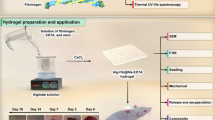
Abstract
Current smart dressings with wound monitoring and electrical stimulation capabilities rely on flexible electronics comprising various sensors and external power sources. Despite increasing efforts to integrate all these components onto flexible, breathable and biocompatible substrates, realizing a zero-power electrical stimulation without compromising the clinical applicability remains challenging. Here we report a solution that harnesses the temperature gradient between the wound and dressing to generate an electric stimulus that provides active wound healing management. This was achieved by a thermogalvanic cell (TGC) dressing composed of Fe2+/Fe3+ cross-linked alginate hydrogel reinforced by nanofibres. The TGC dressing exhibits biocompatibility, antibacterial performance, easy re-shaping and moisture permeability. Moreover, the TGC dressing generates an exogenous electric field, promoting the spontaneous acceleration of wound healing. We additionally integrate a sensing system that can monitor respiration rate. In the large porcine wound model, the wound healing rate of a TGC-bandaged group is improved by about 20.6% on day 14 compared with an untreated group. Our wireless wound monitoring system may facilitate real-time monitoring of common wound models at different wound development stages.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The main data supporting the results of this study are available in the paper and its Supplementary Information. The RNA-seq data are available from the Sequence Read Archive (SRA) database (accession number PRJNA1244933). The raw and processed datasets generated during the study are available from the corresponding author upon reasonable request. Source data are provided with this paper.
Code availability
The custom codes used in smart wound analysis training are available via DR-NTU at https://doi.org/10.21979/N9/ELSU4C (ref. 75).
Change history
12 February 2026
A Correction to this paper has been published: https://doi.org/10.1038/s41551-026-01631-9
References
Yazdanpanah, L. et al. Incidence and risk factors of diabetic foot ulcer: a population-based diabetic foot cohort (ADFC study)—two-year follow-up study. Int. J. Endocrinol. 2018, 1–9 (2018).
Werdin, F., Tenenhaus, M. & Rennekampff, H.-O. Chronic wound care. Lancet 372, 1860–1862 (2008).
Eming, S. A., Martin, P. & Tomic-Canic, M. Wound repair and regeneration: mechanisms, signaling, and translation. Sci. Transl. Med. 6, 265sr6 (2014).
Han, G. & Ceilley, R. Chronic wound healing: a review of current management and treatments. Adv. Ther. 34, 599–610 (2017).
Liang, Y., He, J. & Guo, B. Functional hydrogels as wound dressing to enhance wound healing. ACS Nano 15, 12687–12722 (2021).
Ding, J. et al. Electrospun polymer biomaterials. Prog. Polym. Sci. 90, 1–34 (2019).
Homaeigohar, S. & Boccaccini, A. R. Antibacterial biohybrid nanofibers for wound dressings. Acta Biomater. 107, 25–49 (2020).
Yamakawa, S. & Hayashida, K. Advances in surgical applications of growth factors for wound healing. Burns Trauma https://doi.org/10.1186/s41038-019-0148-1 (2019).
Farahani, M. & Shafiee, A. Wound healing: from passive to smart dressings. Adv. Healthc. Mater. 10, 2100477 (2021).
McLister, A., McHugh, J., Cundell, J. & Davis, J. New developments in smart bandage technologies for wound diagnostics. Adv. Mater. 28, 5732–5737 (2016).
Gao, Y. et al. A flexible multiplexed immunosensor for point-of-care in situ wound monitoring. Sci. Adv. 7, eabg9614 (2021).
Tu, J. et al. A wireless patch for the monitoring of C-reactive protein in sweat. Nat. Biomed. Eng. 7, 1293–1306 (2023).
Yang, S. M. et al. Soft, wireless electronic dressing system for wound analysis and biophysical therapy. Nano Today 47, 101685 (2022).
Xu, G. et al. Battery-free and wireless smart wound dressing for wound infection monitoring and electrically controlled on-demand drug delivery. Adv. Funct. Mater. 31, 2100852 (2021).
Pang, Q. et al. Smart flexible electronics-integrated wound dressing for real-time monitoring and on-demand treatment of infected wounds. Adv. Sci. 7, 1902673 (2020).
Lou, D. et al. Flexible wound healing system for pro-regeneration, temperature monitoring and infection early warning. Biosens. Bioelectron. 162, 112275 (2020).
Zhang, M. et al. Super-ductile, injectable, fast self-healing collagen-based hydrogels with multi-responsive and accelerated wound-repair properties. Chem. Eng. J. 405, 126756 (2021).
Swisher, S. L. et al. Impedance sensing device enables early detection of pressure ulcers in vivo. Nat. Commun. 6, 6575 (2015).
Jiang, Y. et al. Wireless, closed-loop, smart bandage with integrated sensors and stimulators for advanced wound care and accelerated healing. Nat. Biotechnol. 41, 652–662 (2023).
Zheng, M. et al. Skin-inspired gelatin-based flexible bio-electronic hydrogel for wound healing promotion and motion sensing. Biomaterials 276, 121026 (2021).
Lin, X. et al. Ultra-conformable ionic skin with multi-modal sensing, broad-spectrum antimicrobial and regenerative capabilities for smart and expedited wound care. Adv. Sci. 8, 2004627 (2021).
Guo, M., Wang, Y., Gao, B. & He, B. Shark tooth-inspired microneedle dressing for intelligent wound management. ACS Nano 15, 15316–15327 (2021).
Xiong, Z. et al. A wireless and battery-free wound infection sensor based on DNA hydrogel. Sci. Adv. 7, eabj1617 (2021).
Wang, X.-F. et al. Flexible electrical stimulation device with Chitosan-Vaseline® dressing accelerates wound healing in diabetes. Bioact. Mater. 6, 230–243 (2021).
Hu, W. et al. Enhancing proliferation and migration of fibroblast cells by electric stimulation based on triboelectric nanogenerator. Nano Energy 57, 600–607 (2019).
Cui, S., Rouabhia, M., Semlali, A. & Zhang, Z. Effects of electrical stimulation on human skin keratinocyte growth and the secretion of cytokines and growth factors. Biomed. Mater. 16, 065021 (2021).
Luo, R. et al. Reshaping the endogenous electric field to boost wound repair via electrogenerative dressing. Adv. Mater. 35, 2208395 (2023).
Song, J. W. et al. Bioresorbable, wireless, and battery-free system for electrotherapy and impedance sensing at wound sites. Sci. Adv. 9, eade4687 (2023).
Barman, S. R. et al. A self-powered multifunctional dressing for active infection prevention and accelerated wound healing. Sci. Adv. 9, eadc8758 (2023).
Wang, C. et al. Wound management materials and technologies from bench to bedside and beyond. Nat. Rev. Mater. https://doi.org/10.1038/s41578-024-00693-y (2024).
Liu, D. et al. Lattice plainification advances highly effective SnSe crystalline thermoelectrics. Science 380, 841–846 (2023).
Qin, B. et al. High thermoelectric efficiency realized in SnSe crystals via structural modulation. Nat. Commun. 14, 1366 (2023).
Qin, Y. et al. Grid-plainification enables medium-temperature PbSe thermoelectrics to cool better than Bi2Te3. Science 383, 1204–1209 (2024).
Lei, Z., Gao, W. & Wu, P. Double-network thermocells with extraordinary toughness and boosted power density for continuous heat harvesting. Joule 5, 2211–2222 (2021).
Kim, D.-H., Akbar, Z. A., Malik, Y. T., Jeon, J.-W. & Jang, S.-Y. Self-healable polymer complex with a giant ionic thermoelectric effect. Nat. Commun. 14, 3246 (2023).
Han, C.-G. et al. Giant thermopower of ionic gelatin near room temperature. Science 368, 1091–1098 (2020).
Yu, B. et al. Thermosensitive crystallization–boosted liquid thermocells for low-grade heat harvesting. Science 370, 342–346 (2020).
Shirzaei Sani, E. et al. A stretchable wireless wearable bioelectronic system for multiplexed monitoring and combination treatment of infected chronic wounds. Sci. Adv. 9, eadf7388 (2023).
Yang, H. et al. Alloying design of biodegradable zinc as promising bone implants for load-bearing applications. Nat. Commun. 11, 401 (2020).
Shen, D. et al. PDLLA-Zn-nitrided Fe bioresorbable scaffold with 53-μm-thick metallic struts and tunable multistage biodegradation function. Sci. Adv. 7, eabf0614 (2021).
Aderibigbe, B. & Buyana, B. Alginate in wound dressings. Pharmaceutics 10, 42 (2018).
Liling, G. et al. Effects of ionic crosslinking on physical and mechanical properties of alginate mulching films. Carbohydr. Polym. 136, 259–265 (2016).
Reilly, D. M. & Lozano, J. Skin collagen through the lifestages: importance for skin health and beauty. Plast. Aesthetic Res. 8, 2 (2021).
Thakur, S. et al. Recent progress in sodium alginate based sustainable hydrogels for environmental applications. J. Clean. Prod. 198, 143–159 (2018).
Wang, Y., He, Y., Yan, S., Yin, X. & Chen, J. Development of alginate hydrogel modified multifunctional filtration membrane with robust anti-fouling property for efficient water purification. Colloids Surf. A 582, 123891 (2019).
McIntosh, C., Ivory, J. D. & Gethin, G. Managing wound exudate in diabetic foot ulcers. Diabet. Foot J. 22, 46–52 (2019).
Iizaka, S. et al. Quantitative estimation of exudate volume for full-thickness pressure ulcers: the ESTimation method. J. Wound Care 20, 453–463 (2011).
Yeoh-Ellerton, S. & Stacey, M. C. Iron and 8-isoprostane levels in acute and chronic wounds. J. Invest. Dermatol. 121, 918–925 (2003).
Foulds, I. S. & Barker, A. T. Human skin battery potentials and their possible role in wound healing. Br. J. Dermatol. 109, 515–522 (1983).
Levin, M. Large-scale biophysics: ion flows and regeneration. Trends Cell Biol. 17, 261–270 (2007).
Reid, B., Nuccitelli, R. & Zhao, M. Non-invasive measurement of bioelectric currents with a vibrating probe. Nat. Protoc. 2, 661–669 (2007).
Guo, A. et al. Effects of physiological electric fields on migration of human dermal fibroblasts. J. Invest. Dermatol. 130, 2320–2327 (2010).
Kloth, L. C. Electrical stimulation for wound healing: a review of evidence from in vitro studies, animal experiments, and clinical trials. Int. J. Low. Extrem. Wounds 4, 23–44 (2005).
Cao, L., Pu, J. & Zhao, M. GSK-3β is essential for physiological electric field-directed Golgi polarization and optimal electrotaxis. Cell. Mol. Life Sci. 68, 3081–3093 (2011).
Zhao, M. Electrical fields in wound healing—an overriding signal that directs cell migration. Semin. Cell Dev. Biol. 20, 674–682 (2009).
Kim, K., Hwang, S. & Lee, H. Unravelling ionic speciation and hydration structure of Fe(III/II) redox couples for thermoelectrochemical cells. Electrochim. Acta 335, 135651 (2020).
Zhou, H., Yamada, T. & Kimizuka, N. Supramolecular thermo-electrochemical cells: enhanced thermoelectric performance by host–guest complexation and salt-induced crystallization. J. Am. Chem. Soc. 138, 10502–10507 (2016).
Kim, T. et al. High thermopower of ferri/ferrocyanide redox couple in organic-water solutions. Nano Energy 31, 160–167 (2017).
Blacklow, S. O. et al. Bioinspired mechanically active adhesive dressings to accelerate wound closure. Sci. Adv. 5, eaaw3963 (2019).
Theocharidis, G. et al. A strain-programmed patch for the healing of diabetic wounds. Nat. Biomed. Eng. 6, 1118–1133 (2022).
Godoy-Gallardo, M. et al. Antibacterial approaches in tissue engineering using metal ions and nanoparticles: from mechanisms to applications. Bioact. Mater. 6, 4470–4490 (2021).
Yan, L.-J. & Wu, J. Streptozotocin-induced type 1 diabetes in rodents as a model for studying mitochondrial mechanisms of diabetic β cell glucotoxicity. Diabetes Metab. Syndr. Obes. Targets Ther. 8, 181 (2015).
Serbu, D. G. Infrared imaging of the diabetic foot. InfraMation Proc. 86, 5–20 (2009).
Landén, N. X., Li, D. & Ståhle, M. Transition from inflammation to proliferation: a critical step during wound healing. Cell. Mol. Life Sci. 73, 3861–3885 (2016).
Chanmugam, A. et al. Relative temperature maximum in wound infection and inflammation as compared with a control subject using long-wave infrared thermography. Adv. Skin Wound Care 30, 406–414 (2017).
Power, G., Moore, Z. & O’Connor, T. Measurement of pH, exudate composition and temperature in wound healing: a systematic review. J. Wound Care 26, 381–397 (2017).
van Netten, J. J. et al. Diagnostic values for skin temperature assessment to detect diabetes-related foot complications. Diabetes Technol. Ther. 16, 714–721 (2014).
Armstrong, D. G. et al. Skin temperature monitoring reduces the risk for diabetic foot ulceration in high-risk patients. Am. J. Med. 120, 1042–1046 (2007).
Yekta, Z., Pourali, R., Nezhadrahim, R., Ravanyar, L. & Ghasemi-Rad, M. Clinical and behavioral factors associated with management outcome in hospitalized patients with diabetic foot ulcer. Diabetes Metab. Syndr. Obes. Targets Ther. 4, 371 (2011).
Mauricio, D., Jude, E., Piaggesi, A. & Frykberg, R. Diabetic foot: current status and future prospects. J. Diabetes Res. 2016, 5691305 (2016).
Chen, J. et al. Wearable self-powered human motion sensors based on highly stretchable quasi-solid state hydrogel. Nano Energy 88, 106272 (2021).
Zhu, Y. et al. Recyclable bimodal polyvinyl alcohol/PEDOT:PSS hydrogel sensors for highly sensitive strain and temperature sensing. ACS Appl. Mater. Interfaces 16, 32466–32480 (2024).
Freedman, B. R. et al. Enhanced tendon healing by a tough hydrogel with an adhesive side and high drug-loading capacity. Nat. Biomed. Eng. 6, 1167–1179 (2022).
Yang, Y. et al. Improved pharmacodynamics of epidermal growth factor via microneedles-based self-powered transcutaneous electrical stimulation. Nat. Commun. 13, 6908 (2022).
Xin, J., Gao, L. & Wei, L. Codes for wound analysis training. DR-NTU https://doi.org/10.21979/N9/ELSU4C (2024).
Taheri, A., MacFarlane, D. R., Pozo-Gonzalo, C. & Pringle, J. M. Quasi-solid-state electrolytes for low-grade thermal energy harvesting using a cobalt redox couple. ChemSusChem 11, 2788–2796 (2018).
Yang, P. et al. Wearable thermocells based on gel electrolytes for the utilization of body heat. Angew. Chem. Int. Ed. 55, 12050–12053 (2016).
Liu, Y. et al. Advanced wearable thermocells for body heat harvesting. Adv. Energy Mater. 10, 2002539 (2020).
Jin, L., Greene, G. W., MacFarlane, D. R. & Pringle, J. M. Redox-active quasi-solid-state electrolytes for thermal energy harvesting. ACS Energy Lett. 1, 654–658 (2016).
Tian, C., Khan, S. A., Zhang, Z., Cui, X. & Zhang, H. Thermoelectric hydrogel electronic skin for passive multimodal physiological perception. ACS Sens. 9, 840–848 (2024).
Bai, C. et al. Wearable electronics based on the gel thermogalvanic electrolyte for self-powered human health monitoring. ACS Appl. Mater. Interfaces 13, 37316–37322 (2021).
Li, X. et al. Self-powered respiratory monitoring strategy based on adaptive dual-network thermogalvanic hydrogels. ACS Appl. Mater. Interfaces 14, 48743–48751 (2022).
Zhang, D. et al. Highly antifreezing thermogalvanic hydrogels for human heat harvesting in ultralow temperature environments. Nano Lett. 23, 11272–11279 (2023).
Acknowledgements
This work was supported by the Singapore Ministry of Education Academic Research Fund Tier 2 (MOE2019-T2-2-127 and MOE-T2EP50120-0002), the Singapore Ministry of Education Academic Research Fund Tier 1 (RG62/22), A*STAR under AME IRG (A2083c0062), and A*STAR under IAF-ICP Programme I2001E0067 and the Schaeffler Hub for Advanced Research at NTU. This work was supported by the IDMxS (Institute for Digital Molecular Analytics and Science) by the Singapore Ministry of Education under the Research Centres of Excellence scheme. This work was supported by the China Postdoctoral Science Foundation (2023M742317 and GZB20240446), Shanghai Municipal Human Resources and Social Security Bureau Postdoctoral Incentive Program (2023478). We also thank Y. Wang from UCLA for his help and discussions. This work was also supported by the NTU-PSL Joint Lab collaboration.
Author information
Authors and Affiliations
Contributions
J.X., L.G. and L. Wei designed the study. J.X. and L.G. conceived the idea. J.X., L.G. and L. Wang developed the materials and methodology for the TGC dressing. J.X. and S.Y. designed and performed the numerical modelling and analysis. J.X., L.G., Wenrui Li, Zhixun Wang, Zhe Wang, B.H., S.W., T.X., T.Z., Y.L. and Wulong Li performed the material characterizations. L.G., Y.Y., X.S. and W.Z. performed the in vivo experimental analysis. J.X., X.Z. and H.Z. performed the integrated circuit design and test. S.C.J.L. participated in the discussions. J.X., L.G. and L. Wei wrote the paper with contributions from all authors.
Corresponding author
Ethics declarations
Competing interests
J.X., L.G. and L.W. have submitted a provisional patent application through Nanyang Technological University and have been assigned PCT patent application number PCT/SG2024/050745. The other authors declare no competing interests.
Peer review
Peer review information
Nature Biomedical Engineering thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Notes 1–3, Figs. 1–36, Tables 1 and 2, and data and video captions.
Supplementary Data 1 (download XLSX )
Complete list of differentially expressed genes from RNA-seq data.
Supplementary Video 1 (download MP4 )
Simulation of the temperature distribution, potential and electric field distribution for the TGC/skin wound model.
Supplementary Video 2 (download MP4 )
Real-time wound monitoring interface for responding to the acute wound model.
Supplementary Video 3 (download MP4 )
Real-time wound monitoring interface for responding to the chronic wound model.
Supplementary Video 4 (download MP4 )
Real-time wound monitoring interface for responding to the recurrent infection model.
Supplementary Video 5 (download MP4 )
Real-time wound monitoring interface for reflecting the exudate.
Supplementary Video 6 (download MP4 )
TGC dressing for electrically stimulated healing of large acute wounds and respiratory monitoring of pigs.
Source data
Source Data Fig. 2 (download XLSX )
Source data.
Source Data Fig. 3 (download XLSX )
Source data.
Source Data Fig. 4 (download XLSX )
Source data.
Source Data Fig. 5 (download XLSX )
Source data.
Source Data Fig. 6 (download XLSX )
Source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xin, J., Gao, L., Zhang, W. et al. A thermogalvanic cell dressing for smart wound monitoring and accelerated healing. Nat. Biomed. Eng 10, 80–93 (2026). https://doi.org/10.1038/s41551-025-01440-6
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41551-025-01440-6