Abstract
Formal thought disorder (FTD) is a core symptom of schizophrenia. The pathophysiology of FTD is still unclear. We focus on multiple cortical measures to capture the exact nature of brain alterations (e.g., plasticity, early brain development) in FTD dimensions. We included 70 schizophrenia patients. We assessed FTD, acquired structural neuroimaging scans, and analyzed cortical thickness, volume, surface area, and local gyrification (IGI). Results reveal negative FTD to be associated with different structural brain correlates compared to the positive and linguistic control FTD dimensions most prominent in markers of early brain development. Severity of positive and linguistic control FTD dimensions correlated positively with IGI of core language regions including temporal, Heschl’s, and inferior frontal gyri. Severity of negative FTD dimension was inversely correlated with lGI of occipital and parietal regions. Findings propose distinguishable changes most prominent in markers of early brain development associated with FTD dimensions suggesting a distinct pathophysiology.
Similar content being viewed by others
Introduction
Formal thought disorder (FTD) describes a multidimensional syndrome evident in multiple psychiatric conditions1,2. FTD is a core symptom in schizophrenia spectrum disorders with up to 91% prevalence3. FTD occurs early in the course of schizophrenia4,5,6. In general, FTD significantly impairs the remission of symptoms7, occupational and social functioning7,8, life satisfaction and wellbeing9,10, and the therapeutic alliance and psychological recovery as well11. The variability seen in FTD has frequently been depicted as a dichotomy between negative and positive FTD, encompassing a quantitative deficiency in speech and thought production on one end and heightened speech and thought production on the other2. While the distinction between negative and positive FTD is well established2,12, factor analytic studies suggested up to five FTD factors explaining more variance than the negative-positive dichotomy3,13,14.
Importantly, the dimensions of FTD have clinical significance3. Positive and negative FTD are linked to different neuropsychological deficits15 across multiple neurocognitive domains16,17,18. Additionally, the negative FTD dimension more accurately predicted the progression to schizophrenia in individuals at risk for psychosis, irrespective of genetic risk6. Similarly, distinct dimensions of FTD (either positive or negative) predicted poor outcomes, unlike general FTD3,19,20.
Although different FTD dimensions can be distinguished on the behavioral level, the underlying neuropathologies are still a matter of debate. In fact, most of the studies focused on general FTD as a single construct and did not differentiate between different FTD dimensions. In detail, several studies linked general FTD severity to reduction of gray matter (GM) volume in frontal language-related regions21,22,23 (inferior frontal gyrus (IFG), Broca’s area, frontal operculum, and prefrontal cortex (PFC)) with some conflicting results24,25,26,27,28. Likewise temporal and parietal regions within the language network have been associated with FTD severity. Specifically, reductions in GM volume have been reported in the superior temporal gyrus (STG), including the planum temporale and temporal pole, either unilaterally (left)21,24,25,29,30 or bilaterally26,31,32,33,34,35, left superior temporal sulcus (STS)24,25, and left angular gyrus (AngG)25. In contrast, GM volume increase within the left22 and right23,36 STG was associated with FTD severity. Moreover, FTD severity has been linked to reduced or reversed asymmetries of the planum temporale37,38 again with some conflicting results39. However, other investigations have failed to identify significant associations between FTD severity and GM volumes of the STG28,36,39,40,41,42,43,44 and inferior parietal lobule45.
Of note, FTD has also been associated with GM volume reductions in cortical regions beyond the traditional language network such as the middle (MFG), medial, and superior frontal gyri (SFG)33, ventromedial PFC21, orbitofrontal cortex (OFC)21,24,46, precuneus25,33, cuneus/lingual gyrus (LingG)24, cingulate gyrus25,33, and the insula28,33. Moreover, alterations in the volumes of the parahippocampal gyrus31 and both reductions and increases in cerebellar volumes47,48 have been found to be significantly linked to FTD. Finally, investigations into GM correlates of FTD in subcortical areas have yielded inconsistent findings. Studies have reported varied results for the hippocampus-amygdala complex26,31,34,43,49,50,51, basal ganglia27,33, and no association was found between FTD and GM volume of the thalamus52. In essence, these studies reported GM volume deficits in the fronto-temporal language network as associated with FTD in schizophrenia24,25,53. In addition, the language network is closely tied to other important brain networks, such as the default mode, salience, and central executive networks and the identified regions, i.e., lateral and medial PFC and parietal, cingulate, and insular cortex, may at least partly reflect closely related processes such as memory, unification, and cognitive control54.
Hitherto only a small number of studies directly investigated GM correlates of FTD dimensions suggesting distinct pathophysiology of FTD dimensions1,55,56. A recent study based on a large multi-site cohort through the ENIGMA Schizophrenia Working Group (752 individuals with schizophrenia and 1,256 controls) investigated the neuroanatomy of positive, negative, and general FTD in schizophrenia55. Likewise, Maderthaner and colleagues investigated structural correlates (cortical thickness (CT) and GM volume) of four different FTD dimensions. In sum, while positive FTD was associated with altered structural correlates such as CT or GM volume of core language areas, i.e., MTG, STG, and IFG as well as with non-language-related areas, negative FTD is probably less clearly associated with language-related areas but with brain areas relevant for higher order control processes, i.e., MFG, anterior cingulate cortex (ACC), and PFC regions. These two studies1,55 suggest differential structural brain correlates for different FTD dimensions and show that non-language-related regions also contribute to FTD in schizophrenia. White matter dysconnectivity associated with the FTD dimensions disorganization and emptiness has also been reported57, mainly for cortical brain regions previously associated with FTD in schizophrenia53,58.
Similarly, a limited number of functional neuroimaging studies have directly explored the correlates of FTD dimensions, again indicating distinct pathophysiological mechanisms59,60,61,62. While positive FTD was linked to changes in task-based functional activation of the STG (Wernicke’s area), IFG, and the parahippocampal gyrus59,60,62 and to perfusion in the supplementary motor area and IFG61, negative FTD involved task-based functional activations in brain regions important for higher-order control processes such as the parietal lobe, cuneus, precuneus, and posterior frontal lobe63,64 but also altered perfusion in the STG61. Therefore, negative FTD may reflect impaired access to semantic memory, while positive FTD likely indicates an inability to suppress irrelevant information during increased speech production61. The severity of alterations in the language dimension was also associated with altered perfusion in the right Heschl’s gyrus, but perfusion did not differentiate between positive and negative FTD dimensions65.
Notable previous structural neuroimaging studies reviewed elsewhere53,58 focused mostly on GM volume only as a structural marker. However, to capture the exact nature of the morphological alterations in FTD dimensions, it is highly recommended to investigate multiple morphological measures. Importantly, voxel-based morphometry studies rely on GM volume as an anatomical marker forming a product of cortical surface area (CSA) and CT. Therefore, if these variables run in opposite directions, measurements can be confounded which may hamper to detect specific effects of FTD. In fact, CSA and CT are independent neuroanatomical traits influenced by different factors during brain development (e.g., CT and CSA appear to be modulated by different genes)66. While CSA increases during late fetal development due to cortical folding, CT alters dynamically across the entire life span because of training, experience, and disease66. Likewise, gyrification is an anatomical trait influenced by early brain development. Thus, CT, CSA, and gyrification are considered as independent markers best measured separately when investigating the neuroarchitecture of the human cortex67. However, to the best of our knowledge, altered gyrification in association with FTD has not been investigated yet. In fact, FTD occurs early in the course of schizophrenia4,5,6 pointing to alterations in early brain development. Thus, changes in neuromorphological markers related to early brain development such as gyrification and CSA68,69 may be particularly prominent in patients with FTD compared to changes in neuromorphological markers subjected to lifelong plastic processes such as CT66.
In the present study, we therefore investigated morphological correlates of FTD dimensions (positive, negative, and linguistic control) in schizophrenia patients using multiple cortical measures (CSA, CT, cortical volume (CV), as well as gyrification) based on surface-based morphometry. We tested whether FTD dimensions were associated with different structural brain correlates and hypothesized to detect prominent altered markers of brain maturation (gyrification, CSA). Furthermore, morphological correlates were expected to be evident rather in language-related brain regions for the positive and linguistic control FTD dimensions and in non-language-related brain regions for the negative FTD dimension.
Methods
Participants
The current study initially enrolled 93 patients with schizophrenia spectrum disorders according to Diagnostic and Statistical Manual of Mental Disorders (DSM-5) criteria. Patients were recruited from the in- and outpatient clinics at the University Hospital of Psychiatry and Psychotherapy in Bern, Switzerland. All patients were between 20 and 65 years old (Supplementary Table S1) and provided written informed consent to participate in the study. The study protocols adhered to the declaration of Helsinki and were approved by the local ethics committee.
General exclusion criteria for all participants were substance abuse/dependence other than nicotine, a history of neurologic disease, head trauma with concurrent loss of consciousness, electroconvulsive treatment, and any magnetic resonance imaging (MRI) counter-indication. Current symptom severity was assessed with the Positive and Negative Syndrome Scale70. All participants completed the Mini International Neuropsychiatric Interview71.
Twenty-three participants (three due to unclear diagnosis, one due to substance abuse, two due to missing clinical values, and 17 due to ringing artefacts or reduced quality of the MRI scans) were excluded resulting in a final sample size of 70 patients with schizophrenia spectrum disorders (51 patients with schizophrenia, 10 with schizoaffective disorder, and 9 with schizophreniform disorder). The demographic and clinical characteristics of the final sample are presented in Table 1.
Thought, language, and communication scale for measuring formal thought disorder dimensions
FTD dimensions were measured using the thought, language, and communication (TLC) scale72 according to a previous study of our group61. The FTD dimensions are based on a factor analysis published elsewhere14. The positive FTD dimension, also termed disorganization, was defined by the mean of the following ten items (derailment, loss of goal, circumstantiality, pressure of speech, tangentiality, distractible speech, self-reference, perseveration, incoherence, and stilted speech). The negative FTD dimension, also termed emptiness, was defined by the mean of the following three items (poverty of speech, blocking, and poverty of content), whereas the linguistic control FTD dimension was defined by the mean of the following three items (neologism, illogicality, and word approximations). Nagels and colleagues named this factor ‘linguistic control’ “since changes on the word basis were most characteristic”14. Phonemic and semantic paraphasia items were not included in the linguistic control FTD dimension because these two items were not part of the TLC scale. In addition, we also used the sum of all 18 TLC items to assess the severity of general FTD.
Magnetic resonance imaging data acquisition
MRI was performed with two different machines: a 3 Tesla Siemens MAGNETOM Prisma and a 3 Tesla MAGNETOM Trio Tim scanner (Siemens Healthineers, Erlangen, Germany), both equipped with a 20-channel radio frequency head coil array and located at the University Hospital of Bern, Switzerland. On both scanners, identical pulse sequences with the same parameters have been applied. A 3D-T1-weighted magnetization prepared 2 rapid acquisition gradient echoes pulse sequence has been applied, providing 176 sagittal slices with 240 × 256 matrix points and a field of view of 240 × 256 mm2, yielding an isotropic voxel resolution of 1 mm3. Further scan parameters for this anatomical scan were 5,000 ms repetition time, 2.98 ms echo time, 700 and 2500 ms inversion time 1 and 2, respectively, and a flip angle 1 and 2 of α = 4 and 5°, respectively. Siemens’s integrated parallel acquisition technique generalized autocalibrating partial parallel acquisition with an acceleration factor 3 for phase encoding has been applied. Total acquisition time was 8 minutes 22 seconds.
Imaging data processing
Cortical surface reconstruction and volumetric segmentation was performed with the FreeSurfer image analysis suite (version 7.1.1, https://surfer.nmr.mgh.harvard.edu/fswiki). Technical details of these procedures are described elsewhere73,74,75,76,77,78,79,80. FreeSurfer is designed to reconstruct surface-based morphological representations of different cortical measures such as CT, CSA, and CV that can be analyzed in isolation. The processing stream followed FreeSurfer’s standard procedure, and its single steps are described in detail in the Supplementary Methods. We analyzed vertex-wise CT, CSA, and CV as well as the local gyrification index (lGI) as implemented in FreeSurfer (https://surfer.nmr.mgh.harvard.edu/fswiki/LGI). The lGI is a metric that quantifies the amount of cortex buried within the sulcal folds as compared with the amount of cortex on the outer visible cortex. A cortex with extensive folding has a large lGI, whereas a cortex with limited folding has a small lGI81. FreeSurfer also provided the global measures mean CT, total white matter CSA, and total CV.
Statistical analyses
Demographic and clinical variables
Descriptive statistics and correlations of the demographic and clinical variables were computed using IBM SPSS Statistics software (version 27, https://www.ibm.com/products/spss-statistics). Correlations of the TLC scores among the different FTD dimensions and between different FTD dimensions and the covariates of no interest were computed using Pearson’s correlation.
Vertex-wise analyses of the different surface-based morphological measures
The different surface-based morphological measures (CT, CSA, CV, and lGI) were analyzed using FreeSurfer’s implemented statistics tools (mri_glmfit). Partial correlations were computed with either the severity of the different FTD dimensions (positive, negative, and linguistic control) or the severity of general FTD (sum of all TLC items) as independent variable and the vertex-wise cortical measures as dependent variables while simultaneously controlling for sex, age, scanner, and corresponding global brain measures.
When calculating brain-behavior associations for the three distinct FTD dimensions, we also corrected for the other FTD dimensions. Due to the strong correlation between the positive and linguistic control FTD dimensions, we used only one FTD dimension that is not correlated with the FTD dimension of interest. Hence, the negative FTD dimension is used as a covariate when investigating the positive and linguistic control FTD dimensions and the positive FTD dimension is used as a covariate when investigating the negative FTD dimension. For CT and CV analyses, mean global CT and total global CV, respectively, were used as global brain covariates, whereas for CSA and lGI analyses total global white matter CSA was used as global brain covariate. These global cortical measures were used as covariates to account for the dependency between the local cortical measures and the global ones and to increase regional specificity of the findings66,82,83. To rule out any influence of medication, we run also analyses additionally controlling for medication (olanzapine equivalents calculated according to Leucht and colleagues41). Vertex-wise analyses were also corrected for multiple comparisons using Monte Carlo simulations (10,000 repetitions) of the cluster size (mri_glmfit-sim with -mczsim option). Cluster-wise p-value was set at p < 0.05 corrected.
Results
Demographic and clinical characteristics
Demographic and clinical characteristics of the final sample (after exclusions, n = 70) are presented in Table 1. The characteristics of the initial sample (before exclusions, n = 93) are presented in Supplementary Table S1. Correlations among FTD dimensions and between different FTD dimensions and the covariates of no interest are reported in Supplementary Results and Table S2, respectively.
Positive formal thought disorder dimension
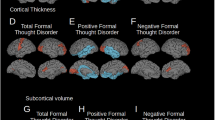
The morphological cortical clusters associated with the severity of the positive FTD dimension are summarized in Table 2 and visualized in Fig. 1. Most prominent results were detected with the lGI. Positive FTD severity was positively correlated with the lGI of core (semantic) language areas including bilateral Heschl’s gyrus and STG as well as core language production areas (i.e., Broca’s area and its homologue). Positive FTD was also positively correlated with the lGI of bilateral insula, MFG, sub- (SubCG), pre- (PreCG), and postcentral gyrus (PostCG), and left AngG and supramarginal gyrus (SupraMG) as well as with CSA of Wernicke’s area. However, we also detected inverse associations of the positive FTD dimension with CV, CT, and/or CSA of brain areas that are not part of the language system (bilateral occipital cortex, left LingG, and right cuneus). Analyses with medication as additional covariate revealed substantially the same results (details of the clusters with medication as covariate see Supplementary Table S4).
Clusters represent partial correlations (positive in red, negative in blue) corrected for sex, age, negative formal thought disorder dimension, scanner, and corresponding global measures as well as corrected for multiple comparisons using Monte Carlo simulations of the cluster size. Cluster information is summarized in Table 2. CSA cortical surface area, CT cortical thickness, CV cortical volume, LGI local gyrification index, LH left hemisphere, RH right hemisphere.
Linguistic control formal thought disorder dimension
The morphological cortical clusters associated with the severity of the linguistic control FTD dimension are summarized in Table 3 and visualized in Fig. 2. There was a considerable overlap of the detected associations with those found for the positive FTD dimension. Specifically, again the lGI of core language areas (i.e., Broca’s area and its homologue, right MTG and STG) was positively associated with linguistic control FTD. In contrast to the positive FTD dimension we did not detect associations of the lGI and the left STG. Linguistic control FTD was also positively correlated with the lGI of bilateral MFG, SubCG, and PostCG, left PreCG and SupraMG, right insula, OFC, frontal pole, ventral ACC, fusiform gyrus, and LingG. As with positive FTD, linguistic control FTD was also positively correlated with CSA of Wernicke’s area. In addition, linguistic control FTD was inversely associated with CV and CSA of brain areas that are not part of the language system such as the bilateral occipital cortex, left LingG, and right cuneus as well as positively correlated with CT of the left OFC, right anterior STG, and insula. Again, analyzing associations with medication as additional covariate only marginally changed the results (details of the clusters with medication as covariate see Supplementary Table S5).
Clusters represent partial correlations (positive in red, negative in blue) corrected for sex, age, negative formal thought disorder dimension, scanner, and corresponding global measures as well as corrected for multiple comparisons using Monte Carlo simulations of the cluster size. Cluster information is summarized in Table 3. CSA cortical surface area, CT cortical thickness, CV cortical volume, LGI local gyrification index, LH left hemisphere, RH right hemisphere.
Negative formal thought disorder dimension
The morphological cortical clusters associated with the severity of the negative FTD dimension are summarized in Table 4 and visualized in Fig. 3. In general, clusters found to be associated with the negative FTD dimension were different from those reported for the severity of the other FTD dimensions. Again, most prominent results were detected with the lGI. While we detected no associations of the negative FTD dimension with core language areas, we found inverse associations of the negative FTD dimension with lGI of bilateral LingG, occipital cortex, cuneus, and precuneus, left OFC and ventral ACC, right medial SFG, middle ACC, superior parietal lobule, intraparietal sulcus, fusiform gyrus, and inferior temporal sulcus. In addition, negative FTD was inversely correlated with CV and CSA of right occipital pole, cuneus, and superior occipital gyrus. As with the other factors associations with medication as additional covariate yielded substantially the same results (details of the clusters with medication as covariate see Supplementary Table S6).
Clusters represent partial correlations (negative in blue) corrected for sex, age, positive formal thought disorder dimension, scanner, and corresponding global measures as well as corrected for multiple comparisons using Monte Carlo simulations of the cluster size. Cluster information is summarized in Table 4. CSA cortical surface area, CV cortical volume, LGI local gyrification index, LH left hemisphere, RH right hemisphere.
Severity of general formal thought disorder
The morphological cortical clusters associated with the severity of general FTD are described in Supplementary Results, summarized in Supplementary Table S3, and visualized in Supplementary Fig. S1. In general, these clusters are very similar to the clusters found to be associated with the positive and partially linguistic control FTD dimensions reported above. The clusters found with medication as an additional covariate are also described in Supplementary Table S7).
Discussion
FTDs form a core symptom dimension evident in multiple psychiatric conditions with unknown pathophysiology. Here we report associations of FTD dimensions with structural brain correlates of multiple morphological markers (CSA, CT, CV) and for the first time with lGI as a marker of early brain development. As hypothesized, we detected prominent altered markers of brain maturation (lGI) and replicated morphological correlates to be evident rather in language-related brain regions for the positive and linguistic control FTD dimensions and in non-language-related brain regions for the negative FTD dimension.
The main novelty are positive correlations between lGI and the severity of the positive and partially also the linguistic control FTD dimensions within fronto-temporal language network regions. Concurrently, we found inverse correlations between the severity of the negative FTD dimension and lGI in occipito-parietal and OFC regions. Thus, results suggest early neurodevelopmental alterations in language areas in subjects who later develop schizophrenia with predominant positive or linguistic control FTD.
Our study also replicated previous published findings of FTD dimension-specific morphological correlates within and beyond the language network. In the following, the associations with different markers investigated, i.e., markers associated with processes of early brain maturation (CSA, lGI) versus those rather associated with processes of neuroplasticity (CT) will be discussed first. We then discuss the different brain regions associated with the different FTD dimensions and how well these regions and their proposed brain functions fit with the literature. Finally we discuss whether our findings support the “dyssemantic hypothesis“84 or rather the “dysexecutive hypothesis“85 of FTD in schizophrenia.
Association with markers influenced by brain development (lGI, CSA) and plasticity (CV, CT), respectively
Remarkably, investigating multiple morphological measures of FTD dimensions revealed distinguishable patterns of brain regions as associated with different markers. In fact, most prominent findings were shown with lGI. Notably, severity of positive and linguistic control FTD was associated with morphological markers in fronto-temporal language regions that are influenced by various factors during brain development (lGI). However, effects were not shown in these brain regions with morphological markers of plasticity changes during the lifespan (CV, CT). Likewise, for negative FTD most prominent findings were present for lGI. Yet findings of CSA only partly support this notion. Still, we may speculate that brain alterations in lGI associated with FTD may at least partly happen already early during (prenatal) brain development68,69 with regional differences, i.e., in fronto-temporal language regions for positive FTD and in occipito-parietal regions for negative FTD. This fits to the clinical observation that FTD can occur early in the course of the disease4,5,6. White and colleagues reviewed several prenatal and early postnatal events (symmetric and asymmetric cell division) that provide a critical foundation for subsequent pathology in gyrification during childhood, adolescence, and into adulthood68. Several factors may impact developmental precursors that contribute to gyrification. Interestingly genetic processes are suggested to play a large role in gyrification, especially early in development68. Kircher and colleagues highlight volume reductions in parts of language-related Wernicke’s and Broca’s areas as a highly consistent finding in structural MRI meta-analyses and mega-analyses in patients with schizophrenia and therefore suggest these alterations as probably of neurodevelopmental, and in particular of genetic, origin2. They suggest risk genes involved in the glutamatergic system led to dysfunctional glutamatergic neurotransmission and synaptic rarefication in the STG as a pathogenic route to positive FTD. Our results support this notion and further build on previous reports and theoretical models applying multiple morphological measures and specifically for the first time lGI measures. However, evidence suggest that general environmental risk factors for schizophrenia (neurodevelopmental, childhood abuse, migration, cannabis, etc.) also contribute to the pathophysiology of FTD in schizophrenia (for review see ref. 2). Furthermore, findings point to the importance to investigate multiple morphological markers to capture the full picture of alterations as associated with FTD dimensions. From a methodological point of view that is of specific relevance if measures go in different directions, which could hinder to detect changes in GM volume measured with voxel-wise morphometric procedures.
Fronto-temporal language network
Here we report that the severity of the general, positive, and partially also of the linguistic control FTD dimensions but not the severity of the negative FTD dimension was associated with morphological features such as lGI of fronto-temporal language regions. In detail, the severity of the positive FTD dimension was positively correlated with lGI in bilateral fronto-temporal regions encompassing Broca’s area and its homologous region but sparing Wernicke’s area and its homologous region. In addition, the severity of the positive FTD dimension was positively correlated with CSA in left temporo-parietal regions partially overlapping with Wernicke’s area. The fronto-temporal morphological correlates of the general and linguistic control FTD dimensions were very similar to those found for the positive FTD dimension. In contrast, no fronto-temporal correlates have been found for the negative FTD dimension. These results are generally in line with the literature on FTD independent of FTD dimensions. In fact, with respect to fronto-temporal language-related regions associated with FTD in general, GM volume decreases or inverse correlations21,22,23,24,25,26,29,30,31,32,33,34,35 as well as increases or positive correlations22,23,33,36 have repeatedly been reported as reviewed elsewhere53,58. Likewise, our results corroborate previous reports that concentrate on FTD dimensions with some discrepancy in the direction of effects1,55,86. While two studies and our study found similar temporal brain regions associated with positive FTD, in our study positive FTD severity was positively correlated with lGI whereas two studies55,86 reported inverse correlations with CT and/or CSA possibly related to the sample, measure of FTD dimension, morphological measure, and/or the covariates of no interest used in the statistical models.
In sum, our study back up previous studies that suggest the positive and linguistic control FTD dimensions as related to both language production (Broca’s area and its homologue) and perception (Wernicke’s area). In addition to these two classical language regions, cortical features of bilateral STG and MTG devoted to semantic processing87 underscored alterations in the functioning of the STG and MTG in FTD88, which are pivotal nodes within the human language processing network89. Thus, results support, the “dyssemantic hypothesis” of FTD84 for positive and linguistic control but not negative FTD dimensions (see below). Importantly, findings further suggest distinguishable changes pointing to distinguishable pathophysiology for FTD dimensions.
Occipito-parietal and occipito-temporal regions
Both different as well as partially overlapping occipito-parietal and occipito-temporal regions were associated with the different FTD dimensions investigated. There were two partially overlapping clusters inversely associated with the severity of all FTD dimensions, albeit with different morphological features (CV, CSA, CT, and/or lGI). One cluster is located in the right cuneus and calcarine sulcus and the other cluster in the left LingG partially extending into the cuneus and calcarine sulcus. Previous studies already reported GM volume reductions of the right cuneus/LingG associated with enhanced FTD severity24, GM volume increases in the right LingG associated with the positive FTD dimension1, and positive correlations between general or positive FTD and CT in bilateral pericalcarine cortex, right cuneus, and left LingG as well as inverse correlations between positive FTD and CSA in bilateral LingG and pericalcarine cortex55. Although an involvement of occipito-parietal and occipito-temporal regions associated with FTD have been reported by some studies, these regions are commonly implicated in visual processing, and it remains unclear whether and how visual processes contribute to FTD. Importantly, occipito-temporal regions were unique for the negative FTD dimension and occipito-parietal correlates were more extensive for the negative compared to the other FTD dimensions. Because FTD typically involves disruptions in the organization, coherence, and logical structure of thought processes, visual information processing might indirectly influence cognitive functions through connections with other brain regions possibly most relevant for the pathophysiology of negative FTD. For example, abnormalities in occipital regions may affect higher-order cognitive processes indirectly linked to FTD, such as attention, memory, and language comprehension.
Orbitofrontal regions
The linguistic control but not the positive FTD dimensions were positively correlated with CT of the left and with lGI of the right OFC. In addition, the negative FTD dimension was negatively correlated with lGI of the left OFC. OFC regions associated with FTD have also been reported by other studies1,21,55. The OFC is associated with cognitive and behavioral control90,91 and with affective processing92, and it has been reported that the OFC is also affected in schizophrenia in general93. Aberrations in OFC morphology could impair cognitive, behavioral, and affective control that indirectly influence thought and language processing and hence contribute to FTD.
Other regions
The present study also found correlates of FTD dimensions in brain regions neither related to primary language nor to other cognitive functions. General, positive, and linguistic control FTD dimensions were positively correlated with lGI in bilateral PreCG and PostCG that are involved in motor and somatosensory processing. Although these regions have been reported to be affected in task-based and resting-state functional59,94,95 and structural55 neuroimaging studies of FTD, it remains unclear how these regions are related to FTD.
Is formal thought disorder related to semantic or executive dysfunctioning?
There has been a prolonged debate regarding whether FTD arises due to impairments in the language processing network, as posited by the “dyssemantic hypothesis“84, or instead stems from deficiencies in higher-order cognitive functions, as proposed by the “dysexecutive hypothesis“85. It has also been proposed that the default mode, salience, and central executive network that interact with the language network might contribute to linguistic disorganization and impoverishment in schizophrenia54. Our findings provide some evidence for the “dyssemantic hypothesis” for the positive and linguistic control FTD dimension but not for the negative FTD dimension because morphological correlates in language-related regions were found only for the positive and linguistic control FTD dimensions but not for the negative FTD dimension. With respect to the “dysexecutive hypothesis”, less evidence is provided by our study because morphological correlates in the classical PFC regions associated with cognitive control and executive functioning96 were rather sparse and limited to the OFC and/or small spots in the lateral and medial PFC and ACC in the positive, linguistic control, and negative FTD dimensions. Hence, our findings suggest that the positive and linguistic control FTD dimensions might result from semantic as well as executive dysfunctions, whereas the pathophysiology of the negative FTD dimension might partially depend on executive dysfunctions and aberration of other than semantic functions. However, due to overlapping morphological correlates in occipital regions among all FTD dimensions, other than semantic and executive dysfunctions might also be involved in all FTD dimensions. What kind of additional mechanism might contribute to FTD is difficult to infer solely from the morphological occipito-parietal and occipito-temporal correlates found in the present study and further research is needed to link these structural alterations to FTD in schizophrenia.
Overlap of clusters across formal thought disorder dimensions and morphological measures
As expected, due to the correlations of the TLC scores among the general, positive, and linguistic control FTD dimensions, there is a considerable overlap of the morphological clusters associated with these FTD dimensions which may indicate shared neurobiological changes associated with these FTD dimensions. In addition to these FTD dimension correlations, there are also correlations among the morphological measures (e.g., CV, CT, and CSA). However, CV is the product of CT times CSA, and we investigated all three measures which at least partly explains the overlap among the morphological clusters found.
Limitations
There are several limitations of the current study worth mentioning. First, most of our patients were medicated, which, in principle, may influence brain structure97. Still, included OLZ equivalence dosage as additional covariate of no interest in our analyses yielded substantially the same results. Second, due to the correlational nature of our study, causality cannot be established. Third, a larger sample size would be needed to confirm the suggested FTD dimensions14 by confirmatory factor analysis98. Forth, due to the dependency of the morphological measures (CV, CT, and CSA) as well as the correlations among FTD dimensions (general, positive, and linguistic control FTD), the clusters found are not fully independent and therefore overlap among morphological measures and FTD dimensions. Last, although the number of items used to define the negative and linguistic control FTD dimension is rather low (three items each) and might therefore limit the validity and reliability of these two FTD dimensions, the FTD dimensions were measured using the well-established TLC scale72 and based on a factor analysis14.
Conclusions
Our findings (I) implies that a marker of early brain development, i.e., local gyrification, might best capture the neuromorphological underpinnings of different FTD dimensions in schizophrenia, (II) support the heterogeneity of FTD in schizophrenia, (III) show FTD dimension-specific neuromorphological correlates, (IV) suggest a distinct pathophysiology at least for the negative compared to other FTD dimensions, (V) reveal that non-language-related brain regions might also contribute to FTD, and (VI) show that neuromorphological correlates are dependent on the morphological marker assessed suggesting that different markers should be investigated.
Importantly, investigating multiple morphological measures reveal distinguishable patterns of brain regions as associated with different markers for different FTD dimensions. In particular, the marker local gyrification associated with early brain development seems to be most sensitive of altered language-related brain regions of the positive and linguistic control FTD dimensions. Our results further reveal that on the one hand compared to the negative FTD dimension the positive and linguistic control FTD dimension are associated with different morphological features (CSA, CT, and/or local gyrification) in language-related (fronto-temporal cortex) as well as non-language-related (mainly OFC and occipital cortex) brain regions. On the other hand, our results also show that the negative FTD dimension is associated with different as well as partially overlapping non-language-related structural brain correlates (mainly occipito-parietal and occipito-temporal cortex) compared to the positive and linguistic control FTD dimensions. Finally, our study provides evidence that the positive and linguistic control FTD dimensions seem to be compatible with the “dyssemantic hypothesis“84 as well as “dysexecutive hypothesis“85 of FTD, whereas the negative FTD dimension is only partially compatible with the “dysexecutive hypothesis“85 but not with the “dyssemantic hypothesis“84 of FTD.
Data availability
The statistical maps that support the findings of this study are available from the corresponding author upon reasonable request. The data are not publicly available due to containing information that could compromise research participant privacy or consent. Explicit consent to deposit raw imaging data was not obtained from the participants.
References
Maderthaner, L. et al. Neural Correlates of Formal Thought Disorder Dimensions in Psychosis. Schizophrenia Bull. 49, S104–S114, https://doi.org/10.1093/schbul/sbac120 (2023).
Kircher, T., Brohl, H., Meier, F. & Engelen, J. Formal thought disorders: from phenomenology to neurobiology. Lancet Psychiatry 5, 515–526, https://doi.org/10.1016/S2215-0366(18)30059-2 (2018).
Roche, E., Creed, L., MacMahon, D., Brennan, D. & Clarke, M. The Epidemiology and Associated Phenomenology of Formal Thought Disorder: A Systematic Review. Schizophr. Bull. 41, 951–962, https://doi.org/10.1093/schbul/sbu129 (2015).
Caplan, R. Thought disorder in childhood. J. Am. Acad. Child Adolesc. Psychiatry 33, 605–615, https://doi.org/10.1097/00004583-199406000-00001 (1994).
Gooding, D. C., Ott, S. L., Roberts, S. A. & Erlenmeyer-Kimling, L. Thought disorder in mid-childhood as a predictor of adulthood diagnostic outcome: findings from the New York High-Risk Project. Psychological Med. 43, 1003–1012, https://doi.org/10.1017/S0033291712001791 (2013).
Ott, S. L., Roberts, S., Rock, D. & Allen, J. & Erlenmeyer-Kimling, L. Positive and negative thought disorder and psychopathology in childhood among subjects with adulthood schizophrenia. Schizophr. Res 58, 231–239, https://doi.org/10.1016/S0920-9964(01)00339-5 (2002).
Yalincetin, B. et al. Relation of formal thought disorder to symptomatic remission and social functioning in schizophrenia. Compr. Psychiatry 70, 98–104, https://doi.org/10.1016/j.comppsych.2016.07.001 (2016).
Tirupati, N. S., Rangaswamy, T. & Raman, P. Duration of untreated psychosis and treatment outcome in schizophrenia patients untreated for many years. Aust. N.Z. J. Psychiatry 38, 339–343, https://doi.org/10.1111/j.1440-1614.2004.01361.x (2004).
Sigaudo, M. et al. Quality of life in stable schizophrenia: The relative contributions of disorganization and cognitive dysfunction. Schizophrenia Res. 153, 196–203, https://doi.org/10.1016/j.schres.2014.01.013 (2014).
Tan, E. J., Thomas, N. & Rossell, S. L. Speech disturbances and quality of life in schizophrenia: Differential impacts on functioning and life satisfaction. Compr. Psychiatry 55, 693–698, https://doi.org/10.1016/j.comppsych.2013.10.016 (2014).
Cavelti, M., Homan, P. & Vauth, R. The impact of thought disorder on therapeutic alliance and personal recovery in schizophrenia and schizoaffective disorder: An exploratory study. Psychiatry Res. 239, 92–98, https://doi.org/10.1016/j.psychres.2016.02.070 (2016).
Zamperoni, G., Tan, E. J., Rossell, S. L., Meyer, D. & Sumner, P. J. Evidence for the factor structure of formal thought disorder: A systematic review. Schizophr. Res 264, 424–434, https://doi.org/10.1016/j.schres.2024.01.006 (2024).
Yalincetin, B. et al. Formal thought disorder in schizophrenia and bipolar disorder: A systematic review and meta-analysis. Schizophr. Res 185, 2–8, https://doi.org/10.1016/j.schres.2016.12.015 (2017).
Nagels, A. et al. The German translation and validation of the scale for the assessment of thought, language and communication: a factor analytic study. Psychopathology 46, 390–395, https://doi.org/10.1159/000345359 (2013).
Nagels, A. et al. Distinct Neuropsychological Correlates in Positive and Negative Formal Thought Disorder Syndromes: The Thought and Language Disorder Scale in Endogenous Psychoses. Neuropsychobiology 73, 139–147, https://doi.org/10.1159/000441657 (2016).
Tan, E. J., Neill, E. & Rossell, S. L. Assessing the Relationship between Semantic Processing and Thought Disorder Symptoms in Schizophrenia. J. Int Neuropsych Soc. 21, 629–638, https://doi.org/10.1017/S1355617715000648 (2015).
Tan, E. J. & Rossell, S. L. Formal thought disorder and neurocognition in schizophrenia: The question of individual mechanisms. Schizophr. Res 190, 189–190, https://doi.org/10.1016/j.schres.2017.03.039 (2017).
Tan, E. J., Yelland, G. W. & Rossell, S. L. Characterising receptive language processing in schizophrenia using word and sentence tasks. Cogn. Neuropsychiatry 21, 14–31, https://doi.org/10.1080/13546805.2015.1121866 (2016).
Roche, E. et al. The prognostic value of formal thought disorder following first episode psychosis. Schizophr. Res 178, 29–34, https://doi.org/10.1016/j.schres.2016.09.017 (2016).
Roche, E. et al. Language disturbance and functioning in first episode psychosis. Psychiat Res 235, 29–37, https://doi.org/10.1016/j.psychres.2015.12.008 (2016).
Sans-Sansa, B. et al. Association of formal thought disorder in schizophrenia with structural brain abnormalities in language-related cortical regions. Schizophrenia Res. 146, 308–313, https://doi.org/10.1016/j.schres.2013.02.032 (2013).
Dieci, M. et al. Brain morphological multidetermination of language and thought disorders in schizophrenia. N. Trends Exp. Clin. Psychiatry 14, 99–102 (1998).
Vita, A. et al. Language and thought disorder in schizophrenia: brain morphological correlates. Schizophrenia Res. 15, 243–251, https://doi.org/10.1016/0920-9964(94)00050-I (1995).
Horn, H. et al. Gray matter volume differences specific to formal thought disorder in schizophrenia. Psychiatry Res. - Neuroimaging 182, 183–186, https://doi.org/10.1016/j.pscychresns.2010.01.016 (2010).
Horn, H. et al. Structural and metabolic changes in language areas linked to formal thought disorder. Br. J. Psychiatry 194, 130–138, https://doi.org/10.1192/bjp.bp.107.045633 (2009).
Marsh, L. et al. Structural magnetic resonance imaging abnormalities in men with severe chronic schizophrenia and an early age at clinical onset. Arch. Gen. Psychiatry 54, 1104–1112, https://doi.org/10.1001/archpsyc.1997.01830240060009 (1997).
Nestor, P. G. et al. A neuropsychological analysis of schizophrenic thought disorder. Schizophrenia Res. 29, 217–225, https://doi.org/10.1016/S0920-9964(97)00101-1 (1998).
Yamasue, H. et al. Localized volume reduction in prefrontal, temporolimbic, and paralimbic regions in schizophrenia: An MRI parcellation study. Psychiatry Res. - Neuroimaging 131, 195–207, https://doi.org/10.1016/j.pscychresns.2004.05.004 (2004).
Rajarethinam, R. P., DeQuardo, J. R., Nalepa, R. & Tandon, R. Superior temporal gyrus in schizophrenia: A volumetric magnetic resonance imaging study. Schizophrenia Res. 41, 303–312, https://doi.org/10.1016/S0920-9964(99)00083-3 (2000).
Shenton, M. E. et al. Abnormalities of the Left Temporal Lobe and Thought Disorder in Schizophrenia: A Quantitative Magnetic Resonance Imaging Study. N. Engl. J. Med. 327, 604–612, https://doi.org/10.1056/NEJM199208273270905 (1992).
Anderson, J. E. et al. An MRI study of temporal lobe abnormalities and negative symptoms in chronic schizophrenia. Schizophrenia Res. 58, 123–134, https://doi.org/10.1016/S0920-9964(01)00372-3 (2002).
Barta, P. E. et al. Planum temporale asymmetry reversal in schizophrenia: Replication and relationship to gray matter abnormalities. Am. J. Psychiatry 154, 661–667, https://doi.org/10.1176/ajp.154.5.661 (1997).
Palaniyappan, L. et al. Structural correlates of formal thought disorder in schizophrenia: An ultra-high field multivariate morphometry study. Schizophrenia Res. 168, 305–312, https://doi.org/10.1016/j.schres.2015.07.022 (2015).
Subotnik, K. L., Bartzokis, G., Green, M. F. & Nuechterlein, K. H. Neuroanatomical correlates of formal thought disorder in schizophrenia. Cogn. Neuropsychiatry 8, 81–88, https://doi.org/10.1080/13546800244000148 (2003).
Weinstein, S., Woodward, T. S. & Ngan, E. T. C. Brain activation mediates the association between structural abnormality and symptom severity in schizophrenia. NeuroImage 36, 188–193, https://doi.org/10.1016/j.neuroimage.2007.02.030 (2007).
Holinger, D. P. et al. Superior temporal gyrus volume abnormalities and thought disorder in left-handed schizophrenic men. Am. J. Psychiatry 156, 1730–1735, https://doi.org/10.1176/ajp.156.11.1730 (1999).
Petty, R. G. et al. Reversal of asymmetry of the planum temporale in schizophrenia. Am. J. Psychiatry 152, 715–721, https://doi.org/10.1176/ajp.152.5.715 (1995).
Rossi, A. et al. Planum temporale asymmetry and thought disorder in schizophrenia. Schizophrenia Res. 12, 1–7, https://doi.org/10.1016/0920-9964(94)90078-7 (1994).
DeLisi, L. E., Hoff, A. L., Neale, C. & Kushner, M. Asymmetries in the superior temporal lobe in male and female first-episode schizophrenic patients: measures of the planum temporale and superior temporal gyrus by MRI. Schizophrenia Res. 12, 19–28, https://doi.org/10.1016/0920-9964(94)90080-9 (1994).
DeLisi, L. E. & Hoff, A. L. Failure to find progressive temporal lobe volume decreases 10 years subsequent to a first episode of schizophrenia. Psychiatry Res. - Neuroimaging 138, 265–268, https://doi.org/10.1016/j.pscychresns.2005.02.005 (2005).
Meisenzahl, E. M. et al. Superior temporal gyrus and P300 in schizophrenia: A combined ERP/structural magnetic resonance imaging investigation. J. Psychiatr. Res. 38, 153–162, https://doi.org/10.1016/S0022-3956(03)00078-5 (2004).
Menon, R. R. et al. Posterior superior temporal gyrus in schizophrenia: grey matter changes and clinical correlates. Schizophrenia Res. 16, 127–135, https://doi.org/10.1016/0920-9964(94)00067-I (1995).
Sallet, P. C. et al. Rightward cerebral asymmetry in subtypes of schizophrenia according to Leonhard’s classification and to DSM-IV: A structural MRI study. Psychiatry Res. - Neuroimaging 123, 65–79, https://doi.org/10.1016/S0925-4927(03)00020-9 (2003).
Shapleske, J., Rossell, S. L., Simmons, A., David, A. S. & Woodruff, P. W. R. Are auditory hallucinations the consequence of abnormal cerebral lateralization? A morphometric MRI study of the sylvian fissure and planum temporale. Biol. Psychiatry 49, 685–693, https://doi.org/10.1016/S0006-3223(00)01006-4 (2001).
Niznikiewicz, M. et al. Abnormal angular gyrus asymmetry in schizophrenia. Am. J. Psychiatry 157, 428–437, https://doi.org/10.1176/appi.ajp.157.3.428 (2000).
Nakamura, M. et al. Orbitofrontal volume deficit in schizophrenia and thought disorder. Brain 131, 180–195, https://doi.org/10.1093/brain/awm265 (2008).
Kühn, S., Romanowski, A., Schubert, F. & Gallinat, J. Reduction of cerebellar grey matter in Crus I and II in schizophrenia. Brain Struct. Funct. 217, 523–529, https://doi.org/10.1007/s00429-011-0365-2 (2012).
Levitt, J. J. et al. Quantitative volumetric MRI study of the cerebellum and vermis in schizophrenia: Clinical and cognitive correlates. Am. J. Psychiatry 156, 1105–1107, https://doi.org/10.1176/ajp.156.7.1105 (1999).
Elfaki, A. et al. Right-left and sex dependent differences of hippocampus and amygdala sizes and their relation to the clinical symptoms of schizophrenia: a comparative brain segmentation study. Folia Med 51, 62–69 (2016).
Fukuzako, H. et al. Reduction in hippocampal formation volume is caused mainly by its shortening in chronic schizophrenia: Assessment by MRI. Biol. Psychiatry 39, 938–945, https://doi.org/10.1016/0006-3223(95)00251-0 (1996).
Rajarethinam, R. et al. Hippocampus and amygdala in schizophrenia: Assessment of the relationship of neuroanatomy to psychopathology. Psychiatry Res. - Neuroimaging 108, 79–87, https://doi.org/10.1016/S0925-4927(01)00120-2 (2001).
Portas, C. M. et al. Volumetric evaluation of the thalamus in schizophrenic male patients using magnetic resonance imaging. Biol. Psychiatry 43, 649–659, https://doi.org/10.1016/S0006-3223(97)00339-9 (1998).
Cavelti, M., Kircher, T., Nagels, A., Strik, W. & Homan, P. Is formal thought disorder in schizophrenia related to structural and functional aberrations in the language network? A systematic review of neuroimaging findings. Schizophr. Res 199, 2–16, https://doi.org/10.1016/j.schres.2018.02.051 (2018).
Palaniyappan, L. Dissecting the neurobiology of linguistic disorganisation and impoverishment in schizophrenia. Semin Cell Dev. Biol. 129, 47–60, https://doi.org/10.1016/j.semcdb.2021.08.015 (2022).
Sharkey, R. J. et al. Differences in the neural correlates of schizophrenia with positive and negative formal thought disorder in patients with schizophrenia in the ENIGMA dataset. Mol. Psychiatry. https://doi.org/10.1038/s41380-024-02563-z (2024).
Stein, F. et al. Dimensions of Formal Thought Disorder and Their Relation to Gray- and White Matter Brain Structure in Affective and Psychotic Disorders. Schizophrenia Bull. 48, 902–911, https://doi.org/10.1093/schbul/sbac002 (2022).
Stein, F. et al. Brain Structural Network Connectivity of Formal Thought Disorder Dimensions in Affective and Psychotic Disorders. Biol. Psychiatry 95, 629–638, https://doi.org/10.1016/j.biopsych.2023.05.010 (2024).
Sumner, P. J., Bell, I. H. & Rossell, S. L. A systematic review of the structural neuroimaging correlates of thought disorder. Neurosci. Biobehav Rev. 84, 299–315, https://doi.org/10.1016/j.neubiorev.2017.08.017 (2018).
Kircher, T. T. J. et al. Neural correlates of formal thought disorder in schizophrenia: Preliminary findings from a functional magnetic resonance imaging study. Arch. Gen. Psychiatry 58, 769–774, https://doi.org/10.1001/archpsyc.58.8.769 (2001).
McGuire, P. K. et al. Pathophysiology of ‘positive’ thought disorder in schizophrenia. Br. J. Psychiatry 173, 231–235, https://doi.org/10.1192/bjp.173.3.231 (1998).
Stegmayer, K. et al. Resting state perfusion in the language network is linked to formal thought disorder and poor functional outcome in schizophrenia. Acta Psychiat Scand. 136, 506–516, https://doi.org/10.1111/acps.12790 (2017).
Kircher, T. T. J. et al. Severity of ‘positive’ formal thought disorder in schizophrenia is inversely correlated with activation in left superior temporal cortex. Neuroimage 13, S1067–S1067 (2001).
Kircher, T., Liddle, P., Brammer, M., Murray, R. & McGuire, P. Neural correlates of negative formal thought disorder. Nervenarzt 74, 748–754, https://doi.org/10.1007/s00115-003-1497-2 (2003).
McGuire, P. K. et al. Distinct neural correlates of ‘positive’ and ‘negative’ thought disorder. Schizophr. Res 29, 111–111, https://doi.org/10.1016/S0920-9964(97)88581-7 (1998).
Stegmayer, K. et al. Specific cerebral perfusion patterns in three schizophrenia symptom dimensions. Schizophr. Res. 190, 96−101, https://doi.org/10.1016/j.schres.2017.03.018 (2017).
Panizzon, M. S. et al. Distinct Genetic Influences on Cortical Surface Area and Cortical Thickness. Cereb. Cortex 19, 2728–2735, https://doi.org/10.1093/cercor/bhp026 (2009).
Meyer, M., Liem, F., Hirsiger, S., Jäncke, L. & Hänggi, J. Cortical Surface Area and Cortical Thickness Demonstrate Differential Structural Asymmetry in Auditory-Related Areas of the Human Cortex. Cereb. Cortex 24, 2541–2552, https://doi.org/10.1093/cercor/bht094 (2014).
White, T., Su, S., Schmidt, M., Kao, C. Y. & Sapiro, G. The development of gyrification in childhood and adolescence. Brain Cogn. 72, 36–45, https://doi.org/10.1016/j.bandc.2009.10.009 (2010).
Dubois, J. et al. Primary cortical folding in the human newborn: an early marker of later functional development. Brain 131, 2028–2041, https://doi.org/10.1093/brain/awn137 (2008).
Kay, S. R., Fiszbein, A. & Opler, L. A. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr. Bull. 13, 261–276 (1987).
Sheehan, D. V. et al. The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J. Clin. Psychiatry 59, 34–57 (1998).
Andreasen, N. C. Scale for the Assessment of Thought, Language, and Communication (Tlc). Schizophrenia Bull. 12, 473–482, https://doi.org/10.1093/schbul/12.3.473 (1986).
Fischl, B. et al. Sequence-independent segmentation of magnetic resonance images. NeuroImage 23, 69–84, https://doi.org/10.1016/j.neuroimage.2004.07.016 (2004).
Fischl, B. et al. Whole brain segmentation: Automated labeling of neuroanatomical structures in the human brain. Neuron 33, 341–355, https://doi.org/10.1016/S0896-6273(02)00569-X (2002).
Dale, A. M., Fischl, B. & Sereno, M. I. Cortical surface-based analysis. I. Segmentation Surf. reconstruction. NeuroImage 9, 179–194, https://doi.org/10.1006/nimg.1998.0395 (1999).
Fischl, B. & Dale, A. M. Measuring the thickness of the human cerebral cortex from magnetic resonance images. PNAS 97, 11050–11055, https://doi.org/10.1073/pnas.200033797 (2000).
Fischl, B., Sereno, M. I. & Dale, A. M. Cortical surface-based analysis. II: Inflat., flattening, a Surf.-based Coord. Syst. NeuroImage 9, 195–207, https://doi.org/10.1006/nimg.1998.0396 (1999).
Fischl, B. et al. Automatically parcellating the human cerebral cortex. Cereb. Cortex 14, 11–22, https://doi.org/10.1093/cercor/bhg087 (2004).
Fischl, B., Sereno, M. I., Tootell, R. B. & Dale, A. M. High-resolution intersubject averaging and a coordinate system for the cortical surface. Hum. Brain Mapp. 8, 272–284, https://doi.org/10.1002/(SICI)1097-0193(1999)8:4<272::AIDHBM10>3.0.CO;2-4 (1999).
Fischl, B., Liu, A. & Dale, A. M. Automated manifold surgery: constructing geometrically accurate and topologically correct models of the human cerebral cortex. IEEE Trans. Med Imaging 20, 70–80, https://doi.org/10.1109/42.906426 (2001).
Schaer, M. et al. A surface-based approach to quantify local cortical gyrification. IEEE Trans. Med Imaging 27, 161–170, https://doi.org/10.1109/TMI.2007.903576 (2008).
Winkler, A. M. et al. Cortical thickness or grey matter volume? The importance of selecting the phenotype for imaging genetics studies. Neuroimage 53, 1135–1146, https://doi.org/10.1016/j.neuroimage.2009.12.028 (2010).
Tamnes, C. K. et al. Cortical surface area and thickness in adult survivors of pediatric acute lymphoblastic leukemia. Pediatr. Blood Cancer 62, 1027–1034, https://doi.org/10.1002/pbc.25386 (2015).
Goldberg, T. E. et al. Cognitive substrates of thought disorder, I: the semantic system. Am. J. Psychiatry 155, 1671–1676, https://doi.org/10.1176/ajp.155.12.1671 (1998).
Barrera, A., McKenna, P. J. & Berrios, G. E. Formal thought disorder in schizophrenia: an executive or a semantic deficit?. Psychol. Med 35, 121–132, https://doi.org/10.1017/s003329170400279x (2005).
Palaniyappan, L., Al-Radaideh, A., Gowland, P. A. & Liddle, P. F. Cortical thickness and formal thought disorder in schizophrenia: An ultra high-field network-based morphometry study. Prog. Neuropsychopharmacol. Biol. Psychiatry 101, 109911, https://doi.org/10.1016/j.pnpbp.2020.109911 (2020).
Price, C. J. The anatomy of language: a review of 100 fMRI studies published in 2009. Ann. N. Y Acad. Sci. 1191, 62–88, https://doi.org/10.1111/j.1749-6632.2010.05444.x (2010).
Wensing, T. et al. Neural correlates of formal thought disorder: An activation likelihood estimation meta-analysis. Hum. Brain Mapp. 38, 4946–4965, https://doi.org/10.1002/hbm.23706 (2017).
Yi, H. G., Leonard, M. K. & Chang, E. F. The Encoding of Speech Sounds in the Superior Temporal Gyrus. Neuron 102, 1096–1110, https://doi.org/10.1016/j.neuron.2019.04.023 (2019).
Bechara, A., Tranel, D. & Damasio, H. Characterization of the decision-making deficit of patients with ventromedial prefrontal cortex lesions. Brain 123, 2189–2202, https://doi.org/10.1093/brain/123.11.2189 (2000).
de la Vega, A., Chang, L. J., Banich, M. T., Wager, T. D. & Yarkoni, T. Large-Scale Meta-Analysis of Human Medial Frontal Cortex Reveals Tripartite Functional Organization. J. Neurosci. 36, 6553–6562, https://doi.org/10.1523/JNEUROSCI.4402-15.2016 (2016).
Rolls, E. T., Cheng, W. & Feng, J. The orbitofrontal cortex: reward, emotion and depression. Brain Commun. 2, fcaa196, https://doi.org/10.1093/braincomms/fcaa196 (2020).
Bellani, M., Cerruti, S. & Brambilla, P. Orbitofrontal cortex abnormalities in schizophrenia. Epidemiol. Psichiatr Soc. 19, 23–25 (2010).
Kircher, T. T. J. et al. Differential activation of temporal cortex during sentence completion in schizophrenic patients with and without formal thought disorder. Schizophrenia Res. 50, 27–40, https://doi.org/10.1016/S0920-9964(00)00042-6 (2001).
Skudlarski, P. et al. Brain Connectivity Is Not Only Lower but Different in Schizophrenia: A Combined Anatomical and Functional Approach. Biol. Psychiatry 68, 61–69, https://doi.org/10.1016/j.biopsych.2010.03.035 (2010).
Friedman, N. P. & Robbins, T. W. The role of prefrontal cortex in cognitive control and executive function. Neuropsychopharmacology 47, 72–89, https://doi.org/10.1038/s41386-021-01132-0 (2022).
Navari, S. & Dazzan, P. Do antipsychotic drugs affect brain structure? A systematic and critical review of MRI findings. Psychol. Med. 39, 1763–1777, https://doi.org/10.1017/S0033291709005315 (2009).
MacCallum, R. C., Widaman, K. F., Zhang, S. & Hong, S. Sample size in factor analysis. Psychol. Methods 4, 84–99, https://doi.org/10.1037/1082-989X.4.1.84 (1999).
Acknowledgements
We acknowledge the schizophrenia patients for participation in the present study. This study received funding from the Swiss National Science Foundation (SNF grant PZPGP3_180022 to K.S.) and from the Adrian und Simone Frutiger Foundation (to K.S.). The funder had no influence on the data, analyses, or decision to publish.
Author information
Authors and Affiliations
Contributions
J.H. analyzed imaging data, data interpretation, and wrote the first draft of the manuscript. S.W. contributed to study design, supervision, data interpretation, and manuscript editing. F.C. and N.G. participated in data collection and manuscript editing. K.S. designed the study, wrote the protocol, acquired funding, supervised the data acquisition, data interpretation, and manuscript editing. All authors discussed findings and edited the final manuscript.
Corresponding author
Ethics declarations
Competing interests
S.W. received honoraria from Lundbeck, Mepha, and Neurolite. All authors declare no competing financial or non-financial interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hänggi, J., Walther, S., Gangl, N. et al. Differential structural cortical correlates of positive, negative, and linguistic control formal thought disorder dimensions in schizophrenia. Schizophr 11, 99 (2025). https://doi.org/10.1038/s41537-025-00644-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41537-025-00644-8