Abstract
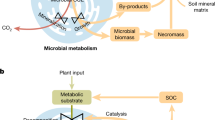
The extent to which microbial processes control soil organic carbon (SOC) dynamics remains uncertain. Carbon use efficiency (CUE), that is, the fraction of assimilated carbon allocated to growth, has been used as a key parameter but its relationship with SOC reflects carbon partitioning rather than the absolute magnitude of microbial fluxes. The microbial growth rate could provide a more mechanistic link to SOC accumulation because it quantifies biomass production and reflects necromass formation. Here we combine a global ¹⁸O–H2O dataset (n = 268 paired observations) with outputs from four land surface models to test whether growth rate predicts SOC more strongly than CUE. In the incubation experiments, growth rates are more closely associated with SOC than CUE, although soil properties and climate explain equal or greater variance. Models reproduce the stronger role of growth rate over CUE but tend to underestimate the abiotic controls. The models also emphasize CUE as the main predictor of the SOC-to-net primary production ratio, in contrast to observations, which indicates the soil’s capacity to retain plant carbon inputs. Together, these findings identify the microbial growth rate as a diagnostic that can help bridge models with empirical data and guide a more balanced representation of microbial and mineral controls in SOC projections.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The global observational dataset is available via figshare at https://doi.org/10.6084/m9.figshare.30070084.v1 (ref. 58). Simulation outputs from the four land surface models (ORCHIDEE-CENTURY, ORCHIDEE-MIMICS, CABLE-CASA and JULES-RothC) are also available via figshare at https://figshare.com/s/95f8435036d7f6825a53 (ref. 69). All custom R scripts used for data processing, statistical analyses and figure generation are openly available via Zenodo at https://doi.org/10.5281/zenodo.17800780 (ref. 70).
References
Janzen, H. H. The soil carbon dilemma: shall we hoard it or use it? Soil Biol. Biochem. 38, 419–424 (2006).
Kuzyakov, Y., Ling, N., Pietramellara, G. & Nannipieri, P. Some new grand questions in soil biology and biochemistry. Soil Biol. Biochem. 212, 109996 (2026).
Xu, X., Thornton, P. E. & Post, W. M. A global analysis of soil microbial biomass carbon, nitrogen and phosphorus in terrestrial ecosystems. Glob. Ecol. Biogeogr. 22, 737–749 (2013).
Angst, G., Mueller, K. E., Nierop, K. G. J. & Simpson, M. J. Plant- or microbial-derived? A review on the molecular composition of stabilized soil organic matter. Soil Biol. Biochem. 156, 108189 (2021).
Liang, C., Amelung, W., Lehmann, J. & Kästner, M. Quantitative assessment of microbial necromass contribution to soil organic matter. Glob. Change Biol. 25, 3578–3590 (2019).
Camenzind, T., Mason-Jones, K., Mansour, I., Rillig, M. C. & Lehmann, J. Formation of necromass-derived soil organic carbon determined by microbial death pathways. Nat. Geosci. 16, 115–122 (2023).
Liang, C., Schimel, J. P. & Jastrow, J. D. The importance of anabolism in microbial control over soil carbon storage. Nat. Microbiol. 2, 17105 (2017).
Xiao, K.-Q. et al. Introducing the soil mineral carbon pump. Nat. Rev. Earth Environ. 4, 135–136 (2023).
Allison, S. D., Wallenstein, M. D. & Bradford, M. A. Soil-carbon response to warming dependent on microbial physiology. Nat. Geosci. 3, 336–340 (2010).
Tang, J. & Riley, W. J. Weaker soil carbon–climate feedbacks resulting from microbial and abiotic interactions. Nat. Clim. Change 5, 56–60 (2015).
Walker, T. W. N. et al. Microbial temperature sensitivity and biomass change explain soil carbon loss with warming. Nat. Clim. Change 8, 885–889 (2018).
Tao, F. et al. Microbial carbon use efficiency promotes global soil carbon storage. Nature 618, 981–985 (2023).
He, X. et al. Model uncertainty obscures major driver of soil carbon. Nature 627, E1–E3 (2024).
Xiao, K.-Q. et al. Beyond microbial carbon use efficiency. Natl Sci. Rev. 11, nwae059 (2024).
Craig, M. E. et al. Fast-decaying plant litter enhances soil carbon in temperate forests but not through microbial physiological traits. Nat. Commun. 13, 1229 (2022).
Hu, J. et al. Microbial carbon use efficiency and growth rates in soil: global patterns and drivers. Glob. Change Biol. 31, e70036 (2025).
Liu, M., Lin, H. & Li, J. Are there links between nutrient inputs and the response of microbial carbon use efficiency or soil organic carbon? A meta-analysis. Soil Biol. Biochem. 201, 109656 (2025).
Liu, X. et al. Long-term soil warming decreases soil microbial necromass carbon by adversely affecting its production and decomposition. Glob. Change Biol. 30, e17379 (2024).
Luo, Z., Ren, J., Manzoni, S. & Fatichi, S. Temperature controls the relation between soil organic carbon and microbial carbon use efficiency. Glob. Change Biol. 30, e17492 (2024).
Schroeder, J. et al. The effect of crop diversification and season on microbial carbon use efficiency across a European pedoclimatic gradient. Eur. J. Soil Sci. 76, e70078 (2025).
Yang, Y. et al. Unlocking mechanisms for soil organic matter accumulation: carbon use efficiency and microbial necromass as the keys. Glob. Change Biol. 31, e70033 (2025).
Zhao, J. et al. Mineral and microbial properties drive the formation of mineral-associated organic matter and its response to increased temperature. Glob. Change Biol. 30, e70004 (2024).
Gao, D., Bai, E., Wasner, D. & Hagedorn, F. Global prediction of soil microbial growth rates and carbon use efficiency based on the metabolic theory of ecology. Soil Biol. Biochem. 190, 109315 (2024).
García-Palacios, P. et al. Dominance of particulate organic carbon in top mineral soils in cold regions. Nat. Geosci. 17, 145–150 (2024).
Varney, R. M. et al. A spatial emergent constraint on the sensitivity of soil carbon turnover to global warming. Nat. Commun. 11, 5544 (2020).
Fan, X. et al. Improved model simulation of soil carbon cycling by representing the microbially derived organic carbon pool. ISME J. 15, 2248–2263 (2021).
Simon, E. et al. Microbial growth and carbon use efficiency show seasonal responses in a multifactorial climate change experiment. Commun. Biol. 3, 584 (2020).
Yin, L. et al. Variation in rhizosphere priming and microbial growth and carbon use efficiency caused by wheat genotypes and temperatures. Soil Biol. Biochem. 134, 54–61 (2019).
Zhang, Q. et al. Effect of field warming on soil microbial carbon use efficiency—a meta-analysis. Soil Biol. Biochem. 197, 109531 (2024).
Zheng, Q. et al. Growth explains microbial carbon use efficiency across soils differing in land use and geology. Soil Biol. Biochem. 128, 45–55 (2019).
Zhou, J., Luo, Y. & Chen, J. Dilemmas in linking microbial carbon use efficiency with soil organic carbon dynamics. Glob. Change Biol. 31, e70047 (2025).
Metze, D. et al. Microbial growth under drought is confined to distinct taxa and modified by potential future climate conditions. Nat. Commun. 14, 5895 (2023).
Qu, L., Wang, C. & Bai, E. Evaluation of the 18O-H2O incubation method for measurement of soil microbial carbon use efficiency. Soil Biol. Biochem. 145, 107802 (2020).
Wasner, D. et al. Environment and microbiome drive different microbial traits and functions in the macroscale soil organic carbon cycle. Glob. Change Biol. 30, e17465 (2024).
Whalen, E. D., Grandy, A. S., Geyer, K. M., Morrison, E. W. & Frey, S. D. Microbial trait multifunctionality drives soil organic matter formation potential. Nat. Commun. 15, 10209 (2024).
Fang, L. Multifaceted links between microbial carbon use efficiency and soil organic carbon sequestration. Glob. Change Biol. 31, e70045 (2025).
He, X. et al. Emerging multiscale insights on microbial carbon use efficiency in the land carbon cycle. Nat. Commun. 15, 8010 (2024).
Geyer, K. M., Dijkstra, P., Sinsabaugh, R. & Frey, S. D. Clarifying the interpretation of carbon use efficiency in soil through methods comparison. Soil Biol. Biochem. 128, 79–88 (2019).
Manzoni, S. & Cotrufo, M. F. Mechanisms of soil organic carbon and nitrogen stabilization in mineral-associated organic matter—insights from modeling in phase space. Biogeosciences 21, 4077–4098 (2024).
Spohn, M. et al. Soil microbial carbon use efficiency and biomass turnover in a long-term fertilization experiment in a temperate grassland. Soil Biol. Biochem. 97, 168–175 (2016).
Georgiou, K. et al. Divergent controls of soil organic carbon between observations and process-based models. Biogeochemistry 156, 5–17 (2021).
Dangal, S. R. S. et al. Improving soil carbon estimates by linking conceptual pools against measurable carbon fractions in the DAYCENT model version 4.5. J. Adv. Model. Earth Syst. 14, e2021MS002622 (2022).
Goll, D. S. et al. Strong dependence of CO2 emissions from anthropogenic land cover change on initial land cover and soil carbon parametrization. Glob. Biogeochem. Cycles 29, 1511–1523 (2015).
Xu, H., Zhang, T., Luo, Y., Huang, X. & Xue, W. Parameter calibration in global soil carbon models using surrogate-based optimization. Geosci. Model Dev. 11, 3027–3044 (2018).
Varney, R. M., Chadburn, S. E., Burke, E. J. & Cox, P. M. Evaluation of soil carbon simulation in CMIP6 Earth system models. Biogeosciences 19, 4671–4704 (2022).
Georgiou, K., Abramoff, R. Z., Harte, J., Riley, W. J. & Torn, M. S. Microbial community-level regulation explains soil carbon responses to long-term litter manipulations. Nat. Commun. 8, 1223 (2017).
Wutzler, T. & Reichstein, M. Colimitation of decomposition by substrate and decomposers—a comparison of model formulations. Biogeosciences 5, 749–759 (2008).
Sulman, B. N. et al. Multiple models and experiments underscore large uncertainty in soil carbon dynamics. Biogeochemistry 141, 109–123 (2018).
Abramoff, R. Z. et al. Improved global-scale predictions of soil carbon stocks with Millennial Version 2. Soil Biol. Biochem. 164, 108466 (2022).
Wieder, W. R., Grandy, A. S., Kallenbach, C. M. & Bonan, G. B. Integrating microbial physiology and physio-chemical principles in soils with the MIcrobial-MIneral Carbon Stabilization (MIMICS) model. Biogeosciences 11, 3899–3917 (2014).
Tang, J. & Riley, W. J. Revising the dynamic energy budget theory with a new reserve mobilization rule and three example applications to bacterial growth. Soil Biol. Biochem. 178, 108954 (2023).
Migliavacca, M. et al. The three major axes of terrestrial ecosystem function. Nature 598, 468–472 (2021).
Marschmann, G. L. et al. Predictions of rhizosphere microbiome dynamics with a genome-informed and trait-based energy budget model. Nat. Microbiol. 9, 421–433 (2024).
Piton, G. et al. Life history strategies of soil bacterial communities across global terrestrial biomes. Nat. Microbiol. 8, 2093–2102 (2023).
Wei, N. et al. Evolution of uncertainty in terrestrial carbon storage in earth system models from CMIP5 to CMIP6. J. Clim. 35, 5483–5499 (2022).
Carvalhais, N. et al. Global covariation of carbon turnover times with climate in terrestrial ecosystems. Nature 514, 213–217 (2014).
Minasny, B. et al. Soil carbon 4 per mille. Geoderma 292, 59–86 (2017).
Hu, J. Database of microbial growth rate outweighs carbon use efficiency in shaping soil carbon dynamics. Dataset. figshare https://doi.org/10.6084/m9.figshare.30070084.v1 (2025).
Running, S. & Zhao, M. MODIS/Terra Net Primary Production Gap-Filled Yearly L4 Global 500m SIN Grid V061. NASA Land Processes Distributed Active Archive Center https://doi.org/10.5067/MODIS/MOD17A3HGF.061 (2021).
Friedlingstein, P. et al. Global carbon budget 2023. Earth Syst. Sci. Data 15, 5301–5369 (2023).
Parton, W. J., Stewart, J. W. B. & Cole, C. V. Dynamics of C, N, P and S in grassland soils: a model. Biogeochemistry 5, 109–131 (1988).
Zhang, H. et al. Microbial dynamics and soil physicochemical properties explain large-scale variations in soil organic carbon. Glob. Change Biol. 26, 2668–2685 (2020).
Potter, C. S. et al. Terrestrial ecosystem production: a process model based on global satellite and surface data. Glob. Biogeochem. Cycles 7, 811–841 (1993).
Coleman, K. & Jenkinson, D. S. in Evaluation of Soil Organic Matter Models (eds Powlson, D. S. et al.) 237–246 (Springer, 1996).
Hengl, T. et al. SoilGrids250m: global gridded soil information based on machine learning. PLoS ONE 12, e0169748 (2017).
Frey, S. D., Lee, J., Melillo, J. M. & Six, J. The temperature response of soil microbial efficiency and its feedback to climate. Nat. Clim. Change 3, 395–398 (2013).
Georgiou, K., Koven, C. D., Riley, W. J. & Torn, M. S. Toward improved model structures for analyzing priming: potential pitfalls of using bulk turnover time. Glob. Change Biol. 21, 4298–4302 (2015).
Breiman, L., Cutler, A., Liaw, A. & Wiener, M. randomForest: Breiman and Cutlers random forests for classification and regression. R package version 4.7-1.2 https://doi.org/10.32614/CRAN.package.randomForest (2002).
Microbial growth better predicts soil carbon than carbon use efficience. figshare https://figshare.com/s/95f8435036d7f6825a53 (2025).
He, X. Microbial growth rate is a stronger predictor of soil organic carbon than carbon use efficiency. Zenodo https://doi.org/10.5281/zenodo.17800781 (2025).
Acknowledgements
We thank Y. Xi and C. Zhou (Laboratoire des Sciences du Climat et de l’Environnement) for their valuable feedback during manuscript preparation and revision. This study was supported by the CALIPSO project funded by Schmidt Sciences. G.M. and D.S.G. acknowledge support from the EJP Soil ICONICA project. E.S. and S.M. were supported by the European Research Council under the European Union’s Horizon 2020 Research and Innovation Programme (grant no. 101001608). H.Z. was supported by the National Natural Science Foundation of China (grant no. 41030052). J.H. was supported by the Natural Science Foundation of Sichuan Province (grant no. 2025ZNSFSC1033) and the Postdoctoral Fellowship Program of the China Postdoctoral Science Foundation (grant no. GZB20250584). Support for Y.-P.W. was provided in part by the Terrestrial Ecosystem Research Network, an Australian Government National Collaborative Research Infrastructure Strategy-enabled project.
Author information
Authors and Affiliations
Contributions
X.H., G.M. and D.S.G. conceived and designed the study. J.H. provided the observational dataset. S.M., P.C., Y.-P.W., R.Z.A. and E.A. contributed to the initial development of the study concept and discussions. Y.C. and E.B. supplied the data that supported the preliminary exploratory analyses. P.C. and D.S.G. secured the project funding. All authors contributed to the writing, discussion and revision of the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Ecology & Evolution thanks Luiz A. Domeignoz-Horta and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
He, X., Marmasse, G., Hu, J. et al. Microbial growth rate is a stronger predictor of soil organic carbon than carbon use efficiency. Nat Ecol Evol 10, 372–381 (2026). https://doi.org/10.1038/s41559-025-02961-8
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41559-025-02961-8
This article is cited by
-
Discrepancies between observations and models highlight how to improve predictions
Nature Ecology & Evolution (2026)