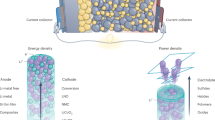
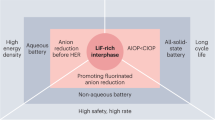
Abstract
Lithium-ion batteries can fail catastrophically through thermal runaway, but the key trigger has remained unclear. Here we show that the most harmful cause is lithium oxidation reaction (LOR). This makes two types of high-energy-density battery surprisingly most dangerous: all-solid-state batteries with cracked solid separators, whether from manufacturing defects, high-pressure assembly or electrochemical cycling, and batteries with non-flammable liquid electrolytes. In both batteries, oxygen evolved from an oxide cathode passes directly to an anode, triggering highly energetic LOR. In contrast, traditional carbonate- and ether-based electrolytes are safer because they can consume O2 in transit, alleviating or avoiding LOR. These findings apply to both lithium metal and lithiated anodes such as graphite. Safe electrolytes are thus either solid ion conductors that stop O2 crossover under all conditions or materials that scavenge O2 through low-exothermic reactions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article and the Supplementary Information.
References
Kamaya, N., Homma, K., Yamakawa, Y., Hirayama, M. & Kanno, R. A lithium superionic conductor. Nat. Mater. 10, 682–686 (2011).
Manthiram, A., Yu, X. & Wang, S. Lithium battery chemistries enabled by solid-state electrolytes. Nat. Rev. Mater. 2, 16103 (2017).
Janek, J. & Zeier, W. G. Challenges in speeding up solid-state battery development. Nat. Energy 8, 230–240 (2023).
Li, Y. et al. A lithium superionic conductor for millimeter-thick battery electrode. Science 381, 50–53 (2023).
Wang, C. & Su, X. The promise of solid-state batteries for safe and reliable energy storage. Engineering 21, 32–35 (2023).
Jarratt, E. New lessons from the epic story of Moli Energy, the Canadian pioneer of rechargeable lithium battery technology. Electric Autonomy Canada (18 September 2020).
Doughty, D. & Roth, E. P. A general discussion of Li ion battery safety. Electrochem. Soc. Interface 21, 37–44 (2012).
Feng, X., Ren, D., He, X. & Ouyang, M. Mitigating thermal runaway of lithium-ion batteries. Joule 4, 743–770 (2020).
Longchamps, R. S., Yang, X. G. & Wang, C. Y. Fundamental insights into battery thermal management and safety. ACS Energy Lett. 7, 1103–1111 (2022).
Bates, A. et al. A multi-scale framework for advancing battery safety through early calorimetric analysis of materials and components. Electrochem. Soc. Interface 33, 69–76 (2024).
Song, I. T. et al. Thermal runaway prevention through scalable fabrication of safety reinforced layer in practical Li-ion batteries. Nat. Commun. 15, 8294 (2024).
Inoue, T. & Mukai, K. Are all-solid-state lithium-ion batteries really safe? Verification by differential scanning calorimetry with an all-inclusive microcell. ACS Appl. Mater. Interfaces 9, 1507–1515 (2017).
Johnson, N. B. et al. Assessing the thermal safety of a Li metal solid-state battery material set using differential scanning calorimetry. ACS Appl. Mater. Interfaces 15, 57134–57143 (2023).
Chen, R. et al. The thermal stability of lithium solid electrolytes with metallic lithium. Joule 4, 812–821 (2020).
Bates, A. M. et al. Are solid-state batteries safer than lithium-ion batteries?. Joule 6, 742–755 (2022).
Ge, S. et al. Quantification of lithium battery fires in internal short circuit. ACS Energy Lett. 9, 5747–5755 (2024).
Qin, K., Ge, S., Gupta, N., Sasaki, T. & Wang, C. Y. High heat tolerance and safety of lithium metal batteries using a high concentration phase change electrolyte. J. Power Sources 630, 236094 (2025).
Ning, Z. et al. Dendrite initiation and propagation in lithium metal solid-state batteries. Nature 618, 287–293 (2023).
Kamphaus, E. P. et al. Role of inorganic surface layer on solid electrolyte interphase evolution at Li-metal anodes. ACS Appl. Mater. Interfaces 11, 31467–31476 (2019).
Cui, Z., Liu, C., Wang, F. & Manthiram, A. Navigating thermal stability intricacies of high-nickel cathodes for high-energy lithium batteries. Nat. Energy 10, 490–501 (2025).
Xu, K. Nonaqueous liquid electrolytes for lithium-based rechargeable batteries. Chem. Rev. 104, 4303–4418 (2004).
Jia, H. et al. Is nonflammability of electrolyte overrated in the overall safety performance of lithium ion batteries? A sobering revelation from a completely nonflammable electrolyte. Adv. Energy Mater. 13, 2203144 (2023).
Hou, J. et al. Thermal runaway of lihthium-ion batteries employing LiN(SO2F)2-based concentrated electrolyte. Nat. Commun. 11, 5100 (2020).
Lee, S. et al. Interplay of cathode−halide solid electrolyte in enhancing thermal stability of charged cathode material in all-solid-state batteries. ACS Energy Lett. 9, 1369–1380 (2024).
Choi, J. K. & Joncich, M. J. Heats of combustion, heats of formation and vapor pressures of some organic carbonates estimation of carbonate group contribution to heat of formation. J. Chem. Eng. Data 16, 87–90 (1971).
Eshetu, G. G. et al. In-depth safety-focused analysis of solvents used in electrolytes for large scale lithium ion batteries. Phys. Chem. Chem. Phys. 15, 9145–9155 (2013).
Heat of combustion of the elements. Periodictable.com https://periodictable.com/Properties/A/CombustionHeat.al.html (2024).
Pilcher, G., Pell, A. S. & Coleman, D. J. Measurements of heats of combustion by flame calorimetry part 2. Dimethyl ether, methyl ethyl ether, methyl n-propyl ether, methyl isopropyl ether. Trans. Faraday Soc. 60, 499–505 (1964).
Heat of combustion for sulfolane. Chemeo https://www.chemeo.com/cid/98-351-4/Thiophene-tetrahydro-1-1-dioxide#ref-joback (2023).
Emmanuel, P. et al. An introduction to the combustion of carbon materials. Chem. Eur. J. 28, e202200117 (2022).
Heat of combustion for SOCl2. Chemeo https://www.chemeo.com/cid/63-179-4/Thionyl-chloride (2023).
Haynes, W. M. (ed.) CRC Handbook of Chemistry and Physics 92nd edn (CRC Press, 2011).
Acknowledgements
Partial financial support from Nissan Motor Co. Ltd and William E. Diefenderfer Endowment is gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
C.-Y.W., K.Q. and N.G. wrote the manuscript. S.G., N.G. and K.Q. designed and built the cells. S.G., K.Q., N.G. and T.S. carried out the experiments. C.-Y.W., K.Q., N.G., T.S. and K.A. participated in discussions and data interpretation. All authors contributed to the development of the manuscript as the project developed.
Corresponding author
Ethics declarations
Competing interests
T.S. and K.A. are employed by Nissan Motor Co. Ltd. The other authors have no competing interests.
Peer review
Peer review information
Nature Energy thanks Xiangming He and Atsuo Yamada for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Illustration of the physical problem and corresponding RISC experimental setup.
Left, Single-layer internal short-circuit in a multilayer battery. Middle, Experimental cell in RISC method. Right, Cell holder with controllable stacking pressure. Figure adapted with permission from ref. 16, American Chemical Society.
Extended Data Fig. 2 Internal short circuiting of liquid-electrolyte graphite/NCM811 LiBs at various state of charge (SOC).
a, Temporal variations of voltage, shorting current, shorting resistance and internal temperature at 100%, 65% and 50% SOC, respectively. b, Photographic images of LiB cells after ISC. The 100% and 65% SOC cells catch fires due to sufficient heat generated from electrolyte oxidation. In contrast, the 50% SOC cell, generating only half of reaction heat from electrolyte oxidation, emits light smokes only.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–11 and Table 1.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, CY., Qin, K., Ge, S. et al. Metrics for evaluating safe electrolytes in energy-dense lithium batteries. Nat Energy 10, 1382–1390 (2025). https://doi.org/10.1038/s41560-025-01887-6
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41560-025-01887-6