Abstract
Neuroimaging studies have revealed that the mechanisms of auditory hallucinations are related to morphological changes in multiple cortical regions, but studies on brain network properties are lacking. This study aims to construct intra-individual structural covariance networks and reveal network changes related to auditory hallucinations. T1-weighted MRI images were acquired from 90 schizophrenia patients with persistent auditory hallucinations (pAH group), 55 schizophrenia patients without auditory hallucinations (non-pAH group), and 83 healthy controls (HC group). Networks were constructed using the voxel-based gray matter volume and the intra-individual structural covariance was based on the similarity between the morphological variations of any two regions. One-way ANCOVA was employed to compare global and local network metrics among the three groups, and edge analysis was conducted via network-based statistics. In the pAH group, Pearson correlation analysis between network metrics and clinical symptoms was conducted. Compared with the HC group, both the pAH group (p = 0.01) and the non-pAH group (p = 3.56 × 10−4) had lower nodal efficiency of the left medial superior frontal gyrus. Compared to the non-pAH group and HC group, the pAH group presented lower nodal efficiency of the temporal pole of the left superior temporal gyrus (p = 1.09 × 10−3; p = 7.67 × 10−4) and right insula (p = 0.02; p = 8.99 × 10−6), and lower degree centrality of the right insula (p = 0.04; p = 1.65 × 10−5). The pAH group had a subnetwork with reduced structural covariance centered by the left temporal pole of the superior temporal gyrus. In the pAH group, the normalized clustering coefficient (r = −0.36, p = 8.45 × 10−3) and small-worldness (r = −0.35, p = 9.89 × 10−3) were negatively correlated with the PANSS positive scale score. This study revealed network changes in schizophrenia patients with persistent auditory hallucinations, and provided new insights into the structural architecture related to auditory hallucinations at the network level.
Similar content being viewed by others
Introduction
Schizophrenia is a disabling psychiatric disorder with a 1% of lifetime prevalence rate where patients have a reduced life span by more than ten years1,2. Schizophrenia is characterized by psychotic symptoms including hallucinations, delusions, disorganized speech, disorderly behavior, and negative symptoms such as apathy or avolition3. Among these common psychotic symptoms, auditory hallucinations are considered as one of the core positive symptoms of schizophrenia, and 60–90% of patients experience such symptoms4. There is no consensus on the definition of persistent auditory hallucinations, and in some studies, they can be defined as symptoms that persist for more than one year after treatment with two different antipsychotic drugs5. Auditory hallucinations are associated with many negative outcomes including emotional problems, cognitive dysfunction, violent behavior, and suicidal attempts6,7,8,9,10,11.
To date, neuroimaging research has been conducted on morphological brain abnormalities related to auditory hallucinations. Surface-based morphometry studies have focused mostly on cortical thickness and have revealed that patients with auditory verbal hallucinations have reduced thickness in the bilateral lateral orbitofrontal cortices, bilateral middle temporal gyri, right inferior temporal gyrus, bilateral postcentral gyri, right Heschl’s gyrus, right rostral anterior cingulate gyrus, right supramarginal gyrus, and right insula12,13,14,15,16. Additionally, patients have increased thickness in the left insula, bilateral anterior and posterior cingulate gyri, dorsal middle frontal gyrus, and parietal lobe13. Voxel-based morphometry studies have focused mostly on the gray matter volume, and meta-analyses have shown that auditory hallucination severity is related to reduced volume of the left insula and bilateral superior temporal gyri17,18,19,20. These studies have focused mainly on morphological changes in certain regions related to auditory hallucinations, but network properties of the human brain as a complex network are still not completely understood.
Brain networks based on structural magnetic resonance imaging (sMRI) data are called structural covariance networks and include the correlation-based approach and the source-based morphometry approach21. In the correlation-based approach, pairwise correlation coefficients are calculated for morphometric measures in regions of interest (ROIs), resulting in one group-level network for each group21. Common morphometric measures include cortical thickness or volume22. In the source-based morphometry approach, the independent components analysis is applied without a priori region selection to create network components for group comparisons21. Between the two methods, the correlation-based approach is commonly used in graph theory analysis. Graph-based networks are constructed by setting brain regions as nodes and structural or functional correlations between nodes as edges22, and they have been shown to have a small-world topology with balanced local specialization and global integration23,24. To date, graph-based network studies have focused mainly on schizophrenia25,26,27, and characteristics related to auditory hallucinations have been less studied. In addition, although traditional network studies are able to depict the brain network in a group, variations at the individual level cannot be reflected. To solve this problem, intra-individual structural covariance network emerges, creating a brain network for each individual in a group. On the basis of the method proposed by Yun et al. 28, the degree of morphological variation (z-score) for each region is first calculated relative to the average level of healthy controls. A z-score > 0 represents morphological increment while a z-score < 0 represents morphological shrinkage. The pairwise regional covariance for creating an intra-individual network is then calculated based on the similarity of morphological variations28. Since patients with schizophrenia have excessive anatomical changes, as mentioned above, this method is suitable for morphological studies of patients with schizophrenia.
This study was designed to construct intra-individual structural covariance networks and conduct graph theory analysis in schizophrenia patients with or without persistent auditory hallucinations and healthy controls. Differences in global, local, and edge metrics were compared, and relationships between network metrics and clinical symptoms were explored. We hypothesized that: (1) patients with persistent auditory hallucinations would show significant differences in the network metrics compared with those without auditory hallucinations and healthy controls and that (2) the altered network metrics would show significant correlations with clinical symptoms in patients with persistent auditory hallucinations.
Methods
Participants
A total of 145 patients with schizophrenia were enrolled from the psychiatric clinic of the Second Xiangya Hospital of Central South University in China, and 83 sex- and age-matched healthy controls (HC group) were enrolled from local communities. All patients were diagnosed by two experienced psychiatrists according to the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5)3 and all participants were evaluated according to the Chinese version of the Mini-International Neuropsychiatric Interview (M.I.N.I.)29. After the evaluation, participants whose abilities to understand the study were confirmed by both psychiatrists were considered for enrollment. Formal estimation of intelligence quotient was not performed in our study. The inclusion criteria for all participants were as follows: (1) of Han Chinese descent and aged between 16 and 45 years; (2) right-handed; and (3) normal hearing. The exclusion criteria for all participants were as follows: (1) a history of other psychiatric disorders; (2) a history of drug or alcohol abuse; (3) a history of head trauma with consciousness disturbances lasting more than 5 min; (4) a history of severe endocrine or other physical illnesses; (5) pregnant or lactating; and (6) MRI incompatibility. The additional exclusion criterion for schizophrenia patients was a history of electroconvulsive treatment, and for healthy controls, the additional exclusion criterion was a family history of psychiatric disorders.
Patients were divided into schizophrenia patients with persistent auditory hallucinations (pAH group) and schizophrenia patients without auditory hallucinations (non-pAH group). The pAH group included 90 patients who: (1) experienced auditory verbal hallucinations at least once daily for at least one year; (2) were unresponsive to at least two types of antipsychotics; and (3) had a score > 3 on PANSS P3 item15,30,31. The non-pAH group included 55 patients who had never experienced AVH and had a PANSS P3 item score = 115,31. The study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of Zhejiang University School of Medicine Sir Run Run Shaw Hospital (No. lunshen2022yandi0081). Each participant provided written informed consent and had the right to withdraw during the study procedure.
Clinical measurements
The Positive and Negative Syndrome Scale (PANSS) is commonly used to evaluate the severity of psychiatric symptoms32. This instrument has 30 items belonging to three subscales, including the Positive Scale (POS, from P1 to P7), Negative Scale (NEG, from N1 to N7), and General Psychopathology Scale (GPS, from G1 to G16). In accordance with previous studies, the P3 hallucination item in the PANSS was used to evaluate the severity of auditory hallucinations33,34.
The Psychotic Symptom Rating Scales (PSYRATS) is a widely used validated scale to assess multiple dimensions of auditory hallucinations and delusions35. This instrument has 17 items belonging to two subscales, including the auditory hallucinations subscale (AHS) and delusions subscale (DS). Each item is rated from 0 to 4 points and a higher score represents more severe symptoms.
MRI acquisition
Within 24 h after enrollment, all participants received an MRI scan via a 3.0 T MRI scanner (Siemens, Munich, Germany) with a 16-channel head coil at the Magnetic Imaging Center of Hunan Children’s Hospital, Changsha, China. Anatomical T1-weighted MRI data were acquired via a 3D magnetization-prepared rapid acquisition gradient echo (3D MPRAGE) sequence with the following parameters: repetition time = 2530 ms, time to echo = 2.33 ms, field of view = 256 × 256 mm, flip angle = 7°, slice thickness = 1 mm, 192 sagittal slices, and voxel size = 1 mm3. There were no major scanner upgrades or instrument replacements during the study period. During the examination, foam pads were used to restrain head movement and earplugs to attenuate noise. The participants were instructed to lie on their backs and keep their head and body motionless.
Image preprocessing
First, the original images were visually inspected to determine whether there was any distortion or motion artifact, and no participant was excluded because of the raw image quality. Then, T1-weighted images of all participants were preprocessed via the New-segment and DARTEL function in the DPABI toolbox (Data Processing Analysis of Brain Imaging toolbox, http://rfmri.org/dpabi)36. In detail, raw images were normalized to Montreal Neurologic Institute (MNI) space, segmented into gray matter, white matter, and cerebrospinal fluid, resampled to 1.5 × 1.5 × 1.5 mm3, and modulated for linear and nonlinear effects to acquire gray matter volume images. The preprocessed images were smoothed by a Gaussian kernel filter with a 6 mm full width at half-maximum (FWHM) and visually checked. Three participants in the pAH group were excluded because of poor segmentation.
Network construction
The AAL 116 template, which included 90 cerebral regions and 26 cerebellar regions, was used to define network nodes. The Graph Analysis Toolbox (GAT, https://www.nitrc.org/projects/gat) was used to extract the mean gray matter volume values of 116 ROIs37. The method of intra-individual network construction by Yun et al. 28 was followed28. The ROI values were corrected for age, sex, years of education, and total intracranial volume (TIV) via linear regression. The resulting residuals were then z-transformed with the mean and standard deviation of each ROI calculated from healthy controls to obtain brain morphological variations per ROI relative to average healthy control values. For the i-th ROI, the z score was calculated as follows:
where Zi is the z score of the i-th ROI, residualsi is the gray matter residual of the i-th ROI, Mi is the mean value of the i-th ROI in the HC group, and SDi is the standard deviation of the i-th ROI in the HC group.
For each participant, the intra-individual structural covariance between 116 ROIs represented the edge weights of the network and each pair was calculated as follows:
where Covariancei,j is the intra-individual structural covariance between the i-th and j-th ROIs, and Zi and Zj are the z scores of the i-th and j-th ROIs.
After the covariance matrix (116 × 116 pairs) for each participant was derived, the GRETNA toolbox (http://www.nitrc.org/projects/gretna) was utilized to construct positive weighted networks38. The range of the sparsity threshold was determined on the basis of the following criteria: (1) the average degree over all nodes of each network was greater than 2 × log(N), where N is the number of nodes; and (2) small-worldness (Sigma) was greater than 1.1 for all participants39. The generated range was 0.04 to 0.38 with an interval of 0.01. Moreover, 1000 random networks were generated for comparison with real networks to test whether real networks possessed non-random topological configurations.
Network metrics
For networks under each sparsity threshold, several key metrics were calculated to examine the topological properties of the structural networks. Specifically, the global measurements included (1) small-world properties, including the clustering coefficient, characteristic path length, normalized clustering coefficient (Gamma), normalized characteristic path length (Lambda), and small-worldness (Sigma), and (2) network efficiency, including the global efficiency and local efficiency. Regional measurements included the nodal clustering coefficient, nodal efficiency, nodal local efficiency, betweenness centrality, and degree centrality. For edge analysis, the network-based statistic (NBS) was used to examine the differences in structural connectivity among different groups, and the methodological principles are described elsewhere40.
Statistical analysis
The area under the curve (AUC) across the threshold range for each network metric was calculated and used for group comparisons because it is less sensitive to thresholding selection41. One-way ANCOVA was used to compare global and local metrics among the three groups with age, sex, and years of education as covariates. The p threshold of the main effect was set as 7.14 × 10−3 (0.05/7) for seven global metrics, and 0.01 (0.05/5) for five local metrics. The main effect of local metrics was additionally corrected via the FDR method (116 nodes). Whenever a significant main effect was found, a post hoc test was conducted by the Bonferroni correction. For the post hoc test, the PANSS-POS, PANSS-NEG, PANSS-GPS, and chlorpromazine equivalent dosage were set as covariates when comparing the pAH and non-pAH groups to rule out the possible nuisance effect of common psychiatric symptoms and medications31.
For edge analysis, the independent samples t tests with the NBS correction (edge level p < 0.001 and 5000 iterations) were performed with age, sex, and years of education as covariates. For the three groups, three respective t tests were conducted; thus, the component level p was set as 0.0167 by the Bonferroni correction (0.05/3 = 0.0167).
Pearson correlation analyses were conducted between network metrics (all global metrics plus local metrics in which the pAH group differed from any other group) and clinical variables (onset age, illness duration, first treatment age, PANSS the P3, PANSS POS, PANSS NEG, PANSS GPS, PANSS total, PSYRATS AHS, PSYRATS DS, and PSYRATS total) in the pAH group. Correlation analyses were also conducted between subnetwork edges and clinical variables in the pAH group. The Bonferroni method was employed to correct for multiple testing.
Results
Demographic and clinical information
The chi-square test revealed that there was no difference in sex distribution among the three groups (χ2 = 2.14, df = 2, p = 0.34). One-way ANOVA revealed that there was no group difference in age (F[2, 222] = 2.64, MSE = 33.71, p = 0.07), whereas years of education differed among the three groups (F[2, 222] = 19.13, MSE = 8.33, p < 0.001). Post hoc tests revealed that the pAH group had fewer years of education than the other two groups did (all ps < 0.05), whereas the non-pAH group had fewer years of education than the HC group did (p = 0.01) (Table 1). Independent samples t tests revealed that, compared with the non-pAH group, the pAH group had a lower onset age and first treatment age; higher PANSS the P3, PANSS POS, and PANSS total score (all ps < 0.01); and no difference in duration, PANSS NEG, or PANSS GPS (all ps > 0.05) (Table 1).
Global network characteristics
One-way ANCOVA revealed that there were no significant main effects of the AUC for the global metrics, including the clustering coefficient, characteristic path length, Gamma, Lambda, Sigma, global efficiency, or local efficiency (all ps > 7.14 × 10−3; the p threshold set as 7.14 × 10−3) (Supplementary Table 1).
Local network characteristics
One-way ANCOVA revealed that the main effects for local metrics, including the nodal clustering coefficient, nodal local efficiency, and betweenness centrality, were not significant after multiple testing correction (all ps > 0.01; the FDR-corrected p threshold set as 0.01), except for the nodal efficiency of the temporal pole of the left superior temporal gyrus (F = 8.88, p = 1.97 × 10−4), right insula (F = 11.53, p = 1.74 × 10−5), and left medial superior frontal gyrus (F = 8.69, p = 2.33 × 10−4), as well as the degree centrality of the right insula (F = 10.92, p = 3.03 × 10−5).
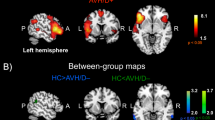
For nodal efficiency, post hoc tests by the Bonferroni correction (p threshold set as 0.05) revealed that the pAH group had lower nodal efficiency of the temporal pole than the non-pAH group (p = 1.09 × 10−3) and the HC group (p = 7.67 × 10−4), whereas there was no difference between the non-pAH group and the HC group (p = 1.00). The pAH group had lower nodal efficiency of the right insula than the non-pAH group (p = 0.02) and the HC group (p = 8.99 × 10−6), whereas there was no difference between the non-pAH group and the HC group (p = 0.13). Both the pAH group (p = 0.01) and the non-pAH group (p = 3.56 × 10−4) had lower nodal efficiency of the left medial superior frontal gyrus than the HC group, whereas there was no difference between the patient groups (p = 0.25) (Fig. 1). The detailed ANCOVA statistics are shown in Table 2.
a Post hoc tests were visualized by the BrainNet Viewer (version1.7, https://www.nitrc.org/projects/bnv). b Post hoc tests were visualized by the histogram; age, sex, and years of education were set as covariates for ANCOVA; PANSS-POS, PANSS-NEG, PANSS-GPS, and chlorpromazine equivalent dosage were set as covariates for post hoc tests when comparing the pAH group and the non-pAH group. SFGmed.L, left medial superior frontal gyrus; TPOsup.L, temporal pole of the left superior temporal gyrus; INS.R, right insula. *p < 0.05; **p < 0.01; ***p < 0.001.
For degree centrality, post hoc tests by the Bonferroni correction (the p threshold set as 0.05) revealed that the pAH group had lower degree centrality of the right insula than the non-pAH group (p = 0.04) and the HC group (p = 1.65 × 10−5), whereas there was no difference between the non-pAH group and the HC group (p = 0.18) (Fig. 2). The detailed ANCOVA statistics are shown in Table 2.
a Post hoc tests were visualized by the BrainNet Viewer. b Post hoc tests were visualized by the histogram. Age, sex, and years of education were set as covariates for ANCOVA; PANSS-POS, PANSS-NEG, PANSS-GPS, and chlorpromazine equivalent dosage were set as covariates for post hoc tests when comparing the pAH group and the non-pAH group. INS.R, right insula. *p < 0.05; ***p < 0.001.
Edge analysis
Network-based statistics revealed a single subnetwork composed of 13 edges with reduced structural covariance in the pAH group compared with the non-pAH group. The majority of connections were associated with the temporal pole of the left superior temporal gyrus (Fig. 3). The statistical details of the 13 edges are listed in Supplementary Table 2.
Edge analysis was visualized by the Circos (version0.69-9, http://circos.ca/software). Brain regions are grouped on the connectogram circumference according to lobes (frontal, temporal, parietal, occipital, subcortical, insula, limbic, cerebellum). The left hemisphere nodes are shown on the left side of the connectogram.
Compared with the HC group, the pAH group presented a subnetwork with reduced structural covariance that was mainly composed of the bilateral frontal, temporal, and cerebellar regions. Compared with the HC group, the non-pAH group also presented a subnetwork with reduced structural covariance, which was composed mainly of the bilateral frontal and cerebellar regions, followed by the temporal and limbic regions (Supplementary Fig. 1).
Correlational analysis
In the pAH group, Pearson correlation analyses were conducted between network metrics/ subnetwork edges and clinical variables. Gamma was negatively correlated with the PANSS POS (r = −0.36, p = 8.45 × 10−3), Sigma was negatively correlated with the PANSS POS (r = −0.35, p = 9.89 × 10−3), and no other significant correlations were found. In particular, the chlorpromazine equivalent dosage was not significantly correlated with any network metrics or subnetwork edges.
Discussion
To the best of our knowledge, this is the first study to apply intra-individual structural covariance networks in schizophrenia patients with persistent auditory hallucinations. Our study revealed a common change in network metrics in schizophrenia patients with or without persistent auditory hallucinations. Both the pAH group and the non-pAH group presented lower nodal efficiency of the left medial superior frontal gyrus. Previous studies have reported similar metrical changes in patients with schizophrenia42 consistent with our results. More importantly, our study revealed distinct changes in network metrics related to auditory hallucinations. The pAH group had lower nodal efficiency of the temporal pole of the left superior temporal gyrus and right insula as well as lower degree centrality of the right insula than the non-pAH group and the HC group did, while the latter groups did not differ from each other. Compared with the non-pAH group, the pAH group had a subnetwork with reduced structural covariance, and the temporal pole served as the main node linking all edges. In the pAH group, the positive scale score of PANSS was negatively correlated with Gamma and Sigma.
Compared with schizophrenia patients without persistent auditory hallucinations and healthy controls, schizophrenia patients with persistent auditory hallucinations had lower nodal efficiency in the temporal pole of the left superior temporal gyrus. Anatomically, the temporal pole lies under the lateral sulcus and occupies the rostral part of the temporal lobe, extending to the superior temporal gyrus43. This area is related to language and semantic processing, binds different high-order information, and processes high-level cognitive treatments in different modalities44. Moreover, the superior temporal gyrus is related to interpretation, production and self-monitoring of language45, and a reduced volume of the left superior temporal gyrus is related to the severity of auditory hallucinations46. On the basis of the inner speech model, self-monitoring refers to one’s ability to distinguish sensations evoked by one’s own actions from those evoked by external factors47. Auditory hallucinations occur when deficits in self-monitoring cause inner speech to be perceived as from externality48. Therefore, structural network metric alterations in the left superior temporal gyrus, especially the temporal pole part, may affect language processing dysfunction and self-monitoring deficit, leading to misinterpretation of inner speech.
Moreover, our study revealed that compared with schizophrenia patients without persistent auditory hallucinations and healthy controls, schizophrenia patients with persistent auditory hallucinations had lower nodal efficiency and degree centrality in the right insula, indicating that the right insula exhibited abnormal structural connection and information transmission within the complex network. One function of the insula is processing of sensory input, which involves self-identification and discriminating between self-generated and external sensory input49. Abnormalities of this function may lead to internally generated information falsely attributed to externally generated information, thus inducing the occurrence of auditory hallucinations. Previous morphological studies also revealed that schizophrenia patients with auditory hallucinations had lower cortical thickness in the right insula than non-hallucinatory patients16, which further indicates that the right insula has not only local morphological abnormalities but also abnormal connections in the complex network, leading to abnormal transmission of auditory information and the generation of auditory hallucinations.
Edge analyses revealed that, compared with healthy controls, both schizophrenia patients with and without persistent auditory hallucinations presented reduced structural covariance in the whole brain, but hallucinatory patients presented wider alterations in the frontal, temporal, parietal, occipital, subcortical, insula, limbic, and cerebellar regions. On the other hand, schizophrenia patients with persistent auditory hallucinations displayed a subnetwork with reduced structural covariance where the temporal pole of the left superior temporal gyrus served as the main node linking the middle temporal gyrus, inferior temporal gyrus, amygdala, parahippocampal gyrus, fusiform gyrus, and cerebellum. Hence, our findings revealed that the intra-individual structural covariance network of schizophrenia patients had distinct structural dysconnectivity. Previous morphological studies revealed that the gray matter volume of the bilateral middle temporal gyri was reduced in patients with schizophrenia or the prodromal phase of illness50,51. A study directly related to auditory hallucinations revealed that the externalization bias of reality-monitoring is negatively correlated with gray matter volume in the bilateral inferior temporal gyri and fusiform52. An fMRI study revealed that the amygdala and parahippocampal gyrus were abnormally activated in schizophrenia patients with auditory hallucinations53. With respect to the cerebellum, schizophrenia patients with treatment-resistant auditory hallucinations were found to have lower gray matter volume in lobule VIII than healthy controls54. These scattered findings implied that regions linking the temporal pole of the left superior temporal gyrus, the main node in the subnetwork, were closely correlated with schizophrenia or auditory hallucinations. Our study provided further evidence that the strength of structural connectivity among these regions was reduced in schizophrenia patients with persistent auditory hallucinations.
Our study also revealed that the positive scale score of the PANSS was negatively correlated with Gamma and Sigma in schizophrenia patients with persistent auditory hallucinations. Gamma, the normalized clustering coefficient, reflects the local efficiency of information processing, whereas Sigma, small-worldness, reflects balanced local efficiency and global efficiency in information communication accompanied by low wiring cost55. Previous study found that patients with first-episode psychosis who became treatment-resistant during the follow-up period presented reduced a clustering coefficient and small-worldness56, which indicates that alterations in global network metrics are strongly associated with poor prognosis. Therefore, we speculate that among patients with persistent auditory hallucinations, those with lower global metrics may have worse clinical efficacy and that symptoms of auditory hallucinations may be treatment-refractory.
However, our study has several limitations. First, the sample size of patients without auditory hallucinations was relatively small, and our findings merit future replication and external validation. Second, the intra-individual structural covariance network created in our study was based on voxel-wise gray matter volumes, and network constructions based on other morphological measures remain to be explored in the future. Third, our study did not utilize individualized brain parcellation for each participant, which would be more suitable for reflecting individual structural variations. Fourth, our study did not recruit first-episode drug-naïve patients, which would be otherwise welcomed for investigating disease mechanisms.
In conclusion, our study revealed distinct network metrical changes related to auditory hallucinations, including lower nodal efficiency of the temporal pole of the left superior temporal gyrus and right insula, lower degree centrality of the right insula, and a unique subnetwork of reduced structural covariance, with the temporal pole serving as the main node. The temporal pole and right insula are two important regions related to the pathogenesis of auditory hallucinations, indicating that these two regions may be of potential targets for clinical interventions. In addition, our results provide evidence for the search for neuroimaging biomarkers for early diagnosis and disease subtyping. Our results encourage future investigations of individualized network changes related to auditory hallucinations at the structural level.
Data availability
The data described in the manuscript will be made available upon request.
References
Saha, S., Chant, D., Welham, J. & McGrath, J. A systematic review of the prevalence of schizophrenia. PLoS Med. 2, e141 (2005).
Hjorthøj, C., Stürup, A. E., McGrath, J. J. & Nordentoft, M. Years of potential life lost and life expectancy in schizophrenia: a systematic review and meta-analysis. Lancet Psychiatry 4, 295–301 (2017).
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition. (American Psychiatric Association, Arlington, VA, 2013).
Johns, L. C. et al. Auditory verbal hallucinations in persons with and without a need for care. Schizophr. Bull. 40, S255–S264 (2014).
González, J. C., Aguilar, E. J., Berenguer, V., Leal, C. & Sanjuan, J. Persistent auditory hallucinations. Psychopathology 39, 120–125 (2006).
McCarthy-Jones, S. et al. A new phenomenological survey of auditory hallucinations: evidence for subtypes and implications for theory and practice. Schizophr. Bull. 40, 231–235 (2014).
Hayashi, N., Igarashi, Y., Suda, K. & Nakagawa, S. Auditory hallucination coping techniques and their relationship to psychotic symptomatology. Psychiatry Clin. Neurosci. 61, 640–645 (2007).
Nayani, T. H. & David, A. S. The auditory hallucination: a phenomenological survey. Psychol. Med. 26, 177–189 (1996).
Shergill, S. S., Murray, R. M. & McGuire, P. K. Auditory hallucinations: a review of psychological treatments. Schizophr. Res. 32, 137–150 (1998).
Hor, K. & Taylor, M. Suicide and schizophrenia: a systematic review of rates and risk factors. J. Psychopharmacol. 24, 81–90 (2010).
Kjelby, E. et al. Suicidality in schizophrenia spectrum disorders: the relationship to hallucinations and persecutory delusions. Eur. Psychiatry 30, 830–836 (2015).
Chen, X. et al. Reduced cortical thickness in right Heschl’s gyrus associated with auditory verbal hallucinations severity in first-episode schizophrenia. BMC Psychiatry 15, 152 (2015).
van Swam, C. et al. Possible dysregulation of cortical plasticity in auditory verbal hallucinations—a cortical thickness study in schizophrenia. J. Psychiatr. Res. 46, 1015–1023 (2012).
Cui, Y. et al. Auditory verbal hallucinations are related to cortical thinning in the left middle temporal gyrus of patients with schizophrenia. Psychol. Med. 48, 115–122 (2018).
Ren, H. et al. Differences in cortical thickness in schizophrenia patients with and without auditory verbal hallucinations. Front. Mol. Neurosci. 15, 845970 (2022).
Shen, X. et al. Cognitive dysfunction and cortical structural abnormalities in first-episode drug-naïve schizophrenia patients with auditory verbal hallucination. Front. Psychiatry 13, 998807 (2022).
Palaniyappan, L., Balain, V., Radua, J. & Liddle, P. F. Structural correlates of auditory hallucinations in schizophrenia: a meta-analysis. Schizophr. Res. 137, 169–173 (2012).
Modinos, G. et al. Neuroanatomy of auditory verbal hallucinations in schizophrenia: a quantitative meta-analysis of voxel-based morphometry studies. Cortex 49, 1046–1055 (2013).
Romeo, Z. & Spironelli, C. Hearing voices in the head: two meta-analyses on structural correlates of auditory hallucinations in schizophrenia. NeuroImage Clin. 36, 103241 (2022).
Barber, L., Reniers, R. & Upthegrove, R. A review of functional and structural neuroimaging studies to investigate the inner speech model of auditory verbal hallucinations in schizophrenia. Transl. Psychiatry 11, 582 (2021).
Prasad, K. et al. Structural covariance networks in schizophrenia: a systematic review Part I. Schizophr. Res. 240, 1–21 (2022).
Bullmore, E. & Sporns, O. Complex brain networks: graph theoretical analysis of structural and functional systems. Nat. Rev. Neurosci. 10, 186–198 (2009).
Watts, D. J. & Strogatz, S. H. Collective dynamics of ‘small-world’ networks. Nature 393, 440–442 (1998).
Chen, Z. J., He, Y., Rosa-Neto, P., Germann, J. & Evans, A. C. Revealing modular architecture of human brain structural networks by using cortical thickness from MRI. Cereb. Cortex 18, 2374–2381 (2008).
Palaniyappan, L. et al. Structural covariance and cortical reorganisation in schizophrenia: a MRI-based morphometric study. Psychol. Med. 49, 412–420 (2019).
Zhang, Y. et al. Abnormal topological organization of structural brain networks in schizophrenia. Schizophr. Res. 141, 109–118 (2012).
Liu, F., Tian, H., Li, J., Li, S. & Zhuo, C. Altered voxel-wise gray matter structural brain networks in schizophrenia: association with brain genetic expression pattern. Brain Imaging Behav. 13, 493–502 (2019).
Yun, J. Y. et al. Brain structural covariance networks in obsessive-compulsive disorder: a graph analysis from the ENIGMA Consortium. Brain 143, 684–700 (2020).
Sheehan, D. V. et al. The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J. Clin. Psychiatry 59, 22–33;quiz 34–57 (1998).
Psomiades, M. et al. N-Acetyl-Aspartate in the dorsolateral prefrontal cortex in men with schizophrenia and auditory verbal hallucinations: a 1.5 T magnetic resonance spectroscopy study. Sci. Rep. 8, 4133 (2018).
Wang, Q. et al. Metabolite differences in the medial prefrontal cortex in schizophrenia patients with and without persistent auditory verbal hallucinations: a (1)H MRS study. Transl. Psychiatry 12, 116 (2022).
Kay, S. R., Fiszbein, A. & Opler, L. A. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr. Bull. 13, 261–276 (1987).
Andreasen, N. C. et al. Remission in schizophrenia: proposed criteria and rationale for consensus. Am. J. Psychiatry 162, 441–449 (2005).
Benetti, S. et al. Auditory verbal hallucinations and brain dysconnectivity in the perisylvian language network: a multimodal investigation. Schizophr. Bull. 41, 192–200 (2015).
Haddock, G., McCarron, J., Tarrier, N. & Faragher, E. B. Scales to measure dimensions of hallucinations and delusions: the psychotic symptom rating scales (PSYRATS). Psychol. Med. 29, 879–889 (1999).
Chao-Gan, Y. & Yu-Feng, Z. DPARSF: a MATLAB toolbox for “pipeline” data analysis of resting-state fMRI. Front. Syst. Neurosci. 4, 13 (2010).
Hosseini, S. M., Hoeft, F. & Kesler, S. R. GAT: a graph-theoretical analysis toolbox for analyzing between-group differences in large-scale structural and functional brain networks. PLoS ONE 7, e40709 (2012).
Wang, J. et al. GRETNA: a graph theoretical network analysis toolbox for imaging connectomics. Front. Hum. Neurosci. 9, 386 (2015).
Zhang, J. et al. Disrupted brain connectivity networks in drug-naive, first-episode major depressive disorder. Biol. Psychiatry 70, 334–342 (2011).
Zalesky, A., Fornito, A. & Bullmore, E. T. Network-based statistic: identifying differences in brain networks. Neuroimage 53, 1197–1207 (2010).
Wang, J. et al. Parcellation-dependent small-world brain functional networks: a resting-state fMRI study. Hum. Brain Mapp. 30, 1511–1523 (2009).
Liu, Z. et al. Distinguishable brain networks relate disease susceptibility to symptom expression in schizophrenia. Hum. Brain Mapp. 39, 3503–3515 (2018).
Chabardès, S., Kahane, P., Minotti, L., Hoffmann, D. & Benabid, A. L. Anatomy of the temporal pole region. Epileptic Disord. 4 (Suppl 1), S9–S15 (2002).
Herlin, B., Navarro, V. & Dupont, S. The temporal pole: from anatomy to function—a literature appraisal. J. Chem. Neuroanat. 113, 101925 (2021).
Pearlson, G. D. Superior temporal gyrus and planum temporale in schizophrenia: a selective review. Prog. Neuropsychopharmacol. Biol. Psychiatry 21, 1203–1229 (1997).
Levitan, C., Ward, P. B. & Catts, S. V. Superior temporal gyral volumes and laterality correlates of auditory hallucinations in schizophrenia. Biol. Psychiatry 46, 955–962 (1999).
Allen, P., Aleman, A. & McGuire, P. K. Inner speech models of auditory verbal hallucinations: evidence from behavioural and neuroimaging studies. Int. Rev. Psychiatry 19, 407–415 (2007).
Frith, C. D. & Done, D. J. Towards a neuropsychology of schizophrenia. Br. J. Psychiatry 153, 437–443 (1988).
Wylie, K. P. & Tregellas, J. R. The role of the insula in schizophrenia. Schizophr. Res. 123, 93–104 (2010).
Cullen, A. E. et al. Temporal lobe volume abnormalities precede the prodrome: a study of children presenting antecedents of schizophrenia. Schizophr. Bull. 39, 1318–1327 (2013).
Onitsuka, T. et al. Middle and inferior temporal gyrus gray matter volume abnormalities in chronic schizophrenia: an MRI study. Am. J. Psychiatry 161, 1603–1611 (2004).
Perret, M. et al. Neuroanatomical correlates of reality monitoring in patients with schizophrenia and auditory hallucinations. Eur. Psychiatry 64, e58 (2021).
Escartí, M. J. et al. Increased amygdala and parahippocampal gyrus activation in schizophrenic patients with auditory hallucinations: an fMRI study using independent component analysis. Schizophr. Res. 117, 31–41 (2010).
Cierpka, M. et al. Cerebellar contributions to persistent auditory verbal hallucinations in patients with Schizophrenia. Cerebellum 16, 964–972 (2017).
Liao, X., Vasilakos, A. V. & He, Y. Small-world human brain networks: perspectives and challenges. Neurosci. Biobehav. Rev. 77, 286–300 (2017).
Ajnakina, O. et al. Structural covariance of cortical gyrification at illness onset in treatment resistance: a longitudinal study of first-episode psychoses. Schizophr. Bull. 47, 1729–1739 (2021).
Acknowledgements
This work was supported by the National Natural Science Foundation of China [grant number 82171495 to J.T.] and the National key R & D plan of China [grant number 2022YFE0103700 to J.T.].
Author information
Authors and Affiliations
Contributions
Xu Shao and Honghong Ren are the co-first authors; Xu Shao, Honghong Ren, and Jinsong Tang designed the research; Xu Shao, Honghong Ren, Jinguang Li, Jingqi He, Lulin Dai, Min Dong, Xiaogang Chen, and Jinsong Tang conducted the research; Xu Shao, Jun Wang, Xiangzhen Kong, and Jinsong Tang analysed the data; and Xu Shao and Jinsong Tang wrote the paper. Xu Shao and Jinsong Tang were primarily responsible for the final content. All the authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Shao, X., Ren, H., Li, J. et al. Intra-individual structural covariance network in schizophrenia patients with persistent auditory hallucinations. Schizophr 10, 92 (2024). https://doi.org/10.1038/s41537-024-00508-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41537-024-00508-7
This article is cited by
-
Morphometric similarity network-based graph convolutional networks for schizophrenia classification
Scientific Reports (2025)