Abstract
Schizophrenia is a severe mental disorder with substantial clinical, economic, and humanistic impacts. This targeted literature review evaluated the burden of schizophrenia on patients and caregivers in Japan. Data were collected from PubMed, Ichushi, CiNii, J-STAGE, and the Cochrane Database (2013–2023) and supplementary materials from medical associations, government agencies, and patient organizations (2018–2023). The review focused on epidemiology, clinical management, societal, humanistic, and economic burdens experienced by patients and caregivers. The review identified 156 journal publications, 73 conference proceedings, and 37 additional data sources. Obesity, depression, and type 2 diabetes were highlighted as frequent comorbidities. Cognitive impairment in schizophrenia, assessed by the Brief Assessment of Cognition in Schizophrenia, indicated severe functional deficits with a Z-score of -2.1. Issues related to long-term hospitalization, including social isolation and inadequate post-discharge support, were also reported. Interventions aimed at improving cognitive function, fostering self-care, and strengthening community cooperation were identified as key factors in reducing early readmission rates. Caregivers experienced significant productivity losses, particularly due to presenteeism, leading to an estimated annual loss of JPY 2.4 million. The hand search further revealed a lack of stakeholder-driven initiatives to address the comprehensive burdens of schizophrenia, such as awareness campaigns, educational programs, and multidisciplinary approaches. This review underscores the multifaceted burdens of schizophrenia in Japan, emphasizing the urgent need for coordinated, evidence-based countermeasures involving multiple stakeholders, including patients, caregivers, healthcare professionals, and policymakers. To reduce burdens and improve healthcare, further research is needed to bridge the gap between required interventions and stakeholder engagement.
Similar content being viewed by others
Introduction
Schizophrenia is a chronic and severe mental disorder that profoundly affects an individual’s thinking, emotions, and behavior. Globally, approximately 23 million people are estimated to live with schizophrenia1, with a lifetime prevalence of approximately 0.3–0.7%2,3. This disorder significantly disrupts patients’ lives, creating challenges in interpersonal relationships and employment4. These difficulties impose substantial social burdens on patients, including stigma and social exclusion, which may hinder reintegration into society and contribute to increasing unemployment and long-term disability.
In Japan, approximately 0.6% of the population is affected by schizophrenia5. The healthcare system is based on universal public insurance, which covers mental healthcare, including outpatient services and some psychological therapies. Psychiatric care remains largely hospital-centered, and long-term hospitalization is common. In recent years, community-based support has gradually expanded, including outreach teams funded by prefectural governments and mental health consultation services at the municipal level6. From a policy perspective, Japan’s 8th Medical Care Plan includes provisions for mental health, emphasizing the promotion of community-based care7. In addition, the government has issued guidelines for the general public titled “The Mental Health Policy for Workplaces,” which aims to improve mental well-being among workers through preventive measures and support systems8.
Despite these institutional frameworks and efforts, significant challenges remain in addressing the burden of schizophrenia and other mental disorders. These include understaffed psychiatric services, inadequate physical healthcare for people with mental illness, slow progress in community transitions, and weak coordination with social services9.
This targeted literature review (TLR) aims to comprehensively assess the burdens of schizophrenia in Japan from epidemiological, clinical, societal, economic, and humanistic perspectives. It also seeks to identify stakeholders’ current activities addressing those burdens of disease and unmet needs and to analyze remaining outcome gaps. We hypothesize that despite ongoing efforts, significant gaps remain between stakeholder activities and the multifaceted burdens of schizophrenia.
Methods
Data sources
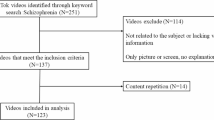
This TLR adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines10, as shown in Fig. 1, and consisted of a biomedical database search and a hand search of gray literature. The database search covering January 1, 2013, to December 31, 2023, was conducted using PubMed, Ichushi, CiNii, J-STAGE, and the Cochrane Database. This 10-year period was chosen to reflect recent trends in research and policy relevant to schizophrenia in Japan.
PRISMA flow diagram of study selection process.
A hand search of gray literature was conducted to identify relevant evidence in Japan from conference abstracts, government statistics, and organizational reports covering January 1, 2018, to December 31, 2023. Fifteen gray literature sources were selected based on consultation with a clinical expert. The list of sources is presented in Supplementary Table S1. Additionally, stakeholders’ activities were reviewed to clarify their roles and contributions to schizophrenia-related challenges over the past 5 years.
Inclusion and exclusion criteria
The inclusion and exclusion criteria were defined using the Population, Intervention, Comparators, Outcomes, and Study Design (PICOS) framework as shown in Table 1. This review examined studies on schizophrenia, focusing on patients and caregivers. It included journal and gray literature covering medical treatments, social support, healthcare programs, and policies. The same PICOS criteria were applied to identify stakeholder activities related to schizophrenia.
Data extraction
The data extraction involved reviewing English and Japanese literature from the specified databases and gray literature. The search strategy of databases, with PubMed as a representative example, is outlined in Supplementary Table S2. The review followed PRISMA guidelines, beginning with defining the review’s scope and consensus on search terms. Searches were implemented systematically, and titles and abstracts were screened for relevance. Articles deemed relevant underwent a full-text review, and data were extracted in alignment with the review objectives to ensure the collection of comprehensive and reliable evidence.
For gray literature, conference abstracts were screened like databases, through review of titles and abstracts, followed by data extraction. Other gray literature and stakeholder activities were identified by hand-searching official websites, capturing government, academic, and organizational efforts over the past 5 years to reflect the multifaceted impact of schizophrenia in Japan. These outcomes included epidemiology, clinical management, societal welfare, humanistic outcomes, and economic impact. Each piece of evidence was reviewed and categorized into the most relevant area to facilitate thematic analysis.
Results
Identified data
From database searches, 156 articles were included in the analysis. Additionally, 110 gray literature articles were identified through conference proceedings (n = 73) and other sources (n = 37). The PRISMA flow of the study selection process is shown in Fig. 1. The selected studies are summarized in Tables 2–4. A complete list of all extracted references is provided in Supplementary Document S1. Searches across 15 organizations identified 746 activities related to schizophrenia, as presented in Table 5.
Characteristics of included studies
Most studies focused on patients (n = 132), followed by caregivers (n = 19) and both (n = 5). Clinical burden was most common (n = 66), followed by epidemiology (n = 41), humanistic burden (n = 27), societal and welfare burden (n = 15), and economic burden (n = 7). Publications were from 2013 to 2017 (n = 79) and 2018 to 2023 (n = 77). Since the peak in 2013 (n = 23), the number of publications has shown a declining trend. In gray literature, conference abstracts mainly targeted patients (n = 60), focusing on clinical management (n = 46), humanistic burden (n = 16), epidemiology (n = 7), and societal and welfare burden (n = 4). Publication years ranged from 2019 to 2023. Other sources also targeted patients (n = 35), addressing clinical management (n = 24), economic burden (n = 8), epidemiology (n = 4), and humanistic burden (n = 1).
Epidemiology
A self-reported online survey from the National Health and Wellness Survey reported a prevalence of 0.59%, primarily among individuals aged 18–64 (89.3%)5. Five studies reported data on mortality11,12,13,14,15. A single-center study by Hattori et al. followed up 59 patients (mean age 64.9) for 10 years; 11 died (mean age 70.6)11. Kimbara et al. examined 134 inpatients (mean age 63); 12 died over 5 years, nearly half (n = 7) from pneumonia12. Ishikawa et al. used data from the Japanese Diagnosis Procedure Combination (DPC) database to assess schizophrenia and cancer stages in acute hospitals; the 30-day in-hospital mortality rate of cancer patients with schizophrenia was 4.2%13. Arai’s dual-center study reported that all 16 patients with schizophrenia and cancer died within a year of the diagnosis; diagnoses were at advanced stages14. Takahashi et al. reported in-hospital mortality rates of 12.9% (acute myocardial infarction: AMI) and 5.8% (pulmonary embolism) among schizophrenia patients using DPC data15.
Comorbidities or complications, such as obesity, hypertension, depression, congestive heart failure, and type 2 diabetes mellitus, were consistently highlighted. A multi-center study found hypertension in 30.5% of outpatients and 19.9% of inpatients; diabetes in 16.8% and 7.1%, respectively16. A single-center study by Haga et al. identified pneumonia risk factors: age, underweight status, smoking habit, use of atypical antipsychotics, and high doses17. A database study by Cheung et al. on medical information from acute care hospitals in Japan reported that 17.2% of 45,201 schizophrenia patients were also diagnosed with depression18.
Clinical management burden
The database study by Cheung et al. (study period between July 2013 and June 2015) reported a mean of 2.0 hospitalizations among 45,201 schizophrenia patients18. Similarly, Kashiwagi et al. analyzed data from the Cognitive Genetics Collaborative Research Organization database (study period: October 2005 and October 2019) and found patients with a violence history had a mean of 2.3 hospitalizations vs. 1.2 without violence19. Furthermore, hospitalization duration varied widely, from 20.7 days to 13,833.5 days. Sugibayashi et al. reported a mean of 3242.1 days among 8379 inpatients (4452 males, 2691 over 65 years old)20. A database study by Kuwabara analyzed 657 patients (222 males) from MDV; the mean index hospitalization was 84.3 days21.
Studies on comorbidities and complications often explored links between antipsychotic drug prescription and comorbidities and complications22,23,24,25. Sugai et al. found that increased prescriptions worsened body mass index, systolic blood pressure, low-density lipoprotein cholesterol, and fasting blood sugar values. They also examined prescribing practices and medication adherence23. Furuhata et al. found multiple sleep medications were associated with age, polypharmacy, and mood stabilizers26. Other studies involved medication adherence, challenges of isolation and physical restraint in psychiatric wards, and challenges in physical comorbidity wards27,28,29,30.
Six studies reported activities of daily living (ADL)20,31,32,33,34,35. A single-center study found that among 770 patients, 513 had mild, 137 moderate, and 81 severe ADL disorders31. In another single-center study, Saito et al. found that 17.6% of 272 schizophrenia patients with dysphagia were “independent” in daily activities32.
Three studies examined cognitive impairment36,37,38. A multi-center study by Fujino et al. reported that the average cognitive impairment score of patients was −16.3, lower than the pivotal score of −10, significantly lower than healthy controls. The score was calculated as the difference between the current Full-Scale Intelligence Quotient (IQ) assessed by the Wechsler Adult Intelligence Scale - Second Edition and the premorbid IQ estimated using the Japanese Adult Reading Test - 2536. A single-center study by Kunizuka et al. found a composite z-score of −2.1 using the Brief Assessment of Cognition in Schizophrenia-Japanese version37. Hori et al. found composite z-scores of −0.7, −1.1, −1.5, and −1.8 among patients with 0, 1, 2, and 3 hospitalizations, respectively38.
According to three gray literature articles, the COVID-19 pandemic had notable impacts on schizophrenia patients. Moriwaki et al. reported increased self-harm and suicidal ideation among hospitalized patients in 2020, although treatment outcomes remained stable and hospitalization duration shortened39. Hanawa et al. identified schizophrenia among suicide attempt diagnoses related to COVID-19-related anxiety and economic stress40. Fukushima et al. reported that in an acute psychiatric ward, a 23% increase in discharged schizophrenia patients in 2020 (from 92 to 113), while the average stay decreased by about 10 days to 58 days41.
Society and welfare burden
Yamada assessed self-stigma using the Perceived Devaluation Discrimination (PDD) scale developed a self-reported scale with 12 items to evaluate perceived stigma, discrimination, stereotyping, and social rejection42,43. The scale ranges from 12 to 60, with higher scores indicating lower levels of stigma. Among 97 inpatients, the mean PDD score was 30.5, lower than those reported in two other studies (mean scores: 32.6 and 31.5). Nagai et al. conducted a cross-sectional study on VAGUS to assess schizophrenia awareness among 148 outpatients, reporting a mean score of 6.6 (standard deviation of 1.5)44. VAGUS is a standardized tool to assess how emotional conditions might influence behavior and decision-making, consisting of 10 self-report and five clinician-rated questions45. A cross-sectional study by Niimura et al. using a patient survey reported a financial burden. Among 21,101 patients with less than 80 days of hospitalization, 19.4% received public assistance. Similarly, among 21,167 patients with over 80 days of hospitalization, 19.1% used public assistance46. A single-center study by Arikawa et al. reported that 151 out of 437 schizophrenia patients used medical expenses for services and support for disabilities. The study mentioned that financial support improves persistence of outpatient treatment for patients with schizophrenia47.
Humanistic burden
Among six studies5,48,49,50,51,52, four quality of life (QoL) assessment tools were identified: EuroQol 5-Dimension (EQ-5D), the Japanese version of the Schizophrenia Quality of Life Scale (JSQLS), MOS 36-Item Short-Form Health Survey (SF-36) v2, and SF-12v2. Baba et al. evaluated Health-related Quality of Life (HRQoL) of schizophrenia patients using SF-12v2, EQ-5D Index, and EQ-5D VAS, dividing patients based on their Patient Health Questionnaire-9 (PHQ-9) scores (<10 vs. ≥10). Patients with PHQ-9 < 10 showed high HRQoL, with EQ-5D VAS scores of 67.8, while those with PHQ-9 ≥ 10 had moderate HRQoL with EQ-5D VAS scores of 44.3. SF-12v2 scores indicated slightly below average physical functioning for both groups, with better mental health for those with PHQ-9 scores < 105. Another study by Ishii et al. with 50 patients used JSQLS to evaluate QoL, with a mean of 45.0 for psychosocial, 47.3 for motivation and energy, and 34.9 for symptoms and side effects scales48. The study also reported the finding that depressive symptoms are key predictors of subjective QoL, highlighting the importance of addressing depression to improve overall well-being in schizophrenia patients.
A qualitative study by Niimura et al. involving 18 discharged patients identified challenges such as “separating life as an inpatient from community life,” including dissatisfaction with inpatient care and a lack of ability to coordinate post-discharge life. Patients reported dissatisfaction with the admission process and treatment in protection rooms, and challenges in managing life after discharge53. Another qualitative study by Tamura et al. with three patients assessed the impact of clozapine treatment, identifying themes like anxiety about a new medication, the hardship of frequent outpatient visits, and joy of symptom attenuation post-treatment54. Qualitative studies also highlighted difficulties faced by caregivers, particularly parents and siblings, in supporting patients. Parents struggled to accept their child’s illness and were anxious about their child’s job continuity despite regular employment55,56,57,58.
Economic burden
An economic evaluation study estimated the cost of schizophrenia in 2008 from a societal perspective. Direct, indirect, and total costs were Japanese yen (JPY) 770.0 billion (US dollars [USD] 5.1 billion), JPY 2.0 trillion (USD 13.2 billion), and JPY 2.8 trillion (USD 18.3 billion), respectively59. Direct costs included social services and healthcare under health insurance, involuntary admission, medical care, and supervision act costs. Indirect costs included morbidity costs (e.g., loss of income due to hospitalization and hospital visits) and mortality costs (future income lost due to early death due to a certain illness). Inpatient and outpatient costs were JPY 602.8 billion (USD 4.0 billion) and 148.0 billion (USD 1.0 billion), respectively59. Kamiura et al. estimated the cost-effectiveness of oral medication by hazard ratios for hospitalization compared to LAI. They found the largest reduction of incremental cost-effectiveness ratios between hazard ratios of 1.1 and 2.060. Baba et al. assessed the productivity loss of schizophrenia patients using the Work Productivity and Activity Impairment (WPAI) tool. The mean total activity impairment was 22.8 for PHQ-9 scores <10 and 53.6 for scores ≥10, indicating moderate to significant impairment5. Nakamura et al. estimated productivity losses due to unemployment or premature death, totaling JPY 3990 million (USD 26.3 million). 4146 patients’ labor was lost due to unemployment, and 151 patients due to premature death61. For caregivers, Sruamsiri et al. analyzed productivity losses among caregivers. Using the WPAI questionnaire with 171 caregivers, they found absenteeism at 4.7% and presenteeism at 25.0%, resulting in per capita annual productivity costs of JPY 2.4 million (USD 16.0 thousand), with presenteeism accounting for 97% (JPY 2.4 million [USD 15.6 thousand]). They also calculated annual productivity losses due to job resignation for caregiving at JPY 1.3 million (USD 8.6 thousand), leading to a total annual productivity loss of JPY 2.4 million (USD 15.8 thousand)62.
The economic outcomes reported in Japanese yen are accompanied by their equivalent values in USD in parentheses, calculated using the exchange rate as of February 28, 2025, which represents the conversion rate at the time of writing this manuscript. The rate applied for conversion was 1 JPY = 0.0066 USD.
Stakeholder activities
Stakeholder activities were categorized into six main types: academic and research activities, education and training, publication, information and communication, policy and advocacy, and community support and services. The Ministry of Health, Labour, and Welfare (MHLW) was the most active organization, with 150 activities, predominantly in policy and advocacy. Efforts also included research on clozapine and medical fees. The National Center of Neurology and Psychiatry was the second-most active organization, with 58 activities focused on academic research, information dissemination, and other areas. Also, they led educational efforts for psychiatrists through the Effectiveness of GUIdeline for Dissemination and Education in psychiatric treatment (EGUIDE) project. The Japanese Society of Neuropsychopharmacology primarily disseminated information and updated treatment guidelines. The Organization of Persons with Psychosocial Disabilities (Porque) was engaged in community-building initiatives and combating stigma through public relations and support activities. For example, they published newspaper articles and appeared on television to raise awareness. Other organizations demonstrated limited activity with the least number of activities. Overall, the more active organizations prioritized policy, research, and communication, while activities related to community support and education were less frequent, indicating potential areas for development.
Discussion
This review identified various psychiatric burdens in Japan, including epidemiological, clinical management, societal welfare, humanistic, and economic perspectives. Although some stakeholder activities address these challenges, many gaps still remain.
Based on the characteristics of the included studies, despite the clinical and societal importance of schizophrenia, Japanese literature remains limited. Notably, publications declined over the past decade, suggesting reduced research attention. While clinical management and epidemiology were well represented, humanistic, societal, and economic burdens were underreported. Similarly, gray literature, including conference abstracts and other sources, was not plentiful. These trends highlight the need for renewed focus and sustained research efforts to capture the multifaceted burden of schizophrenia in Japan.
Schizophrenia’s prevalence in Japan (0.6%5) is comparable to global rates (0.3–0.6%2,63,64,65). Additionally, a systematic review by Saha et al. demonstrated that the mortality gap between schizophrenia patients and the general population is worsening globally, underscoring the need for targeted interventions66. Reported mortality data11,12,13,14,15 appears higher than the global average (~2.5%67), possibly due to study populations with comorbid conditions such as cancer and AMI, or emergency admissions. These patients have a higher mortality risk compared to general schizophrenia patients. Comorbidities, such as hypertension, diabetes, and depression, further complicate health and contribute to disease burden16,17,18. In Australia, national surveys by Morgan et al. emphasized the importance of integrating mental health services with physical healthcare to address comorbidities and improve patient outcomes68. To address these issues in Japan, more epidemiological research is needed to inform public health strategies, early interventions, and equitable healthcare policies.
In addition to physical health issues, suicide is a major contributor to mortality in schizophrenia. Several studies in this review addressed suicide and suicide attempts, underscoring the importance of suicide prevention in this population. While motives were not detailed in the reviewed literature, previous research has identified hallucinations and delusions as common triggers, along with social factors such as living alone and unemployment69. These findings highlight the need for both clinical and social support strategies to reduce excess mortality.
Clinical management of schizophrenia presents challenges due to the complex treatment regimens, long-term hospitalization, and medication side effects. Functional impairments, such as those related to ADLs and cognitive dysfunction, also added to the clinical management burden. However, stakeholder activities targeting these were limited in this review. Holistic care and expanded insurance coverage for cognitive assessments and counseling are needed to close these gaps. Clinical guidelines help standardize care, but implementation remains inconsistent. This is a global issue—despite their benefits, full adoption of guidelines is still challenging in the US and Europe3,70,71. The search of stakeholders’ activities revealed that, in Japan, the EGUIDE project has promoted guideline use by training psychiatrists. A multi-center study showed higher rates of key prescribing indicators, suggesting education improves practice72. Ongoing efforts like EGUIDE are essential to strengthen care quality.
The COVID-19 pandemic had measurable impacts on individuals with schizophrenia, as shown in gray literature. These included increased self-harm and suicidal ideation, shorter hospital stays, and an increase in discharge cases. Such findings highlight the vulnerability of this population during public health crises. To mitigate these effects, healthcare service and policy adaptations are essential. In Japan, early intervention of healthcare services for psychosis is still developing, with efforts focused on reducing the duration of untreated psychosis and building community-based networks for early detection and support73. Additionally, telemedicine has shown promise in improving treatment adherence and reducing relapse rates among patients with schizophrenia, especially during periods of restricted access to in-person care74. Strengthening these healthcare services could help maintain continuity of care and reduce the burden on patients, caregivers, and healthcare systems during future crises.
The stigma influences patients’ willingness to seek treatment and social integration, hindering access to public assistance and increasing emotional and social toll. Welfare support was insufficient, with stakeholders providing additional support. Although stigma is a global issue, New Zealand progressed through nationwide anti-stigma campaigns like “Like Minds, Like Mine,” which could guide Japan75. In Japan, patient organizations advocate through media or events. Kutcher et al. suggested stigma reduction requires context-specific, developmentally appropriate mental health literacy integrated into schools and communities76. Adopting similar strategies in Japan could help improve care access and welfare services.
Strengthening caregiver support systems and ensuring healthcare continuity during crises are essential. The review found that family members bear a disproportionate caregiving burden, risking mental health issues. Stakeholders’ activities were identified to address these challenges. According to Chien and Norman, mutual support groups for caregivers have been shown to reduce stress and improve coping mechanisms77. However, in the review, the burden during disasters/pandemics was identified but not addressed by stakeholder activities. The caregiver’s burden is especially relevant given the earlier discussion on COVID-19’s impact on patients, which likely also intensified the caregiving burden on families. Community-based disaster preparedness programs in the Philippines implemented programs that specifically address vulnerable populations78, could be adapted to Japan’s disaster-prone environment.
The economic burden of schizophrenia is substantial. Indirect costs, such as productivity loss from hospitalization or outpatient visits, and future income lost from premature death, exceed direct costs59. Despite this, our review did not identify any stakeholder activities addressing productivity losses or healthcare-related costs. Mitigating economic burden requires sustainable and comprehensive healthcare systems with public funding and insurance. Assessment of national and regional healthcare cost management is critical, as in the US case, where the economic burden of schizophrenia has been extensively documented, with estimates including both direct healthcare costs and indirect societal costs such as lost productivity79,80.
Limitations and strengths of research
This review leverages gray literature, often excluded from traditional literature reviews, thereby adding unique value by capturing a broader range of real-world evidence and activities. While hand searches for gray literature may introduce bias or overlook less publicized initiatives, this approach ensures a more comprehensive understanding of the current landscape. Although limited to 2013–2023, focusing on recent developments ensures that the findings are highly relevant to current healthcare policies. The research does not aim to evaluate the effectiveness of interventions by stakeholders’ activities for schizophrenia but instead highlights existing initiatives and identifies gaps between stakeholders’ activities and unmet needs, serving as a foundation for future intervention studies. Further research with broader methodologies like implementation science is necessary for a comprehensive understanding of patient circumstances and healthcare policies, but this review provides critical groundwork for future studies to address these gaps.
Conclusions
This review revealed the multifaceted and complex burden of schizophrenia from epidemiological, clinical, societal, welfare, humanistic, and economic perspectives. Policy recommendations include enhancing early diagnosis, standardizing treatment protocols, increasing public health funding, and improving welfare support systems to reduce the overall burden of schizophrenia. Effective strategies require collaborative approaches, including community-integrated care frameworks, multidisciplinary teams, and inclusive social welfare systems. However, the limited evidence underscores the need for evidence-based strategies, requiring further research, such as longitudinal studies and real-world data analyses, to monitor trends and evaluate healthcare interventions. These efforts will ensure that future strategies are grounded in robust evidence and tailored to the needs of patients and caregivers.
Data availability
Minimal data needed to interpret, replicate, or build upon findings can be made available upon reasonable request to the corresponding author.
References
Global Health Data Exchange (GHDx). [cited 12 December 2024]. Available from: https://vizhub.healthdata.org/gbd-results/ (2021).
World Health Organization. World Mental Health Report: Transforming Mental Health for All (World Health Organization, 2022).
Keepers, G. A. et al. The American Psychiatric Association Practice Guideline for the Treatment of Patients With Schizophrenia Vol. 177, 868–872 (American Psychiatric Association Publishing, 2020).
Galderisi, S. et al. Schizophrenia—Time to Commit to Policy Change (Oxford Health Policy Forum, 2024).
Baba, K., Guo, W., Chen, Y., Nosaka, T. & Kato, T. Burden of schizophrenia among Japanese patients: a cross-sectional National Health and Wellness Survey. BMC Psychiatry 22, 410 (2022).
Notice from the Director of the Regional Medical Planning Division, Medical Policy Bureau, Ministry of Health, Labour and Welfare. Medical system for diseases, businesses, and home healthcare. [cited 2025 Apr 4]. Available from: https://www.mhlw.go.jp/content/001103126.pdf (2023).
Notice from the Director of the Medical Policy Bureau of the Ministry of Health, Labour and Welfare. 8th Medical Care Plan. [cited 2025 Apr 4]. Available from: https://www.mhlw.go.jp/content/001108169.pdf (2023).
Occupational Health Division, Department of Safety and Health, Labour Standards Bureau, Ministry of Health, Labour and Welfare. The mental health policy for workplaces—guidelines for improvement of preservation of worker’s mental health. [cited 2025 Apr 4]. Available from: https://www.mhlw.go.jp/content/001579077.pdf (2025).
Japanese Society of Psychiatry and Neurology. The Vision of Psychiatric Care and Health Welfare in Japan. [cited 2025 Apr 4]. Available from: https://www.jspn.or.jp/uploads/uploads/files/activity/20230520.pdf (2023).
Moher, D., Liberati, A., Tetzlaff, J. & Altman, D. G. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 6, e1000097 (2009).
Hattori, S. et al. Association between dysfunction of autonomic nervous system activity and mortality in schizophrenia. Compr. Psychiatry 86, 119–122 (2018).
Kimbara, Y. et al. A survey of clinical characteristics of medical and psychiatric comorbidity patients at Emergency Medical Center, Nagoya Ekisaikai Hospital. J. Jpn. Soc. Emerg. Med. 17, 675–679 (2014).
Ishikawa, H., Yasunaga, H., Matsui, H., Fushimi, K. & Kawakami, N. Differences in cancer stage, treatment and in-hospital mortality between patients with and without schizophrenia: retrospective matched-pair cohort study. Br. J. Psychiatry 208, 239–244 (2016).
Arai, H. A clinical psychiatric perspective on palliative care (Part 2): considering pain in patients with schizophrenia complicated by cancer: a survey using the support team assessment schedule. Jpn. J. Psychiatr. Nurs. 41, 41–48 (2014).
Takahashi, K., Uchida, H., Suzuki, T., Mimura, M. & Ishida, T. Comorbid psychiatric disorders in patients hospitalized for pulmonary embolism and acute myocardial infarction: a Japanese Nationwide Database Study. J. Clin. Psychiatry 83, 21m13962 (2021).
Sugai, T. et al. High prevalence of obesity, hypertension, hyperlipidemia, and diabetes mellitus in Japanese outpatients with schizophrenia: a nationwide survey. PLoS ONE 11, e0166429 (2016).
Haga, T. et al. Risk factors for pneumonia in patients with schizophrenia. Neuropsychopharmacol. Rep. 38, 204–209 (2018).
Cheung, S. et al. Drug utilization of Japanese patients diagnosed with schizophrenia: an administrative database analysis. Clin. Drug Investig. 37, 559–569 (2017).
Kashiwagi, H. et al. Neurocognitive features, personality traits, and social function in patients with schizophrenia with a history of violence. J. Psychiatr. Res. 147, 50–58 (2022).
Sugibayashi, Y., Yoshimura, K., Yamauchi, K., Inagaki, A. & Ikegami, N. Influence of patient characteristics on care time in patients hospitalized with schizophrenia. Neuropsychiatr. Dis. Treat. 10, 1577–1584 (2014).
Kuwabara, H., Saito, Y. & Mahlich, J. Adherence and rehospitalizations in patients with schizophrenia: evidence from Japanese claims data. Neuropsychiatr. Dis. Treat. 11, 935–940 (2015).
Kida, N. et al. Report of 13 cases of agranulocytosis appearing during clozapine treatment. In Proc. Annual Meeting of the Japanese Society of Psychiatry and Neurology Vol. 117, 566 (The Japanese Society of Psychiatry and Neurology, 2021).
Sugai, T. et al. Association between multiple antipsychotic drug use and risk of lifestyle-related diseases in patients with schizophrenia. In Proc. Annual Meeting of the Japanese Society of Psychiatry and Neurology Vol. 116, 209 (The Japanese Society of Psychiatry and Neurology, 2020).
Inakuma, T., Nadatani, S., Ikemoto, S. & Kae, I. Association of paralytic ileus with psychotropic drugs in patients with schizophrenia. In Proc. Annual Meeting of the Japanese Society of Psychiatry and Neurology Vol. 116, 552 (The Japanese Society of Psychiatry and Neurology, 2020).
Kusumi, I., Inoue, S., Baba, K., Nosaka, T. & Anzai, T. Validation of the risk of cardiovascular events with schizophrenia medications: a cohort study using a medical information database. In Proc. Annual Meeting of the Japanese Society of Psychiatry and Neurology Vol. 115, 444 (The Japanese Society of Psychiatry and Neurology, 2019).
Furuhata, R. et al. Factors associated with the actual prescribing of sleeping pills and multiple prescribing: analysis of prescribing data for schizophrenia and depression patients. In Proc. Annual Meeting of the Japanese Society of Psychiatry and Neurology Vol. 117, 575 (The Japanese Society of Psychiatry and Neurology, 2021).
Goto, T., Toda, Y., Yamada, H., Kato, H. & Kodama, Y. Changes in individuals with schizophrenia who relapsed after discontinuation of medication up to continuation of medication. In Proc. Annual Conference of the Japanese Academy of Psychiatric and Mental Health Nursing Vol. 33, 99 (Japanese Academy of Psychiatric and Mental Health Nursing, 2023).
Hirota, N. et al. Clinical and demographic characteristics of the use of seclusion and restraint in NMU Psychiatric Institute (NMU-PI). Psychiatry 37, 440–447 (2020).
Taniguchi, N. et al. Have the Problems in Psychiatric Complex Wards Been Solved? -Compared to 10 years ago. In Proc. Annual Meeting of the Japanese Society of Psychiatry and Neurology Vol. 117, 635 (The Japanese Society of Psychiatry and Neurology, 2021).
Ishii, J. et al. Association between functional prognosis and medication adherence in schizophrenia. In Proc. Annual Meeting of the Japanese Society of Psychiatry and Neurology Vol. 116, 443 (The Japanese Society of Psychiatry and Neurology, 2020).
Sado, J. et al. Socio-environmental factors associated with diabetes mellitus among patients hospitalized with schizophrenia in Japan. Environ. Health Prev. Med. 21, 460–469 (2016).
Saito, T., Koike, S., Ozawa, T. & Usui, Y. Dysphagia among schizophrenic patients: multivariate analysis of risk factors for impairment of swallowing ability. Jpn. J. Dysphagia Rehabil. 17, 201–208 (2013).
Nakagawa, Y., Kumoi, H., Sasaki, H. & Yamada, S. Positional stability of the non-dominant hand is associated with difficulties in daily functioning in schizophrenia. Asian J. Occup. Ther. 19, 124–131 (2023).
Hosoi, T., Komatsu, T. & Ishibashi, Y. Conclusion of literature to schizophrenia inpatient concerned with physical exercise in Japan. Phys. Ther. Jpn. 47, 354–362 (2020).
Ikeda, T. & Makabe, H. The physical functionality of long-term schizophrenic inpatients at our hospital. Annu. Rep. Tohoku Sect. Jpn. Phys. Ther. Assoc. 27, 14–17 (2015).
Fujino, H. et al. Estimated cognitive decline in patients with schizophrenia: a multicenter study. Psychiatry Clin. Neurosci. 71, 294–300 (2017).
Kunizuka, T., Abe, H., Suzuki, Y. & Someya, T. Relationship between clinical features and cognitive function of patients with schizophrenia in Niigata University Medical and Dental Hospital. Psychiatr. Neurol. Jpn. 121, 344–355 (2019).
Hori, H., Atake, K., Katsuki, A. & Yoshimura, R. Effects of the number of hospitalizations on cognitive function in Japanese patients with stable schizophrenia. CNS Spectr. 26, 658–663 (2021).
Moriwaki, K. et al. Impact of the COVID-19 pandemic on psychiatric emergency inpatient care: a report from a core hospital. In Proc. Annual Meeting of the Japanese Society of Psychiatry and Neurology Vol. 117, 605 (The Japanese Society of Psychiatry and Neurology, 2021).
Hanawa, K. et al. Association between COVID-19 and suicide attempts. In Proc. Annual Meeting of the Japanese Society of Psychiatry and Neurology Vol. 117, 612 (The Japanese Society of Psychiatry and Neurology, 2021).
Fukushima, S. & Matsumoto, K. Report and discussion of outpatient and inpatient trends before and after the COVID-19 pandemic at our hospital. In Proc. Annual Meeting of the Japanese Society of Psychiatry and Neurology Vol. 118, 639 (The Japanese Society of Psychiatry and Neurology, 2022).
Yamada, M. Impact of self-stigma on the self-esteem of patients with schizophrenia. J. Jpn. Soc. Nurs. Res. 38, 1_85–1_91 (2015).
Link, B. G., Cullen, F. T., Struening, E. L., Shrout, P. E. & Dohrenwend, B. P. A modified labeling theory approach to mental disorders: an empirical assessment. Am. Sociol. Rev. 54, 400–423 (1989).
Nagai, N. et al. Drug attitude, insight, and patient’s knowledge about prescribed antipsychotics in schizophrenia: a cross-sectional survey. Neuropsychiatr. Dis. Treat. 16, 781–787 (2020).
Gerretsen, P. et al. The VAGUS insight into psychosis scale-self-report and clinician-rated versions. Psychiatry Res. 220, 1084–1089 (2014).
Niimura, J., Nakanishi, M., Yamasaki, S. & Nishida, A. Regional supply of outreach service and length of stay in psychiatric hospital among patients with schizophrenia: national case mix data analysis in Japan. Psychiatry Res. 258, 295–298 (2017).
Arikawa, M. et al. The use of the Japanese public financial support has positive impact on persistence with outpatient treatments for schizophrenia: single-center retrospective cohort study in Japan. Patient Prefer. Adherence 15, 169–175 (2021).
Ishii, Y. et al. Relationship between quality of life and clinical factors in inpatients with schizophrenia. J. Med. Investig. 69, 80–85 (2022).
Sugawara, N. et al. Attitudes toward long-acting injectable antipsychotics among patients with schizophrenia in Japan. Neuropsychiatr. Dis. Treat. 15, 205–211 (2019).
Sugawara, N. et al. Body mass index and quality of life among outpatients with schizophrenia in Japan. BMC Psychiatry 13, 108 (2013).
Yoshida, K. et al. Resilience in schizophrenia: a comparative study between a remote island and an urban area in Japan. Schizophr. Res. 171, 92–96 (2016).
Sato, K. et al. Comprehensive care for first-episode schizophrenia. The continuity from inpatient psychiatric treatment to community care. Jpn. J. Prev. Psychiatry 1, 68–79 (2016).
Niimura, J., Tanoue, M. & Nakanishi, M. Challenges following discharge from acute psychiatric inpatient care in Japan: patients’ perspectives. J. Psychiatr. Ment. Health Nurs. 23, 576–584 (2016).
Tamura, S., Takai, M., Masuoka, T. & Manabe, R. Impact on daily life of patients who accepted clozapine treatment. Jpn. Psychiatr. Nurs. Soc. 57, 384–388 (2014).
Kojima, Y., Yokoyama, K., Hayashi, H. & Morita, M. Thoughts and involvement of parents regarding “employment” of their children with schizophrenia. In Proc. Annual Conference of the Japanese Academy of Psychiatric and Mental Health Nursing Vol. 31, 73 (Japanese Academy of Psychiatric and Mental Health Nursing, 2021).
Kimura, S., Yokoyama, K. & Morita, M. Experiences of siblings of patients with schizophrenia who became mental health care welfare supporters. In Proc. Annual Conference of the Japanese Academy of Psychiatric and Mental Health Nursing Vol. 31, 109 (Japanese Academy of Psychiatric and Mental Health Nursing, 2021).
Yabuta, A. Family coping processes for help-seeking behavior in persons with schizophrenia. In Proc. Annual Conference of the Japanese Academy of Psychiatric and Mental Health Nursing Vol. 29, 90 (Japanese Academy of Psychiatric and Mental Health Nursing, 2019).
Fujisawa, Y. Awareness and attitude of mothers who continue to interact with their children with schizophrenia. In Proc. Annual Conference of the Japanese Academy of Psychiatric and Mental Health Nursing Vol. 29, 91 (Japanese Academy of Psychiatric and Mental Health Nursing, 2019).
Sado, M. et al. The cost of schizophrenia in Japan. Neuropsychiatr. Dis. Treat. 9, 787–798 (2013).
Kamiura, Y., Sakurai, M., Inagaki, A. & Yamauchi, K. Cost effectiveness analysis of aripiprazole once-monthly and oral formulation for the treatment of schizophrenia in Japan: a Markov model simulation study. Jpn. J. Clin. Psychopharmacol. 21, 955–964 (2018).
Nakamura, Y. & Mahlich, J. Productivity and deadweight losses due to relapses of schizophrenia in Japan. Neuropsychiatr. Dis. Treat. 13, 1341–1348 (2017).
Sruamsiri, R., Mori, Y. & Mahlich, J. Productivity loss of caregivers of schizophrenia patients: a cross-sectional survey in Japan. J. Ment. Health 27, 583–587 (2018).
Kessler, R. C. et al. The prevalence and correlates of nonaffective psychosis in the National Comorbidity Survey Replication (NCS-R). Biol. Psychiatry 58, 668–676 (2005).
Wu, E. Q., Shi, L., Birnbaum, H., Hudson, T. & Kessler, R. Annual prevalence of diagnosed schizophrenia in the USA: a claims data analysis approach. Psychol. Med. 36, 1535–1540 (2006).
Desai, P. R., Lawson, K. A., Barner, J. C. & Rascati, K. L. Estimating the direct and indirect costs for community-dwelling patients with schizophrenia. J. Pharm. Health Serv. Res. 4, 187–194 (2013).
Saha, S., Chant, D., Welham, J. & McGrath, J. A systematic review of the prevalence of schizophrenia. PLoS Med. 2, e141 (2005).
Correll, C. U. et al. Mortality in people with schizophrenia: a systematic review and meta-analysis of relative risk and aggravating or attenuating factors. World Psychiatry 21, 248–271 (2022).
Morgan, V. A. et al. Psychosis prevalence and physical, metabolic and cognitive co-morbidity: data from the second Australian national survey of psychosis. Psychol. Med. 44, 2163–2176 (2014).
Koeda, A. et al. Characteristics of suicide attempts in patients diagnosed with schizophrenia in comparison with depression: a study of emergency room visit cases in Japan. Schizophr. Res. 142, 31–39 (2012).
National Institute for Health and Care Excellence: Guidelines. Psychosis and Schizophrenia in Adults: Prevention and Management (National Institute for Health and Care Excellence (NICE), 2014).
Gaebel, W., Weinmann, S., Sartorius, N., Rutz, W. & McIntyre, J. S. Schizophrenia practice guidelines: international survey and comparison. Br. J. Psychiatry 187, 248–255 (2005).
Hasegawa, N. et al. Effect of education regarding treatment guidelines for schizophrenia and depression on the treatment behavior of psychiatrists: a multicenter study. Psychiatry Clin. Neurosci. 77, 559–568 (2023).
Mizuno, M. et al. Early psychosis initiative in Japan: challenges and opportunities. In Early Intervention in Psychiatric Disorders Across Cultures: Oxford Cultural Psychiatry (eds Chen, E. Y. H., Ventriglio, A. & Bhugra, D.) 109–116 (Oxford University Press, 2019).
Keshvardoost, S. et al. Telemedicine for patients with schizophrenia: a systematic literature review on applications and outcomes. Iran. J. Psychiatry Behav. Sci. 16, e122144 (2022).
Thornicroft, C., Wyllie, A., Thornicroft, G. & Mehta, N. Impact of the “Like Minds, Like Mine” anti-stigma and discrimination campaign in New Zealand on anticipated and experienced discrimination. Aust. N.Z. J. Psychiatry 48, 360–370 (2014).
Kutcher, S., Wei, Y. & Coniglio, C. Mental health literacy: past, present, and future. Can. J. Psychiatry 61, 154–158 (2016).
Chien, W.-T. & Norman, I. The effectiveness and active ingredients of mutual support groups for family caregivers of people with psychotic disorders: a literature review. Int. J. Nurs. Stud. 46, 1604–1623 (2009).
Center for Disaster Preparedness Foundation. Inclusive Data Management System: A Guide to Disability-Inclusive and Data-Driven Local Government Planning https://3b723566-4dad-4654-af88-4aa05c744db7.filesusr.com/ugd/cb2848_1af9e3187ecb4a0fb843ca8782f68e1b.pdf.
Cloutier, M. et al. The economic burden of schizophrenia in the United States in 2013. J. Clin. Psychiatry 77, 764–771 (2016).
Kadakia, A. et al. The economic burden of schizophrenia in the United States. J. Clin. Psychiatry 83, 22m14458 (2022).
Acknowledgements
Nippon Boehringer Ingelheim Co., Ltd. participated in the study design, research, analysis, data collection, data interpretation, and the presentation’s review and approval. Members of Syneos Health Japan contributed to study planning, data curation, analysis, and manuscript development. Russell Miller, MHS, ISMPP CMPP™ of Syneos Health Japan, provided medical writing support. The author(s) meet criteria for authorship as recommended by the International Committee of Medical Journal Editors (ICMJE). This study was funded by Nippon Boehringer Ingelheim Co., Ltd.
Author information
Authors and Affiliations
Contributions
Conception or design of the work, acquisition, analysis, and interpretation of data: F.O. and M.O. Drafting the work and revising it critically: F.O. and M.O. Final approval of the completed manuscript: F.O. and M.O. All authors had access to relevant data, participated in drafting, reviewing, and approving this publication, and approved the final manuscript, taking public responsibility for appropriate portions of the content.
Corresponding author
Ethics declarations
Competing interests
F.O. is an employee of Nippon Boehringer Ingelheim Co., Ltd. M.O. is an employee of Syneos Health Japan, a contract research organization with clients in the biotech/pharmaceutical industry. This work was solely supported by Nippon Boehringer Ingelheim Co., Ltd., which provided funding for the study and service fees to Syneos Health Japan. No honoraria or payments were made for authorship.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ono, F., Okamura, M. Clinical, social, and economic burdens of schizophrenia in Japan: a targeted literature review. Schizophr 12, 27 (2026). https://doi.org/10.1038/s41537-025-00716-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41537-025-00716-9