risk individuals or population controls have associations between altered hippocampal subfield or total hippocampal volumes and glucose parameters at the two time points during 1-year follow-up. Secondly, we tested whether subfield volumes or the measurements of glucose homeostasis change during the one-year follow-up in these groups. Thirdly, we tested whether the cross-sectional associations or the longitudinal changes in patient groups are related to clinical outcome trajectories, such as remission, functioning, and transition to psychosis. We hypothesized that the hippocampal volume reductions are associated with progressive glucose metabolism disturbances, and that the effect is most pronounced in patients with first-episode psychosis and in CHR patients who developed psychosis during follow-up.
Methods
Participants
Participants at baseline
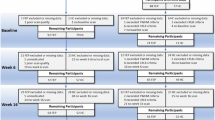
The intent-to-study groups consisted of age- and sex-matched 88 first-episode psychosis patients (FEP), 56 clinical high-risk for psychosis patients (CHR) and 96 randomly selected general population controls (CTR) between 18 and 50 years of age. Patients were recruited from psychiatric services of the Hospital District of Southwest Finland. The general population control group was recruited from the same geographic area using a random sample of the national population register. The study protocol was approved by the Ethics Committee of the Hospital District of Southwest Finland and the study was conducted in accordance with the Declaration of Helsinki. Written informed assent and consent were given by from all the participants.
The clinical high-risk status was defined by the ultra-high-risk criteria: Attenuated Psychotic Symptoms (APS), Brief Limited Psychotic Symptoms, and Genetic risk and reduction of function assessed by the 3.0/5.0 version of the Structured Interview for Prodromal Syndromes (SIPS/SOPS). Psychotic and non-psychotic diagnoses of all participants were evaluated using the Structured Clinical Interview for DSM-IV disorders (SCID-I/NP) Eight population control participants who had a non-psychotic DSM-IV diagnosis, were included in the study. Diagnoses are listed in Supplementary table 1.
Participants with an IQ under 70, a significant somatic or neurological illness that might affect brain structure or function, earlier head injury with loss of consciousness for over five minutes, or alcohol dependence during the preceding 6 months were excluded.
At baseline, one FEP, two CHRs and five CTRs with neurological findings in MRI (Supplementary table 2), and two FEPs and one CHR scanned with incompatible MRI scan parameters (non-iso T1 sequence) were excluded. One control participant who was later found to be a 1st degree relative of a psychosis spectrum disorder patient was excluded. We also excluded two FEPs, two CHRs and one CTR due to excessive motion in their T1 weighted images. The hippocampal segmentation failed in 12 subjects (five FEPs, two CHRs, five CTRs), who were also excluded. Further, one control participant was excluded due to a CA3 volume being a statistical outlier (standard residual: (SD) = 3.5 ≥ ± 3.5 SD; statistical influence: Cook’s mean x3 = 0.005 x3 = 0.015; Cook’s distance = 0.033 > 0.015) and one CHR participant was excluded due to the whole hippocampus volume being a statistical outlier (standard residual: (SD) = −3.5 ≥ ± 3.5 SD; statistical influence: Cook’s mean x3 = 0.005 x3 = 0.015; Cook’s distance = 0.051 > 0.015).
Six FEPs, ten CHRs and eight CTRs did not participate in blood sampling at one of the two time points. From analyses related to glucose metabolism variables, two FEPs, one CHR and two CTRs with diabetes mellitus diagnosis (type 1), and three CHRs and one CTR whose insulin sample was affected by hemolysis were excluded. Further, one FEP with an outlier value of insulin at follow-up (standard residual: (SD) = 3.5 ≥ ± 4.6 SD; statistical influence: Cook’s mean x3 = 0.019 x3 = 0.057; Cook’s distance = 0.823 > 0.057) was excluded from analyses of the glucose parameters.
The final baseline sample consisted of 78 first-episode psychosis patients, 48 clinical high-risk for patients for psychosis patients and 83 randomly selected general population controls of similar age and sex. The baseline demographical comparisons are presented in Table 1. At the baseline, FEP group consisted of 56 non-affective psychoses (NAP) and 22 affective psychoses (AP).
Participants at one-year follow-up
At follow-up, the number of retained participants in each group were 34 FEPs, 23 CHRs, and 53 CTRs. We excluded two FEPs, one CHR and two CTRs with failed hippocampal segmentation, and two FEPs and one CHR with excess movement in the T1 sequence, in addition to the subjects who had dropped out from the study.
Clinical outcome trajectories
For analyses of clinical outcome trajectories, we divided patient groups to two groups based on follow-up evaluation on level of functioning, remission status and transition to psychosis.
The CHR group was divided into two groups based on follow-up time transition to psychosis (CHR converting (CHR-C n = 11) and CHR non-converting (CHR-NC n = 37)), resulting in a 23% psychosis transition rate in the CHR group during the one-year follow-up. Only seven out of these eleven CHR converting subjects were available for the analyses due to dropouts.
Level of functioning was measured using global assessment of functioning (GAF) that was binarized to poor (GAF < 65) or good performance (GAF ≥ 65) based on follow-up evaluation. Remission status (remission or non-remission) was defined using the scores of the Brief Psychiatric Rating Scale (BPRS) and three added items from Scale for the Assessment of Negative Symptoms (SANS) or the Positive and Negative Symptom Scale (SCI-PANSS) scores at the follow-up time point46. Sample sizes based on outcome measures are presented in Tables 5, 6.
MRI acquisition and processing
The participants were scanned with a Philips Ingenuity TF 3-Tesla PET/MR scanner. A T1-weighted (Ultrafast Gradient Echo 3D, TR = 8.1 ms, TE-time = 3.7 ms, flip angle 7°, FOV = 256 x 256 x 176 mm3 and voxel size 1 x 1 x 1 mm3) image was acquired from all subjects. The T1 images were preprocessed, and the 12 hippocampal subfield volumes were segmented using the longitudinal FreeSurfer pipeline in version 7.1.147,48. (https://surfer.nmr.mgh.harvard.edu/fswiki/HippocampalSubfieldsAndNucleiOfAmygdala). Quality control was done by visual inspection for all T1-weighted images and all segmented hippocampus in coronal, axial and sagittal planes by R-L A. The small head and body subdivisions were combined to correspond to each whole subfield and to reduce the number of comparisons. Eight subfields were chosen for statistical testing: the subiculum, cornu ammonis 1 (CA1), presubiculum, the molecular layer of the subiculum and the cornu ammonis -fields (ML), the granule cell layer and the molecular layer of the dentate gyrus (GCMLDG), cornu ammonis 2 and 3 (CA2 and CA3), cornu ammonis 4 (CA4) and hippocampal tail (Fig. 1). The hippocampal-amygdaloid transition region (HATA), hippocampal fissure and parasubiculum were excluded due to low reliability of segmenting regions with relatively small volumes compared to the voxel size.
Upper: 3D-visualisation of hippocampal subfields generated with FreeSurfer. Lower: Hippocampal subfield segmentation masks shown overlaid on a representative coronal plane of T1-weighted MRI image.
Clinical measures
Blood samples including fasting plasma glucose and fasting plasma insulin were acquired from the subjects (Table 2). The fasting glucose values included in the analysis were at non-diabetic level ranges (below 7.0 mmol/l; ranging from 4.0 mmol/l to 6.5 mmol/l). The homeostatic Model Assessment, HOMA2-IR (https://www.dtu.ox.ac.uk/homacalculator/) was used as a proxy for insulin resistance instead of HOMA1-IR49,50 since HOMA2-IR is considered to be a more accurate measure of insulin resistance and beta-cell function due to it representing both hepatic glucose output and peripheral glucose uptake.
All antipsychotic daily doses preceding each scanning date were documented using available medical records. The daily doses were then converted to chlorpromazine (CPZ) equivalent daily dosages51 after which they were summed up to obtain the total cumulative lifetime antipsychotic exposures at each study time point. The information on the use of antidepressive and mood stabilizing medications were also collected (Supplementary table 3).
Symptom severity was assessed using either SCI-PANSS52 or BPRS (24- items, version 4.0). The PANSS scores were converted to correspond to the BPRS 18-item scores and the BPRS 24-item scores reduced to correspond to the BPRS 18-item scores53.
Statistical analyses
Analyses of hippocampal volumetry and glucose parameters based on clinical groups of FEP, CHR and CTR
Between group differences of whole hippocampal and subfield volumes at baseline
Differences of whole hippocampus volumes between FEP, CHR and CTR were tested using a linear mixed-effects model with age, sex, body mass index (BMI) and scaled total intracranial volume (TIV) as covariates. Between-group comparisons of whole hippocampal volumes were done post-hoc using estimated marginal means using alpha levels corrected for false discovery rates.
Differences of hippocampal subfield volumes were tested between the three groups using a linear mixed-effects model: volume ~ intercept + β1 (group) + β2 (subfield) + β3 (group by subfield) + β4 (age) + β5 (sex) + β6 (TIV) + β7 (BMI) + random (subject) + ε. In the analysis, subfields were used as a within subject repeated measure. Volumes were deemed a dependent variable, while group status, subfield, interactions between group and subfield, age, sex, BMI, and TIV were the independent variables. Pairwise repeated measure between-group comparisons of subfield volumes were done post-hoc using estimated marginal means while correcting for false discovery rates.
Between group differences in parameters of glucose metabolism
Pairwise group differences of measured fasting plasma glucose and insulin, and calculated insulin resistance (HOMA2-IR) were tested using Student’s t-test in FEP, CHR and CTR both the baseline and follow-up time points. (Table 2)
Effect of fasting glucose, insulin and insulin resistance on hippocampal volumetry
Associations between parameters of glucose metabolism and volumes (subfield and total hippocampal volumes) were tested separately for FEP, CHR and CTR, for all metabolic indexes and for all volumes as well as in both time points using a linear model: volume ~ intercept + β1 (glucose parameter value) + β2 (age) + β4 (sex) + β5 (BMI) + β6 (total intracranial volume) + ε.
Linear mixed-effects model of longitudinal changes in subfield volumes and glucose metabolism parameters
We analyzed the baseline and one-year follow-up time points in the same linear mixed-effects model analysis to detect longitudinal changes in subfield volumes, total hippocampal volumes or glucose parameter values across time between FEP, CHR and CTR. These models included also subjects who did not have measurements at both time points. The linear mixed-effect models were performed separately for each volume and glucose parameter. Subject ID was used as a random effects variable. Post-hoc comparisons of volume or glucose parameter changes within each group between the baseline and one-year follow-up were performed using estimated marginal means.
The model included a binary independent variable that indicated the time point and its interaction with the group variable. The model used for the volume changes in time was: volume ~ intercept + β1 (group by time) + β2 (time) + β3 (group) + β4 (sex) + β5 (age) + β6 (total intracranial volume) + β7 (BMI) + random (subject) + ε. Model used for the glucose parameter value changes in time was: glucose parameter value ~ intercept + β1 (group by time) + β2 (time) + β3 (group) + β4 (sex) + β5 (age) + β7 (BMI) + random (subject) + ε. The post-hoc comparisons of volume or glucose parameter changes within each group between the baseline and one-year follow-up were performed using estimated marginal means.
Analyses of hippocampal volumetry and glucose parameters based on clinical outcomes
Transition to psychosis, remission and functioning during follow-up
A mixed-effects linear model was used to examine whether the reductions in subfield volumes at baseline, the baseline glucose parameter levels, or the longitudinal change in volumes or glucose parameters during the follow-up period, were associated with the clinical outcome trajectories, such as transition to psychosis, level of functioning or remission status at the follow-up.
With linear model, we also tested whether associations, between parameters of glucose metabolism and volumes, were based on clinical outcome trajectories.
Exploratory analyses
In the exploratory analyses we used linear mixed-effects model to detect differences in subfield or whole hippocampal volumes between non-affective psychosis and affective psychosis at baseline. Also, we explored possible differences in associations between hippocampal volumes and glucose parameters separately in NAP and AP at the two time points.
We collected the demographic information of the use of antidepressant and mood-stabilizing medications of the participants at the two time points. Due to the small sample size of the users of mood stabilizing medication, only linear model analyses of associations between lifetime antipsychotic exposure or the use of antidepressant medication and hippocampal total and subfield volumes were tested separately for studied clinical groups of FEP and CHR at the two time points. Further similar associations were studied in groups based on clinical outcome trajectories at the baseline.
The effect of lifetime antipsychotic exposure at each MRI scan time point was controlled by using it as a covariate for each test of this study separately. In all statistical analyses, two-tailed p-values < 0.05 were considered statistically significant. Baseline pairwise multiple comparisons of volumetric differences and analyses on associations between volumes and glucose parameters were corrected using false discovery rate (FDR) correction at p-value < 0.05.
Results
Analyses of hippocampal volumetry and glucose parameters based on clinical groups of FEP, CHR and CTR
Group differences of whole hippocampal and subfield volumes at baseline
There were no significant interactions between subfield volumes and hemispheres in single groups (hemisphere by subfield), nor were there interactions between the three groups (hemisphere by subfield by group). Consequently, all following tests were done using means of bilateral volumes as there was no hemispheric bias. All mean volumes were normally distributed (Shapiro Wilk’s test p > 0.05). There was no multicollinearity between the independent variables used in each model (Variance Inflation Factor, VIF < 2) (age, sex, TIV, BMI and fasting plasma glucose, fasting plasma insulin or insulin resistance).
The mean volume of the whole hippocampus was different between the three groups (F2,202 = 9.158, p < 0.001). Post-hoc pairwise tests showed that the mean whole hippocampus volume was significantly smaller in FEP compared to controls (estimated marginal means ± standard error: FEP = 3509 ± 38 mm3; CTR = 3645 ± 37 mm3), but not in FEP compared to CHR (CHR = 3603 ± 49 mm3). The difference between CHR and the control group was not significant.
The subfield volumes did not differ significantly between FEPs, CHRs and CTRs (subfield by group interaction F14,1442 = 1.33, p = 0.183). However, pairwise repeated measures analysis showed that in the FEP group, the volumes of the subiculum, presubiculum, molecular layer, CA1, GCMLDG and tail were significantly reduced compared to CTRs, whereas no statistically significant differences were observed in the combined CA2 and CA3 subfield or CA4 after FDR corrections (8 subfields and 3 contrasts per subfield; total 24 tests per FDR correction). FEPs had also significantly smaller CA1 volumes compared to CHRs, but no differences in other subfields were observed between FEP and CHR. Also, hippocampal tail was significantly smaller in CHR compared to CTR. (Table 3)
Between group differences in parameters of glucose metabolism
FEPs had significantly higher baseline fasting plasma and fasting insulin levels compared to CTR, while insulin resistance was higher only at a trend-level when compared to CTR. CHR had significantly higher fasting glucose values compared to CTR.
At the follow-up time point insulin and insulin resistance were higher in CHR while comparing to CTR. No such differences were seen between FEP and CHR at any time point. (Table 2)
Associations between parameters of glucose and hippocampal subfield volumetry
There was a subfield specific effect of fasting insulin and insulin resistance in FEP at the follow-up time point (subfield by insulin F3.4,72 = 3.73, p = 0.011; subfield by insulin resistance F3.4,71 = 3.45, p = 0.017). We observed a significant inverse association between fasting plasma insulin or insulin resistance and hippocampal tail (insulin: t = −3.42, beta = −7.41, 95% CI [−11.9, −2.9], p = 0.003 FDR p = 0.024; insulin resistance: t = −17.85, beta = −58.90, 95% CI [−96.0, −21.8], p = 0.003, FDR p = 0.024) and between insulin resistance and molecular layer (t = −2.14, beta = −25.74, 95% CI [−50.8, −0.7], p = 0.044, FDR p = 0.186) in non-diabetic FEP patients, at the follow-up time point, but not at the baseline. After FDR correction for multiple comparisons (8 subfields and 2 glucose parameters; total 16 tests per FDR correction), only associations on hippocampal tail remained statistically significant (Fig. 2). Similar associations were not observed in CHR or CTR. Adjusting for lifetime exposure to antipsychotic medication, or the use of antidepressants did not change these results.
There is a significant subfield-specific inverse association between fasting plasma insulin (A) or insulin resistance (B) and hippocampal tail volumes in first-episode psychosis patients at the one-year follow-up time point. The model is adjusted for age, sex, TIV and BMI. Adjusting for lifetime antipsychotic exposure did not change these results.
Also, there was a significant inverse association between insulin or insulin resistance and total hippocampal volume of non-diabetic FEPs at the follow-up (insulin: t = −2.43, beta = −21.31, 95% CI [−39.6, −3.0], p = 0.024; insulin resistance: t = −2.46, beta = −175.53, 95% CI [−323.8, −27.3], p = 0.023) (Supplementary fig. 1). Adjusting for lifetime exposure to antipsychotic medication or the use of antidepressants did not change these results. However, these results of total hippocampal volume did not survive the FDR correction (3 groups and 3 measures of glucose parameters; total 9 tests for FDR correction).
There were no significant associations between subfield volumes or total hippocampal volume and glucose parameters in any group at the baseline, or in CHR and CTR at the follow-up time point.
Longitudinal changes in subfield volumes and glucose metabolism parameters
There was a statistically significant increase in insulin (t74 = 3.334, p = 0.0013, estimate = 4.49, 95% CI [1.8, 7.2], ES (Cohen’s d) = 1.12, ES 95% CI [0.4, 1.8],) and insulin resistance (t74 = 3.232, p = 0.0018, estimate = 0.549, 95% CI [0.2, 0.9], ES (Cohen’s d) = 1.08, ES 95% CI [0.4,1.8]) levels in CHR, but not in FEP or CTR during the follow-up period.
When we compared the difference of the change of insulin and insulin resistance between groups, we found that the increase in fasting insulin and insulin resistance was significantly different in CHR compared to CTR (insulin: Group by time t74 = 3.119, p = 0.003; insulin resistance: Group by time t74 = 2.970, p = 0.004), and in CHR compared to FEP (insulin: Group by time t74 = 2.447, p = 0.016; Group by time t74 = 2.380, p = 0.020) but not in FEP compared to CTR. Adjusting for lifetime exposure to antipsychotic medication at in each time point did not change these results.
There were no changes in whole hippocampal or subfield volumes within the follow-up period in FEP, CHR or CTR in a longitudinal linear mixed-effects model.
Analyses of hippocampal volumetry and glucose parameters based on clinical outcome groups
Baseline subfield volumetry and plasma glucose parameter levels based on outcome measures of transition, remission and functioning
The hippocampal tail volume was statistically significantly smaller in CHR-NC (p = 0.0002, FDR p = 0.0025), but not in CHR-C compared to CTR (p = 0.970, FDR p = 0.970). The difference in hippocampal tail volume between CHR-C and CHR-NC did not survive the FDR correction (p = 0.033, FDR p = 0.142). Also, the volumes of CA1 in the FEP group differed from those of CHR-NCs (p = 0.0099, FDR p = 0.079) but not from those of CHR-Cs (FDR p = 0.672). The tail volume was bigger in CHR-C compared to CHR-NC and CA1 was volume bigger in CHR-NC compared to FEP, but overall, these baseline differences did not survive FDR correction for multiple comparisons (Table 4 and Fig. 3). There were no differences between CHR-C and CHR-NC in whole hippocampal volumes at the baseline. The total hippocampal volume of CHR-NR, FEP-NR and FEP-R was smaller compared to CTR (Table 5). Comparisons of baseline volumes based on outcome measures are listed in Tables 4, 5 and 6.
Illustration of differences in CA1 and tail volumes of FEP, CHR-C, CHR-NC and CTR at baseline (A) and at one-year follow-up (B). The volumes are adjusted for age, sex, TIV and BMI.
There were no significant differences in baseline glucose parameter measures between CHR-C and CHR-NC, between FEPs with poor and good GAF or between CHRs with poor and good GAF. Further, no differences in the glucose parameters between nonremitting and remitting FEPs or nonremitting and remitting CHRs.
Associations between hippocampal volumetry and glucose parameters based on clinical outcomes
There was a trend of negative association between fasting plasma insulin or insulin resistance and hippocampal tail, but also between fasting plasma insulin or insulin resistance and total hippocampal volume at the follow-up time point of CHR converted to psychosis (Fig. 2 and Supplementary figure 1). However, the group of CHR-C was too small to study associations between glucose parameters and volumetry at the two time points. No associations between glucose parameters and hippocampal subfields or total volumes were observed in the subgroups of remission and functioning of FEP or CHR at either time point.
Longitudinal changes in glucose parameters and hippocampal volumetry based on outcome trajectories
During the follow-up period, insulin (t73 = 3.705, p = 0.0004, estimate = 9.97, 95% CI [4.6, 15.3], ES (Cohen’s d) = 2.5, ES 95% CI [1.1, 3.9],) and insulin resistance (t73 = 3.539, p = 0.0007, estimate = 1.205, 95% CI [0.5, 1.9], ES (Cohen’s d) = 2.4, ES 95% CI [1.0, 3.8],) levels significantly increased in CHR-C, but not in CHR-NC (Fig. 4). This result was not affected by adjusting the model for lifetime antipsychotic exposure. The longitudinal change in insulin and insulin resistance levels of CHR-C differed significantly from the change of CTRs (insulin: group by time t73 = 3.684, p < 0.001 and insulin resistance: group by time t73 = 3.501, p < 0.001). Similar differences were not observed between CHR-NCs and CTRs or FEPs and CTRs.
The change of fasting plasma insulin (A) and insulin resistance (B) during one-year follow-up. The increase in fasting plasma insulin and insulin resistance is statistically significant only in CHR-C. The model is adjusted for age, sex, and BMI. Adjusting for lifetime antipsychotic exposure did not change these results.
Also, insulin and insulin resistance increased significantly in CHR-NRs (insulin: t68 = 2.886, p = 0.0052, estimate = 7.513, 95% CI [2.3, 12.7], ES (Cohen’s d) = 1.9, ES 95% CI [0.5, 3.2]; insulin resistance: t68 = 2.849, p = 0.0058, estimate = 0.9321, 95% CI [0.3, 1.6], ES (Cohen’s d) = 1.8, ES 95% CI [0.5, 3.1]) and in CHRs with poor GAF (insulin: t72 = 3.573, p = 0.0006, estimate = 5.784, 95% CI [2.6, 9.0], ES (Cohen’s d) = 1.4, ES 95% CI [0.6, 2.2]); insulin resistance: t72 = 3.477, p = 0.0009, estimate = 0.7092, 95% CI [0.3, 1.1], ES (Cohen’s d) = 1.4, ES 95% CI [0.6, 2.2]) during the follow-up period. This was not observed in other groups. Adjusting for lifetime antipsychotic exposure did not change these results.
Further, the volume of the presubiculum in FEPs with good GAF increased significantly during the follow-up period (presubiculum: t91 = 3.079 p = 0.0027, estimate = 5.314, 95% CI [1.9, 8.7], ES (Cohen’s d) = 1.2, ES 95% CI [0.4, 1.9]). This result remained significant after adjusting for lifetime antipsychotic exposure. There were no significant longitudinal changes in subfield or total hippocampal volumes in CHR-C, CHR-NC, nonremitters, remitters or patients with poor or good GAF in FEP or CHR during the follow-up period.
Exploratory analyses
Non-affective psychosis had significantly smaller hippocampal tail volume compared to CTR (estimate = 42.05, 95% CI [25.9, 58.2] t201 = 5.14 p < 0.0001, FDR p < 0.0001). Similar difference was not observed between affective psychosis and CTR in the exploratory analyses using linear mixed-effects model. The hippocampal tail volume was significantly smaller in NAP compared to AP, but it did not survive FDR correction for multiple comparisons. There were no differences in other subfields or whole hippocampal volumes between NAP and AP. However, it seemed that there were more widespread reductions in the hippocampus in NAP compared to CTR than AP compared to CTR (Supplementary Table 4).
There were no significant associations between hippocampal total volume and glucose parameters, or subregion specific effects of glucose parameters on subfields in NAP or AP at the two time points. There was no significant difference in associations between fasting insulin or insulin resistance and hippocampal tail volume between the groups of NAP and AP at the follow-up.
The demographic information of the use of antidepressant and mood stabilizing medication at the two time points, and the results of the exploratory analyses of associations between lifetime antipsychotic exposure or antidepressant usage and hippocampal volumes using linear model, are presented in Supplementary table 3.
Discussion
Our study supports the view that hippocampal subfield morphology deficits are partly developmental and stable near the onset of first psychosis, being more robust in non-affective psychoses, but less prominent in the CHR group, that includes both patients transitioning and not transitioning to psychosis. This highlights the temporospatial dimension of hippocampal abnormalities in the development of psychotic disorders. A novel finding in this study is the significant inverse association between fasting plasma insulin or insulin resistance and hippocampal tail in non-diabetic FEP patients, at the one-year follow-up time point, independent of antipsychotic and antidepressant medications. Also, we found that insulin and insulin resistance worsened in CHR during the one-year follow-up period. In CHR, the worsening was related to clinical outcome trajectories such as transition to psychosis, non-remission and poor level of functioning. Similar longitudinal findings of the changes in glucose metabolism were not observed in CTR, FEP, non-converting CHR, or related to clinical outcome in FEP.
Our MRI imaging analysis of hippocampus subfields in early psychosis are mainly in line with earlier studies20,23,54. There was an overall reduction of subfield hippocampus volumes in patients with first-episode psychosis compared to population controls. This volume defect stayed stable during the one-year follow-up. The most pronounced volume reductions in the FEP group were observed in tail, CA1 and molecular layer of CA fields, independent of lifetime antipsychotic exposure. A similar trend of reduction in CA1 was also observed in subgroup of CHRs converting to psychosis. Instead, in CHR, only the hippocampal tail was smaller compared to CTRs, especially in those not converting to psychosis. This might mean that volume reduction in tail of CHR is not specific to psychosis or it might be related to the effect of antidepressants55,56 However, there was no association between the use of antidepressants and tail volume in the patient groups. Also, it is possible that some CHR-NC transition to psychosis only after the one-year follow-up period. Thus, these glucometabolic and subfield-specific findings in CHR converting to psychosis, and those in FEP that relate to glucose metabolism, may provide additional temporospatial information that represents traits and states differently in the progression of psychotic syndrome.
Temporospatial subfield defects are stable in early psychosis
The hippocampus is considered a key hub region in schizophrenia pathophysiology. The observed changes in hippocampus morphology are affected by the interaction of genes and the environment (GxE). The total volume of the hippocampus is moderately heritable in schizophrenia57 but there may be differences in the developmental trajectories of the separate hippocampal subfields58 and between posterior and anterior parts, but also between head-body-tail-division of the hippocampus59,60. These different divisions are partly overlapping which complicates the comparison and interpretation of the earlier studies on hippocampal morphology and related phenomena. Heritability of volume seems to be highest in the molecular layer, CA1, tail and DG, whereas smaller volume subfields, which were excluded from these analyses, seem to be less influenced by genetic factors in healthy twin studies61. Additionally, the reduced volume of the composite anterior CA subfields appears as a manifestation of the genetic vulnerability to schizophrenia also in unaffected relatives of schizophrenia patients62. A greater impact of genetic factors on the volumes of CA1 and molecular layer of CA fields is in line with our findings of a stable and possibly a developmentally early reduction of hippocampal subfield volumes. There is also evidence of hippocampal tail volume showing enrichment for schizophrenia-related genes58. In our sample, the majority of CHR-C converted to non-affective psychosis, and the majority of FEPs had a diagnosis of non-affective psychosis. Thus, these groups can be interpreted to primarily represent schizophrenia-spectrum psychoses, that have been shown to inherit a genetic liability to relatively smaller hippocampal volumes63,64. However, it is possible that the CA1 deficits are even more specific for schizophrenia22,54. Volume changes in the anterior hippocampus15, the region that is dominated by CA1 and subiculum subfield volumes65 have also been previously reported in genetic58,66 and MRI studies54,67,68 of psychoses. However, the relation between the anterior parts of hippocampus and schizophrenia seems to be more genetically driven, whereas the global reduction of hippocampal volume seen in later disease stages might be more strongly influenced by environmental factors and the disease process58.
Hypofunction of the N-methyl-D-aspartate (NMDA) glutamate receptor, as well as glutamate itself, are proposed to play a key role in the pathophysiology of schizophrenia69. There is evidence that the NMDA receptor, particularly in the CA1 subfield, has specific role in the function of the hippocampal comparator system involved in the processing of the match-mismatch of neural activity70, as well as in behavioral inhibition71. In addition, there is evidence of glutamate as a driver of hypermetabolism and atrophy starting in CA1 and spreading to the subiculum8,67. The CA1 subfield seems to be involved already in the prodromal phase of psychosis as implicated by elevated cerebral blood flow, hypermetabolism and volume loss in clinical high-risk patients who later convert to psychosis23,72,73 These findings further indicate that the changes in CA1 volumes seen in psychotic disorders are developmental in nature. In addition, the functions usually linked to the anterior region of the hippocampus such as regulation of anxiety and stress, sense of novelty, encoding, decision making, perception, imagination and episodic memory65, are all dimensions highly relevant for the clinical phenotypes of a patient with psychotic disorder.
Also, it has been shown that hippocampal function is altered in psychotic states with early life stress74,75. This might be due to abnormal input from the basolateral amygdala76. Volume reductions of the lateral nucleus of the amygdala are also related to childhood adversities in FEP77. The hippocampus is involved in the regulation of stress responses78, and stress on the other hand is associated with altered glucose metabolism79,80. Stress has also been suggested to induce changes in the morphology and dendrite spine density of the subiculum, CA1, CA3 and dentate gyrus76,78,81,82,83. Out of these regions, the CA1 and CA3 seem to be the primary hippocampal subfields influencing stress responses, including HPA axis activity78,84. On the other hand, hyperglycemia induces changes in synaptic function and neuronal loss particularly in the CA1 and CA3 subfields, in the studies on diabetic rats27,29,85,86. Hippocampal tail was not studied in these publications. Thus, the possible developmental defects in these subregions, especially in CA1, might be related to the development of dysregulation of glucose metabolism in patients who develop psychosis.
Glucose metabolism near the onset of psychosis
In this study, we found that insulin resistance worsened during the prodromal period near the onset of first psychosis. This dysregulation of glucose metabolism may be explained by genetic factors but also by the combined effect of non-specific factors such as insomnia, poor nutrition and stress. However, previous studies87,88 support our results that the changes in glucose homeostasis seem to increase during the prodrome and intensify when the onset of the first psychotic episode draws closer. Since we found worsening of insulin and insulin resistance in CHR, most of whom do not develop psychosis, it would support the view that worsening of glucose metabolism is more commonly related to non-psychotic mental illness progression40. However, the worsening of glucose metabolism was statistically significant only in CHR-C, but not in CHR-NC. Also, the volumetric association with insulin and insulin resistance was observed only in FEP, which suggests a different mechanism in relation to psychotic illness.
Previous studies have shown that insulin signaling, and glucose homeostasis are dysregulated in first-episode psychosis independent of antipsychotic treatment11,35,89,90. This is in line with the results of this study: FEPs had the most severe deviation of glucose parameters compared to CHRs and CTRs. Further, there is evidence suggesting that insulin signaling is involved in regulation of metabolism and behavior through affecting the functioning of the hippocampus and amygdala91. The involvement of the brain hippocampus, as one part of the circuitry25, in regulating normoglycemia has been well established26,92,93. The hippocampus has connections to the hypothalamus, that is considered a part of the brain-centered glucoregulatory system26,94,95, but it is also a part of stress-related regulatory systems, such as HPA-axis96. Both of these systems affect blood glucose and insulin levels, but possibly through different circuitries. Thus, it is possible that the hippocampus is a mediator of these functionally related, but anatomically separate circuitries97. To conclude, it is possible that the developmental deficits in the hippocampus may be related to its ability to regulate glucose metabolism, and based on our results, the impairment of this regulation manifests near the onset of first psychosis.
Hippocampus subfield morphology associates with insulin resistance in early psychosis
We found that higher insulin and insulin resistance associated statistically significantly with smaller hippocampal tail volume in FEP at one-year follow-up. Hereby, our result might indicate the bidirectional mechanism; the developmental and progressive deficits in the hippocampus may be related to its ability to regulate glucose metabolism, but also, that the dysfunctional hippocampus might further be more sensitive to the effects of glucose as the psychotic illness progress. This may lead to circular effect of progressive dysregulation of glucose metabolism, and increased volume loss in hippocampus in the progression of psychosis.
There is evidence that the anterior hippocampus is more strongly linked to the temporal pole and lateral temporal cortex, with only a small number of nerve fibres from these cortical areas projecting to the hippocampal tail. However, hippocampal tail has multiple connections to the primary visual cortex and the medial parietal cortex. These connections gradually decrease towards the head of the hippocampus98. Increased parietal and occipital lobe gyrification have been recently suggested to predict conversion to psychosis in clinical high-risk patients99. Gyrification index is considered a proxy marker for early cortical neurodevelopmental abnormalities100,101. These connections between hippocampal tail, parietal and occipital cortex, with possible different temporal manifestations in psychotic disorders, need to be studied further.
Widespread reductions in subfields, including hippocampal tail have been previously reported in type 2 diabetes, but not in prediabetes34. In our sample, fasting glucose values ranged from normal to prediabetic levels. Thus, it is possible that the mechanisms related to the tail volume defects associate with the worsening of glucose metabolism and are more related to the pathophysiology of psychotic illness itself, rather than to co-morbid diabetes. Also, these associations seem to emerge only after the onset of first psychosis. This association might also indirectly reflect other phenomena related to psychosis such as disruption of the blood brain barrier102. There is evidence of progressive volumetric changes of the hippocampal tail in chronic schizophrenia23, but also of a potential shared role of inflammation in insulin resistance and schizophrenia103. In our study, there was no difference in the associations between insulin and insulin resistance and tail volume while comparing first episode NAP and AP groups. Although the tail volume was smaller only in NAP compared to CTR, there was no significant changes in the tail volumes in FEPs in our longitudinal analyses. It is possible that progressive decline in tail volume associating with dysfunctional glucose metabolism may start after the first psychotic episode, but this might be related to a more chronic non-affective psychosis, or more specifically to chronic schizophrenia.
Clinical outcome trajectories, hippocampus morphometry and glucose metabolism
We found associations between increased insulin or insulin resistance and impaired measures of clinical outcome, such as nonremission, poor daily functioning and transition to psychosis, in CHR. Hypothetically, this could be due to individual health behaviors, such as exercise and diet, which are subject to be influenced by symptomatology. However, a recent longitudinal study found worsening of fasting glucose levels in recent-onset psychosis, despite of improving lifestyle habits, clinical improvement and thus decreasing antipsychotic medication44. This might mean that deteriorating glucose metabolism is related rather to psychotic illness progression than poor health behaviors. Also, since we observed a worsening of insulin resistance near the onset of psychosis, it is possible, that the association between the deteriorating glucose metabolism and measures of outcome indicates that the metabolic processes seen in psychotic disorders progress during the prodromal period. This would support the current view of psychotic disorder as a systemic disease.
Notably, the FEPs with good level of functioning at follow-up had an increase in presubiculum volume. This might be evidence that the presubiculum is subject to state rather than trait influences, or it could alternatively be a result of therapeutic effects104, exposure to antipsychotic medication, or other confounding factors105,106. It is possible that the volume increases of the presubiculum are related to specific types of antipsychotic or antidepressant medication107. However, antipsychotic exposure or the use of antidepressants was not significantly associated with presubiculum morphology. Although our results of an association between presubiculum volumes and better functional outcome should be ultimately regarded as preliminary, it should be noted that they are in line with earlier reports of better GAF associating with bilateral increases108 or unchanged volumes109 of the hippocampus in FEP. In CHR, the results from previous studies of the associations between outcome and hippocampus volumes have been inconsistent110.
Strengths and limitations
This study included characterizations of two clinical samples at baseline and follow-up, as well as across a one-year follow-up period. We used state-of-the-art methodology (FreeSurfer 7.1.1) for segmentation of hippocampus subfield volumes. We were also able to control for the confounding effects of medication by determining lifetime cumulative antipsychotic exposure and daily use of antidepressants. The clinical groups were compared to a random population sample control group. This study has two main limitations. Firstly, we were able to include only modest sample sizes. This is particularly true for the analyses including smaller subgroups of CHR-C, CHR-NC, NAP and AP. The follow-up period for determining the division into CHR-C and CHR-NC groups was adequate, but not optimal; the CHR-C group could possibly have been increased by extending the follow-up time. Secondly, the position of the internal boundaries between the hippocampal substructures segmented using scans with 1mm isometric voxel size relies heavily on prior knowledge from ex vivo training data. For this reason, the volumes of the internal subfields (dentate gyrus, molecular layer and CA4) must be interpreted with caution.
Conclusions
Our longitudinal study suggests that higher fasting plasma insulin and insulin resistance associates with smaller hippocampal tail volumes in non-diabetic first-episode psychosis patients. Also, we found that insulin and insulin resistance worsened in CHR during the follow-up period. This effect was driven by high-risk patients converting to psychosis. In CHR, the glucometabolic deterioration was related to clinical outcome trajectories, such as transition to psychosis, non-remission, and poor level of functioning.
Our findings support the idea that psychotic disorders have heterogenous temporospatial subfield defects, but also common systemic manifestations near the onset of first psychosis, which could be relevant for understanding the etiology and progression of these disorders. Also, clinical attention should increasingly be diverted to the somatic health and metabolic state in the context of high clinical risk for psychosis and first episode of psychosis, as glucose metabolism appears to be related to both clinical outcome trajectories as well as hippocampal morphology in early psychosis.
Data availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Code availability
R code available upon request. All analyses were carried out with R version 4.2.1 (2022-06-23) “Funny-Looking Kid”111
Change history
02 September 2024
A Correction to this paper has been published: https://doi.org/10.1038/s41537-024-00495-9
References
Haug, J. O. Pneumoencephalographic evidence of brain atrophy in acute and chronic schizophrenic patients. Acta Psychiatr. Scand. 66, 374–383 (1982).
DeLisi, L. E. Defining the course of brain structural change and plasticity in schizophrenia. Psychiatry Res Neuroimaging 92, 1–9 (1999).
Wright, I. C. et al. Meta-Analysis of Regional Brain Volumes in Schizophrenia. Am. J. Psychiatry 157, 16–25 (2000).
Geijselaers, S. L. C., Sep, S. J. S., Stehouwer, C. D. A. & Biessels, G. J. Glucose regulation, cognition, and brain MRI in type 2 diabetes: a systematic review. Lancet Diab. Endocrinol. 3, 75–89 (2015).
Sim, K. et al. Hippocampal and Parahippocampal Volumes in Schizophrenia: A Structural MRI Study. Schizophr. Bull. 32, 332–340 (2006).
van Erp, T. G. M. et al. Subcortical brain volume abnormalities in 2028 individuals with schizophrenia and 2540 healthy controls via the ENIGMA consortium. Mol. Psychiatry 21, 547–553 (2016).
Okada, N. et al. Abnormal asymmetries in subcortical brain volume in schizophrenia. Mol. Psychiatry 21, 1460–1466 (2016).
Lieberman, J. A. et al. Hippocampal dysfunction in the pathophysiology of schizophrenia: a selective review and hypothesis for early detection and intervention. Mol Psychiatry 1–9 23, 1764–1772 (2018) https://doi.org/10.1038/mp.2017.249.
Baglivo, V. et al. Hippocampal Subfield Volumes in Patients With First-Episode Psychosis. Schizophr. Bull. 44, 552–559 (2018).
Ho, N. F. et al. Progressive Decline in Hippocampal CA1 Volume in Individuals at Ultra-High-Risk for Psychosis Who do not Remit: Findings from the Longitudinal Youth at Risk Study.Neuropsychopharmacology i, 1–10 (2017)..
Pillinger, T. et al. Impaired Glucose Homeostasis in First-Episode Schizophrenia. JAMA Psychiatry 74, 261 (2017).
Pillinger, T., McCutcheon, R. A. & Howes, O. D. Variability of glucose, insulin, and lipid disturbances in first-episode psychosis: A meta-analysis. Psychol. Med 53, 3150–3156 (2023).
Roeske, M. J. et al. Incomplete hippocampal inversion in schizophrenia: prevalence, severity, and impact on hippocampal structure. Mol. Psychiatry 26, 5407–5416 (2021).
Knight, S. et al. Hippocampal circuit dysfunction in psychosis. Translational Psychiatry 12, 344 (2022).
Chopra, S. et al. Network-Based Spreading of Gray Matter Changes Across Different Stages of Psychosis. JAMA Psychiatry, https://doi.org/10.1001/jamapsychiatry.2023.3293 (2023).
Adriano, F., Caltagirone, C. & Spalletta, G. Hippocampal volume reduction in first-episode and chronic schizophrenia: a review and meta-analysis. Neuroscientist 18, 180–200 (2012).
Velakoulis, D. et al. Hippocampal volume in first-episode psychoses and chronic schizophrenia: A high-resolution magnetic resonance imaging study. Arch. Gen. Psychiatry 56, 133–141 (1999).
Walter, A. Hippocampal volume in subjects at clinical high-risk for psychosis: A systematic review and meta-analysis. Neurosci Biobehav Rev 71, 680–690 (2016).
Jalbrzikowski, M. et al. Association of Structural Magnetic Resonance Imaging Measures with Psychosis Onset in Individuals at Clinical High Risk for Developing Psychosis: An ENIGMA Working Group Mega-analysis. JAMA Psychiatry 78, 753–766 (2021).
Hinney, B., Walter, A., Aghlmandi, S., Andreou, C. & Borgwardt, S. Does Hippocampal Volume Predict Transition to Psychosis in a High-Risk Group?. A Meta-Analysis 14, 614659 (2021).
Wood, S. J. et al. Hippocampal pathology in individuals at ultra-high risk for psychosis: A multi-modal magnetic resonance study. Neuroimage 52, 62–68 (2010).
McHugo, M. et al. Hippocampal volume in early psychosis: a 2-year longitudinal study. Transl. Psychiatry 10(1), 306 (2020).
Sasabayashi, D. et al. Reduced Hippocampal Subfield Volume in Schizophrenia and Clinical High-Risk State for Psychosis. Front. Psychiatry 12, 642048 (2021).
Adel K. Afifi & Ronald A. Bergman. Functional Neuroanatomy: Text and Atlas, 2nd Edition. (2005).
Tingley, D., McClain, K., Kaya, E., Carpenter, J. & Buzsáki, G. A metabolic function of the hippocampal sharp wave-ripple. Nature 597, 82–86 (2021).
Schwartz, M. W. et al. Cooperation between brain and islet in glucose homeostasis and diabetes. Nature 503, 59–66 (2013).
Sima, A. A. F., Kamiya, H. & Li, Z. G. Insulin, C-peptide, hyperglycemia, and central nervous system complications in diabetes. Eur J Pharmacol vol. 490, 187–197 (2004).
Hayashi, K. et al. Association of cognitive dysfunction with hippocampal atrophy in elderly Japanese people with type 2 diabetes. Diab. Res Clin. Pr. 94, 180–185 (2011).
Kamal, A., Biessels, G. J., Urban, I. J. A. & Gispen, W. H. Hippocampal Synaptic Plasticity in Streptozotocin- Diabetic Rats: Impairment of Long-Term Potentiation and Facilitation of Long-Term Depression. Neuroscience 90, 737–745 (1999).
Biessels, G. J., Staekenborg, S., Brunner, E., Brayne, C. & Scheltens, P. Risk of dementia in diabetes mellitus: a systematic review. Lancet Neurol. 5, 64–74 (2006).
Bruehl, H., Wolf, O. T. & Convit, A. A blunted cortisol awakening response and hippocampal atrophy in type 2 diabetes mellitus. Psychoneuroendocrinology 34, 815–821 (2009).
Frangou, S. et al. Hippocampal volume reduction is associated with direct measure of insulin resistance in adults. Neurosci. Res 174, 19–24 (2022).
Li, M. et al. Altered Hippocampal Subfields Volumes Is Associated With Memory Function in Type 2 Diabetes Mellitus. Front. Neurol. 12, 756500 (2021).
Monereo-Sánchez, J. et al. The association of prediabetes and type 2 diabetes with hippocampal subfields volume: The Maastricht study. Neuroimage Clin 39, 103455 (2023).
Perry, B. I., McIntosh, G., Weich, S., Singh, S. & Rees, K. The association between first-episode psychosis and abnormal glycaemic control: systematic review and meta-analysis. Lancet Psychiatry 3, 1049–1058 (2016).
Petrikis, P. et al. Parameters of glucose and lipid metabolism at the fasted state in drug-naïve first-episode patients with psychosis: Evidence for insulin resistance. Psychiatry Res. 229, 901–904 (2015).
Tosato, S. et al. Childhood trauma and glucose metabolism in patients with first-episode psychosis. Psychoneuroendocrinology 113, 104536 (2020).
Misiak, B. et al. A meta-analysis of blood and salivary cortisol levels in first-episode psychosis and high-risk individuals. Front Neuroendocrinol 62, 100930 (2021).
Pillinger, T., D’Ambrosio, E., McCutcheon, R. & Howes, O. D. Is psychosis a multisystem disorder? A meta-review of central nervous system, immune, cardiometabolic, and endocrine alterations in first-episode psychosis and perspective on potential models. Mol. Psychiatry, https://doi.org/10.1038/s41380-018-0058-9 (2018).
Kucukgoncu, S. et al. Glucose metabolism dysregulation at the onset of mental illness is not limited to first episode psychosis: A systematic review and meta-analysis. Early Intervention Psychiatry 13, 1021–1031 (2019).
Ryan, M. C., Collins, P., Thakore, J. H. Impaired Fasting Glucose Tolerance in First-Episode, Drug-Naive Patients With Schizophrenia. Am. J. Psychiatry 160 http://ajp.psychiatryonline.org (2003).
Pillinger, T., Beck, K., Stubbs, B. & Howes, O. D. Cholesterol and triglyceride levels in first-episode psychosis: systematic review and meta-analysis. Br. J. Psychiatry bjp.bp.117.200907, https://doi.org/10.1192/bjp.bp.117.200907 (2017).
Nettis, M. A. et al. Metabolic-inflammatory status as predictor of clinical outcome at 1-year follow-up in patients with first episode psychosis. Psychoneuroendocrinology 99, 145–153 (2019).
Alonso, Y. et al. Risk factors for metabolic syndrome in individuals with recent-onset psychosis at disease onset and after 1-year follow-up. Sci Rep 12, (2022).
Qi, W. et al. Hippocampal Subfield Volumes Predict Disengagement from Maintenance Treatment in First Episode Schizophrenia. Schizophr. Bull. 49, 34–42 (2023).
Andreasen, N. C. et al. Reviews and Overviews Remission in Schizophrenia: Proposed Criteria and Rationale for Consensus. Am. J. Psychiatry 162 http://ajp.psychiatryonline.org (2005).
Iglesias, J. E. et al. A computational atlas of the hippocampal formation using ex vivo, ultra-high resolution MRI: Application to adaptive segmentation of in vivo MRI. Neuroimage 115, 117–137 (2015).
Sämann, P. G. et al. FreeSurfer-based segmentation of hippocampal subfields: A review of methods and applications, with a novel quality control procedure for ENIGMA studies and other collaborative efforts. Hum. Brain Mapping 43, 207–233 (2022).
Wallace, T. M., Levy, J. C. & Matthews, D. R. Use and Abuse of HOMA Modeling. http://diabetesjournals.org/care/article-pdf/27/6/1487/645883/zdc00604001487.pdf (2004).
Song, Y. S., Hwang, Y. C., Ahn, H. Y. & Park, C. Y. Comparison of the usefulness of the updated homeostasis model assessment (HOMA2) with the original HOMA1 in the prediction of type 2 diabetes mellitus in Koreans. Diab. Metab. J. 40, 318–325 (2016).
Leucht, S. et al. Dose Equivalents for Second-Generation Antipsychotics: The Minimum Effective Dose Method. Schizophr. Bull. 40, 314–326 (2014).
Opler, L. A., Kay, S. R., Lindenmayer, J. P. & Fiszbein, A. Structured clinical interview: The positive and negative syndrome scale (SCI-PANSS). North Tonawanda, NY: Multi-Health Systems (1999).
Leucht, S., Rothe, P., Davis, J. M. & Engel, R. R. Equipercentile linking of the BPRS and the PANSS. Eur. Neuropsychopharmacol. 23, 956–959 (2013).
McHugo, M. et al. Smaller anterior hippocampal subfields in the early stage of psychosis. Transl Psychiatry 14, (2024).
Nogovitsyn, N. et al. Hippocampal tail volume as a predictive biomarker of antidepressant treatment outcomes in patients with major depressive disorder: a CAN-BIND report. Neuropsychopharmacology 45, 283–291 (2020).
Qi, W. et al. Effect of citalopram on hippocampal volume in first-episode schizophrenia: Structural MRI results from the DECIFER trial. Psychiatry Res. Neuroimaging 312, 111286 (2021).
Roalf, D. R. et al. Heritability of subcortical and limbic brain volume and shape in multiplex-multigenerational families with schizophrenia. Biol. Psychiatry 77, 137–146 (2015).
van der Meer, D. et al. Brain scans from 21,297 individuals reveal the genetic architecture of hippocampal subfield volumes. Mol. Psychiatry 25, 3053–3065 (2020).
Gogtay, N. et al. Dynamic mapping of normal human hippocampal development. Hippocampus 16, 664–672 (2006).
Lynch, K. M., Shi, Y., Toga, A. W. & Clark, K. A. Hippocampal shape maturation in childhood and adolescence. Cereb. Cortex 29, 3651–3665 (2019).
Vilor-Tejedor, N. et al. Genetic Influences on Hippocampal Subfields: An Emerging Area of Neuroscience Research. Neurology: Genetics. 7, e591 (2021).
Choi, S. et al. Volume deficits in hippocampal subfields in unaffected relatives of schizophrenia patients with high genetic loading but without any psychiatric symptoms. Schizophr. Res 240, 125–131 (2022).
Cheng, W. et al. Shared genetic architecture between schizophrenia and subcortical brain volumes implicates early neurodevelopmental processes and brain development in childhood. Mol. Psychiatry https://doi.org/10.1038/s41380-022-01751-z (2022).
Li, Z. & Chen, X. Comprehensive analysis of shared genetic loci between hippocampal volume and schizophrenia. Psychiatry Res. 316, 114795 (2022).
Zeidman, P. & Maguire, E. A. Anterior hippocampus: The anatomy of perception, imagination and episodic memory. Nat. Rev. Neurosci. 17, 173–182 (2016).
Trubetskoy, V. et al. Mapping genomic loci implicates genes and synaptic biology in schizophrenia. Nature 604, 502–508 (2022).
Schobel, S. A. et al. Differential targeting of the CA1 subfield of the hippocampal formation by schizophrenia and related psychotic disorders. Arch. Gen. Psychiatry 66, 938–946 (2009).
Ho, N. F. et al. Progression from selective to general involvement of hippocampal subfields in schizophrenia. Mol. Psychiatry 22, 142–152 (2017).
Nakazawa, K. & Sapkota, K. The origin of NMDA receptor hypofunction in schizophrenia. Pharmacol. Ther. 205, 107426 (2020).
Kumaran, D. & Maguire, E. A. Match-mismatch processes underlie human hippocampal responses to associative novelty. J. Neurosci. 27, 8517–8524 (2007).
Taylor, A. M. et al. Hippocampal NMDA receptors are important for behavioural inhibition but not for encoding associative spatial memories. Philosophical Transac. Royal Soc. B: Biol. Sci. 369, 20130149 (2014).
Schobel, S. A. et al. Imaging Patients with Psychosis and a Mouse Model Establishes a Spreading Pattern of Hippocampal Dysfunction and Implicates Glutamate as a Driver. Neuron 78, 81–93 (2013).
Provenzano, F. A. et al. Hippocampal Pathology in Clinical High-Risk Patients and the Onset of Schizophrenia. Biol. Psychiatry 87, 234–242 (2020).
Borges, S., Gayer-Anderson, C. & Mondelli, V. A systematic review of the activity of the hypothalamic–pituitary–adrenal axis in first episode psychosis. Psychoneuroendocrinology 38, 603–611 (2013).
Murphy, F. et al. Childhood Trauma, the HPA Axis and Psychiatric Illnesses: A Targeted Literature Synthesis. Front. Psychiatry 13, 748372 (2022).
Kim, J. J. & Diamond, D. M. The stressed hippocampus, synaptic plasticity and lost memories. Nat. Rev. Neurosci. 3, 453–462 (2002).
Armio, R. L. et al. Amygdala subnucleus volumes in psychosis high-risk state and first-episode psychosis: Amygdala subnuclei and psychosis. Schizophr. Res 215, 284–292 (2020).
Lowry, C. A. Functional Subsets of Serotonergic Neurones: Implications for Control of the Hypothalamic-Pituitary-Adrenal Axis. J. Neuroendocrinol. 14, 911–923 (2002).
Armario, A. Acute stress markers in humans: Response of plasma glucose, cortisol and prolactin to two examinations differing in the anxiety they provoke. Psychoneuroendocrinology 21, 17–24 (1996).
Gagliano, H., Ortega-Sanchez, J. A., Nadal, R. & Armario, A. Psychostimulants and forced swim stress interaction: how activation of the hypothalamic-pituitary-adrenal axis and stress-induced hyperglycemia are affected. Psychopharmacol. (Berl.) 234, 2859–2869 (2017).
Conrad, C. D. What Is the Functional Significance of Chronic Stress-Induced CA3 Dendritic Retraction Within the Hippocampus? Behav. Cogn. Neurosci. Rev. 5, 41–60 (2006).
Belujon, P. & Grace, A. A. Hippocampus, amygdala, and stress: interacting systems that affect susceptibility to addiction. Ann. N. Y Acad. Sci. 1216, 114–121 (2011).
Belujon, P. & Grace, A. A. Regulation of dopamine system responsivity and its adaptive and pathological response to stress. Proc. Roy. Soc. B: Biol. Sci. 282, 20142516 (2015).
Steullet, P. et al. Redox Dysregulation Affects the Ventral But Not Dorsal Hippocampus: Impairment of Parvalbumin Neurons, Gamma Oscillations, and Related Behaviors. J. Neurosci. 30, 2547–2558 (2010).
Biessels, G. J. et al. Place learning and hippocampal synaptic plasticity in streptozotocin-induced diabetic rats. Diabetes 45, 1259–1266 (1996).
Li, Z.-G., Zhang, W., Grunberger, G. & Sima, A. A. F. Hippocampal neuronal apoptosis in type 1 diabetes. Brain Res 946, 221–231 (2002).
Sormunen, E. et al. Metabolic trajectories in childhood and adolescence: Effects on risk for schizophrenia. Schizophrenia 8, 82 (2022).
Walker, E. F. et al. Cortisol levels and risk for psychosis: Initial findings from the North American Prodrome Longitudinal Study. Biol. Psychiatry 74, 410–417 (2013).
Zaki, J. K. et al. Diagnostic model development for schizophrenia based on peripheral blood mononuclear cell subtype-specific expression of metabolic markers. Transl. Psychiatry 12, 457 (2022).
Chouinard, V. A. et al. Impaired insulin signaling in unaffected siblings and patients with first-episode psychosis. Mol. Psychiatry 24, 1513–1522 (2019).
Soto, M., Cai, W., Konishi, M. & Kahn, C. R. Insulin signaling in the hippocampus and amygdala regulates metabolism and neurobehavior. Proc. Natl Acad. Sci. USA 116, 6379–6384 (2019).
Garfield, A. S. et al. A parabrachial-hypothalamic cholecystokinin neurocircuit controls counterregulatory responses to hypoglycemia. Cell Metab. 20, 1030–1037 (2014).
Deem, J. D., Muta, K., Scarlett, J. M., Morton, G. J. & Schwartz, M. W. How should we think about the role of the brain in glucose homeostasis and diabetes? in Diabetes vol. 66 1758–1765 (American Diabetes Association Inc., 2017).
Toda, C. et al. UCP2 Regulates Mitochondrial Fission and Ventromedial Nucleus Control of Glucose Responsiveness. Cell 164, 872–883 (2016).
Scarlett, J. M. & Schwartz, M. W. Gut-brain mechanisms controlling glucose homeostasis. F1000Prime Rep 7, 12 (2015).
Herman, J. P., Ostrander, M. M., Mueller, N. K. & Figueiredo, H. Limbic system mechanisms of stress regulation: Hypothalamo-pituitary- adrenocortical axis. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 29, 1201–1213 (2005).
Valli, I. et al. HPA-axis function and grey matter volume reductions: imaging the diathesis-stress model in individuals at ultra-high risk of psychosis. Transl. Psychiatry 6, e797–e797 (2016).
Strange, B. A. Functional organization of the hippocampal longitudinal axis, https://doi.org/10.1038/nrn3785. (2014).
Basavaraju, R. et al. Increased parietal and occipital lobe gyrification predicts conversion to syndromal psychosis in a clinical high-risk cohort. Schizophr. Res 255, 246–255 (2023).
White, T., Su, S., Schmidt, M., Kao, C. Y. & Sapiro, G. The development of gyrification in childhood and adolescence. Brain and Cogn 72, 36–45 (2010).
Kalantar-Hormozi, H. et al. A cross-sectional and longitudinal study of human brain development: The integration of cortical thickness, surface area, gyrification index, and cortical curvature into a unified analytical framework. Neuroimage 268, 119885 (2023).
Najjar, S. et al. Neurovascular Unit Dysfunction and Blood-Brain Barrier Hyperpermeability Contribute to Schizophrenia Neurobiology: A Theoretical Integration of Clinical and Experimental Evidence. Front. Psychiatry 8, 83, https://doi.org/10.3389/fpsyt.2017.00083 (2017).
Perry, B. I. et al. The potential shared role of inflammation in insulin resistance and schizophrenia: A bidirectional two-sample mendelian randomization study. PLoS Med 18, e1003455 (2021).
Lahti, A. C., Weiler, M. A., Holcomb, H. H., Tamminga, C. A. & Cropsey, K. L. Modulation of limbic circuitry predicts treatment response to antipsychotic medication: A functional imaging study in schizophrenia. Neuropsychopharmacology 34, 2675–2690 (2009).
Power, P. Outcome and recovery in first-episode psychosis. British J. Psychiatry 211, 331–333 (2017).
Salokangas, R. K. R. et al. Short-term functional outcome in psychotic patients: results of the Turku early psychosis study (TEPS). BMC Psychiatry 21, (2021).
Seo, M. K. et al. Effects of antidepressant drugs on synaptic protein levels and dendritic outgrowth in hippocampal neuronal cultures. Neuropharmacology 79, 222–233 (2014).
Lappin, J. M. et al. Bilateral hippocampal increase following first-episode psychosis is associated with good clinical, functional and cognitive outcomes. Psychol. Med 44, 1279–1291 (2014).
Anvari, A. A. et al. Hippocampal volume change relates to clinical outcome in childhood-onset schizophrenia. Psychol. Med 45, 2667–2674 (2015).
Tognin, S. et al. The Relationship Between Grey Matter Volume and Clinical and Functional Outcomes in People at Clinical High Risk for Psychosis. Schizophr Bull Open 3, sgac040, (2022).
R Core Team. R Core Team: A language and environment for statistical computing. R Foundation for Statistical Computing. Vienna, Austria. URL https://www.R-project.org/ (2022).
Acknowledgements
This work was supported by funding for the VAMI-project (Turku University Hospital, state research funding, no. P3848), partly supported by EU FP7 grants (PRONIA, grant a # 602152 and METSY grant #602478). Dr. Armio received personal funding from Doctoral Programme in Clinical Research at the University of Turku, grants from State Research. Funding, Turunmaa Duodecim Society, Finnish Psychiatry Research Foundation, Finnish University Society of Turku (Valto Takala Foundation), Tyks-foundation, The Finnish Medical Foundation (Maija and Matti Vaskio fund), University of Turku, The Alfred Kordelin Foundation, Finnish Cultural Foundation (Terttu Enckell fund and Ritva Helminen fund) and The Alfred Kordelin foundation. Further, Dr. Tuominen received personal grant from Sigrid Juselius and Orion research foundation and NARSAD Young Investigator Grant from the Brain & Behavior Research Foundation. We thank Dr. Harri Merisaari for help with the MRI analyses and the Turku PET center and VAMI group personnels for great technical support.
Author information
Authors and Affiliations
Contributions
Reetta-Liina Armio collected most of the MRI data and attended to design the analysis. Also, she performed all data processing, quality control and most of the statistical tests and wrote the manuscript. Study conception and design: Lauri Tuominen, Reetta-Liina Armio, Jarmo Hietala. Contribution in data collection: Reetta-Liina Armio, Heikki Laurikainen, Maija Walta, Elina Sormunen, Tuula Ilonen, Raimo KR Salokangas, Lauri Tuominen, Nikolaos Koutsouleris. Draft manuscript preparation: Reetta-Liina Armio, Heikki Laurikainen, Lauri Tuominen, Jarmo Hietala, Raimo KR Salokangas, Arvi Tolvanen. Statistical analysis Reetta-Liina Armio, Arvi Tolvanen, Lauri Tuominen. Interpretation of results: Reetta-Liina Armio, Lauri Tuominen, Jarmo Hietala, Arvi Tolvanen.
Corresponding author
Ethics declarations
Competing interests
We declare that none of the authors have competing financial or non-financial interests as defined by Nature Portfolio.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Armio, RL., Laurikainen, H., Ilonen, T. et al. Longitudinal study on hippocampal subfields and glucose metabolism in early psychosis. Schizophr 10, 66 (2024). https://doi.org/10.1038/s41537-024-00475-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41537-024-00475-z