Abstract
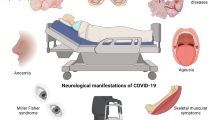
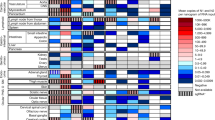
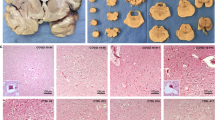
Severe coronavirus disease 2019 and post-acute sequelae of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection are associated with neurological complications that may be linked to direct infection of the central nervous system (CNS), but the selective pressures ruling neuroinvasion are poorly defined. Here we assessed SARS-CoV-2 evolution in the lung versus CNS of infected mice. Higher levels of viral divergence were observed in the CNS than the lung after intranasal challenge with a high frequency of mutations in the spike furin cleavage site (FCS). Deletion of the FCS significantly attenuated virulence after intranasal challenge, with lower viral titres and decreased morbidity compared with the wild-type virus. Intracranial inoculation of the FCS-deleted virus, however, was sufficient to restore virulence. After intracranial inoculation, both viruses established infection in the lung, but dissemination from the CNS to the lung required the intact FCS. Cumulatively, these data suggest a critical role for the FCS in determining SARS-CoV-2 tropism and compartmentalization.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data supporting the findings of this study are available within the paper, extended data or in the source data files. Viral whole-genome sequencing data have been deposited to NCBI as Bioproject PRJNA1017824. SARS-CoV-2 reference genome accession MN908947.3. Source data are provided with this paper.
Code availability
No unique code was generated in the course of the data acquisition. To determine consensus RNA sequence, reads were trimmed to remove adaptors and low-quality sequences using Trimmomatic v0.36. Trimmed reads were aligned to the reference genome sequence of SARS-CoV-2 (accession MN908947.3) using bwa v0.7.15. Pile-ups were generated from the alignment using samtools v1.9 and consensus sequence determined using iVar v1.2.2. For phylogenetic analysis, consensus sequences assembled for each sample were aligned using MAFFT v7.453 software. A maximum likelihood (ML) phylogeny with all consensus sequences were inferred with IQ-Tree v2.0.5. We performed probabilistic inference of intra-host viral quasispecies of the spike gene for each sample using QuasiRecomb. All final tree representation was performed with the R package ggtree v3.2.1. Viral diversity was determined using lme4 version 1.1-34 in R version 4.0.3.
References
Chi, W.-Y. et al. COVID-19 vaccine update: vaccine effectiveness, SARS-CoV-2 variants, boosters, adverse effects, and immune correlates of protection. J. Biomed. Sci. 29, 82 (2022).
Hu, B., Guo, H., Zhou, P. & Shi, Z.-L. Characteristics of SARS-CoV-2 and COVID-19. Nat. Rev. Microbiol. 19, 141–154 (2021).
Stein, S. R. et al. SARS-CoV-2 infection and persistence in the human body and brain at autopsy. Nature 612, 758–763 (2022).
Hartard, C. et al. Multiorgan and vascular tropism of SARS-CoV-2. Viruses 14, 515 (2022).
Gupta, A. et al. Extrapulmonary manifestations of COVID-19. Nat. Med. 26, 1017–1032 (2020).
Jackson, C. B., Farzan, M., Chen, B. & Choe, H. Mechanisms of SARS-CoV-2 entry into cells. Nat. Rev. Mol. Cell Biol. 23, 3–20 (2022).
Essalmani, R. et al. Distinctive roles of furin and TMPRSS2 in SARS-CoV-2 infectivity. J. Virol. 96, e00128–22 (2022).
Peng, R., Wu, L.-A., Wang, Q., Qi, J. & Gao, G. F. Cell entry by SARS-CoV-2. Trends Biochem. Sci 46, 848–860 (2021).
Jaimes, J. A., Millet, J. K. & Whittaker, G. R. Proteolytic cleavage of the SARS-CoV-2 spike protein and the role of the novel S1/S2 site. iScience 23, 101212 (2020).
Pišlar, A. et al. The role of cysteine peptidases in coronavirus cell entry and replication: the therapeutic potential of cathepsin inhibitors. PLoS Pathog. 16, e1009013 (2020).
Davies, N. G. et al. Estimated transmissibility and impact of SARS-CoV-2 lineage B.1.1.7 in England. Science 372, eabg3055 (2021).
Plante, J. A. et al. Spike mutation D614G alters SARS-CoV-2 fitness. Nature 592, 116–121 (2021).
Lorenzo-Redondo, R. et al. A clade of SARS-CoV-2 viruses associated with lower viral loads in patient upper airways. EBioMedicine 62, 103112 (2020).
Yuan, S. et al. Pathogenicity, transmissibility, and fitness of SARS-CoV-2 Omicron in Syrian hamsters. Science 377, 428–433 (2022).
Korber, B. et al. Tracking changes in SARS-CoV-2 spike: evidence that D614G increases infectivity of the COVID-19 virus. Cell 182, 812–827.e19 (2020).
Lorenzo-Redondo, R., de Sant’Anna Carvalho, A. M., Hultquist, J. F. & Ozer, E. A. SARS-CoV-2 genomics and impact on clinical care for COVID-19. J. Antimicrob. Chemother. 78, ii25–ii36 (2023).
Carabelli, A. M. et al. SARS-CoV-2 variant biology: immune escape, transmission and fitness. Nat. Rev. Microbiol. 21, 162–177 (2023).
Yurkovetskiy, L. et al. Structural and functional analysis of the D614G SARS-CoV-2 spike protein variant. Cell 183, 739–751.e8 (2020).
Hou, Y. J. et al. SARS-CoV-2 D614G variant exhibits efficient replication ex vivo and transmission in vivo. Science 370, 1464–1468 (2020).
Liu, Y. et al. Delta spike P681R mutation enhances SARS-CoV-2 fitness over alpha variant. Cell Rep. 39, 110829 (2022).
Chan, Y. A. & Zhan, S. H. The emergence of the spike furin cleavage site in SARS-CoV-2. Mol. Biol. Evol. 39, msab327 (2022).
Liu, S. et al. The PRRA insert at the S1/S2 site modulates cellular tropism of SARS-CoV-2 and ACE2 usage by the closely related Bat RaTG13. J. Virol. 95, e01751-20 (2021).
Johnson, B. A. et al. Loss of furin cleavage site attenuates SARS-CoV-2 pathogenesis. Nature 591, 293–299 (2021).
Dangi, T., Class, J., Palacio, N., Richner, J. M. & Penaloza MacMaster, P. Combining spike- and nucleocapsid-based vaccines improves distal control of SARS-CoV-2. Cell Rep. 36, 109664 (2021).
Dangi, T. et al. Improved control of SARS-CoV-2 by treatment with a nucleocapsid-specific monoclonal antibody. J. Clin. Invest. 132, e162282 (2022).
Dangi, T. et al. Pre-existing immunity modulates responses to mRNA boosters. Cell Rep. 42, 112167 (2023).
Sanchez, S., Palacio, N., Dangi, T., Ciucci, T. & Penaloza-MacMaster, P. Fractionating a COVID-19 Ad5-vectored vaccine improves virus-specific immunity. Sci. Immunol. 6, eabi8635 (2021).
Dangi, T. et al. Cross-protective immunity following coronavirus vaccination and coronavirus infection. J. Clin. Invest. 131, e151969 (2021).
Ke, R. et al. Longitudinal analysis of SARS-CoV-2 vaccine breakthrough infections reveals limited infectious virus shedding and restricted tissue distribution. Open Forum Infect. Dis. 9, ofac192 (2022).
Simons, L. M. et al. Assessment of virological contributions to COVID-19 outcomes in a longitudinal cohort of hospitalized adults. Open Forum Infect. Dis. 9, ofac027 (2022).
Simons, L. M. et al. De novo emergence of SARS-CoV-2 spike mutations in immunosuppressed patients. Transpl. Infect. Dis. 24, e13914 (2022).
Winkler, E. S. et al. SARS-CoV-2 infection of human ACE2-transgenic mice causes severe lung inflammation and impaired function. Nat. Immunol. 21, 1327–1335 (2020).
Dong, W. et al. The K18-Human ACE2 transgenic mouse model recapitulates non-severe and severe COVID-19 in response to an infectious dose of the SARS-CoV-2 virus. J. Virol. 96, e0096421 (2022).
Leist, S. R. et al. A mouse-adapted SARS-CoV-2 induces acute lung injury and mortality in standard laboratory mice. Cell 183, 1070–1085.e12 (2020).
Hossain, M. G., Tang, Y.-D., Akter, S. & Zheng, C. Roles of the polybasic furin cleavage site of spike protein in SARS-CoV-2 replication, pathogenesis, and host immune responses and vaccination. J. Med. Virol. 94, 1815–1820 (2022).
Li, M.-Y., Li, L., Zhang, Y. & Wang, X.-S. Expression of the SARS-CoV-2 cell receptor gene ACE2 in a wide variety of human tissues. Infect. Dis. Poverty 9, 45 (2020).
Piva, F., Sabanovic, B., Cecati, M. & Giulietti, M. Expression and co-expression analyses of TMPRSS2, a key element in COVID-19. Eur. J. Clin. Microbiol. Infect. Dis. 40, 451–455 (2021).
Gkogkou, E., Barnasas, G., Vougas, K. & Trougakos, I. P. Expression profiling meta-analysis of ACE2 and TMPRSS2, the putative anti-inflammatory receptor and priming protease of SARS-CoV-2 in human cells, and identification of putative modulators. Redox Biol. 36, 101615 (2020).
Prasad, K., Ahamad, S., Gupta, D. & Kumar, V. Targeting cathepsins: A potential link between COVID-19 and associated neurological manifestations. Heliyon 7, e08089 (2021).
Wrobel, A. G. et al. SARS-CoV-2 and bat RaTG13 spike glycoprotein structures inform on virus evolution and furin-cleavage effects. Nat. Struct. Mol. Biol. 27, 763–767 (2020).
Le Coupanec, A. et al. Cleavage of a neuroinvasive human respiratory virus spike glycoprotein by proprotein convertases modulates neurovirulence and virus spread within the central nervous system. PLoS Pathog. 11, e1005261 (2015).
Schnell, G., Price, R. W., Swanstrom, R. & Spudich, S. Compartmentalization and clonal amplification of HIV-1 variants in the cerebrospinal fluid during primary infection. J. Virol. 84, 2395–2407 (2010).
Adami, C. et al. Evolution of mouse hepatitis virus (MHV) during chronic infection: quasispecies nature of the persisting MHV RNA. Virology 209, 337–346 (1995).
Bender, S. J. & Weiss, S. R. Pathogenesis of murine coronavirus in the central nervous system. J. Neuroimmune Pharmacol. 5, 336–354 (2010).
Normandin, E. et al. High-depth sequencing characterization of viral dynamics across tissues in fatal COVID-19 reveals compartmentalized infection. Nat. Commun. 14, 574 (2023).
Bertelli, A. et al. Role of Q675H mutation in improving SARS-CoV-2 spike interaction with the furin binding pocket. Viruses 13, 2511 (2021).
Vanderheiden, A. & Klein, R. S. Neuroinflammation and COVID-19. Curr. Opin. Neurobiol. 76, 102608 (2022).
Joag, V. et al. Cutting edge: mouse SARS-CoV-2 epitope reveals infection and vaccine-elicited CD8 T cell responses. J. Immunol. 206, 931–935 (2021).
Richner, J. M. et al. Modified mRNA vaccines protect against Zika virus infection. Cell 168, 1114–1125 (2017).
Acknowledgements
Infectious clones provided by the Vineet Menachery lab from University of Texas Medical Branch. This research was supported, in part, through the computational resources and staff contributions provided by the Quest high-performance computing facility at Northwestern University, which is jointly supported by the Office of the Provost, the Office for Research and Northwestern University Information Technology. Tissue embedding and imaging was performed by the Research Histology and Tissue Imaging Core at UIC Research Resources Center. Funding for this work was provided by: NIH grants R01 AI150672 and R56 DE033249 (J.M.R.), DOD grant MS200290 (S.E.L.), NIH grant R21 AI163912 (J.F.H.), NIH grant U19 AI135964 (J.F.H., E.A.O. and R.L.R.), and through institutional support for the Center for Pathogen Genomics and Microbial Evolution (J.F.H., E.A.O. and R.L.R.). The funding sources had no role in the study design, data collection, analysis, interpretation or writing of the report. Figures 4a and 5a were created with BioRender.com.
Author information
Authors and Affiliations
Contributions
Conceptualization was performed by J.F.H., J.M.R. and P.P.-M.; methodology was carried out by J.C., L.M.S., L.C., J.G.A., S.E.L, T.D. and R.L.-R.; software and validation was carried out by L.M.S.; formal analysis was performed by J.C., L.M.S. and R.L.-R.; investigation was carried out by J.C., L.C., T.D. and L.M.S.; resources were provided by J.M.R., L.R., P.P.-M., J.F.H., R.L.-R. and E.A.O.; data curation was carried out by J.C., L.M.S. and R.L.-R.; writing of the original draft was performed by J.C., J.F.H. and J.M.R.; writing, review and editing, was performed by L.M.S., J.F.H., L.C., P.P.-M., L.R. and J.M.R; visualization was carried out by R.L.-R., J.F.H., S.E.L., J.C. and J.M.R.; supervision was carried out by J.F.H., R.L.-R., E.A.O., J.M.R. and L.R.; project administration was carried out by J.M.R., J.F.H., R.L.-R., E.A.O. and L.R.; and funding acquisition was carried out by J.M.R., J.F.H., R.L.-R., E.A.O. and L.R.
Corresponding authors
Ethics declarations
Competing interests
J.F.H. has received research support paid to Northwestern University from Gilead Sciences and is a paid consultant for Merck. All other authors declare no conflicts of interest.
Peer review
Peer review information
Nature Microbiology thanks Kei Sato, Shan-Lu Liu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Ad5-S and Ad5-N vaccines elicit robust humoral and cellular immunity against target antigens.
Mice were vaccinated with PBS, or Ad5 vaccines encoding for spike or nucleocapsid viral proteins (see reference below). 21 days post vaccination, mice were euthanized, and serum and spleens were collected. a, Antigen specific CD8 T cells were quantified with tetramers to the spike or nucleocapsid protein. Serum was analyzed for antibodies against spike (b) or nucleocapsid (c) recombinant protein via ELISA assay. P-values of Student’s t-test is reported24.
Supplementary information
Source data
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download PDF )
Unprocessed western blots.
Source Data Fig. 4 (download PDF )
Unprocessed histology images.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download PDF )
Unprocessed histology and in vitro images.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 1 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Class, J., Simons, L.M., Lorenzo-Redondo, R. et al. Evolution of SARS-CoV-2 in the murine central nervous system drives viral diversification. Nat Microbiol 9, 2383–2394 (2024). https://doi.org/10.1038/s41564-024-01786-8
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41564-024-01786-8