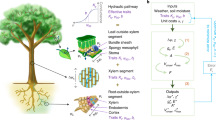
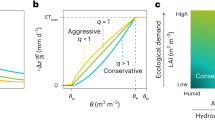
Abstract
Understanding how plants adjust their hydraulic system to the environment is essential to predict how these organisms will respond to global change. Here we compiled a dataset and performed meta-analysis on 223 studies on plastic and evolutionary adjustments of hydraulic traits to air temperature, CO2 concentration, irradiance, soil nutrient and water availability. On average, species plastically increased embolism resistance and sapwood area per leaf area under drier conditions, with a decrease in stem-specific hydraulic conductivity and water potential at the turgor loss point, which are consistent with adaptive responses. However, the average increased embolism resistance was not sufficient to compensate the reduction in the minimum water potential, implying a lower safety margin from lethal hydraulic failure under drought. These results point towards a general critical increase in the risk of hydraulic failure in future drier environments. Plastic responses to increased soil nutrient content and irradiance did not always align with those to drought, highlighting the potential for changes in light and nutrient conditions to modify plant hydraulic responses to climate-change-driven droughts.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The dataset used in this Article with the metadata information is available via Zenodo at https://doi.org/10.5281/zenodo.14582442 (ref. 86).
Code availability
The code used is available via GitHub at https://github.com/ramirezval/Ramirez-Valiente-etal-2025.
Change history
16 September 2025
In the version of the article initially published, the second affiliation of Jordi Martínez-Vilalta was incorrect and has now been amended to affiliation 3 (Universitat Autònoma de Barcelona, Cerdanyola del Vallès, Spain) in the HTML and PDF versions of the article.
References
Vitousek, P. M., Mooney, H. A., Lubchenco, J. & Melillo, J. M. Human domination of Earth’s ecosystems. Science 277, 494–499 (1997).
Sardans, J. et al. Changes in nutrient concentrations of leaves and roots in response to global change factors. Glob. Change Biol. 23, 3849–3856 (2017).
Hammond, W. M. et al. Global field observations of tree die-off reveal hotter-drought fingerprint for Earth’s forests. Nat. Commun. 13, 1761 (2022).
Hoffmann, A. A. & Sgrò, C. M. Climate change and evolutionary adaptation. Nature 470, 479–485 (2011).
Shaw, R. G. & Etterson, J. R. Rapid climate change and the rate of adaptation: insight from experimental quantitative genetics. New Phytol. 195, 752–765 (2012).
Anderegg, W. R. L. Spatial and temporal variation in plant hydraulic traits and their relevance for climate change impacts on vegetation. New Phytol. 205, 1008–1014 (2015).
Sperry, J. S. Hydraulic constraints on plant gas exchange. Agric. For. Meteorol. 104, 13–23 (2000).
McCulloh, K. A., Domec, J., Johnson, D. M., Smith, D. D. & Meinzer, F. C. A dynamic yet vulnerable pipeline: Integration and coordination of hydraulic traits across whole plants. Plant Cell Environ. 42, 2789–2807 (2019).
Torres‐Ruiz, J. M. et al. Plant hydraulics at the heart of plant, crops and ecosystem functions in the face of climate change. New Phytol. 241, 984–999 (2024).
Cochard, H., Pimont, F., Ruffault, J. & Martin-StPaul, N. SurEau: a mechanistic model of plant water relations under extreme drought. Ann. For. Sci. 78, 55 (2021).
Liu, Y., Holtzman, N. M. & Konings, A. G. Global ecosystem-scale plant hydraulic traits retrieved using model–data fusion. Hydrol. Earth Syst. Sci. 25, 2399–2417 (2021).
Rowland, L., Martínez‐Vilalta, J. & Mencuccini, M. Hard times for high expectations from hydraulics: predicting drought‐induced forest mortality at landscape scales remains a challenge. New Phytol. 230, 1685–1687 (2021).
Venturas, M. D., Todd, H. N., Trugman, A. T. & Anderegg, W. R. L. Understanding and predicting forest mortality in the western United States using long‐term forest inventory data and modeled hydraulic damage. New Phytol. 230, 1896–1910 (2021).
Paschalis, A., De Kauwe, M. G., Sabot, M. & Fatichi, S. When do plant hydraulics matter in terrestrial biosphere modelling? Glob. Change Biol. 30, e17022 (2024).
Eller, C. B. et al. Stomatal optimization based on xylem hydraulics (SOX) improves land surface model simulation of vegetation responses to climate. New Phytol. 226, 1622–1637 (2020).
Sabot, M. E. B. et al. Plant profit maximization improves predictions of European forest responses to drought. New Phytol. 226, 1638–1655 (2020).
Koven, C. D. et al. Benchmarking and parameter sensitivity of physiological and vegetation dynamics using the Functionally Assembled Terrestrial Ecosystem Simulator (FATES) at Barro Colorado Island, Panama. Biogeosciences 17, 3017–3044 (2020).
Sperry, J. S. & Tyree, M. T. Mechanism of water stress-induced xylem embolism. Plant Physiol. 88, 581–587 (1988).
Tyree, M. T. & Sperry, J. S. Vulnerability of xylem to cavitation and embolism. Annu. Rev. Plant Physiol. Plant Mol. Biol. 40, 19–36 (1989).
Domec, J., Smith, D. D. & McCulloh, K. A. A synthesis of the effects of atmospheric carbon dioxide enrichment on plant hydraulics: implications for whole‐plant water use efficiency and resistance to drought. Plant Cell Environ. 40, 921–937 (2017).
Liang, X. et al. Stomatal responses of terrestrial plants to global change. Nat. Commun. 14, 2188 (2023).
Zhang, H. et al. Responses of woody plant functional traits to nitrogen addition: a meta-analysis of leaf economics, gas exchange, and hydraulic traits. Front. Plant Sci. 9, 683 (2018).
Lamy, J. et al. Limited genetic variability and phenotypic plasticity detected for cavitation resistance in a Mediterranean pine. New Phytol. 201, 874–886 (2014).
Skelton, R. P. et al. No local adaptation in leaf or stem xylem vulnerability to embolism, but consistent vulnerability segmentation in a North American oak. New Phytol. 223, 1296–1306 (2019).
Fuchs, S., Leuschner, C., Mathias Link, R. & Schuldt, B. Hydraulic variability of three temperate broadleaf tree species along a water availability gradient in central Europe. New Phytol. 231, 1387–1400 (2021).
Martínez‐Vilalta, J. et al. Hydraulic adjustment of Scots pine across Europe. New Phytol. 184, 353–364 (2009).
Bartlett, M. K. et al. Global analysis of plasticity in turgor loss point, a key drought tolerance trait. Ecol. Lett. 17, 1580–1590 (2014).
Mencuccini, M. et al. Leaf economics and plant hydraulics drive leaf: wood area ratios. New Phytol. 224, 1544–1556 (2019).
Huang, R. et al. Robust hydraulic traits correlation in woody species despite large trait variation along natural and experimental environmental gradients. Funct. Ecol. 38, 1835–1847 (2024).
Bartlett, M. K., Klein, T., Jansen, S., Choat, B. & Sack, L. The correlations and sequence of plant stomatal, hydraulic, and wilting responses to drought. Proc. Natl Acad. Sci. USA 113, 13098–13103 (2016).
Rowland, L., Ramírez‐Valiente, J., Hartley, I. P. & Mencuccini, M. How woody plants adjust above‐ and below‐ground traits in response to sustained drought. New Phytol. 239, 1173–1189 (2023).
Challis, A. et al. Adaptive plasticity in plant traits increases time to hydraulic failure under drought in a foundation tree. Tree Physiol. 42, 708–721 (2022).
Ramírez‐Valiente, J. A. et al. Genetically based trait coordination and phenotypic plasticity of growth, gas exchange, allometry, and hydraulics across the distribution range of Pinus pinaster. New Phytol. 246, 984–1000 (2025).
Meinzer, F. C., Johnson, D. M., Lachenbruch, B., McCulloh, K. A. & Woodruff, D. R. Xylem hydraulic safety margins in woody plants: coordination of stomatal control of xylem tension with hydraulic capacitance. Funct. Ecol. 23, 922–930 (2009).
Delzon, S. & Cochard, H. Recent advances in tree hydraulics highlight the ecological significance of the hydraulic safety margin. New Phytol. 203, 355–358 (2014).
Limousin, J. et al. Drought acclimation of Quercus ilex leaves improves tolerance to moderate drought but not resistance to severe water stress. Plant Cell Environ. 45, 1967–1984 (2022).
Choat, B. et al. Global convergence in the vulnerability of forests to drought. Nature 491, 752–755 (2012).
Sanchez-Martinez, P. et al. Increased hydraulic risk in assemblages of woody plant species predicts spatial patterns of drought-induced mortality. Nat. Ecol. Evol. 7, 1620–1632 (2023).
Fichot, R. et al. Common trade-offs between xylem resistance to cavitation and other physiological traits do not hold among unrelated Populus deltoides × Populus nigra hybrids: xylem resistance to cavitation and water relations in poplar. Plant Cell Environ. 33, 1553–1568 (2010).
Sorek, Y., Greenstein, S. & Hochberg, U. Seasonal adjustment of leaf embolism resistance and its importance for hydraulic safety in deciduous trees. Physiol. Plant. 174, e13785 (2022).
Schumann, K., Schuldt, B., Fischer, M., Ammer, C. & Leuschner, C. Xylem safety in relation to the stringency of plant water potential regulation of European beech, Norway spruce, and Douglas-fir trees during severe drought. Trees 38, 607–623 (2024).
Sterck, F. J., Song, Y. & Poorter, L. Drought- and heat-induced mortality of conifer trees is explained by leaf and growth legacies. Sci. Adv. 10, eadl4800 (2024).
Martínez‐Vilalta, J., García‐Valdés, R., Jump, A., Vilà‐Cabrera, A. & Mencuccini, M. Accounting for trait variability and coordination in predictions of drought‐induced range shifts in woody plants. New Phytol. 240, 23–40 (2023).
Valladares, F. et al. The effects of phenotypic plasticity and local adaptation on forecasts of species range shifts under climate change. Ecol. Lett. 17, 1351–1364 (2014).
Des Roches, S. et al. The ecological importance of intraspecific variation. Nat. Ecol. Evol. 2, 57–64 (2017).
Benito Garzón, M., Robson, T. M. & Hampe, A. ΔTrait SDMs: species distribution models that account for local adaptation and phenotypic plasticity. New Phytol. 222, 1757–1765 (2019).
Lamy, J., Plomion, C., Kremer, A. & Delzon, S. QST < FST As a signature of canalization. Mol. Ecol. 21, 5646–5655 (2012).
Torres-Ruiz, J. M. et al. Genetic differentiation in functional traits among European sessile oak populations. Tree Physiol. 39, 1736–1749 (2019).
Hacke, U. G., Spicer, R., Schreiber, S. G. & Plavcová, L. An ecophysiological and developmental perspective on variation in vessel diameter. Plant Cell Environ. 40, 831–845 (2017).
Bartlett, M. K., Scoffoni, C. & Sack, L. The determinants of leaf turgor loss point and prediction of drought tolerance of species and biomes: a global meta‐analysis. Ecol. Lett. 15, 393–405 (2012).
Eziz, A. et al. Drought effect on plant biomass allocation: a meta‐analysis. Ecol. Evol. 7, 11002–11010 (2017).
Poorter, H. et al. Biomass allocation to leaves, stems and roots: meta‐analyses of interspecific variation and environmental control. New Phytol. 193, 30–50 (2012).
Cui, E., Weng, E., Yan, E. & Xia, J. Robust leaf trait relationships across species under global environmental changes. Nat. Commun. 11, 2999 (2020).
Poorter, H. & Nagel, O. The role of biomass allocation in the growth response of plants to different levels of light, CO2, nutrients and water: a quantitative review. Funct. Plant Biol. 27, 1191 (2000).
Cochard, H., Martin, R., Gross, P. & Bogeat‐Triboulot, M. B. Temperature effects on hydraulic conductance and water relations of Quercus robur L. J. Exp. Bot. 51, 1255–1259 (2000).
Zwieniecki, M. A., Boyce, C. K. & Holbrook, N. M. Hydraulic limitations imposed by crown placement determine final size and shape of Quercus rubra L. leaves. Plant Cell Environ. 27, 357–365 (2004).
Carins Murphy, M. R., Jordan, G. J. & Brodribb, T. J. Differential leaf expansion can enable hydraulic acclimation to sun and shade. Plant Cell Environ. 35, 1407–1418 (2012).
Graciano, C., Goya, J. F., Frangi, J. L. & Guiamet, J. J. Fertilization with phosphorus increases soil nitrogen absorption in young plants of Eucalyptus grandis. For. Ecol. Manag. 236, 202–210 (2006).
Villagra, M., Campanello, P. I., Bucci, S. J. & Goldstein, G. Functional relationships between leaf hydraulics and leaf economic traits in response to nutrient addition in subtropical tree species. Tree Physiol. 33, 1308–1318 (2013).
Gorska, A., Zwieniecka, A., Michele Holbrook, N. & Zwieniecki, M. A. Nitrate induction of root hydraulic conductivity in maize is not correlated with aquaporin expression. Planta 228, 989–998 (2008).
Heineman, K. D., Turner, B. L. & Dalling, J. W. Variation in wood nutrients along a tropical soil fertility gradient. New Phytol. 211, 440–454 (2016).
Cunha, H. F. V. et al. Direct evidence for phosphorus limitation on Amazon forest productivity. Nature 608, 558–562 (2022).
Tong, X. et al. Reforestation policies around 2000 in southern China led to forest densification and expansion in the 2010s. Commun. Earth Environ. 4, 260 (2023).
Seidl, R. & Senf, C. Changes in planned and unplanned canopy openings are linked in Europe’s forests. Nat. Commun. 15, 4741 (2024).
Craine, J. M. et al. Isotopic evidence for oligotrophication of terrestrial ecosystems. Nat. Ecol. Evol. 2, 1735–1744 (2018).
Borer, E. T. & Stevens, C. J. Nitrogen deposition and climate: an integrated synthesis. Trends Ecol. Evol. 37, 541–552 (2022).
Wild, M. et al. Global dimming and brightening: an update beyond 2000. J. Geophys. Res. Atmospheres 114, 2008JD011382 (2009).
Matesanz, S. & Ramírez‐Valiente, J. A. A review and meta‐analysis of intraspecific differences in phenotypic plasticity: Implications to forecast plant responses to climate change. Glob. Ecol. Biogeogr. 28, 1682–1694 (2019).
Solé‐Medina, A., Robledo‐Arnuncio, J. J. & Ramírez‐Valiente, J. A. Multi‐trait genetic variation in resource‐use strategies and phenotypic plasticity correlates with local climate across the range of a Mediterranean oak (Quercus faginea). New Phytol. 234, 462–478 (2022).
Gleason, S. M. et al. Weak tradeoff between xylem safety and xylem‐specific hydraulic efficiency across the world’s woody plant species. New Phytol. 209, 123–136 (2016).
Sanchez‐Martinez, P., Martínez‐Vilalta, J., Dexter, K. G., Segovia, R. A. & Mencuccini, M. Adaptation and coordinated evolution of plant hydraulic traits. Ecol. Lett. 23, 1599–1610 (2020).
Isasa, E. et al. Addressing controversies in the xylem embolism resistance–vessel diameter relationship. New Phytol. 238, 283–296 (2023).
Lajeunesse, M. J. in Handbook of Meta-analysis in Ecology and Evolution (eds Koricheva, J. et al.) 195–206 (Princeton Univ. Press, 2013); https://doi.org/10.1515/9781400846184-015
Hedges, L. V., Gurevitch, J. & Curtis, P. S. The meta-analysis of response ratios in experimental ecology. Ecology 80, 1150–1156 (1999).
Viechtbauer, W. Conducting meta-analyses in R with the metafor package. J. Stat. Softw. 36, 1–47 (2010).
R Core Team. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2021).
Viechtbauer, W. Confidence intervals for the amount of heterogeneity in meta‐analysis. Stat. Med. 26, 37–52 (2007).
Collins, R., Yusuf, S. & Peto, R. Overview of randomised trials of diuretics in pregnancy. Br. Med. J. 290, 17–23 (1985).
Adams, D. C., Gurevitch, J. & Rosenberg, M. S. Resampling tests for meta-analysis of ecological data. Ecology 78, 1277–1283 (1997).
Efron, B. Better bootstrap confidence intervals. J. Am. Stat. Assoc. 82, 171–185 (1987).
Deeks, J. J., Higgins, J. P. & Altman, D. G. in Cochrane Handbook for Systematic Reviews of Interventions (eds Higgins, J. P. T. et al.) 241–284 (Wiley, 2019); https://doi.org/10.1002/9781119536604.ch10
Bates, D., Mächler, M., Bolker, B. & Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 67, 1–48 (2015).
Warton, D., Duursma, R., Falster, D. & Taskinen, S. smatr: (Standardised) Major Axis Estimation and Testing Routines. 3.4-8 (R Project, 2006); https://doi.org/10.32614/CRAN.package.smatr
Wickham, H. Ggplot2: Elegant Graphics for Data Analysis (Springer, 2016); https://doi.org/10.1007/978-3-319-24277-4
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B 57, 289–300 (1995).
Ramírez-Valiente, J. A. et al. Data for: Limited adaptive responses in safety traits support greater hydraulic risk under drier conditions. Zenodo https://doi.org/10.5281/zenodo.14582442 (2024).
Acknowledgements
M.M. acknowledges support by the Spanish Ministry of Economy and Competitiveness (MINECO) via grant CGL2017-89149-C2-1-R (DRESS) and EU-2020 programme via grant 862221 (FORGENIUS). J.A.R.-V. was supported by a research contract financed by the AEI (Spanish Research Agency) and Ministry of Science through the Severo Ochoa Program for Centres of Excellence in R+D+I (CEX2018-000828-S) and competitive grants TED2021-129570B-I00 and PID2021-126399NB-I00. J.M.-V. acknowledges support from competitive grants CGL2017-89149-C2-1-R and PID2021-127452NB-I00 funded by MCIN/AEI (https://doi.org/10.13039/501100011033), by grant 2021 SGR 00849 funded by AGAUR and by an ICREA Academia award.
Author information
Authors and Affiliations
Contributions
R.P., C.J.B., A.C., E.C., H.C., D.C., S.D., R.G.-V., J.-M.L., R.L., N.M.-S. My. Mo., L.S.S., B.S., J.M.T.-R., A.V., J.M.-V. and Ma. Me. conceptualized the idea and compiled the database. J.A.R.-V. and R.P. conducted the analyses. J.A.R.-V. wrote the first draft, and R.P., C.J.B., A.C., E.C., H.C., D.C., S.D., R.G.-V., J.-M.L., R.L., N.M.-S. My. Mo., L.R., L.S.S., B.S., J.M.T.-R., A.V., J.M.-V. and Ma. Me contributed with substantial revisions.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Ecology & Evolution thanks Mauro Santos and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Table 1, Figs. 1–8 and Methods 1.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ramírez-Valiente, J.A., Poyatos, R., Blackman, C.J. et al. Limited plastic responses in safety traits support greater hydraulic risk under drier conditions. Nat Ecol Evol 9, 1825–1836 (2025). https://doi.org/10.1038/s41559-025-02830-4
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41559-025-02830-4