Abstract
The functioning of high-diversity ecosystems, such as coral reefs, is intrinsically tied to the integrity and efficiency of the trophic pathways within these systems. Coral reef productivity depends, in part, on the input of external nutrients, primarily zooplankton, that is assimilated by extraordinarily diverse fish communities. The plankton–planktivore trophic pathway is thus crucial for sustaining the productivity that exemplifies coral reef ecosystems; however, it remains poorly understood at large spatial scales. Here we explore global patterns in reef fish community structure, revealing a major discrepancy between the Indo-Pacific and Caribbean in the productivity and fisheries potential of planktivorous reef fishes. Indo-Pacific reefs support 6.6 times more planktivorous fish biomass and 3.4 times greater productivity than the Caribbean, a difference largely due to the marked contribution of species that feed on gelatinous plankton in the Indo-Pacific. Although species that feed on gelatinous plankton constitute only 4% of the planktivorous fish abundance in the Indo-Pacific, they account for one-third of the biomass and one-quarter of the productivity. This divergence reflects the contrasting biogeographic histories of the two realms, with Indo-Pacific oceanography fostering diversification, while repeated extinction events and trophic erosion may have constrained planktivory in the Caribbean. Ultimately, these differences in energy flow translate into fundamental differences in coral reef functioning and, potentially, their capacity to support ecosystem services, including fisheries.
Similar content being viewed by others
Main
Ecosystems often rely on external energy sources to sustain populations, with energy mediated through trophic connections linking donor and recipient systems1. In tropical marine environments, energy frequently flows from the open ocean into continental shelf ecosystems, where it supplements and enhances productivity2,3,4. Coral reefs exemplify this dynamic: they are among the most productive ecosystems on Earth, yet their productivity is heavily dependent on pelagic subsidies, particularly zooplankton1,2,3,5,6. Despite their importance, the link between ecosystem productivity—the rate at which biomass is generated—and the assimilation of the pelagic subsidies that fuel productivity remains largely unresolved.
Pelagic subsidies are assimilated into coral reef ecosystems through feeding by planktivores, including corals and fishes7,8,9,10. Planktivorous fishes are especially prominent, often dominating reef fish assemblages. In the Indo-Pacific, they can account for to 90% of all surveyed fishes1. The plankton–planktivore pathway is, therefore, central to the productivity of coral reefs1,2,3,5,6. However, despite frequently being considered a uniform group (that is, ‘planktivores’), detailed small-scale studies have revealed that planktivorous fishes do not consume incoming plankton indiscriminately5,11,12. Instead, fishes select and consume specific components of the incoming plankton assemblage, with a marked division between species that feed on gelatinous versus non-gelatinous plankton5,11,12.
Tropical near-reef zooplankton communities are typically dominated by five broad taxonomic groups—copepods (~60%), appendicularians (13.2%), chaetognaths (11.3%), non-copepod crustaceans (10.2%) and thaliaceans (4.4%)—which together account for ~98% of the assemblage13,14,15,16. Gelatinous zooplankton, such as appendicularians, thaliaceans and cnidarians, are characterized by very high body-water content17,18, whereas non-gelatinous taxa, including copepods, chaetognaths and euphausiids, have a comparatively lower body-water content19. Despite their apparent lower nutritional content, gelatinous taxa are readily consumed by planktivorous fishes, with the advantage of a gelatinous diet likely related to digestibility, prey size, and their capacity for rapid, high-abundance blooms20,21. Yet, the broader implications of this prey selectivity for coral reef productivity remains unexplored.
Here, we demonstrate that the selection and use of different trophic resources may have far-reaching effects that reverberate through entire food chains. By combining the extensive Reef Life Survey (RLS) dataset of underwater visual surveys22, encompassing over 2.5 million planktivorous fish observations, we explore the energetic potential of planktivorous reef fish assemblages. Using these data to compare two major coral reef realms (the Indo-Pacific versus the Caribbean; Fig. 1a) we reveal a major, global, between-realm division in planktivorous fish productivity, largely driven by the use of gelatinous pelagic subsidies.
a, Survey locations (n = 1,131) mapped using an Eckert IV projection, with points jittered for clarity. Points are coloured and shaped by realm, with blue circles representing the Indo-Pacific and brown triangles representing the Caribbean. b,c, Fish biomass (b) and fish productivity (c). Circles represent the median value of the posterior distribution; thick coloured bars indicate the 50% credible intervals, and thin coloured bars indicate the 95% credible intervals. Sites: Indo-Pacific n = 1,052, and Caribbean n = 79. Each plot (b and c) shows the PP of the Indo-Pacific (IP) being greater than the Caribbean (Car). d,e, Examples of planktivorous fish species from each realm, Caesio cuning, (Lutjanidae) (d) and Chromis multilineata (Pomacentridae) (e). Photographs in d and e courtesy of François Libert, under a Creative Commons licence CC BY-NC-SA 2.0.
Results
Between-realm differences
We investigated differences in the plankton–planktivore trophic pathway between two of the major coral reef biogeographic realms, the Indo-Pacific and Caribbean23,24. These two realms have a long history of direct comparisons in terms of trophic structure25, benthic processes and ecosystem change24,26, and species diversity27, reflecting their distinct biogeographic histories. Initially, we explored variation in planktivorous reef fish density, biomass and productivity between these two realms using individual Bayesian generalized linear mixed-effects models (GLMMs). We found that, while planktivore density in the Indo-Pacific was on average 2.2 times greater than in the Caribbean, there was only moderate evidence to support this difference (posterior probability (PP) 87.0%; Extended Data Fig. 1). However, there was strong evidence in support of higher biomass and productivity on Indo-Pacific reefs compared with the Caribbean reefs (PP 96.9% and 91.7%, respectively; Fig. 1b,c). The biomass and productivity of planktivorous reef fish assemblages was 6.6 and 3.4 times greater on Indo-Pacific reefs compared with Caribbean reefs, respectively (Fig. 1b,c). In addition, ecoregion-level analyses show that these trends were largely consistent at this smaller spatial scale between realms (Supplementary Text 1 and Supplementary Fig. 1).
To further disentangle the basis of these differences in planktivore density, biomass and productivity between the two realms, we specifically evaluated how these metrics were associated with planktivore size distributions. Body size influences predation on zooplankton and the assimilation and transfer of off-reef nutrients to the on-reef energy budget14,28,29. Therefore, variations in fish size spectra may reveal differences in how the plankton–planktivore trophic pathway operates in each realm. Using additional Bayesian GLMMs, we modelled variations in the density, biomass and productivity of planktivorous reef fishes across size spectra in each realm. Our analyses revealed a notable divergence: Caribbean planktivores were increasingly rare above 10 cm in total length compared with their Indo-Pacific counterparts (Fig. 2a–c). This suggests that the functional capacity of Caribbean planktivores to contribute to total biomass and productivity may be more limited than in the Indo-Pacific.
a–c, Density (a), biomass (b) and productivity (c). Black lines represent the mean prediction from the posterior distribution, with the shaded ribbons representing the 95% credible intervals. Note both axes are on the log10 scale, and the x axis is restricted to observed realm-size distribution. All figures are coloured by realm: blue for Indo-Pacific and brown for Caribbean. Within-site size-class observations: Indo-Pacific n = 5,857, and Caribbean n = 234.
The role of trophic resource division
To further explore differences in how the plankton–planktivore trophic pathway functions between realms, and to provide insights into the potential drivers of between-realm differences, we focused on a major division in plankton use: gelatinous versus non-gelatinous planktivory. Using species identification guides30,31,32 and dietary information reported in FishBase33,34, along with published gut content information11,12,35,36,37,38,39,40,41,42, we categorized 336 planktivorous fish species as primarily gelatinous (n = 16) or non-gelatinous feeders (n = 320) (Supplementary Data 1). In doing so, we revealed that gelatinous planktivory is an entirely Indo-Pacific phenomenon. No Caribbean species were identified to primarily consume gelatinous zooplankton (Supplementary Data 1). To ensure the robustness of this classification, we also conducted a non-conservative analysis categorizing species where genus-level and prey preference data were lacking based on phylogenetic similarity (Methods). This alternative approach resulted in 278 non-gelatinous and 50 gelatinous feeders. Nevertheless, it produced similar patterns (Extended Data Figs. 2–4), providing strong support for the reliability of the conservative results (Methods).
Using Bayesian GLMMs, we also modelled the influence of prey preference (that is, gelatinous versus non-gelatinous) on Indo-Pacific planktivorous fish contributions to density, biomass and productivity (Fig. 3a,c,e). In the Indo-Pacific, gelatinous planktivores averaged only 5.4 individuals per 250 m−2 (median 5.0, highest posterior density (HPD) 1.9–10.5) compared with non-gelatinous planktivores at 142.4 individuals per 250 m−2 (median 130.3, HPD 48.7–274.1) (Fig. 3a), a 26.4-fold difference. However, these density values conceal the contribution of gelatinous planktivores to energetics. Despite accouting for only 4% of all individuals in the Indo-Pacific, the gelatinous planktivores account for 32.5% and 24.2% of all biomass stored and produced, respectively, by planktivorous fishes in the Indo-Pacific. Furthermore, size-spectra analysis revealed a clear dichotomy in the contributions of the two planktivore trophic groups: gelatinous planktivores clearly dominate density, biomass and productivity at fish body sizes above 20–30 cm (Fig. 3d–f). Ecoregion-level analyses revealed the persistence of these patterns across the Indo-Pacific (Supplementary Text 1 and Supplementary Fig. 2); one of the key contributors to Indo-Pacific reef processes is large-body fishes that feed on gelatinous plankton.
a–f, Density (a), biomass (c) and productivity (e), as well as their corresponding predicted total length size distributions (b, d and f). In a, c and e, circles represent the median value of the posterior distribution, thick coloured bars represent the 50% credible intervals, and thin coloured bars represent the 95% credible intervals. Each plot (a, c and e) shows the PP of non-gelatinous planktivores (NG) being greater than the gelatinous planktivores (G). In b, d and f, black lines represent the mean prediction from the posterior distribution, with the shaded ribbons representing the 95% credible intervals. Note that both axes are on the log10 scale, and the x axis is restricted to observed planktivore size distributions. All figures are coloured by primary prey type: red for non-gelatinous and purple for gelatinous. Values do not sum directly to those in Fig. 1, as these panels represent a subset of the data (Indo-Pacific gelatinous versus non-gelatinous planktivores). This division introduces additional zero values where one group is absent, lowering both means and medians. Sites: in a, c and e, non-gelatinous n = 1,052, and gelatinous n = 1,052, and within-site size-class observations: in b, d and f, non-gelatinous n = 5,121, and gelatinous n = 2,135.
The different contributions of planktivores that feed on gelatinous versus non-gelatinous plankton to biomass and productivity highlighted the importance of gelatinous-feeding planktivores in the Indo-Pacific. To investigate the extent of this role, we reran the analyses standardizing by density to model the per-capita contribution to biomass and productivity between realms, and between Indo-Pacific gelatinous and non-gelatinous planktivores. These analyses revealed strong evidence that planktivorous fishes in the Indo-Pacific contribute 4.3 times more biomass and are 2.3 times more productive on a per-capita level than their Caribbean counterparts (PP 96% and 90.3%, respectively; Fig. 4a,b). Furthermore, when prey preference in the Indo-Pacific was investigated, we found decisive evidence that gelatinous planktivores have greater per-capita biomass and productivity than non-gelatinous planktivores (PP 100%), with gelatinous-feeding individuals representing 7.3 times more biomass and producing 5.7 times more biomass than non-gelatinous-feeding fishes (Fig. 4c,d).
a–d, Comparison of the per-capita contribution to biomass (a) and productivity (b) across the Indo-Pacific and Caribbean and prey preference in the Indo-Pacific (biomass (c) and productivity (d)). Circles represent the median value from the posterior distribution; thick coloured bars represent the 50% credible intervals, and thin coloured bars represent the 95% credible intervals. Each plot shows the PP of the Indo-Pacific (IP) being greater than the Caribbean (Car) (a and b) and non-gelatinous planktivores (NG) being greater than the gelatinous planktivores (G) (c and d). Panels a and b are coloured by realm: blue for the Indo-Pacific and brown for the Caribbean, while panels c and d are coloured by primary prey type: red for non-gelatinous and purple for gelatinous. Sites: Indo-Pacific n = 1,052, and Caribbean n = 79 (a and b), and non-gelatinous n = 1,052, and gelatinous n = 1,052 (c and d).
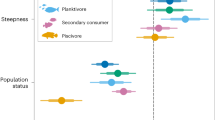
Finally, we investigated potential taxonomic differences that may influence the between-realm dichotomy. We used Bayesian GLMMs to separately model how realm-specific planktivore density, biomass and productivity varied at the family level (Supplementary Tables 2 and 3). Using the top five families by biomass in each realm, we calculated the relative contribution to biomass and productivity (Fig. 5). We revealed that, in the Indo-Pacific, planktivore biomass is dominated by the contribution of Lutjanidae (including caesionines43), while in the Caribbean, biomass is mainly composed of smaller members of the family Labridae. In the Indo-Pacific, larger-bodied families (that is, Lutjanidae and Acanthuridae) are primarily composed of gelatinous planktivores (Supplementary Data 1). Meanwhile, in the Caribbean there are no planktivorous acanthurids and only one species of non-gelatinous planktivorous lutjanid (Ocyurus chrysurus; Supplementary Data 1). Critically, the scarcity of larger-bodied planktivores in the Caribbean appears to limit the assimilation of pelagic subsidies, with the bulk of the functional capacity restricted to smaller planktivores such as Labridae and Pomacentridae.
a,b, The median relative contribution to total planktivore biomass (solid) and productivity (outline) of the top five fish families (based on biomass) in the Indo-Pacific (blue) (a) and Caribbean (brown) (b). Family silhouettes indicate the top five planktivorous fish families (biomass). Note that values represent the standardized median relative contributions, calculated as a proportion of total planktivore biomass and productivity within each region. See Supplementary Data 2 and Supplementary Table 1 for non-standardized values.
Discussion
We reveal how a fundamental trophic division in planktivorous fishes underpins the relatively limited productivity of planktivorous fish communities on Caribbean reefs. In the Caribbean, reefs appear to lack gelatinous-feeding planktivorous fishes and exhibit planktivore biomass and productivity levels that are only 15% and 19%, respectively, of those estimated for Indo-Pacific reefs. By contrast, despite accounting for just 4% of all individuals on Indo-Pacific reefs, gelatinous-feeding planktivores disproportionately contributed to biomass and productivity, accounting for nearly a third of the total accumulated planktivore biomass and a quarter of the total planktivore productivity. The importance of gelatinous planktivory to Indo-Pacific reefs may be explained through the alternate size spectra of non-gelatinous and gelatinous planktivores, with the former predominantly composed of smaller (<20 cm) individuals. Ultimately, these differences in energy flow between realms translate into fundamental differences in coral reef functioning and, potentially, their capacity to support ecosystem services, including fisheries.
The capacity for gelatinous-feeding planktivores to disproportionately contribute to reef productivity is likely underpinned, at least in part, by the nature of their target food resources. Gelatinous zooplankton, such as appendicularians and thaliaceans19, have historically been viewed as an inferior nutritional resource compared to non-gelatinous plankton44. However, recent evidence suggests that the two major plankton-based food resources are nutritionally comparable based on standardized dry weight14. Yet, gelatinous zooplankton possess two other key traits: (1) it is generally larger14,17, and (2) it has a very high body-water content14,17,18,19. The large size of gelatinous plankton means it is readily visible to fishes, facilitating targeted consumption even by larger individuals45. Moreover, the high body-water content facilitates faster digestion in comparison with non-gelatinous taxa46 (over 20 times greater47). Indeed, rapid digestion promotes postprandial (that is, after-meal) appetite revival, which may facilitate elevated ingestion rates of gelatinous prey and a greater uptake of nutrients in comparison with non-gelatinous zooplankton48. These plankton traits potentially underpin a positive feedback loop, wherein a gelatinous diet facilitates growth to larger body mass, which in turn allows these planktivores to feed further off-reef, granting them access to greater quantities of gelatinous plankton, further enabling growth5. However, confirming this would require growth rate trials across planktivorous reef fish species, probably involving gelatinous zooplankton culturing for live feeding. Indeed, future research is required to uncover the mechanistic basis of this global-scale pattern. Regardless of the mechanism, a select group of fishes that target gelatinous zooplankton contribute disproportionately to productivity on Indo-Pacific reefs, with this contribution underpinning high planktivore productivity relative to Caribbean reefs.
This discrepancy raises the question: if gelatinous planktivory is such a successful strategy, why are there no predominantly gelatinous planktivores in the Caribbean? Zooplankton appears to be a readily abundant food source available across all marine environments5,49,50,51, with the non-gelatinous component accounting for over 80% and 88% of the zooplankton community and the gelatinous one accounting for less than 20% and 12% in the Indo-Pacific and Caribbean, respectively13,16. Given that gelatinous plankton is available to Caribbean planktivores, the answer probably lies in the disparate biogeographic history of the two realms. In brief, the Indo-Pacific and Caribbean were formed following the Terminal Tethyan Event and the closure of the Isthmus of Panama, which split the circumglobal tropics into east and west realms (presently the Indo-Pacific and Atlantic)52. This catalysed either speciation or extinction in these newly formed realms27,53. In the Indo-Pacific, this oceanographic reshuffle encompassed areas of complex bathymetry (especially in the Indo-Australia Archipelago), where interactions among currents, island geometry and vertical mixing processes promote a constant flow of abundant planktonic resources to Indo-Pacific reefs43,54,55. These ‘sweet spots’ likely sparked the diversification and ecological dominance (in terms of density in fish communities) observed in Indo-Pacific planktivores1,27.
By contrast, the Caribbean inherited a relatively simple oceanography, with less complex island archipelagos, and water movement generally restricted to a unidirectional flow from southeast to northwest56. This less dynamic oceanography likely results in lower production or a less consistent supply of plankton. When combined with other differences in contextual constraints (for example, enhanced iron inputs in the Caribbean enhancing benthic macroalgae dominance24), differences in plankton production or supply could have driven the evolution of Caribbean fishes towards benthic rather than planktonic trophic pathways. Instead of the planktivorous fish diversification seen in the Indo-Pacific, Caribbean planktivores have endured 10 million years of environmental changes, consistently punctuated by extinction events37,57. These events probably eroded the trophic foundation inherited from the ancient Tethys Sea, which is typified in this case by the presence of an extinct unicornfish (Eonaso58) that potentially could have been a gelatinous zooplanktivore based on the diets of some unicornfish in the Indo-Pacific12. The current species richness of the Caribbean planktivorous fish assemblage is less than 6% that of the Indo-Pacific (Supplementary Data 1). Indeed, gelatinous planktivory is just one of a number of trophic links missing or degraded in the Caribbean (for example, corals and herbivorous fishes59,60).
The broader utilization of pelagic energy by fishes on Indo-Pacific reefs potentially buffers the energetic demands of higher trophic groups, distributing energy flow more evenly across trophic pathways and enhancing overall ecosystem stability and resilience compared to the Caribbean61,62,63. Indeed, the relative reduction of pelagic nutrients assimilated by Caribbean reef fish communities through the planktivore pathway has the potential to place additional pressure on the services provided by other trophic groups, particularly benthic-feeding pathways64,65,66,67. In this respect, it is notable that high-taxonomic-resolution fisheries data suggests that planktivores, especially gelatinous-feeding individuals, can make major contributions to fisheries catch on some Indo-Pacific reefs68,69, while on Caribbean reefs such contributions appear to be lacking70,71 (Extended Data Fig. 5, Supplementary Text 2 and Supplementary Data 3 and 4). Although the spatial and temporal extent of such high-resolution data is limited, it suggests that gelatinous plankton may ultimately fuel the productivity of fish species directly relevant to fisheries and human nutrition68. Unfortunately, the lack of high-taxonomic-resolution fisheries data from the world’s coral reefs currently stymies a broad-scale quantitative comparison of this potential link between planktivorous reef fish productivity and fisheries. Given the major, global, between-realm division in planktivore productivity that we identify, unravelling the extent to which this division translates into between-realm differences in coral reef fisheries yields represents an exciting avenue for future research.
Overall, the contrast is stark: the Caribbean, with its low biomass of predominantly small planktivores, yields limited productivity of planktivorous fishes. In the Indo-Pacific, however, substantial pelagic subsidies underpin the high productivity of large planktivores. Clearly, biogeographic legacies may have far-reaching effects, shaping the functional deck that each of the biogeographic realms has been dealt. The absence of gelatinous planktivores in the Caribbean highlights that, globally, coral reefs are far from uniform. Fundamental differences in how energy flows through these ecosystems are evident, with the functional capacity of Caribbean planktivores vastly lower than that of their Indo-Pacific counterparts. This diminished trophic capacity in the Caribbean has direct implications for ecosystem functioning. Ultimately, in terms of planktivorous fish assemblages, Caribbean reefs support a depauperate and relatively unproductive version of their Indo-Pacific counterparts, appearing to have only a limited capacity to channel pelagic energy to higher trophic levels.
Methods
Survey dataset
To investigate how planktivore productivity varies globally across the Indo-Pacific and Caribbean, as well as the role of gelatinous versus non-gelatinous prey preference in the Indo-Pacific, we used the open-access database of global reef fish counts from RLS (accessed on 16 December 2021)22,72. In brief, RLS fish counts are conducted by two divers on parallel ‘blocks’, with each block measuring 50 m × 5 m (250 m2)72. All fishes observed within these blocks are recorded by number and estimated size category72. Fish total length is estimated to the nearest standard predetermined size increment: 2.5, 5, 7.5, 10, 12.5, 15, 20, 25, 30, 35, 40 and 50 cm total length, and to the nearest 12.5 cm for larger fishes above 50 cm (ref. 72).
The complete RLS dataset was filtered to include tropical locations between the latitudes of 30˚ S and 30˚ N, facilitating comparison between the two major biogeographic coral reef realms: the Indo-Pacific and the Tropical Western Atlantic (Caribbean)23,24. Specifically, Indo-Pacific included locations from 34.92° E to 105.36° W longitude, while the Tropical Western Atlantic focused exclusively on Caribbean locations between 88.10° W and 68.25° W. To focus the analysis to the Caribbean, ten Brazilian locations were excluded from the analysis. These locations accounted for only 20 surveys, compared with the 157 surveys across 79 locations included for the Caribbean. Importantly, excluding the Brazilian locations did not alter the results (Supplementary Fig. 3).
Following Morais et al.1, we also removed surveys shallower than 4 m or deeper than 15 m depth, to minimize habitat-related variation (for example, lagoons versus reef slopes) and the chance of including surveys located in places naturally scarce in plankton and planktivorous fishes (that is, the reef flat and back reef)1. Lastly, counts were averaged between adjacent blocks owing to the lack of spatial independence72, with the collated dataset comprising 42 ecoregions and 1,131 unique sites (Indo-Pacific: 36 and 1,052; Caribbean: 6 and 79; Supplementary Fig. 4). We conducted Bayesian bootstrapped analyses to specifically test whether our inferences were influenced by the sampling idiosyncrasies between the Indo-Pacific and Caribbean (Supplementary Text 3). Critically, these analyses confirmed that our results and inferences are robust to the sampling imbalance (Supplementary Fig. 5).
Defining planktivores
From the RLS survey data, all coral reef planktivorous fish species were identified and categorized into primarily gelatinous or non-gelatinous feeders. Classification was conducted through a comprehensive analysis of species identification guides30,31,32, dietary information reported in FishBase33,34, along with published gut content information11,12,35,36,37,38,39,40,41,42. Notably, our definition of predominately gelatinous feeders is inherently conservative as, historically, planktivory has been seen as a largely non-gelatinous affair. While many fishes have been observed consuming gelatinous zooplankton5,21,73,74,75,76, we restricted the classification of primarily gelatinous feeders to planktivorous fishes whose diets predominantly consist of this prey type. Importantly, we do not consider these species obligate gelatinous feeders, as occasional or incidental consumption of other prey taxa may occur, although gelatinous plankton is, on average, the dominant dietary item. In addition, any species described as planktivorous and lacking associated dietary information were grouped into the default category of primarily non-gelatinous feeders. This conservative analysis led to the categorization of 336 planktivores of which 320 were non-gelatinous and 16 gelatinous feeders (Supplementary Data 1). In addition, we ran non-conservative analyses, where genus-level data and prey preference for species that lacked dietary information was categorized on the basis of phylogenetically similar species, resulting in 313 non-gelatinous and 54 gelatinous feeders (Supplementary Data 1). The results of these analyses closely mirrored those from the conservative approach, providing strong support for the reliability of our main results (Extended Data Figs. 2 and 3). Importantly, this expanded classification included four potentially gelatinous-feeding species in the Caribbean: Canthidermis sufflamen, Chaetodipterus faber, Inermia vittata and Ocyurus chrysurus, classified based on their ecological and morphological similarities to gelatinous-feeding taxa in the Indo-Pacific30,35,77,78. However, even with this expanded non-conservative classification, the contributions of potential gelatinous-feeding Caribbean planktivores remain negligible (Extended Data Fig. 4).
Biomass and productivity calculation
We calculated the biomass of the planktivorous fish assemblage, converting length measurements to biomass using Bayesian length–weight regression coefficients from FishBase33,34. Planktivorous fish productivity was subsequently calculated following the methods developed by Morais and Bellwood79,80. In short, we combined somatic growth and mortality estimates with fish surveys to calculate productivity (that is, the biomass arising from the somatic growth of all surviving individuals) for each individual. Specifically, we used a suite of ecological (maximum body size, dietary preference and position in the water column) and environmental covariates (sea surface temperature) to calculate and predict Kmax, a standardization of the von Bertalanffy growth parameter K (sensu Morais and Bellwood79), using extreme gradient boosting models for all surveyed individuals. Ecological traits were derived from FishBase33,34 or the peer-reviewed literature; estimates of sea surface temperature were extracted for each coordinate for each survey year from the National Ocean and Atmospheric Administration’s Optimum Interpolation Sea Surface Temperature V2 (https://psl.noaa.gov/data/gridded/data.noaa.oisst.v2.html). Due to the stochastic model-building procedure of boosted regression trees81, we used 1,000 bootstrapped iterations to generate Kmax estimates. For each simulated iteration, somatic growth was calculated by drawing Kmax values from a truncated normal distribution bounded by the 95% quantile interval of predicted Kmax values. We then used estimates of Kmax to predict the expected body size arising from somatic growth the following day. Following this, we used the Bayesian length–weight regression coefficients82 to convert expected body size into standing stock biomass (g). Individual productivity was calculated as the biomass expected given each individual’s growth trajectory minus their observed biomass on the survey. We therefore calculated the total community productivity (g per day) as the sum of the somatic growth of all individuals after one day over 1,000 bootstrapped simulations to account for the stochasticity of mortality80.
Statistical analyses
Individual Bayesian GLMMs were used to analyse the data. Initially, we used separate models to examine variation in planktivore abundance, biomass and productivity (response variables) across realm (fixed factors, two levels; Indo-Pacific n = 1,052 and Caribbean n = 79) and Indo-Pacific prey preference (fixed factors, two levels; gelatinous n = 1,052 and non-gelatinous n = 1,052). Additional models were used to examine how the contribution to planktivore abundance, biomass and productivity (response variables) varied across the size spectra (scaled continuous interaction) across realm (fixed factors, two levels; Indo-Pacific n = 5,857 and Caribbean n = 234) and Indo-Pacific prey preference (fixed factors, two levels; gelatinous n = 2,135 and non-gelatinous n = 5,121). To account for the unequal bin widths of size estimates in the RLS data (2.5, 5, 10 and 12.5 cm), density, biomass and productivity values within each size bin were normalized by dividing them by the actual width of the corresponding size bin83,84. We then ran separate models to assess the per-capita contribution of planktivores, standardizing biomass and productivity (response variables) by dividing abundance across realm (fixed factors, two levels; Indo-Pacific n = 1,052 and Caribbean n = 79) and Indo-Pacific prey preference (fixed factors, two levels; gelatinous n = 1,052 and non-gelatinous n = 1,052). Lastly, we assessed the relative family-level contributions to abundance, biomass and productivity (response variables) separately for each realm (fixed factor, 28 levels Indo-Pacific n = 30,508, 11 levels Caribbean n = 948).
All models included site nested within ecoregion as random factors to account for any spatial non-independence at these scales. All models used a Gamma distribution with a log link. To accommodate zero values, 10% of the smallest value for the response variable was added to the entire dataset. Due to the large number of zeroes, this resulted in a suboptimal model fit. Therefore, we chose to repeat the analyses using a hurdle Gamma distribution, modelling zeroes and non-zeroes independently. Although model fit was improved and gave visually similar results (albeit at a higher magnitude), we chose to retain the Gamma distribution for the main text, as many of the zeroes likely represented a lack of detection (because of low densities) rather than a true absence (see Supplementary Text 4 and Supplementary Figs. 6 and 7 for hurdle gamma distributions). All models incorporated weakly informative priors using the brms package85; some priors were not centred on zero to improve model fit and convergence. Full code and prior specifications are provided (Supplementary Data 5 and 6). Each model included three chains each of 10,000 iterations (2,000 warmup, thinning rate of 10). The models were validated through visual inspection to ensure that the priors were sufficiently weakly informative, chains were well mixed and converged on a stable posterior (\(\hat{R} < 1.05\)), and there was no evidence of within-chain autocorrelation. Furthermore, posterior predictive checks and simulated residuals using the DHARMa package86 were also used to assess model fits.
The median, lower and upper 50% and 95% HPD intervals, as well as PPs for each model, were calculated. Evidence of an effect was determined through the PPs. While the interpretation of what constitutes the strength of effect is inherently subjective, we chose to categorize the levels of effect as: no evidence (<50%), weak (50–75%), moderate (75–90%), strong (90–97.5%) and decisive evidence (>97.5%). All statistical analyses were performed within the R 4.3.1 Statistical and Graphical Environment87, using the brms85, emmeans88 and rstan89 packages.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The raw dataset analysed during the current study is publicly available at https://reeflifesurvey.com/survey-data/, and the derived dataset is available via ‘Research Data JCU’ at https://doi.org/10.25903/wz7d-yz51 (ref. 90). Source data are provided with this paper.
Code availability
The code required to reproduce all the analyses and results is available via ‘Research Data JCU’ at https://doi.org/10.25903/wz7d-yz51 (ref. 90).
References
Morais, R. A., Siqueira, A. C., Smallhorn-West, P. F. & Bellwood, D. R. Spatial subsidies drive sweet spots of tropical marine biomass production. PLoS Biol. 19, e3001435 (2021).
Morais, R. A. & Bellwood, D. R. Pelagic subsidies underpin fish productivity on a degraded coral reef. Curr. Biol. 29, 1521–1527 (2019).
Skinner, C. et al. Offshore pelagic subsidies dominate carbon inputs to coral reef predators. Sci. Adv. 7, 1–12 (2021).
Fox, M. D. et al. Gradients in primary production predict trophic strategies of mixotrophic corals across spatial scales. Curr. Biol. 28, 3355–3363 (2018).
Hamner, W. M., Jones, M. S., Carleton, J. H., Hauri, I. R. & Williams, D. M. Zooplankton, planktivorous fish, and water currents on a windward reef face: Great Barrier Reef, Australia. Bull. Mar. Sci. 42, 459–479 (1988).
Hamner, W., Colin, P. & Hamner, P. Export import dynamics of zooplankton on a coral reef in Palau. Mar. Ecol. Prog. Ser. 334, 83–92 (2007).
Houlbrèque, F. & Ferrier-Pagès, C. Heterotrophy in tropical scleractinian corals. Biol. Rev. 84, 1–17 (2009).
Glynn, P. W. & Enochs, I. C. in Coral Reefs: An Ecosystem in Transition (eds Dubinsky, Z. & Stambler, N.) 273–325 (Springer, 2011); https://doi.org/10.1007/978-94-007-0114-4_18
Hobson, E. & Chess, J. Trophic relationships among fishes and plankton in the lagoon at Enewetak Atoll, Marshall Islands. Fish. Bull. 76, 133–153 (1978).
Lazzaro, X. A review of planktivorous fishes: their evolution, feeding behaviours, selectivities, and impacts. Hydrobiologia 146, 97–167 (1987).
Huertas, V. & Bellwood, D. R. Trophic separation in planktivorous reef fishes: a new role for mucus?. Oecologia 192, 813–822 (2020).
Choat, J. H., Clements, K. D. & Robbins, W. D. The trophic status of herbivorous fishes on coral reefs. Mar. Biol. 140, 613–623 (2002).
Gahan, J., Bellwood, D. R., Nankervis, L. & Tebbett, S. B. Spatial and temporal variability in tropical off-reef zooplankton across broad spatial and temporal scales. Mar. Environ. Res. 191, 106169 (2023).
Gahan, J., Bellwood, D. R., Bellwood, O. & Schlaefer, J. Gelatinous versus non-gelatinous zooplankton: their value as food for planktivorous coral reef fishes. Coral Reefs 43, 243–252 (2024).
Sale, P. F., McWilliam, P. S. & Anderson, D. T. Composition of the near-reef zooplankton at Heron Reef, Great Barrier Reef. Mar. Biol. 34, 59–66 (1976).
Carrillo Baltodano, A. M. & Morales Ramírez, Á. Changes in abundance and composition of a Caribbean coral reef zooplankton community after 25 years. Rev. Biol. Trop. 64, 1029–1040 (2016).
Hamner, W. M., Madin, L. P., Alldredge, A. L., Gilmer, R. W. & Hamner, P. P. Underwater observations of gelatinous zooplankton: sampling problems, feeding biology, and behavior. Limnol. Oceanogr. 20, 907–917 (1975).
Jaspers, C., Acuña, J. L. & Brodeur, R. D. Interactions of gelatinous zooplankton within marine food webs. J. Plankton Res. 37, 985–988 (2015).
Sommer, U., Stibor, H., Katechakis, A., Sommer, F. & Hansen, T. Pelagic food web configurations at different levels of nutrient richness and their implications for the ratio fish production:primary production. Hydrobiologia 484, 11–20 (2002).
Boero, F. et al. Gelatinous plankton: irregularities rule the world (sometimes). Mar. Ecol. Prog. Ser. 356, 299–310 (2008).
Purcell, J. E. & Arai, M. N. Interactions of pelagic cnidarians and ctenophores with fish: a review. Hydrobiologia 451, 27–44 (2001).
Edgar, G. J. et al. Establishing the ecological basis for conservation of shallow marine life using Reef Life Survey. Biol. Conserv. 252, 108855 (2020).
Kulbicki, M. et al. Global biogeography of reef fishes: a hierarchical quantitative delineation of regions. PLoS ONE 8, e81847 (2013).
Roff, G. & Mumby, P. J. Global disparity in the resilience of coral reefs. Trends Ecol. Evol. 27, 404–413 (2012).
Birkeland, C. Geographic comparisons of coral-reef community processes. In Proc. Sixth International Coral Reef Symposium Vol. 1 (1988).
Tebbett, S. B., Connolly, S. R. & Bellwood, D. R. Benthic composition changes on coral reefs at global scales. Nat. Ecol. Evol. 7, 71–81 (2023).
Siqueira, A. C., Morais, R. A., Bellwood, D. R. & Cowman, P. F. Trophic innovations fuel reef fish diversification. Nat. Commun. 11, 2669 (2020).
Brooks, J. L. & Dodson, S. I. Predation, body size, and composition of plankton: the effect of a marine planktivore on lake plankton illustrates theory of size, competition, and predation. Science 150, 28–35 (1965).
Hall, D. J., Threlkeld, S. T., Burns, C. W. & Crowley, P. H. The size-efficiency hypothesis and the size structure of zooplankton communities. Annu. Rev. Ecol. Syst. 7, 177–208 (1976).
Randall, J. E., Allen, G. R. & Steene, R. C. Fishes of the Great Barrier Reef and Coral Sea (Univ. Hawaii Press, 1988).
Myers, R. F. Micronesian Reef Fishes: A Comprehensive Guide to the Coral Reef Fishes of Micronesia (Coral Graphics, 1999).
Hobson, E. Feeding relationships of teleostean fishes on coral reefs in Kona, Hawaii. Collect. Repr. 72, 915 (1982).
Froese, R. & Pauly, D. FishBase 2000: Concepts, Designs and Data Sources Vol. 1594 (WorldFish, 2000).
Froese, R. & Pauly, D. FishBase. FishBase https://www.fishbase.org/ (2024).
Randall, J. E. Food habits of reef fishes of the West Indies. Stud. Trop. Oceanogr. 5, 665–847 (1967).
Randall, J. E. Unicornfishes of the subgenus Axinurus (Perciformes: Acanthuridae: Naso), with description of a new species. Copeia 1994, 116–124 (1994).
Friedman, S. T., Price, S. A., Hoey, A. S. & Wainwright, P. C. Ecomorphological convergence in planktivorous surgeonfishes. J. Evol. Biol. 29, 965–978 (2016).
Milton, D. A., Blaber, S. J. M. & Rawlinson, N. J. F. Diet and prey selection of six species of tuna baitfish in three coral reef lagoons in the Solomon Islands. J. Fish Biol. 37, 205–224 (1990).
Hiatt, R. W. & Strasburg, D. W. Ecological relationships of the fish fauna on coral reefs of the Marshall Islands. Ecol. Monogr. 30, 65–127 (1960).
Randall, J. E. & Dooley, J. K. Revision of the Indo-Pacific branchiostegid fish genus Hoplolatilus, with descriptions of two new species. Copeia 1974, 457 (1974).
Randall, J. E. & Lubbock, R. A revision of the serranid fishes of the subgenus Mirolabrichthys (Anthiinae: Anthias): with descriptions of five new species. Contrib. Sci. 333, 1–27 (1982).
Wilson, S. K. Diversity in the diet and feeding habits of blennies. in The Biology of Blennies (ed Patzner, R. A.) 139–162 (Science Publishers, 2009).
Gordon, A. L. & Fine, R. A. Pathways of water between the Pacific and Indian oceans in the Indonesian seas. Nature 379, 146–149 (1996).
Arai, M. N. Predation on pelagic coelenterates: a review. J. Mar. Biol. Assoc. UK. 85, 523–536 (2005).
Gardner, M. B. Mechanisms of size selectivity by planktivorous fish: a test of hypotheses. Ecology 62, 571–578 (1981).
Henschke, N., Everett, J. D., Richardson, A. J. & Suthers, I. M. Rethinking the role of salps in the ocean. Trends Ecol. Evol. 31, 720–733 (2016).
Arai, M. N., Welch, D. W., Dunsmuir, A. L., Jacobs, M. C. & Ladouceur, A. R. Digestion of pelagic Ctenophora and Cnidaria by fish. Can. J. Fish. Aquat. Sci. 60, 825–829 (2003).
Sterner, R. W. & George, N. B. Carbon, nitrogen, and phosphorus stoichiometry of cyprid fishes. Ecology 81, 127–140 (2000).
Heidelberg, K. B., Sebens, K. P. & Purcell, J. E. Composition and sources of near reef zooplankton on a Jamaican forereef along with implications for coral feeding. Coral Reefs 23, 263–274 (2004).
Roman, M. R., Furnas, M. J. & Mullin, M. M. Zooplankton abundance and grazing at Davies Reef, Great Barrier Reef, Australia. Mar. Biol. 105, 73–82 (1990).
Yahel, R., Yahel, G. & Genin, A. Near- bottom depletion of zooplankton over coral reefs: I: diurnal dynamics and size distribution. Coral Reefs 24, 75–85 (2005).
Van Der Spoel, S. & Pierrot-Bults, A. C. Zoogeography and Diversity of Plankton (Bunge Scientific Publishers, 1979).
Pellissier, L. et al. Quaternary coral reef refugia preserved fish diversity. Science 344, 1016–1019 (2014).
Siqueira, A. C., Morais, R. A., Bellwood, D. R. & Cowman, P. F. Planktivores as trophic drivers of global coral reef fish diversity patterns. Proc. Natl Acad. Sci. USA 118, e2019404118 (2021).
Robertson, R. & Ffield, A. M2 baroclinic tides in the Indonesian Seas. Oceanography 18, 62–73 (2005).
Richardson, P. L. Caribbean Current and eddies as observed by surface drifters. Deep Sea Res. Pt. II 52, 429–463 (2005).
Cooper, W. J. & Westneat, M. W. Form and function of damselfish skulls: rapid and repeated evolution into a limited number of trophic niches. BMC Evol. Biol. 9, 24 (2009).
Siqueira, A. C., Bellwood, D. R. & Cowman, P. F. Historical biogeography of herbivorous coral reef fishes: the formation of an Atlantic fauna. J. Biogeogr. 46, 1611–1624 (2019).
Bellwood, D. R., Hughes, T. P., Folke, C. & Nyström, M. Confronting the coral reef crisis. Nature 429, 827–833 (2004).
Siqueira, A. C., Bellwood, D. R. & Cowman, P. F. The evolution of traits and functions in herbivorous coral reef fishes through space and time. Proc. R. Soc. B 286, 20182672 (2019).
Feng, J. et al. Mediating role of food web structure in linking diversity to multidimensional stability: evidence from global marine ecosystems. Sci. Adv. 11, 1–13 (2025).
Crisp, S. K., Tebbett, S. B. & Bellwood, D. R. A critical evaluation of benthic phase shift studies on coral reefs. Mar. Environ. Res. 178, 105667 (2022).
Liu, Y., Hu, J. & Gore, J. Ecosystem stability relies on diversity difference between trophic levels. Proc. Natl Acad. Sci. USA 121, e2416740121 (2024).
Shantz, A. A., Ladd, M. C. & Burkepile, D. E. Overfishing and the ecological impacts of extirpating large parrotfish from Caribbean coral reefs. Ecol. Monogr. 90, e01403 (2020).
Mumby, P. J. Stratifying herbivore fisheries by habitat to avoid ecosystem overfishing of coral reefs. Fish Fish. 17, 266–278 (2016).
Mumby, P. J. The impact of exploiting grazers (Scaridae) on the dynamics of Caribbean coral reefs. Ecol. Appl. 16, 747–769 (2006).
Mumby, P. et al. Fishing down a Caribbean food web relaxes trophic cascades. Mar. Ecol. Prog. Ser. 445, 13–24 (2012).
Bellwood, D. Seasonal changes in the size and composition of the fish yield from reefs around Apo Island, Central Philippines, with notes on methods of yield estimation. J. Fish Biol. 32, 881–893 (1988).
Abesamis, R. A., Russ, G. R. & Alcala, A. C. Gradients of abundance of fish across no-take marine reserve boundaries: evidence from Philippine coral reefs. Aquat. Conserv. Mar. Freshw. Ecosyst. 16, 349–371 (2006).
Munro, J. L. Caribbean Coral Reef Fishery Resources Vol. 7 (ICLARM Studies and Reviews, International Center for Living Aquatic Resources Management, 1983).
Olsen, D. A., Nellis, D. W. & Wood, R. S. Ciguatera in the Eastern Caribbean. Mar. Fish. Rev. 46, 13–18 (1984).
Edgar, G. J. & Stuart-Smith, R. D. Systematic global assessment of reef fish communities by the Reef Life Survey program. Sci. Data 1, 140007 (2014).
Diaz Briz, L., Sánchez, F., Marí, N. & Genzano, G. Seasonal variation of gelatinous plankton consumption by fish in the South-western Atlantic Ocean: a question of strategy. Mar. Biol. Res. 14, 739–751 (2018).
Diaz Briz, L., Sánchez, F., Marí, N., Mianzan, H. & Genzano, G. Gelatinous zooplankton (ctenophores, salps and medusae): an important food resource of fishes in the temperate SW Atlantic Ocean. Mar. Biol. Res. 13, 630–644 (2017).
Mianzan, H., Pájaro, M., Colombo, G. A. & Madirolas, A. Feeding on survival-food: gelatinous plankton as a source of food for anchovies. Hydrobiologia 451, 45–53 (2001).
Ates, R. M. L. Medusivorous fishes, a review. Zool. Meded. 62, 29–42 (1988).
Hayse, J. Feeding habits, age, growth, and reproduction of Atlantic spadefish Chaetodipterus faber (Pisces: Ephippidae) in South Carolina. Fish. Bull. 88, 67–83 (1990).
Carpenter, K. E. FAO Species Catalogue. Vol. 8. Fusilier Fishes of the World. An Annotated and Illustrated Catalogue of Caesionid Species Known to Date FAO Fisheries Synopsis No. 125 (Food and Agriculture Organization of the United Nations, 1988).
Morais, R. A. & Bellwood, D. R. Global drivers of reef fish growth. Fish Biol. 19, 874–889 (2018).
Morais, R. A. & Bellwood, D. R. Principles for estimating fish productivity on coral reefs. Coral Reefs 39, 1221–1231 (2020).
Elith, J., Leathwick, J. R. & Hastie, T. A working guide to boosted regression trees. J. Anim. Ecol. 77, 802–813 (2008).
Froese, R., Thorson, J. T. & Reyes, R. B. Jr. A Bayesian approach for estimating length–weight relationships in fishes. J. Appl. Ichthyol. 30, 78–85 (2014).
Platt, T. & Denman, K. Organisation in the pelagic ecosystem. Helgoländer Wiss. Meeresunters. 30, 575–581 (1977).
Heather, F. J., Blanchard, J. L., Edgar, G. J., Trebilco, R. & Stuart-Smith, R. D. Globally consistent reef size spectra integrating fishes and invertebrates. Ecol. Lett. 24, 572–579 (2021).
Bürkner, P.-C. brms: An R package for Bayesian multilevel models using Stan. R package version 2.20.4. CRAN https://CRAN.R-project.org/package=brms (2021).
Hartig, F. DHARMa: residual diagnostics for hierarchical (multi-level / mixed) regression models. R package version 0.4.6. CRAN https://CRAN.R-project.org/package=DHARMa (2022).
R Core Team R: A Language and Environment for Statistical Computing (R Core Team, 2023).
Lenth, R. V. emmeans: Estimated marginal means, aka least-squares means. R package version 1.8.8. CRAN https://CRAN.R-project.org/package=emmeans (2023).
Stan Development Team. RStan: the R interface to Stan. R package version 2.32.7. CRAN https://cran.r-project.org/web/packages/rstan/index.html (2023).
Gahan, J. et al. Data for: Missing planktivore functions drive global variation in reef fish productivity. James Cook University https://doi.org/10.25903/wz7d-yz51 (2026).
Acknowledgements
We thank the late William ‘Bill’ Hamner both for his insightful correspondence and for the pioneering work that inspired this study, and F. Libert for photographs. We also thank the Reef Life Survey Team and their volunteers for collecting and supplying the data used for the study. These data are managed by, and were sourced from, Australia’s Integrated Marine Observing System (IMOS), which is enabled by the National Collaborative Research Infrastructure Strategy (NCRIS). This work was funded by the Australian Research Council (D.R.B., grant number FL190100062; A.C.S., grant number DE250101047) and through an Australian Government Research Training Program Scholarship (J.G.).
Author information
Authors and Affiliations
Contributions
J.G., D.R.B., L.N., A.C.S. and S.B.T. conceived the study. G.J.E. and R.D.S.-S. collected RLS data. H.F.Y. collated the data and conducted productivity and biomass calculations. J.G. performed the analyses, produced the figures and wrote the first draft of the paper. All authors interpreted the results and contributed substantially to revisions.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Ecology & Evolution thanks James Robinson and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Between-realm comparison of planktivorous fishes.
Circles represent the median value from the posterior distribution, thick coloured bars represent the 50% credible intervals, and the thin coloured bars the 95% credible intervals. Text shows the posterior probability (PP) of the Indo-Pacific (IP) being greater than the Caribbean (Car). Figure is coloured by realm; blue Indo-Pacific and brown the Caribbean. Sites: Indo-Pacific n = 1052, and Caribbean n = 79.
Extended Data Fig. 2 Non-conservative analyses of between realm comparison of planktivore metrics.
a) density, c) biomass, and e) productivity, as well as their corresponding predicted b, d, and f) size distribution. a, c, and e) Circles represent the median value from the posterior distribution, thick coloured bars represent the 50% credible intervals, and the thin coloured bars the 95% credible intervals. Each plot a, c, e) shows the posterior probability (PP) of the Indo-Pacific (IP) being greater than the Caribbean (Car). b, d, f) Black lines represent the mean prediction from the posterior distribution, with the shaded ribbons representing the 95% credible interval, note both axes are on the log10 scale, and x axis restricted to observed realm-size distribution. All figures are coloured by realm; blue Indo-Pacific and brown the Caribbean. Sites: a, c, e) Indo-Pacific n = 1052 and Caribbean n = 79; Within-site size-class observations: b, d, f) Indo-Pacific n = 5906 and Caribbean n = 234. See Supplementary Table 1 for non-conservative species categorisation.
Extended Data Fig. 3 Non-conservative analyses of gelatinous versus non-gelatinous Indo-Pacific planktivorous fishes.
a) Density, c) biomass, and e) productivity, as well as their corresponding predicted b, d, f) size distribution. a, c, d) Circles represent the median value from the posterior distribution, thick coloured bars represent the 50% credible intervals, and the thin coloured bars the 95% credible intervals. Each plot a, c, and d) shows the posterior probability (PP) of non-gelatinous planktivores (NG) being greater than the gelatinous planktivores (G). b, d, f) Black lines represent the mean prediction from the posterior distribution, with the shaded ribbons representing the 95% credible interval, note both axes are on the log10 scale, and x axis restricted to observed planktivore size distribution. All figures are coloured by primary prey type; red non-gelatinous and purple gelatinous. Sites: a, c, e) non-gelatinous n = 1052 and gelatinous n = 1052; Within-site size-class observations: b, d, f) non-gelatinous n = 4826 and gelatinous n = 2820. See Supplementary Table 1 for non-conservative species categorisation.
Extended Data Fig. 4 Non-conservative analyses of gelatinous versus non-gelatinous Caribbean planktivorous fishes.
a) Density, c) biomass, and e) productivity, as well as their corresponding predicted b, d, and f) size distribution. a, c, and d) Circles represent the median value from the posterior distribution, thick coloured bars represent the 50% credible intervals, and the thin coloured bars the 95% credible intervals. Each plot a, c, d) shows the posterior probability (PP) of non-gelatinous planktivores (NG) being greater than the gelatinous planktivores (G). b, d, f) Black lines represent the mean prediction from the posterior distribution, with the shaded ribbons representing the 95% credible interval, note both axes are on the log10 scale, and x axis restricted to observed planktivore size distribution. All figures are coloured by primary prey type; red non-gelatinous and purple gelatinous. Sites: a, c, e) non-gelatinous n = 79 and gelatinous n = 79; Within-site size-class observations: b, d, f) non-gelatinous n = 169 and gelatinous n = 51. See Supplementary Table 1 for non-conservative species categorisation.
Extended Data Fig. 5 Relative fishery yields by trophic level.
Supplementary information
Supplementary Information (download PDF )
Supplementary Texts 1–4, Figs. 1–9, Table 1 and References.
Supplementary Data 1 (download XLSX )
Workbook containing Supplementary Data 1–7 in individual tabs.
Source data
Source Data Fig. 1 (download XLSX )
Processed data for Fig. 1b,c.
Source Data Fig. 2 (download XLSX )
Processed data for Fig. 2.
Source Data Fig. 3 (download XLSX )
Processed data for Fig. 3a–f.
Source Data Fig. 4 (download XLSX )
Processed data for Fig. 4a–f.
Source Data Fig. 5 (download XLSX )
Processed data for Fig. 5.
Source Data Extended Data Fig. 1 (download XLSX )
Processed data for Extended Data Fig. 1.
Source Data Extended Data Fig. 2 (download XLSX )
Processed data for Extended Data Fig. 2a–f.
Source Data Extended Data Fig. 3 (download XLSX )
Processed data for Extended Data Fig. 3a–f.
Source Data Extended Data Fig. 4 (download XLSX )
Processed data for Extended Data Fig. 4a–f.
Source Data Extended Data Fig. 5 (download XLSX )
Processed data for Extended Data Fig. 5.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Gahan, J., Yan, H.F., Bellwood, D.R. et al. Missing planktivore functions drive global variation in reef fish productivity. Nat Ecol Evol (2026). https://doi.org/10.1038/s41559-026-03029-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41559-026-03029-x