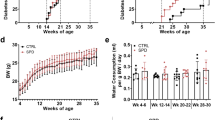
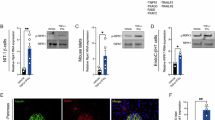
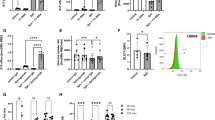
Abstract
It has been established that N-acetyltransferase (murine NAT1 (mNAT1) and human NAT2 (hNAT2)) mediates insulin sensitivity in type 2 diabetes. Here we show that mNAT1 deficiency leads to a decrease in cellular spermidine—a natural polyamine exhibiting health-protective and anti-ageing effects—but understanding of its mechanism is limited. We identify that mNAT1 and hNAT2 modulate a type of post-translational modification involving acetylated spermidine, which we name acetylhypusination, on receptor-interacting serine/threonine-protein kinase 1 (RIPK1)—a key regulator of inflammation and cell death. Spermidine supplementation decreases RIPK1-mediated cell death and diabetic phenotypes induced by NAT1 deficiency in vivo. Furthermore, insulin resistance and diabetic kidney disease mediated by vascular pathology in NAT1-deficient mice can be blocked by inhibiting RIPK1. Finally, we demonstrate a decrease in spermidine and activation of RIPK1 in the vascular tissues of human patients with diabetes. Our study suggests a role for vascular pathology in diabetes onset and progression and identifies the inhibition of RIPK1 kinase as a potential therapeutic approach for the treatment of type 2 diabetes.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium (http://proteomecentral.proteomexchange.org) via the iProX partner repository63,64 (https://www.iprox.cn//page/SCV017.html?query=PXD047079) with the dataset identifier PXD047079. The raw LC−MS-based untargeted metabolomics data files generated in this study have been deposited in the National Omics Data Encyclopedia under accession code OEP004847 and are publicly available. Source data are provided with this paper. All of the other data supporting the findings of this study are available from the corresponding author upon reasonable request.
References
Ogurtsova, K. et al. IDF Diabetes Atlas: global estimates of undiagnosed diabetes in adults for 2021. Diabetes Res. Clin. Pract. 183, 109118 (2022).
Knowles, J. W. et al. Identification and validation of N-acetyltransferase 2 as an insulin sensitivity gene. J. Clin. Invest. 125, 1739–1751 (2015).
Chennamsetty, I. et al. Nat1 deficiency is associated with mitochondrial dysfunction and exercise intolerance in mice. Cell Rep. 17, 527–540 (2016).
Camporez, J. P. et al. Mechanism by which arylamine N-acetyltransferase 1 ablation causes insulin resistance in mice. Proc. Natl Acad. Sci. USA 114, E11285–E11292 (2017).
Zou, C. et al. Reduction of mNAT1/hNAT2 contributes to cerebral endothelial necroptosis and Aβ accumulation in Alzheimer’s disease. Cell Rep. 33, 108447 (2020).
Conway, L. P. et al. Unexpected acetylation of endogenous aliphatic amines by arylamine N-acetyltransferase NAT2. Angew. Chem. Int. Ed. Engl. 59, 14342–14346 (2020).
Hein, D. W. & Millner, L. M. Arylamine N-acetyltransferase acetylation polymorphisms: paradigm for pharmacogenomic-guided therapy—a focused review. Expert Opin. Drug Metab. Toxicol. 17, 9–21 (2021).
Pegg, A. E. Recent advances in the biochemistry of polyamines in eukaryotes. Biochem. J. 234, 249–262 (1986).
Scalabrino, G. & Ferioli, M. E. Polyamines in mammalian ageing: an oncological problem, too? A review. Mech. Ageing Dev. 26, 149–164 (1984).
Hofer, S. J. et al. Spermidine-induced hypusination preserves mitochondrial and cognitive function during aging. Autophagy 17, 2037–2039 (2021).
Eisenberg, T. et al. Induction of autophagy by spermidine promotes longevity. Nat. Cell Biol. 11, 1305–1314 (2009).
Partridge, L., Fuentealba, M. & Kennedy, B. K. The quest to slow ageing through drug discovery. Nat. Rev. Drug Discov. 19, 513–532 (2020).
Liang, Y. et al. eIF5A hypusination, boosted by dietary spermidine, protects from premature brain aging and mitochondrial dysfunction. Cell Rep. 35, 108941 (2021).
Ma, L. et al. Spermidine improves gut barrier integrity and gut microbiota function in diet-induced obese mice. Gut Microbes 12, 1–19 (2020).
Eisenberg, T. et al. Cardioprotection and lifespan extension by the natural polyamine spermidine. Nat. Med. 22, 1428–1438 (2016).
Chen, Z. et al. Spermidine activates RIP1 deubiquitination to inhibit TNF-α-induced NF-κB/p65 signaling pathway in osteoarthritis. Cell Death Dis. 11, 503 (2020).
Park, M. H., Cooper, H. L. & Folk, J. E. Identification of hypusine, an unusual amino acid, in a protein from human lymphocytes and of spermidine as its biosynthetic precursor. Proc. Natl Acad. Sci. USA 78, 2869–2873 (1981).
Tauc, M. et al. The eukaryotic initiation factor 5A (eIF5A1), the molecule, mechanisms and recent insights into the pathophysiological roles. Cell Biosci. 11, 219 (2021).
Shan, B., Pan, H., Najafov, A. & Yuan, J. Necroptosis in development and diseases. Genes Dev. 32, 327–340 (2018).
Mifflin, L., Ofengeim, D. & Yuan, J. Receptor-interacting protein kinase 1 (RIPK1) as a therapeutic target. Nat. Rev. Drug Discov. 19, 553–571 (2020).
Zhu, K. et al. Necroptosis promotes cell-autonomous activation of proinflammatory cytokine gene expression. Cell Death Dis. 9, 500 (2018).
Li, W. et al. Nuclear RIPK1 promotes chromatin remodeling to mediate inflammatory response. Cell Res. 32, 621–637 (2022).
Al-Habsi, M. et al. Spermidine activates mitochondrial trifunctional protein and improves antitumor immunity in mice. Science 378, eabj3510 (2022).
Casero, R. A. Jr., Murray Stewart, T. & Pegg, A. E. Polyamine metabolism and cancer: treatments, challenges and opportunities. Nat. Rev. Cancer 18, 681–695 (2018).
Degterev, A. et al. Identification of RIP1 kinase as a specific cellular target of necrostatins. Nat. Chem. Biol. 4, 313–321 (2008).
Meng, H. et al. Discovery of a cooperative mode of inhibiting RIPK1 kinase. Cell Discov. 7, 41 (2021).
Ofengeim, D. et al. Activation of necroptosis in multiple sclerosis. Cell Rep. 10, 1836–1849 (2015).
He, S. et al. Receptor interacting protein kinase-3 determines cellular necrotic response to TNF-α. Cell 137, 1100–1111 (2009).
Sun, L. et al. Mixed lineage kinase domain-like protein mediates necrosis signaling downstream of RIP3 kinase. Cell 148, 213–227 (2012).
Duprez, L. et al. RIP kinase-dependent necrosis drives lethal systemic inflammatory response syndrome. Immunity 35, 908–918 (2011).
Chandler, C. S. & Ballard, F. J. Multiple biotin-containing proteins in 3T3-L1 cells. Biochem. J. 237, 123–130 (1986).
Kirkeby, S., Moe, D., Bog-Hansen, T. C. & van Noorden, C. J. Biotin carboxylases in mitochondria and the cytosol from skeletal and cardiac muscle as detected by avidin binding. Histochemistry 100, 415–421 (1993).
Joe, Y. A., Wolff, E. C. & Park, M. H. Cloning and expression of human deoxyhypusine synthase cDNA. Structure–function studies with the recombinant enzyme and mutant proteins. J. Biol. Chem. 270, 22386–22392 (1995).
Lee, Y., Kim, H. K., Park, H. E., Park, M. H. & Joe, Y. A. Effect of N1-guanyl-1,7-diaminoheptane, an inhibitor of deoxyhypusine synthase, on endothelial cell growth, differentiation and apoptosis. Mol. Cell. Biochem. 237, 69–76 (2002).
Wolff, E. C., Lee, S. B. & Park, M. H. Assay of deoxyhypusine synthase activity. Methods Mol. Biol. 720, 195–205 (2011).
Yi, C. H. et al. Metabolic regulation of protein N-alpha-acetylation by Bcl-xL promotes cell survival. Cell 146, 607–620 (2011).
Degterev, A. et al. Chemical inhibitor of nonapoptotic cell death with therapeutic potential for ischemic brain injury. Nat. Chem. Biol. 1, 112–119 (2005).
Xie, T. et al. Structural basis of RIP1 inhibition by necrostatins. Structure 21, 493–499 (2013).
Tan, S. et al. Hepatocyte-specific TAK1 deficiency drives RIPK1 kinase-dependent inflammation to promote liver fibrosis and hepatocellular carcinoma. Proc. Natl Acad. Sci. USA 117, 14231–14242 (2020).
Kubota, N. et al. PPARγ mediates high-fat diet-induced adipocyte hypertrophy and insulin resistance. Mol. Cell 4, 597–609 (1999).
Hosseinkhani, S. et al. Targeted metabolomics analysis of amino acids and acylcarnitines as risk markers for diabetes by LC–MS/MS technique. Sci. Rep. 12, 8418 (2022).
Ducker, G. S. & Rabinowitz, J. D. One-carbon metabolism in health and disease. Cell Metab. 25, 27–42 (2017).
Pavkov, M. E., Collins, A. J., Coresh, J. & Nelson, R. G. in Diabetes in America 3rd edn. (eds Cowie, C. C. et al.) Ch. 22 (NIDDK, 2018).
Sepehri, Z. et al. Inflammasomes and type 2 diabetes: an updated systematic review. Immunol. Lett. 192, 97–103 (2017).
Hotamisligil, G. S. Inflammation and metabolic disorders. Nature 444, 860–867 (2006).
Ito, Y. et al. RIPK1 mediates axonal degeneration by promoting inflammation and necroptosis in ALS. Science 353, 603–608 (2016).
Christofferson, D. E. et al. A novel role for RIP1 kinase in mediating TNFα production. Cell Death Dis. 3, e320 (2012).
Wakamatsu, K. et al. Metabolites and biomarker compounds of neurodegenerative diseases in cerebrospinal fluid. Metabolites 12, 343 (2022).
Mastracci, T. L., Robertson, M. A., Mirmira, R. G. & Anderson, R. M. Polyamine biosynthesis is critical for growth and differentiation of the pancreas. Sci. Rep. 5, 13269 (2015).
Levasseur, E. M. et al. Hypusine biosynthesis in beta cells links polyamine metabolism to facultative cellular proliferation to maintain glucose homeostasis. Sci. Signal. 12, eaax0715 (2019).
Kar, R. K. et al. Neuron-specific ablation of eIF5A or deoxyhypusine synthase leads to impairments in growth, viability, neurodevelopment, and cognitive functions in mice. J. Biol. Chem. 297, 101333 (2021).
Zeyda, M. & Stulnig, T. M. Obesity, inflammation, and insulin resistance—a mini-review. Gerontology 55, 379–386 (2009).
Geng, J. et al. Regulation of RIPK1 activation by TAK1-mediated phosphorylation dictates apoptosis and necroptosis. Nat. Commun. 8, 359 (2017).
Li, X. et al. Ubiquitination of RIPK1 regulates its activation mediated by TNFR1 and TLRs signaling in distinct manners. Nat. Commun. 11, 6364 (2020).
Casero, R. A. Jr. & Pegg, A. E. Spermidine/spermine N1-acetyltransferase—the turning point in polyamine metabolism. FASEB J. 7, 653–661 (1993).
Niiranen, K. et al. Mice with targeted disruption of spermidine/spermine N1-acetyltransferase gene maintain nearly normal tissue polyamine homeostasis but show signs of insulin resistance upon aging. J. Cell. Mol. Med. 10, 933–945 (2006).
Pietrocola, F. et al. Spermidine induces autophagy by inhibiting the acetyltransferase EP300. Cell Death Differ. 22, 509–516 (2015).
Cox, J. & Mann, M. MaxQuant enables high peptide identification rates, individualized p.p.b.-range mass accuracies and proteome-wide protein quantification. Nat. Biotechnol. 26, 1367–1372 (2008).
Tyanova, S. et al. The Perseus computational platform for comprehensive analysis of (prote)omics data. Nat. Methods 13, 731–740 (2016).
Wang, R. et al. Global stable-isotope tracing metabolomics reveals system-wide metabolic alternations in aging Drosophila. Nat. Commun. 13, 3518 (2022).
Zhou, Z. et al. Metabolite annotation from knowns to unknowns through knowledge-guided multi-layer metabolic networking. Nat. Commun. 13, 6656 (2022).
Shen, X. et al. Metabolic reaction network-based recursive metabolite annotation for untargeted metabolomics. Nat. Commun. 10, 1516 (2019).
Ma, J. et al. iProX: an integrated proteome resource. Nucleic Acids Res. 47, D1211–D1217 (2019).
Chen, T. et al. iProX in 2021: connecting proteomics data sharing with big data. Nucleic Acids Res. 50, D1522–D1527 (2022).
Acknowledgements
This project was supported in part by the National Natural Science Foundation of China (82188101, 92049303, 21837004 and 91849204), Strategic Priority Research Program of the Chinese Academy of Sciences (XDB39030200), Shanghai Municipal Science and Technology Major Project (grant number 2019SHZDZX02), Shanghai Basic Research Pioneer Project and Shanghai Key Laboratory of Aging Studies (19DZ2260400) (all to J.Y.), National Natural Science Foundation of China (32370796 to B.S.) and Major Program of the National Natural Science Foundation of China (92357303 to Junli Liu).
Author information
Authors and Affiliations
Contributions
This project was directed by J.Y. J.Y., B.S. and T.Z. conceived of the concept and designed the experiments. T.Z. performed the majority of the experiments. Z.-J.Z. directed H. Zhang, B.X. and Y.C. to conduct the metabolomics experiments. Y.Z. and B.S. directed J.Li and Mengmeng Zhang to perform the mass spectrometry experiments. Z.W. directed X.H., H. Zhu, S.Y. and Min Zhang in the probe synthesis. Jianping Liu analysed K140 in the RIPK1 kinase structure. W.F., L.Q., C.Q., G.L., Yang Li., R.X., Z.Q., J.H. and C.Z. generated the Nat1fl/fl mice and helped with the animal experiments. X.L. helped with some of the western blot experiments. Q.W., X.J., R.P. and P.L. prepared the clinical samples. Ying Li and C.Q. performed the immunostaining experiments on the clinical samples. Junli Liu provided guidance on the GTT and ITT experiments. J.Y., B.S., Z.-J.Z. and T.Z. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
J.Y., B.S. and T.Z. have filed a patent (Use of RIPK1 inhibitors in the prevention and treatment of vascular injury and diabetes. Patent application no. 202410916336.6; 2024). The other authors declare no competing interests.
Peer review
Peer review information
Nature Cell Biology thanks David James and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Spermidine is decreased in NAT1-deficient cells.
(a) Western blotting analysis of Nat1 knockout and WT MEFs. (b) Quantification of the relative levels of isoniazid (1 mM, 4 hours) and acetylated isoniazid in wild-type and Nat1 knockout MEFs using HPLC-MS. N = 6 in each group. P values were determined using two-tailed Student’s t-test. Data are presented as means ± SEM. (c) WT and Nat1 knockout MEFs pretreated with polyamine oxidase inhibitor MDL72527 (20 μM) for 1 hour were treated with TNFɑ (10 ng/ml) and SM164 (50 nM) to induce RDA with or without putrescine (50 or 100 μM) pretreatment for 4 hours or Nec-1s (10 μM) pretreatment for 1 hour for indicated periods of time and cell death was measured by SytoxGreen positivity. N = 3 in each group. P values were determined using two-way ANOVA post hoc Bonferroni’s test. Data are presented as means ± SEM. (d) WT and Nat1 knockout MEFs were treated with TNFɑ (10 ng/ml)/SM164 (50 nM)/zVAD-fmk (25 μM) to induce necroptosis with or without putrescine (50 or 100 μM) pretreatment for 4 hours or Nec-1s (10 μM) pretreatment for 1 hour for indicated periods of time and cell death was measured by SytoxGreen positivity. N = 3 in each group. P values were determined using two-way ANOVA post hoc Bonferroni’s test. Data are presented as means ± SEM.
Extended Data Fig. 2 RIPK1 is hypusinated by spermidine in DHPS-dependent manner.
(a) A schematic depicting the procedure used to identify hypusinated proteome by click chemistry. MEFs were incubated with the alkyne-spermidine probe (400 μM) for 4 hours to mark hypusinated proteome and the cells were lysed. A CuAAC click-chemistry reaction was performed in the cell lysates with biotin-azide to couple the alkyne-spermidine with biotin. The hypusinated protein targets were pulled down with streptavidin beads and then trypsin-digested peptides were identified and quantified by mass spectrometry. (b) Venn diagram of results for hypusinated proteome by click chemistry reactions. The red circle represents the number of differentially biotin-labeled proteins depicted in red in Fig. 2b. The blue circle represents the number of differentially biotin-labeled proteins depicted in red in Fig. 2c. The orange circle represents the number of differentially biotin-labeled proteins depicted in red in Fig. 2d. (c) Principal component analysis of results for hypusinated proteome by click chemistry reactions. (d) Mass spectrometry analysis of RIPK1 hypusination sites of the in vitro hypusination assay. (e) Western blotting analysis of in vitro hypusination assay using recombinant RIPK1 (1-330aa) kinase domain and eIF5A protein with NAD+, purified DHPS and DOHH proteins with spermidine or vehicle as indicated. Experiments were repeated independently twice with similar outcomes.
Extended Data Fig. 3 Spermidine reduces RIPK1 activation in DHPS-dependent manner.
(a-b) WT and Dhps knockout MEFs pretreated with polyamine oxidase inhibitor MDL72527 (20 μM) for 1 hour were treated with (a) TNFɑ (10 ng/ml)/SM164 (50 nM) to induce RDA or (b) TNFɑ (10 ng/ml)/ SM164 (50 nM)/ zVAD-fmk (25 μM) to induce necroptosis in the presence or absence of spermidine (400 μM), or Nec-1s (10 μM) for the indicated periods of time. Cell death was measured by SytoxGreen positivity. N = 3 in each group. P values were determined using two-way ANOVA post hoc Bonferroni’s test. Data are presented as means ± SEM. (c-d) Western blotting analysis of WT and Dhps knockout MEFs treated with (c) TNFɑ (10 ng/ml)/SM164 (50 nM) to induce RDA or (d) TNFɑ (10 ng/ml)/ SM164 (50 nM)/ zVAD-fmk (25 μM) to induce necroptosis in the presence or absence of spermidine (400 μM) or Nec-1s (10 μM) for 2 and 4 hours. All MEFs were pretreated with polyamine oxidase inhibitor MDL72527 (20 μM) for 1 hour. Experiments were repeated independently twice with similar outcomes. (e) HEK293T cells were transfected with expression vectors of Flag-mRIPK1-WT for 12 hrs, and then treated with indicated concentrations of spermidine for 6 hours. The cell lysates were harvested and then analysed by Western blotting using indicated antibodies. Experiments were repeated independently twice with similar outcomes. (f) HEK293T cells were transfected with expression vectors of Flag-mRIPK1-WT for 12 hrs, and then treated with indicated concentrations of DHPS inhibitor GC7(N1-guanyl-1,7-diamineohepatane) for 6 hrs. Nec-1s (10 μM) was used as a positive control. The cell lysates were harvested and then analysed by Western blotting using indicated antibodies. Experiments were repeated independently twice with similar outcomes.
Extended Data Fig. 4 Nat1 knockout efficiency and the levels of RIPK1 K140 acetylhypusination in conditional Nat1 KO mice.
(a) Strategy of generating a floxed Nat1 allele (Nat1fl/fl). (b) Western blotting analysis of protein levels of Nat1 in different tissues of mice at 8 weeks of age. Experiments were repeated independently twice with similar outcomes. (c) Western blotting analysis of knockout efficiency of Nat1 after Tamoxifen mediated cre-induction in different tissues of indicated conditional knockout mice at 8 weeks of age including Nat1fl/fl mice, Nat1fl/fl; UBCcreERT2/+ mice, Nat1fl/fl; Ripk1D138N/D138N mice and Nat1fl/fl; UBCcreERT2/+; Ripk1D138N/D138N mice. Experiments were repeated independently twice with similar outcomes. (d) Food intake of normal diet-fed mice at 14 weeks of age with indicated genotypes per day. N = 5 in each group. P values were determined using two-way ANOVA post hoc Bonferroni’s test. Data are presented as means ± SEM. (e) Mice were fed with spermidine in the drinking water at the indicated concentrations for 2 weeks. Western blotting analysis of isolated endothelial cells from the pancreas of Nat1fl/fl WT mice and Nat1fl/fl; UBCcreERT2/+ mice at 14 weeks of age was then performed. Cre was activated at 4 weeks of age with daily Tamoxifen injection for 5 consecutive days. Experiments were repeated independently twice with similar outcomes.
Extended Data Fig. 5 Food intake and body weight of endothelial NAT1-deficient mice.
(a) Western blotting analysis of knockout efficiency of Nat1 in isolated CD31 positive endothelial cells of Nat1fl/fl mice, Nat1fl/fl; Cdh5creERT2/+ mice, Nat1fl/fl; Ripk1D138N/D138N mice and Nat1fl/fl; Cdh5creERT2/+; Ripk1D138N/D138N mice. (b) Food intake of normal diet-fed mice at 14 weeks of age with indicated genotypes per day. N = 5 in each group. P values were determined using two-way ANOVA post hoc Bonferroni’ s test. Data are presented as means ± SEM. (c) Body weights of normal diet-fed Nat1fl/fl mice, Nat1fl/fl; UBCcreERT2/+ mice, Nat1fl/fl; Cdh5creERT2/+ mice, Nat1fl/fl; Ripk1D138N/D138N mice, Nat1fl/fl; UBCcreERT2/+; Ripk1D138N/D138N mice and Nat1fl/fl; Cdh5creERT2/+; Ripk1D138N/D138N recorded at 12 weeks of age. N = 5 in each group. P values were determined using one-way ANOVA post hoc Bonferroni’s test. Data are presented as means ± SEM.
Extended Data Fig. 6 Myeloid lineage or microglia conditional Nat1 knockout does not lead to insulin resistance.
(a) Western blotting analysis of the knockout efficiency of Nat1 in primary myeloid cells of Nat1fl/fl; Lyz2cre/+ mice at 8 weeks of age compared with that of Nat1fl/fl mice as a control. b,c, Body weights of normal diet-fed (b) or high-fat-diet-fed (c) Nat1fl/fl mice and Nat1fl/fl; Lyz2cre/+ mice recorded from 8 to 14 weeks of age. N = 5 in each group. P values were determined using two-way ANOVA post hoc Bonferroni’s test. Data are presented as means ± SEM. d,e, GTT (d) or ITT (e) of normal diet-fed Nat1fl/fl mice and Nat1fl/fl; Lyz2cre/+ mice on normal diet at 14 weeks of age. N = 5 in each group. Two-way ANOVA post hoc Bonferroni’s test. Data are presented as means ± SEM. (f) Western blotting analysis of the knockout efficiency of Nat1 in primary microglia of Nat1fl/fl; Cx3cr1creERT2/+ mice at 8 weeks of age compared with that of Nat1fl/fl mice as a control. Tamoxifen-mediated cre-induction was performed at 4 weeks of age for 5 consecutive days. g,h, Body weights of normal diet-fed (g) or high-fat-diet-fed (h) Nat1fl/fl mice and Nat1fl/fl; Cx3cr1creERT2/+ mice recorded from 8 to 14 weeks of age. Tamoxifen-mediated cre-induction was performed at 4 weeks of age for 5 consecutive days. N = 5 in each group. P values were determined using two-way ANOVA post hoc Bonferroni’s test. Data are presented as means ± SEM. i,j, GTT (i) or ITT (j) of normal diet-fed Nat1fl/fl mice and Nat1fl/fl; Cx3cr1creERT2/+ mice on normal diet at 14 weeks of age. Tamoxifen-mediated cre-induction was performed at 4 weeks of age for 5 consecutive days. N = 5 in each group. P values were determined using two-way ANOVA post hoc Bonferroni’s test. Data are presented as means ± SEM.
Extended Data Fig. 7 Liver or muscle conditional Nat1 knockout does not lead to insulin resistance.
(a) Western blotting analysis of the knockout efficiency of Nat1 in liver of Nat1fl/fl; Albcre/+ mice at 8 weeks of age compared with that of Nat1fl/fl mice as a control. b,c, Body weight of normal diet-fed (b) or high-fat-diet-fed (c) Nat1fl/fl and Nat1fl/fl; Albcre/+ mice recorded from 8 to 14 weeks of age. N = 5 in each group. P values were determined using two-way ANOVA post hoc Bonferroni’s test. Data are presented as means ± SEM. d,e, GTT (d) or ITT (e) of normal diet-fed Nat1fl/fl mice and Nat1fl/fl; Albcre/+ mice with normal diet at 14 weeks of age. N = 5 in each group. P values were determined using two-way ANOVA post hoc Bonferroni’s test. Data are presented as means ± SEM. (f) Inflammatory cytokine expression in livers from Nat1fl/fl and Nat1fl/fl; Albcre/+ mice at 14 weeks of age. N = 4 in each group. P values were determined using one-way ANOVA post hoc Bonferroni’s test. Data are presented as means ± SEM. (g) Western blotting analysis of the knockout efficiency of Nat1 in muscle of Nat1fl/fl; Ckmmcre/+ mice at 8 weeks of age compared with that of Nat1fl/fl mice as a control. h,i, Body weights of normal diet-fed (h) or high-fat-diet-fed (i) Nat1fl/fl mice and Nat1fl/fl; Ckmmcre/+ mice were recorded from 8 to 14 weeks of age. N = 5 in each group. P values were determined using two-way ANOVA post hoc Bonferroni’s test. Data are presented as means ± SEM. j,k, GTT (j) or ITT (k) of normal diet-fed Nat1fl/fl mice and Nat1fl/fl; Ckmmcre/+ mice on normal diet at 14 weeks of age. N = 5 in each group. P values were determined using two-way ANOVA post hoc Bonferroni’s test. Data are presented as means ± SEM. (l) Inflammatory cytokines expression in muscles from Nat1fl/fl mice and Nat1fl/fl; Ckmmcre/+ mice at 14 weeks of age. N = 4 in each group. P values were determined using one-way ANOVA post hoc Bonferroni’s test. Data are presented as means ± SEM.
Extended Data Fig. 8 The representative metabolites quantified in metabolic analysis.
(a) Abundances of citrulline and ornithine in the urea cycle pathway and serine and methionine in one-carbon metabolism in plasma in Nat1fl/fl mice, Nat1fl/fl; UBCcreERT2/+ mice, Nat1fl/fl; Cdh5creERT2/+ mice, Nat1fl/fl; Ripk1D138N/D138N mice, Nat1fl/fl; UBCcreERT2/+; Ripk1D138N/D138N mice and Nat1fl/fl; Cdh5creERT2/+; Ripk1D138N/D138N mice at 14 weeks of age. Cre was induced by Tamoxifen injection at 4 weeks of age for 5 consecutive days. P values were determined using one-way ANOVA post hoc Bonferroni’s test. N = 6 in each group. Data are presented as means ± SEM. (b) Abundances of metabolites in nucleotide metabolism, including adenosine, inosine, thymidine, cytidine and NAD, as well as amino acids, including leucine, valine and asparagine, in plasma in mouse groups with the indicated genotypes. P values were determined using one-way ANOVA post hoc Bonferroni’s test. N = 6 in each group. Data are presented as means ± SEM.
Extended Data Fig. 9 NAT1-deficient mice develop RIPK1-dependent vascular damage and cell death in pancreas.
(a) Immunostaining and quantification (below) of pericyte marker NG2 in pancreas from mice with the indicated genotypes at 14 weeks of age. Scale bars, 50 μm. N = 3 in each group. P values were determined using one-way ANOVA post hoc Dunnett’s test. Data are presented as means ± SEM. (b) Co-immunostaining and quantification (below) of cleaved-casp3 and endothelial marker CD31 in pancreas from mice at 14 weeks of age with the indicated genotypes. Cre was activated at 4 weeks of age with daily Tamoxifen injection for 5 consecutive days. Scale bars, 50 μm. N = 4 in each group. P values were determined using one-way ANOVA post hoc Dunnett’s test. Data are presented as means ± SEM.
Extended Data Fig. 10 Flow chart of the collection and processing of clinical samples.
(a) Flow chart of the collection and processing of clinical cardiovascular samples from the T2D group and non-T2D group. (b) Flow chart of the collection and processing of clinical kidney samples from the diabetic nephropathy group and non-diabetic nephropathy group.
Supplementary information
Supplementary Tables (download XLSX )
Supplementary Table 1. Absolute quantification of the intracellular levels of polyamines, including spermidine, spermine and putrescine and the acetylated isoforms, in WT and Nat1 KO MEFs using high-performance LC–MS. WT and Nat1 KO MEFs were treated with spermidine (400 μM) or Nec-1s (10 μM) for 4 h. n = 6 per group. P values were determined by one-way ANOVA with post-hoc Bonferroni’s test. The data are presented as means ± s.e.m. Supplementary Table 2. Quantification of the relative levels of isoniazid (1 mM; 4 h) and acetylated isoniazid in WT and Nat1 KO MEFs using high-performance LC–MS. n = 6 per group. P values were determined by two-tailed Student’s t-test. The data are presented as means ± s.e.m. Supplementary Table 3. [U-13C] arginine tracing analysis in WT and Nat1 KO MEFs measured by LC–MS. WT and Nat1 KO MEFs were treated with [U-13C] arginine (400 μM) for 16 h and tracing into polyamines was measured by LC–MS. n = 6 (WT group) and n = 3 (Nat1 KO group). P values were determined by two-tailed Student’s t-test. The data are presented as means ± s.e.m. Supplementary Table 4. Mass spectrometry for the identification and quantification of proteins pulled down by alkyne–spermidine probe. An excel file of the proteomics data showing quantification of the abundance of each protein enriched by alkyne–spermidine probe under different conditions. n = 4 per group. P values were determined by two-tailed Student’s t-test. Supplementary Table 5. Mass spectrometry analysis of RIPK1 hypusination sites identified by in vitro hypusination assay. An excel file of the quantified hypusination sites identified by in vitro hypusination assay using the recombinant RIPK1 kinase domain (amino acids 1–330) and eIF5A protein along with NAD+, purified DHPS and DOHH proteins and either spermidine or vehicle. Supplementary Table 6. Mass spectrometry analysis of immunoprecipitated endogenous RIPK1 in WT MEFs and Nat1 KO MEFs for acetylhypusination site(s) and hypusination site(s). n = 3 per group. Supplementary Table 7. PCA of the metabolome of plasma in Nat1fl/fl, Nat1fl/fl;UBCcreERT2/+, Nat1fl/fl;Cdh5creERT2/+, Nat1fl/fl;Ripk1D138N/D138N, Nat1fl/fl;UBCcreERT2/+;Ripk1D138N/D138N and Nat1fl/fl;Cdh5creERT2/+;Ripk1D138N/D138N mice at 14 weeks of age. n = 6 per group. Supplementary Table 8. 136 significantly changed plasma metabolites between Nat1 KO mice (inducible global KO or endothelium-specific inducible KO) and other mouse groups with the indicated genotypes (P < 0.05; two-tailed Student’s t-test). Supplementary Table 9. Absolute high-performance LC–MS quantification of the levels of polyamines, including spermidine, spermine and putrescine and the acetylated isoforms, in salvaged vascular tissue from patients with or without T2D who had undergone coronary artery bypass graft surgery. n = 3 in control patients without T2D and n = 4 in patients with T2D. P values were determined by two-tailed Student’s t-test. The data are presented as means ± s.e.m.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Fig. 1 (download PDF )
Unprocessed western blots and/or gels.
Source Data Fig. 2 (download PDF )
Unprocessed western blots and/or gels.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download PDF )
Unprocessed western blots and/or gels.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download PDF )
Unprocessed western blots and/or gels.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download PDF )
Unprocessed western blots and/or gels.
Source Data Fig. 7 (download XLSX )
Statistical source data.
Source Data Fig. 8 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 1 (download PDF )
Unprocessed western blots and/or gels.
Source Data Extended Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 2 (download PDF )
Unprocessed western blots and/or gels.
Source Data Extended Data Fig. 3 (download PDF )
Unprocessed western blots and/or gels.
Source Data Extended Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 4 (download PDF )
Unprocessed western blots and/or gels.
Source Data Extended Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 5 (download PDF )
Unprocessed western blots and/or gels.
Source Data Extended Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 6 (download PDF )
Unprocessed western blots and/or gels.
Source Data Extended Data Fig. 6 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 7 (download PDF )
Unprocessed western blots and/or gels.
Source Data Extended Data Fig. 7 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 8 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 9 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, T., Fu, W., Zhang, H. et al. Spermidine mediates acetylhypusination of RIPK1 to suppress diabetes onset and progression. Nat Cell Biol 26, 2099–2114 (2024). https://doi.org/10.1038/s41556-024-01540-6
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41556-024-01540-6
This article is cited by
-
The transition from monocyte to tissue-resident macrophage requires DHPS
Nature (2026)
-
Receptor-Interacting Protein Kinase 1 (RIPK1): A Potential Therapeutic Target in Ischemic Stroke
Molecular Neurobiology (2026)
-
Metabolism of Polyamine in Vascular Diseases: An Prospective Therapeutic Target
Bratislava Medical Journal (2025)
-
The functional roles and mechanisms of polyamines in age-related bone diseases
Cellular and Molecular Life Sciences (2025)
-
Spermidine Alleviates Sepsis-Induced Acute Lung Injury through AMPK-Mediated Improvement of Necroptosis
Inflammation (2025)