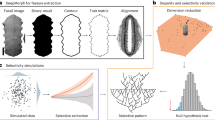
Abstract
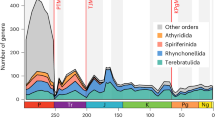
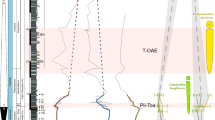
Environmental stressors have exacerbated the collapse of marine ecosystems during mass extinctions. However, the survival strategies of marine species during mass extinctions remain unclear. Here, we investigated morphological evolution of brachiopods across the Permian–Triassic mass extinction (PTME) using a database of 3,225 specimens representing 1,061 species and foraminifera across the PTME and early Toarcian oceanic anoxic event (T-OAE) using a database of 757 specimens representing 12 species. We found a significant reduction in the number and proportion (plicae length/shell length) of shell plicae of brachiopods (36.4% and 60.0%, respectively) across the PTME and a significant decrease in the shell thickness of foraminifera (18.9% and 42.4% across the PTME and 36.9–61.8% across the T-OAE). We calculated that these adaptive strategies could reduce the energetic costs of calcification by more than half for brachiopods across the PTME, and by ~20–62% for foraminifera across the PTME and T-OAE, to compensate for the elevated cost of calcification due to environmental and ecological pressures. We propose that simplification of morphological features, such as reduced shell ornamentation and shell thinning, serves as a potential economic strategy for calcifying organisms to cope with extinction events by reducing energy demands, but further studies with a broader range of taxa and extinction events are needed to confirm the generality of this bioenergetic strategy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data supporting the findings of this study are available within the article, Extended Data Figs. 1–5, Extended Data Tables 1–5 and Supplementary Information. Source data are provided with this paper.
Code availability
The code needed to run the cGENIE model is available via Github at https://github.com/derpycode/cgenie.muffin and additional configuration codes to replicate the model results are available via Zenodo at https://doi.org/10.5281/zenodo.14169287 (ref. 107).
References
Raup, D. M. The role of extinction in evolution. Proc. Natl Acad. Sci. USA 91, 6758–6763 (1994).
Bambach, R. K. Phanerozoic biodiversity mass extinctions. Annu. Rev. Earth Planet. Sci. 34, 127–155 (2006).
Stanley, S. M. Estimates of the magnitudes of major marine mass extinctions in Earth history. Proc. Natl Acad. Sci. USA 113, E6325–E6334 (2016).
Wagner, P. J., Kosnik, M. A. & Lidgard, S. Abundance distributions imply elevated complexity of post-Paleozoic marine ecosystems. Science 314, 1289–1292 (2006).
Muscente, A. D. et al. Quantifying ecological impacts of mass extinctions with network analysis of fossil communities. Proc. Natl Acad. Sci. USA 115, 5217–5222 (2018).
Jablonski, D. Lessons from the past: evolutionary impacts of mass extinctions. Proc. Natl Acad. Sci. USA 98, 5393–5398 (2001).
Hull, P. Life in the aftermath of mass extinctions. Curr. Biol. 25, R941–R952 (2015).
Pelejero, C., Calvo, E. & Hoegh-Guldberg, O. Paleo-perspectives on ocean acidification. Trends Ecol. Evol. 25, 332–344 (2010).
Hönisch, B. et al. The geological record of ocean acidification. Science 335, 1058–1063 (2012).
Kiessling, W. & Simpson, C. On the potential for ocean acidification to be a general cause of ancient reef crises. Glob. Change Biol. 17, 56–67 (2011).
McGhee, G. R. The Geometry of Evolution: Adaptive Landscapes and Theoretical Morphospaces (Cambridge Univ. Press, 2006).
Reddin, C. J., Nätscher, P. S., Kocsis, Á. T., Pörtner, H.-O. & Kiessling, W. Marine clade sensitivities to climate change conform across timescales. Nat. Clim. Change 10, 249–253 (2020).
Malanoski, C. M., Farnsworth, A., Lunt, D. J., Valdes, P. J. & Saupe, E. E. Climate change is an important predictor of extinction risk on macroevolutionary timescales. Science 383, 1130–1134 (2024).
Fox, C. P. et al. Two-pronged kill mechanism at the end-Triassic mass extinction. Geology 50, 448–453 (2022).
Keller, G. et al. Environmental changes during the Cretaceous–Paleogene mass extinction and Paleocene–Eocene thermal maximum: implications for the Anthropocene. Gondwana Res. 56, 69–89 (2018).
Schaal, E. K., Clapham, M. E., Rego, B. L., Wang, S. C. & Payne, J. L. Comparative size evolution of marine clades from the Late Permian through Middle Triassic. Paleobiology 42, 127–142 (2016).
Barnosky, A. D. et al. Has the Earth’s sixth mass extinction already arrived? Nature 471, 51–57 (2011).
Finnegan, S. et al. Using the fossil record to understand extinction risk and inform marine conservation in a changing world. Annu. Rev. Mar. Sci. 16, 307–333 (2024).
Penn, J. L., Deutsch, C., Payne, J. L. & Sperling, E. A. Temperature-dependent hypoxia explains biogeography and severity of end-Permian marine mass extinction. Science 362, eaat1327 (2018).
Knoll, A. H., Barnbach, R. K., Payne, J. L., Pruss, S. & Fischer, W. W. Paleophysiology and end-Permian mass extinction. Earth Planet. Sci. Lett. 256, 295–313 (2007).
Pohl, A. et al. Why the Early Paleozoic was intrinsically prone to marine extinction. Sci. Adv. 9, eadg7679 (2023).
Strotz, L. C., Saupe, E. E., Kimmig, J. & Lieberman, B. S. Metabolic rates, climate and macroevolution: a case study using Neogene molluscs. Proc. R. Soc. B 285, 20181292 (2018).
Tajika, A. et al. Ammonoid extinction versus nautiloid survival: is metabolism responsible? Geology 51, 621–625 (2023).
Carlson, S. J. The evolution of Brachiopoda. Annu. Rev. Earth Planet. Sci. 44, 409–438 (2016).
Chen, Z. Q., Kaiho, K. & George, A. D. Survival strategies of brachiopod faunas from the end-Permian mass extinction. Paleogeogr. Paleoclimatol. Paleoecol. 224, 232–269 (2005).
Song, H. J., Tong, J. N., Zhang, K. X., Wang, Q. X. & Chen, Z. Q. Foraminiferal survivors from the Permian–Triassic mass extinction in the Meishan section, South China. Palaeoworld 16, 105–119 (2007).
Song, H. J., Tong, J. N. & Chen, Z. Q. Two episodes of foraminiferal extinction near the Permian–Triassic boundary at the Meishan section, South China. Aust. J. Earth Sci. 56, 765–773 (2009).
Jiang, S. Y., Song, H. J., Kemp, D. B., Dai, X. & Liu, X. K. Two pulses of extinction of larger benthic foraminifera during the Pliensbachian–Toarcian and early Toarcian environmental crises. Paleogeogr. Paleoclimatol. Paleoecol. 560, 109998 (2020).
Gilbert, P. U. et al. Biomineralization: integrating mechanism and evolutionary history. Sci. Adv. 8, eabl9653 (2022).
Spalding, C., Finnegan, S. & Fischer, W. W. Energetic costs of calcification under ocean acidification. Glob. Biogeochem. Cycles 31, 866–877 (2017).
Wu, Y. Y. et al. Volcanic CO2 degassing postdates thermogenic carbon emission during the end-Permian mass extinction. Sci. Adv. 9, eabq4082 (2023).
Ye, F. C. et al. Variation in brachiopod microstructure and isotope geochemistry under low-pH-ocean acidification conditions. Biogeosciences 16, 617–642 (2019).
Valentine, J. W. Evolutionary Paleoecology of the Marine Biosphere (Prentice Hall, 1973).
Guex, J. Reinitialization of evolutionary clocks during sublethal environmental stress in some invertebrates. Earth Planet. Sci. Lett. 242, 240–253 (2006).
Guex, J. Retrograde Evolution During Major Extinction Crises (Springer, 2016).
Forel, M. B., Kershaw, S., Lord, A. R. & Crasquin, S. Applications of fossil taxonomy in palaeoenvironmental reconstruction: a case study of ostracod identification and diversity in Permian–Triassic boundary microbialites. Facies 67, 23 (2021).
Dai, X., Korn, D. & Song, H. J. Morphological selectivity of the Permian–Triassic ammonoid mass extinction. Geology 49, 1112–1116 (2021).
Stanley, G. D. Recovery of corals and reefs after the end-Permian and the “naked coral” Lazarus effect. J. Earth Sci. 21, 161 (2010).
Keller, G., Abramovich, S., Berner, Z. & Adatte, T. Biotic effects of the Chicxulub impact, K–T catastrophe and sea level change in Texas. Paleogeogr. Paleoclimatol. Paleoecol. 271, 52–68 (2009).
Keller, G. et al. Paleoecology of the Cretaceous–Tertiary mass extinction in planktonic foraminifera. Paleogeogr. Paleoclimatol. Paleoecol. 178, 257–297 (2002).
Nützel, A. & Schulbert, C. Facies of two important Early Triassic gastropod lagerstatten: implications for diversity patterns in the aftermath of the end-Permian mass extinction. Facies 51, 480–500 (2005).
O’Dea, S. A. et al. Coccolithophore calcification response to past ocean acidification and climate change. Nat. Commun. 5, 5363 (2014).
Barker, S. & Elderfield, H. Foraminiferal calcification response to glacial-interglacial changes in atmospheric CO2. Science 297, 833–836 (2002).
McGhee, G. R. Geometry of evolution in the biconvex Brachiopoda: morphological effects of mass extinction. Neues Jahrb. Geol. Paläontol. Abh. 197, 357–382 (1995).
McGhee, G. R. Shell geometry and stability strategies in the biconvex Brachiopoda. Neues Jahrb. Geol. Paläontol. Abh. 3, 155–184 (1980).
Wang, F. Y., Chen, J., Dai, X. & Song, H. J. A new Early Triassic brachiopod fauna from southern Tibet, China: implications on brachiopod recovery and the late Smithian extinction in southern Tethys. J. Paleontol. 96, 1–32 (2022).
Garbelli, C. Shell microstructures in Lopingian brachiopods: implications for fabric evolution and calcification. Riv. Ital. Paleontol. Stratigr. 123, 541–560 (2017).
Palmer, A. R. Calcification in marine molluscs: how costly is it? Proc. Natl Acad. Sci. USA 89, 1379–1382 (1992).
Garbelli, C., Angiolini, L. & Shen, S. Z. Biomineralization and global change: a new perspective for understanding the end-Permian extinction. Geology 45, 19–22 (2017).
Tyszka, J., Godos, K., Goleń, J. & Radmacher, W. Foraminiferal organic linings: functional and phylogenetic challenges. Earth Sci. Rev. 220, 103726 (2021).
Dubicka, Z. et al. Biocalcification in porcelaneous foraminifera. eLife 13, RP91568 (2024).
Pan, T. C. F., Applebaum, S. L. & Manahan, D. T. Experimental ocean acidification alters the allocation of metabolic energy. Proc. Natl Acad. Sci. USA 112, 4696–4701 (2015).
Melzner, F., Mark, F. C., Seibel, B. A. & Tomanek, L. Ocean acidification and coastal marine invertebrates: tracking CO2 effects from seawater to the cell. Annu. Rev. Mar. Sci. 12, 499–523 (2020).
Garilli, V. et al. Physiological advantages of dwarfing in surviving extinctions in high-CO2 oceans. Nat. Clim. Change 5, 678–682 (2015).
Zhao, X. G. et al. CO2-driven ocean acidification weakens mussel shell defense capacity and induces global molecular compensatory responses. Chemosphere 243, 125415 (2020).
Chadwick, M., Harper, E. M., Lemasson, A., Spicer, J. I. & Peck, L. S. Quantifying susceptibility of marine invertebrate biocomposites to dissolution in reduced pH. R. Soc. Open Sci. 6, 190252 (2019).
Cui, Y., Kump, L. R. & Ridgwell, A. in Volcanism and Global Environmental Change (ed. Schmidt, A. et al.) 291–307 (Cambridge Univ. Press, 2015).
Jurikova, H. et al. Permian–Triassic mass extinction pulses driven by major marine carbon cycle perturbations. Nat. Geosci. 13, 745–750 (2020).
Muller, T. et al. Ocean acidification during the early Toarcian extinction event: evidence from boron isotopes in brachiopods. Geology 48, 1184–1188 (2020).
Dal Corso, J. et al. Environmental crises at the Permian–Triassic mass extinction. Nat. Rev. Earth Environ. 3, 197–214 (2022).
Foster, W. J. et al. Bioindicators of severe ocean acidification are absent from the end-Permian mass extinction. Sci. Rep. 12, 1202 (2022).
Clarkson, M. et al. Ocean acidification and the Permo–Triassic mass extinction. Science 348, 229–232 (2015).
Hofmann, G. E. et al. The effect of ocean acidification on calcifying organisms in marine ecosystems: an organism-to-ecosystem perspective. Annu. Rev. Ecol. Evol. Syst. 41, 127–147 (2010).
Gazeau, F. et al. Impact of elevated CO2 on shellfish calcification. Geophys. Res. Lett. https://doi.org/10.1029/2006GL028554(2007).
Pörtner, H. O. Ecosystem effects of ocean acidification in times of ocean warming: a physiologist’s view. Mar. Ecol. Prog. Ser. 373, 203–217 (2008).
Martin, R. E. Secular increase in nutrient levels through the Phanerozoic: implications for productivity, biomass, and diversity of the marine biosphere. Palaios 11, 209–219 (1996).
Schobben, M. et al. Flourishing ocean drives the end-Permian marine mass extinction. Proc. Natl Acad. Sci. USA 112, 10298–10303 (2015).
Zhao, X. G. et al. Ocean acidification adversely influences metabolism, extracellular pH and calcification of an economically important marine bivalve, Tegillarca granosa. Mar. Environ. Res. 125, 82–89 (2017).
Stumpp, M. et al. Digestion in sea urchin larvae impaired under ocean acidification. Nat. Clim. Change 3, 1044–1049 (2013).
Frieder, C. A., Applebaum, S. L., Pan, T.-C. F., Hedgecock, D. & Manahan, D. T. Metabolic cost of calcification in bivalve larvae under experimental ocean acidification. ICES J. Mar. Sci. 74, 941–954 (2017).
Waldbusser, G. G. et al. Saturation-state sensitivity of marine bivalve larvae to ocean acidification. Nat. Clim. Change 5, 273–280 (2015).
Osborne, E. B., Thunell, R. C., Gruber, N., Feely, R. A. & Benitez-Nelson, C. R. Decadal variability in twentieth-century ocean acidification in the California current ecosystem. Nat. Geosci. 13, 43–49 (2020).
Chaabane, S. et al. Migrating is not enough for modern planktonic foraminifera in a changing ocean. Nature 636, 390–396 (2024).
Fox, L., Stukins, S., Hill, T. & Miller, C. G. Quantifying the effect of anthropogenic climate change on calcifying plankton. Sci. Rep. 10, 1620 (2020).
Dong, S., Lei, Y., Li, T., Cao, Y. & Xu, K. J. G. F. Biocalcification crisis in the continental shelf under ocean acidification. Geosci. Front. 14, 101622 (2023).
Leung, J. Y. S., Zhang, S. & Connell, S. D. Is ocean acidification really a threat to marine calcifiers? A systematic review and meta-analysis of 980+ studies spanning two decades. Small 18, 2107407 (2022).
Davis, C. V. et al. Ocean acidification compromises a planktic calcifier with implications for global carbon cycling. Sci. Rep. 7, 2225 (2017).
Deutsch, C. et al. Impact of warming on aquatic body sizes explained by metabolic scaling from microbes to macrofauna. Proc. Natl Acad. Sci. USA 119, e2201345119 (2022).
Pörtner, H. O. Oxygen- and capacity-limitation of thermal tolerance: a matrix for integrating climate-related stressor effects in marine ecosystems. J. Exp. Biol. 213, 881–893 (2010).
Brown, J. H., Gillooly, J. F., Allen, A. P., Savage, V. M. & West, G. B. Toward a metabolic theory of ecology. Ecology 85, 1771–1789 (2004).
Rubalcaba, J. G., Verberk, W. C., Hendriks, A. J., Saris, B. & Woods, H. A. Oxygen limitation may affect the temperature and size dependence of metabolism in aquatic ectotherms. Proc. Natl Acad. Sci. USA 117, 31963–31968 (2020).
Pörtner, H. O. Integrating climate-related stressor effects on marine organisms: unifying principles linking molecule to ecosystem-level changes. Mar. Ecol. Prog. Ser. 470, 273–290 (2012).
Pörtner, H. O. & Farrell, A. P. Physiology and climate change. Science 322, 690–692 (2008).
Higgins, J. A., Fischer, W. & Schrag, D. Oxygenation of the ocean and sediments: consequences for the seafloor carbonate factory. Earth Planet. Sci. Lett. 284, 25–33 (2009).
Knoll, A. H., Fischer, W. W., Gattuso, J. & Hansson, L. in Ocean Acidification Vol. 4 (eds Gattuso, J.-P. & Hansson, L.) 67–82 (Oxford Univ. Press, 2011).
Rodolfo-Metalpa, R. et al. Coral and mollusc resistance to ocean acidification adversely affected by warming. Nat. Clim. Change 1, 308–312 (2011).
Wijgerde, T., Silva, C. I. F., Scherders, V., van Bleijswijk, J. & Osinga, R. Coral calcification under daily oxygen saturation and pH dynamics reveals the important role of oxygen. Biol. Open 3, 489–493 (2014).
Vermeij, G. J. A Natural History of Shells Vol. 15 (Princeton Univ. Press, 1995).
Williams, A., Brunton, C. H. C., Carlson, S. J., Alvarez, F. & Blodgett, R. B. Treatise on Invertebrate Paleontology, Part H, Brachiopoda, Revised, Volume 4: Rhynchonelliformea (part) (Geological Society of America and Univ. of Kansas, 2002).
Williams, A., Brunton, C. H. C., Carlson, S. J., Baker, P. G. & Carter, J. L. Treatise on Invertebrate Paleontology, Part H, Brachiopoda, Revised, Volume 5: Rhynchonelliformea (part) (Geological Society of America and Univ. of Kansas, 2006).
Yin, H. F., Zhang, K. X., Tong, J. N., Yang, Z. Y. & Wu, S. B. The global stratotype section and point (GSSP) of the Permian–Triassic boundary. Episodes J. Int. Geosci. 24, 102–114 (2001).
Song, H. J. et al. Early Triassic disaster and opportunistic foraminifers in South China. Geol. Mag. 153, 298–315 (2016).
Han, Z., Hu, X. M., Kemp, D. B. & Li, J. Carbonate-platform response to the Toarcian oceanic anoxic event in the southern hemisphere: implications for climatic change and biotic platform demise. Earth Planet. Sci. Lett. 489, 59–71 (2018).
Ye, F. C., Garbelli, C., Shen, S. & Angiolini, L. The shell fabric of Palaeozoic brachiopods: patterns and trends. Lethaia 54, 419–439 (2021).
Rudwick, M. J. S. Living and Fossil Brachiopods (Hutchinson, 1970).
Pérez-Huerta, A. et al. Brachiopod punctae: a complexity in shell biomineralisation. J. Struct. Biol. 167, 62–67 (2009).
Bryan, A. K., Goranov, A., Amon, A. & Manalis, S. R. Measurement of mass, density, and volume during the cell cycle of yeast. Proc. Natl Acad. Sci. USA 107, 999–1004 (2010).
Immel, F. et al. Shell proteome of rhynchonelliform brachiopods. J. Struct. Biol. 190, 360–366 (2015).
Feijó Delgado, F. et al. Intracellular water exchange for measuring the dry mass, water mass and changes in chemical composition of living cells. PLoS ONE 8, e67590 (2013).
Peck, L. S. & Edwards, T. M. in Brachiopods (eds Cooper, P. & Jin, J.) 203–207 (CRC, 1996).
Edwards, N. R. & Marsh, R. Uncertainties due to transport-parameter sensitivity in an efficient 3-D ocean-climate model. Clim. Dynam. 24, 415–433 (2005).
Ridgwell, A. et al. Marine geochemical data assimilation in an efficient Earth System Model of global biogeochemical cycling. Biogeosciences 4, 87–104 (2007).
Hülse, D. et al. End-Permian marine extinction due to temperature-driven nutrient recycling and euxinia. Nat. Geosci. 14, 862–867 (2021).
Pohl, A. et al. Continental configuration controls ocean oxygenation during the Phanerozoic. Nature 608, 523–527 (2022).
Wu, Y. Y. et al. Six-fold increase of atmospheric pCO2 during the Permian–Triassic mass extinction. Nat. Commun. 12, 2137 (2021).
Nordt, L., Breecker, D. & White, J. Jurassic greenhouse icesheet fluctuations sensitive to atmospheric CO2 dynamics. Nat. Geosci. 15, 54–59 (2022).
Wang, F. et al. Bioeconomy in extinction events: reducing calcification costs through morphological simplification. Zenodo https://doi.org/10.5281/zenodo.14169288 (2024).
Scotese, C. R., Song, H. J., Mills, B. J. W. & van der Meer, D. G. Phanerozoic paleotemperatures: the Earth’s changing climate during the laet 540 million years. Earth Sci. Rev. 215, 103503 (2021).
Kiessling, W. et al. Pre–mass extinction decline of latest Permian ammonoids. Geology 46, 283–286 (2018).
Acknowledgements
This study was supported by the National Natural Science Foundation of China (42325202, 42072010, 92155201, 92255303), State Key R&D Project of China (2023YFF0804000), Natural Science Foundation of Hubei (2023AFA006) and the Fundamental Research Funds for the Central Universities, China University of Geosciences (Wuhan).
Author information
Authors and Affiliations
Contributions
F.W. and Haijun Song designed the research. F.W. collected brachiopod data. Haijun Song, J.T. and S.J. provided modern brachiopods and Permian, Triassic and Jurassic foraminifera data. F.W., Haijun Song, X.D. and Y.W. analysed the data. F.W., Haijun Song, S.F., J.D.C., Y.W. and X.D. discussed the data. F.W. and Haijun Song wrote the paper with input from all authors. F.W., Haijun Song, S.F., J.D.C., F.Y., Y.W., X.D., S.J., J.C., L.T., D.C., Huyue Song and J.T. further revised the original manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Ecology & Evolution thanks Nicholas Hebdon, Pedro Monarrez and Anshuman Swain for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Relationships between shell mass and the number and proportion of plicae.
Different colors and shapes of symbols represent shells with different proportions of Plicae. PP represents the proportion of plicae.
Extended Data Fig. 2 Relationships between shell length and shell thickness and change in shell thickness of foraminifera during the PTME and T-OAE.
a, Scaling relationship of Diplosphaerina inaequalis. b, Shell thickness of D. inaequalis. c, Scaling relationship of Globivalvulina lukachiensis. d, Shell thickness of G. lukachiensis. e, Scaling relationship of Siphovalvulina colomi. f, Shell thickness of S.a colomi. g, Scaling relationship of Siphovalvulina gibraltarensis. h, Shell thickness of S. gibraltarensis. i, Scaling relationship of Glomospira tetragona. j, Shell thickness of G. tetragona. k, Scaling relationship of Glomospira tingriensis. l, Shell thickness of G. tingriensis. m, Scaling relationship of Glomospira sinensis. n, Shell thickness of G. sinensis. o, Scaling relationship of Glomospirella pavida. p, Shell thickness of G. pavida. q, Scaling relationship of Textularia dollfussi. r, Shell thickness of T. dollfussi. s, Scaling relationship of Mesoendothyra croatica. t, Shell thickness of M. croatica. u, Scaling relationship of Valvulina triangularis. v, Shell thickness of V. triangularis. w, Scaling relationship of Duotaxis birmanica. x, Shell thickness of D. birmanica. The shaded area around the regression curve represents the 95% confidence interval. Statistical test: two-sided Mann–Whitney U -test. Each boxplot displays the 25th percentile, median, and 75th percentile, with whiskers extending to the 5th and 95th percentiles.
Extended Data Fig. 3 Species-level distribution of percentage of calcium shell mass of living brachiopods and bivalves.
The percentage of calcium shell mass is calculated from the ratio of ash-free dry mass to total dry mass. All data collected from published literatures. Each boxplot displays the 25th percentile, median, and 75th percentile, with whiskers extending to the 5th and 95th percentiles.
Extended Data Fig. 4 Illustration of statistical indicators.
a, Brachiopoda, NP, the number of plicae; PP, the proportion of plicae; L1, the length of plicae; L2, the length of ventral or dorsal valve; PHP, the relative height of plicae; H1, the maximum height of plicae; H2, the height of shell. b, Foraminifera, ST, shell thickness, L, shell length, RST, relative shell thickness.
Extended Data Fig. 5 The relationship between the number of plicae and the relative height of plicae.
Red dashed line represents regression line, which is plotted for all species in the Changhsingian.
Supplementary information
Supplementary Information (download PDF )
Supplementary Table 1.
Source data
Source Data Fig. 1 (download XLSX )
Source data of brachiopod plicae and calcification energy.
Source Data Fig. 2 (download XLSX )
Source data of foraminiferal relative shell thickness.
Source Data Fig. 3 (download XLSX )
Source data of foraminiferal calcification energy.
Source Data Extended Data Fig. 1 (download XLSX )
Source data of simulated shell mass of brachiopods.
Source Data Extended Data Fig. 2 (download XLSX )
Source data of foraminiferal shell length and thickness.
Source Data Extended Data Fig. 3 (download XLSX )
Source data of calcium shell mass of living brachiopods and bivalves.
Source Data Extended Data Fig. 5 (download XLSX )
Source data of brachiopod plicae during the Changhsingian.
Source Data Extended Data Table 2 (download XLSX )
Source data of living brachiopod microstructure.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, F., Finnegan, S., Dal Corso, J. et al. Brachiopods and forams reduced calcification costs through morphological simplification during mass extinction events. Nat Ecol Evol 9, 1456–1468 (2025). https://doi.org/10.1038/s41559-025-02749-w
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41559-025-02749-w