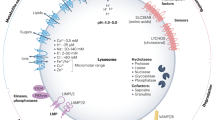
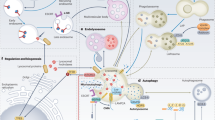
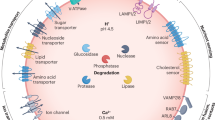
Abstract
Polymers are endocytosed and hydrolysed by lysosomal enzymes to generate transportable solutes. While the transport of diverse organic solutes across the plasma membrane is well studied, their necessary ongoing efflux from the endocytic fluid into the cytosol is poorly appreciated by comparison. Myeloid cells that employ specialized types of endocytosis, that is, phagocytosis and macropinocytosis, are highly dependent on such transport pathways to prevent the build-up of hydrostatic pressure that otherwise offsets lysosomal dynamics including vesiculation, tubulation and fission. Without undergoing rupture, we found that lysosomes incurring this pressure owing to defects in solute efflux, are unable to retain luminal Na+, which collapses its gradient with the cytosol. This cation ‘leak’ is mediated by pressure-sensitive channels resident to lysosomes and leads to the inhibition of mTORC1, which is normally activated by Na+-coupled amino acid transporters driven by the Na+ gradient. As a consequence, the transcription factors TFEB/TFE3 are made active in macrophages with distended lysosomes. In addition to their role in lysosomal biogenesis, TFEB/TFE3 activation causes the release of MCP-1/CCL2. In catabolically stressed tissues, defects in efflux of solutes from the endocytic pathway leads to increased monocyte recruitment. Here we propose that macrophages respond to a pressure-sensing pathway on lysosomes to orchestrate lysosomal biogenesis as well as myeloid cell recruitment.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
Mass spectrometry data have been deposited in the MassIVE database as MSV000094949 (https://massive.ucsd.edu/ProteoSAFe/dataset.jsp?task=327454621fae453d9df1e5e21bfa972e). Source data are provided with this paper. All other data supporting the findings of this study are available from the corresponding author on reasonable request.
References
Davies, L. C., Jenkins, S. J., Allen, J. E. & Taylor, P. R. Tissue-resident macrophages. Nat. Immunol. 14, 986–995 (2013).
Haldar, M. et al. Heme-mediated SPI-C induction promotes monocyte differentiation into iron-recycling macrophages. Cell 156, 1223–1234 (2014).
Kohyama, M. et al. Role for Spi-C in the development of red pulp macrophages and splenic iron homeostasis. Nature 457, 318–321 (2009).
Manwani, D. & Bieker, J. J. The erythroblastic island. Curr. Top. Dev. Biol. 82, 23–53 (2008).
Freeman, S. A. et al. Lipid-gated monovalent ion fluxes regulate endocytic traffic and support immune surveillance. Science 367, 301–305 (2020).
Saric, A. & Freeman, S. A. Endomembrane tension and trafficking. Front. Cell Dev. Biol. 8, 611326 (2020).
Zeziulia, M., Blin, S., Schmitt, F. W., Lehmann, M. & Jentsch, T. J. Proton-gated anion transport governs macropinosome shrinkage. Nat. Cell Biol. 24, 885–895 (2022).
Mercier, V. et al. Endosomal membrane tension regulates ESCRT-III-dependent intra-lumenal vesicle formation. Nat. Cell Biol. 22, 947–959 (2020).
Swanson, J., Bushnell, A. & Silverstein, S. C. Tubular lysosome morphology and distribution within macrophages depend on the integrity of cytoplasmic microtubules. Proc. Natl Acad. Sci. USA 84, 1921–1925 (1987).
Saric, A. et al. mTOR controls lysosome tubulation and antigen presentation in macrophages and dendritic cells. Mol. Biol. Cell 27, 321–333 (2016).
Chow, A., Toomre, D., Garrett, W. & Mellman, I. Dendritic cell maturation triggers retrograde MHC class II transport from lysosomes to the plasma membrane. Nature 418, 988–994 (2002).
Nakamura, N. et al. Endosomes are specialized platforms for bacterial sensing and NOD2 signalling. Nature 509, 240–244 (2014).
Steinman, R. M., Brodie, S. E. & Cohn, Z. A. Membrane flow during pinocytosis. A stereologic analysis. J. Cell Biol. 68, 665–687 (1976).
Chen, C. C., Krogsaeter, E. & Grimm, C. Two-pore and TRP cation channels in endolysosomal osmo-/mechanosensation and volume regulation. Biochim. Biophys. Acta Mol. Cell. Res. 1868, 118921 (2021).
Chen, C. C. et al. TRPML2 is an osmo/mechanosensitive cation channel in endolysosomal organelles. Sci. Adv. 6, eabb5064 (2020).
Skowyra, M. L., Schlesinger, P. H., Naismith, T. V. & Hanson, P. I. Triggered recruitment of ESCRT machinery promotes endolysosomal repair. Science 360, eaar5078 (2018).
Colom, A. et al. A fluorescent membrane tension probe. Nat. Chem. 10, 1118–1125 (2018).
Bright, N. A., Davis, L. J. & Luzio, J. P. Endolysosomes are the principal intracellular sites of acid hydrolase activity. Curr. Biol. 26, 2233–2245 (2016).
Bosch, M. E. & Kielian, T. Neuroinflammatory paradigms in lysosomal storage diseases. Front. Neurosci. 9, 417 (2015).
Alayoubi, A. M. et al. Systemic ceramide accumulation leads to severe and varied pathological consequences. EMBO Mol. Med. 5, 827–842 (2013).
DeCourcy, K. & Storrie, B. Osmotic swelling of endocytic compartments induced by internalized sucrose is restricted to mature lysosomes in cultured mammalian cells. Exp. Cell. Res. 192, 52–60 (1991).
Cohn, Z. A. & Ehrenreich, B. A. The uptake, storage, and intracellular hydrolysis of carbohydrates by macrophages. J. Exp. Med. 129, 201–225 (1969).
Rilla, K. et al. Plasma membrane residence of hyaluronan synthase is coupled to its enzymatic activity. J. Biol. Chem. 280, 31890–31897 (2005).
Freeman, S. A. et al. Transmembrane pickets connect cyto- and pericellular skeletons forming barriers to receptor engagement. Cell 172, 305–317 e310 (2018).
Cowman, M. K., Lee, H. G., Schwertfeger, K. L., McCarthy, J. B. & Turley, E. A. The content and size of hyaluronan in biological fluids and tissues. Front. Immunol. 6, 261 (2015).
Lee-Sayer, S. S. et al. The where, when, how, and why of hyaluronan binding by immune cells. Front. Immunol. 6, 150 (2015).
Dong, Y. et al. Endotoxin free hyaluronan and hyaluronan fragments do not stimulate TNF-alpha, interleukin-12 or upregulate co-stimulatory molecules in dendritic cells or macrophages. Sci. Rep. 6, 36928 (2016).
McKee, C. M. et al. Hyaluronan (HA) fragments induce chemokine gene expression in alveolar macrophages. The role of HA size and CD44. J. Clin. Invest. 98, 2403–2413 (1996).
Noble, P. W., McKee, C. M., Cowman, M. & Shin, H. S. Hyaluronan fragments activate an NF-kappa B/I-kappa B alpha autoregulatory loop in murine macrophages. J. Exp. Med. 183, 2373–2378 (1996).
Reimer, R. J. SLC17: a functionally diverse family of organic anion transporters. Mol. Asp. Med. 34, 350–359 (2013).
Dillon, S. et al. Yeast zymosan, a stimulus for TLR2 and dectin-1, induces regulatory antigen-presenting cells and immunological tolerance. J. Clin. Invest. 116, 916–928 (2006).
Gantner, B. N., Simmons, R. M., Canavera, S. J., Akira, S. & Underhill, D. M. Collaborative induction of inflammatory responses by dectin-1 and Toll-like receptor 2. J. Exp. Med. 197, 1107–1117 (2003).
Levin-Konigsberg, R. & Grinstein, S. Phagosome–endoplasmic reticulum contacts: kissing and not running. Traffic 21, 172–180 (2020).
Aguilar-Uscanga, B. & Francois, J. M. A study of the yeast cell wall composition and structure in response to growth conditions and mode of cultivation. Lett. Appl. Microbiol. 37, 268–274 (2003).
Aula, N. et al. The spectrum of SLC17A5-gene mutations resulting in free sialic acid-storage diseases indicates some genotype-phenotype correlation. Am. J. Hum. Genet 67, 832–840 (2000).
Verheijen, F. W. et al. A new gene, encoding an anion transporter, is mutated in sialic acid storage diseases. Nat. Genet. 23, 462–465 (1999).
Robak, L. A. et al. Excessive burden of lysosomal storage disorder gene variants in Parkinson’s disease. Brain 140, 3191–3203 (2017).
Prolo, L. M., Vogel, H. & Reimer, R. J. The lysosomal sialic acid transporter sialin is required for normal CNS myelination. J. Neurosci. 29, 15355–15365 (2009).
Lee, Y. S. et al. CX(3)CR1 differentiates F4/80(low) monocytes into pro-inflammatory F4/80(high) macrophages in the liver. Sci. Rep. 8, 15076 (2018).
Sardiello, M. et al. A gene network regulating lysosomal biogenesis and function. Science 325, 473–477 (2009).
Rusmini, P. et al. Trehalose induces autophagy via lysosomal-mediated TFEB activation in models of motoneuron degeneration. Autophagy 15, 631–651 (2019).
Pastore, N. et al. TFEB and TFE3 cooperate in the regulation of the innate immune response in activated macrophages. Autophagy 12, 1240–1258 (2016).
Wang, X. et al. TPC proteins are phosphoinositide- activated sodium-selective ion channels in endosomes and lysosomes. Cell 151, 372–383 (2012).
Zou, J. et al. A DNA nanodevice for mapping sodium at single-organelle resolution. Nat. Biotechnol. 10.1038/s41587-023-01950-1 (2023).
Saito, M., Hanson, P. I. & Schlesinger, P. Luminal chloride-dependent activation of endosome calcium channels: patch clamp study of enlarged endosomes. J. Biol. Chem. 282, 27327–27333 (2007).
Radin, I. et al. Plant PIEZO homologs modulate vacuole morphology during tip growth. Science 373, 586–590 (2021).
Li, K. et al. Drosophila TMEM63 and mouse TMEM63A are lysosomal mechanosensory ion channels. Nat. Cell Biol. 26, 393–403 (2024).
Murthy, S. E. et al. OSCA/TMEM63 are an evolutionarily conserved family of mechanically activated ion channels. Elife 7, e41844 (2018).
Parenti, G., Medina, D. L. & Ballabio, A. The rapidly evolving view of lysosomal storage diseases. EMBO Mol. Med. 13, e12836 (2021).
Davis, O. B. et al. NPC1–mTORC1 signaling couples cholesterol sensing to organelle homeostasis and is a targetable pathway in Niemann–Pick type C. Dev. Cell 56, 260–276 e267 (2021).
Hasegawa, J. et al. PP2A-dependent TFEB activation is blocked by PIKfyve-induced mTORC1 activity. Mol. Biol. Cell 33, ar26 (2022).
Efeyan, A. et al. Regulation of mTORC1 by the Rag GTPases is necessary for neonatal autophagy and survival. Nature 493, 679–683 (2013).
Song, H. L., Demirev, A. V., Kim, N. Y., Kim, D. H. & Yoon, S. Y. Ouabain activates transcription factor EB and exerts neuroprotection in models of Alzheimer’s disease. Mol. Cell Neurosci. 95, 13–24 (2019).
Rebsamen, M. et al. SLC38A9 is a component of the lysosomal amino acid sensing machinery that controls mTORC1. Nature 519, 477–481 (2015).
Verdon, Q. et al. SNAT7 is the primary lysosomal glutamine exporter required for extracellular protein-dependent growth of cancer cells. Proc. Natl Acad. Sci. USA 114, E3602–E3611 (2017).
Wyant, G. A. et al. mTORC1 activator SLC38A9 is required to efflux essential amino acids from lysosomes and use protein as a nutrient. Cell 171, 642–654 e612 (2017).
Zhou, X. et al. Circuit design features of a stable two-cell system. Cell 172, 744–757 e717 (2018).
Medina, D. L. et al. Lysosomal calcium signalling regulates autophagy through calcineurin and TFEB. Nat. Cell Biol. 17, 288–299 (2015).
Sava, I., Davis, L. J., Gray, S. R., Bright, N. A. & Luzio, J. P. Reversible assembly and disassembly of V-ATPase during the lysosome regeneration cycle. Mol. Biol. Cell 35, ar63 (2024).
Durgan, J. & Florey, O. Many roads lead to CASM: diverse stimuli of noncanonical autophagy share a unifying molecular mechanism. Sci. Adv. 8, eabo1274 (2022).
Xu, H. & Ren, D. Lysosomal physiology. Annu. Rev. Physiol. 77, 57–80 (2015).
Choy, C. H. et al. Lysosome enlargement during inhibition of the lipid kinase PIKfyve proceeds through lysosome coalescence. J. Cell Sci. 131, jcs213587 (2018).
Wang, W. et al. Up-regulation of lysosomal TRPML1 channels is essential for lysosomal adaptation to nutrient starvation. Proc. Natl Acad. Sci. USA 112, E1373–E1381 (2015).
Bussi, C. et al. Lysosomal damage drives mitochondrial proteome remodelling and reprograms macrophage immunometabolism. Nat. Commun. 13, 7338 (2022).
Westman, J. et al. Lysosome fusion maintains phagosome integrity during fungal infection. Cell Host Microbe 28, 798–812 e796 (2020).
Hornung, V. et al. Silica crystals and aluminum salts activate the NALP3 inflammasome through phagosomal destabilization. Nat. Immunol. 9, 847–856 (2008).
Ono, K., Kim, S. O. & Han, J. Susceptibility of lysosomes to rupture is a determinant for plasma membrane disruption in tumor necrosis factor alpha-induced cell death. Mol. Cell. Biol. 23, 665–676 (2003).
Bandyopadhyay, D., Cyphersmith, A., Zapata, J. A., Kim, Y. J. & Payne, C. K. Lysosome transport as a function of lysosome diameter. PLoS ONE 9, e86847 (2014).
Saric, A. et al. SNX19 restricts endolysosome motility through contacts with the endoplasmic reticulum. Nat. Commun. 12, 4552 (2021).
Li, X. et al. A molecular mechanism to regulate lysosome motility for lysosome positioning and tubulation. Nat. Cell Biol. 18, 404–417 (2016).
Uusi-Rauva, K. et al. Neuronal ceroid lipofuscinosis protein CLN3 interacts with motor proteins and modifies location of late endosomal compartments. Cell. Mol. Life Sci. 69, 2075–2089 (2012).
Johnson, D. E., Ostrowski, P., Jaumouille, V. & Grinstein, S. The position of lysosomes within the cell determines their luminal pH. J. Cell Biol. 212, 677–692 (2016).
Freeman, S. A. & Grinstein, S. Resolution of macropinosomes, phagosomes and autolysosomes: Osmotically driven shrinkage enables tubulation and vesiculation. Traffic 19, 965–974 (2018).
Suresh, B. et al. Tubular lysosomes harbor active ion gradients and poise macrophages for phagocytosis. Proc. Natl Acad. Sci. USA 118, e2113174118 (2021).
Moreland, L. W. Intra-articular hyaluronan (hyaluronic acid) and hylans for the treatment of osteoarthritis: mechanisms of action. Arthritis Res. Ther. 5, 54–67 (2003).
Petrey, A. C. & de la Motte, C. A. Hyaluronan, a crucial regulator of inflammation. Front. Immunol. 5, 101 (2014).
Romo, M. et al. Small fragments of hyaluronan are increased in individuals with obesity and contribute to low-grade inflammation through TLR-mediated activation of innate immune cells. Int J. Obes. 46, 1960–1969 (2022).
Triggs-Raine, B., Salo, T. J., Zhang, H., Wicklow, B. A. & Natowicz, M. R. Mutations in HYAL1, a member of a tandemly distributed multigene family encoding disparate hyaluronidase activities, cause a newly described lysosomal disorder, mucopolysaccharidosis IX. Proc. Natl Acad. Sci. USA 96, 6296–6300 (1999).
Alessi, D. R. & Sammler, E. LRRK2 kinase in Parkinson’s disease. Science 360, 36–37 (2018).
Cookson, M. R. The role of leucine-rich repeat kinase 2 (LRRK2) in Parkinson’s disease. Nat. Rev. Neurosci. 11, 791–797 (2010).
Langston, R. G. et al. Association of a common genetic variant with Parkinson’s disease is mediated by microglia. Sci. Transl. Med. 14, eabp8869 (2022).
Santaella, A. et al. Cerebrospinal fluid monocyte chemoattractant protein 1 correlates with progression of Parkinson’s disease. NPJ Parkinsons Dis. 6, 21 (2020).
Russo, I. et al. Transcriptome analysis of LRRK2 knock-out microglia cells reveals alterations of inflammatory- and oxidative stress-related pathways upon treatment with alpha-synuclein fibrils. Neurobiol. Dis. 129, 67–78 (2019).
Liao, Y. C. et al. RNA granules hitchhike on lysosomes for long-distance transport, using annexin A11 as a molecular tether. Cell 179, 147–164 e120 (2019).
Belkacemi, A. et al. The TRPV2 channel mediates Ca2+ influx and the Delta9-THC-dependent decrease in osmotic fragility in red blood cells. Haematologica 106, 2246–2250 (2021).
Colucci, A. M., Campana, M. C., Bellopede, M. & Bucci, C. The Rab-interacting lysosomal protein, a Rab7 and Rab34 effector, is capable of self-interaction. Biochem. Biophys. Res. Commun. 334, 128–133 (2005).
Harrison, R. E., Bucci, C., Vieira, O. V., Schroer, T. A. & Grinstein, S. Phagosomes fuse with late endosomes and/or lysosomes by extension of membrane protrusions along microtubules: role of Rab7 and RILP. Mol. Cell. Biol. 23, 6494–6506 (2003).
Kaniuk, N. A. et al. Salmonella exploits Arl8B-directed kinesin activity to promote endosome tubulation and cell-to-cell transfer. Cell Microbiol 13, 1812–1823 (2011).
Keren-Kaplan, T. et al. RUFY3 and RUFY4 are ARL8 effectors that promote coupling of endolysosomes to dynein–dynactin. Nat. Commun. 13, 1506 (2022).
Acknowledgements
We thank A. Belkacemi and V. Flockerzi for helpful discussions and Y. Li and B. Fubara at the NINDS Proteomics Core Facility for technical assistance. S.A.F. and R.J.B. are recipients of Canada Research Chairs. S.A.F. was supported by grants PJT-169180 and PJT-190244 from the Canadian Institutes of Health Research (CIHR). R.C. is the recipient of a CIHR fellowship. S.U. is supported by Deutsche Forschungsgemeinschaft (DFG) grants (448121430, 405969122 and 447268119), the Hightech Agenda Bavaria and by an ERC starting grant (101039438). R.J.B. is supported by CIHR PJT-166047 and research support by Toronto Metropolitan University.
Author information
Authors and Affiliations
Contributions
R.C., A.S., S.U. and S.A.F. designed the study. B.A.S., R.J.B., A.S., S.U. and S.A.F. supervised the experiments. R.C., O.S., G.Y., T.L., E.S., W.K., S.I., A.S., S.U. and S.A.F. performed the experiments. R.C. and S.A.F. wrote the paper; all authors reviewed and edited the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Cell Biology thanks the anonymous reviewers for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 The slow accumulation of solutes in lysosomes impairs their tubulation and motility without rupture.
a) BMDM pulsed for 4 h with 3 kDa TMR-Dextran and chased for >4 h in normal medium then given a hypotonic solution for 5 min. b-c) BMDM control or those treated with 200 μM GPN for 10 min were evaluated for their tubulation and scored. > 200 cells. d-f) BMDM as in a, with or without 20 mM sucrose or trehalose overnight, then chased for >2 h followed by incubation with 0.5 mg/mL invertase for 30 min. Lysosome volume determined using confocal microscopy. Cell volume for >10000 cells per n determined by Coulter Counter. In e, p = 2.8 × 10−18 and 2.6 × 10−20. In f, p = 1.1 × 10−10. n = 4 independent experiments. g-j) RAW264.7 cells incubated with 20 mM sucrose for indicated times or 5 mM LLOMe for 15 min, pulsed for 5 min with lysotracker red and imaged live or fixed and immunostained as indicated. In h, the lysotracker intensity was determined for individual fields of >5 cells, p = 4.0 × 10−9. n = 3 independent experiments. For i and j, quantification of puncta is graphed in Fig. 1. k-m) HeLa cells expressing tubulin-GFP and LAMP-1-RFP. LAMP-1-RFP compartments were tracked over 60 s and analyzed for motion type (see Methods). Cells were then given 200 μM GPN for 10 min and LAMP-1-RFP compartments were tracked again. The % of tracks identified as being directed in their motion (c) and the diffusion coefficient (d) is shown for >20 cells, >1000 lysosomes. Data are presented as mean values ± SD (c, e, f, h, l, m). ns, not significant. For panels c, e, f, h, l, and m, the statistical significance was determined by two-sided Student’s t-test. Source numerical data are available in source data. n = 3 independent experiments.
Extended Data Fig. 2 Organic solutes are taken up by macropinocytosis and fluxed into the cytosol.
a) 10 μM 2-NBDG and 70 kDa TMR-dextran given to BMDM for 10 min before staining their surface with AlexaFluor647-wheat germ agglutin for 1 min to indicate the plasma membrane (PM). b) 10 μM 2-NBDG and FM4-64 given to BMDM for 10 min before washing 5 times and imaging live. c) TMR-High molecular weight HA was treated with 1 units/mL hyaluronidase for 30 min then incubated with BMDM together with 70 kDa FITC-dextran for 20 min. n = 3 independent experiments. d) Cells treated as in a, but chased for 5 min and then imaged. The fluorescence intensity of the 2-NBDG compared to the TMR-dextran for individual macropinosomes is shown. >100 macropinosomes. e) BMDM pulsed with 10 μM 2-NBDG for 10 min imaged immediately or after 15 min chase. f) RAW264.7 cells pulsed with 10 μM 2-NBDG and 70 kDa TMR-dextran for 10 min imaged immediately (0 min) or after 15 min chase before staining their surface with AlexaFluor647-WGA. g) BMDM incubated with 70 kDa dextran together with fragmented TMR-hyaluronan for 10 min.
Extended Data Fig. 3 GLUT localization in macrophages.
a) QPCR. n = 3 independent experiments. Data are presented as mean values ± SD. b) BMDM incubated with 3 kDa TMR-dextran and 20 mM sucrose overnight, then chased for > 2 h followed by incubation with 0.5 mg/mL invertase for 30 min ± BAY876 (10 μM). Quantitation as in b. p = 2.6 × 10−10. n = 3 independent experiments. Data are presented as mean values ± SD. c) Western blotting of BMDM with or without fragmented HA overnight. d) immunostaining of BMDM. e) Lysosomes of indicated cells for 1 h ± BAY876 (10 μM). f) RAW264.7 macrophages expressing SLC2A6-GFP (cyan) with or without LAMP1-RFP challenged with 20 mM sucrose overnight or C. albicans expressing RFP. g-h) BMDM challenged with heat-killed TMR-conjugated C. albicans for 6 h and 0.5 mg/mL zymolase for the last 3 h. The % of phagosomes undergoing tubulation is graphed in h. Each dot represents one field, >15 fields, p = 3.0 × 10−7, n = 3 independent experiments. Data are presented as mean values ± SD (a, h). For panels a, b, and h, the statistical significance was determined by two-sided Student’s t-test. Source numerical data and unprocessed blots are available in source data.
Extended Data Fig. 4 Solute accumulation in lysosomes activates TFEB dependent on TMEM63A.
a) MCP-1 immunostaining of liver tissues in WT and SLC17A5−/− mice. Scale bar, 20 μm. b) MCP-1 collected from culture medium of indicated BMDM with or without 20 mM sucrose overnight. n = 3 independent experiments. c) TFEBexplorer software (https://tfeb.tigem.it) to determine coordinated lysosomal expression and regulation (CLEAR) elements upstream of the MCP-1/CCL2 promotor region. d) Western blotting as indicated. e) WT and SLC17A5 KO RAW264.7 cells immunostained for galectin-3 or CHMP-3. The number of puncta per cell (numbers indicate individual cells) was quantified. n = 3 independent experiments. f) Example images of TFE3 and DAPI in BMDM with or without 20 mM sucrose overnight followed by the TRPML antagonist ML-SI3 for 2 h. g) QPCR for TMEM63A, n = 3 independent experiments. h-i) WT or SLC17A5 KO RAW264.7 cells expressing scramble or siRNA against TMEM63A, challenged with 20 mM sucrose overnight and stained for TFE3 and quantified. Individual cells are plotted. In h, p = 5.5 × 10−33. In i, p = 4.0 × 10−44. n = 3 independent experiments. j-k) Re-expression of TMEM63A-OFP in TMEM63A KO cells. Individual cells quantified in k (dots). p = 5.2 × 10−33. n = 3 independent experiments. l) Lysosomes from indicated RAW264.7 cells isolated using magnetic nanoparticles. [Na+] was determined using atomic absorption spectroscopy (AAS) and normalized to LAMP-1 protein from the isolates (below). p = 3.5 × 10−4. n = 3 independent experiments. m-n) Lysosomal Natrium Green/cresyl violet with sucrose for indicated cells. Dots represent individual cells for >150 cells. p = 1.3 × 10−36. n = 3 independent experiments. o-q) RAW264.7 cells loaded with sucrose overnight, dialyzed, and analyzed by AAS. p = 6.6 × 10−5 (o), p = 2.4 × 10−4 (p), p = 3.5 × 10−2 (q). n = 3 independent experiments. r) ELISA. n = 3 independent experiments. s) Filipin staining of U18666A-treated cells before and after 5 min shift to hypertonic medium. All scale bars unless otherwise indicated, 10 μm. Data are presented as mean values ± SD (b, e, g, h, i, k, l, n-r). For these panels, the statistical significance was determined by two-sided Student’s t-test. Source numerical data and unprocessed blots are available in source data. The data are presented as mean values ± SD for panels k-m and o-r.
Extended Data Fig. 5 Lysosomes in Salla fibroblasts are swollen and less motile.
a-c) Primary fibroblasts pulsed with 3 kDa TMR-dextran for 4 h, chased for 4 h, then imaged. In b, the volume of the lysosome compartment is determined by z stacks for single cells (dots) > 20, for 2 controls and 3 patient cultures. p = 2.3 × 10−15 (control 1 vs patient 1), p = 4.7 × 10−16 (control 2 vs patient 1), p = 1.1 × 10−14 (control 1 vs patient 2), p = 3.1 × 10−15 (control 2 vs patient 2), p = 3.3 × 10−18 (control 1 vs patient 3), p = 6.0 × 10−19 (control 2 vs patient 3). In c, the position of the lysosomes can be observed relative to the nuclei. n = 3 independent experiments. d-g) Tracking of single lysosomes. The % of tracks identified as being confined to linear trajectories (e; ****, p = 5.2 × 10−15) and directed in their motion (f; ****, p = 3.9 × 10−11) and the diffusion coefficient (c; ****, p = 1.9 × 10−11) is shown for >20 cells, >1000 lysosomes. n = 3 independent experiments. Data are presented as mean values ± SD (b, e-g). For these panels, the statistical significance was determined by two-sided Student’s t-test. Source numerical data are available in source data.
Extended Data Fig. 6 Sucrosomes recruit functional motor complexes.
a) Model of lysosomal anterograde and retrograde transport. b) iPSC-expressing LAMP-1-APEX cells were given 40 mM sucrose overnight followed by invertase treatment (‘invertase’) or no treatment (‘sucrose’), biotin-phenol was added for 30 minutes and pulsed for 30 s with hydrogen peroxide. Biotinylated proteins were isolated on Streptavidin beads and identified by mass spectrometry (see Methods). n = 3 independent experiments. c-e) HeLa cells expressing indicated fusion proteins given 3 kDa TMR-dextran overnight together with 20 mM sucrose where indicated. Cells were chased for >4 h before imaging. Invertase or hypertonic medium were added where indicated.
Supplementary information
Supplementary Video 1 (download MOV )
SLC17A5−/− and littermate control (WT) at 3.5 weeks of age.
Source data
Unprocessed western blots Fig 2 (download PDF )
Unprocessed western blots and gels.
Unprocessed western blots Fig 3 (download PDF )
Unprocessed western blots and gels.
Unprocessed western blots Fig 5 (download PDF )
Unprocessed western blots and gels.
Unprocessed western blots Fig 6 (download PDF )
Unprocessed western blots and gels.
Unprocessed western blots Fig 7 (download PDF )
Unprocessed western blots and gels.
Source Data Fig. 1 (download XLSX )
Source data for Fig. 1.
Source Data Fig. 2 (download XLSX )
Source data for Fig. 2.
Source Data Fig. 3 (download XLSX )
Source data for Fig. 3.
Source Data Fig. 4 (download XLSX )
Source data for Fig. 4.
Source Data Fig. 5 (download XLSX )
Source data for Fig. 5.
Source Data Fig. 6 (download XLSX )
Source data for Fig. 6.
Source Data Fig. 7 (download XLSX )
Source data for Fig. 7.
Source Data S Figures (download XLSX )
Source data for Extended Data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cai, R., Scott, O., Ye, G. et al. Pressure sensing of lysosomes enables control of TFEB responses in macrophages. Nat Cell Biol 26, 1247–1260 (2024). https://doi.org/10.1038/s41556-024-01459-y
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41556-024-01459-y
This article is cited by
-
p38 mitogen-activated protein kinase drives senescence in CD4+ T lymphocytes and increases their pathological potential
Immunity & Ageing (2025)
-
The spectrum of lysosomal stress and damage responses: from mechanosensing to inflammation
EMBO Reports (2025)
-
Structural basis for mTORC1 activation on the lysosomal membrane
Nature (2025)
-
Lysosome heterogeneity and diversity mapped through its distinct cellular functions
Cellular and Molecular Life Sciences (2025)