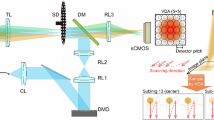
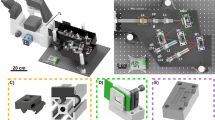
Abstract
Despite its proven value for biomedical research, super-resolution structured illumination microscopy still faces challenges in both fidelity of image reconstruction and imaging speed. Substantial background interference introduces artefacts and degrades resolution, while computationally intensive image reconstruction and illumination pattern switching limit imaging throughput. Here we present digital array modulation microscopy (DaMo), which combines digital array modulation with a single-spectrum reconstruction algorithm. Gaussian illumination modulation combined with digital detection modulation enables heterodyne detection with a 100% contrast. Therefore, DaMo achieves high-fidelity reconstruction (Pearson correlation coefficient 0.99 ± 0.01) under substantial background interference, with a 102× faster reconstruction speed than state-of-the-art super-resolution structured illumination microscopy processing. DaMo offers an axial resolution of 300 nm and a lateral resolution of 100 nm while achieving a 1,284-fold improvement in the signal-to-background ratio in whole-cell three-dimensional imaging. DaMo operates without additional modulators or extra image enhancement, providing artefact-free precision with a streamlined workflow. We demonstrate the versatility of DaMo via quantitative live-cell imaging of actin dynamics, tracking of cascaded filopodia fusion events, multicolour whole-smear imaging for cell cycle profiling, and tissue pathology assessment of intestinal epithelial injury in mitochondria. DaMo paves the way for large-scale, background-suppressed super-resolution imaging across diverse biological systems.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Several example datasets are available via Figshare at https://doi.org/10.6084/m9.figshare.29825972 (ref. 40). The same datasets can also be accessed via GitHub at https://github.com/MOSTYuan/DaMo (ref. 41). All the data that support the findings of this study are available from the corresponding author upon reasonable request.
Code availability
The reconstruction algorithm developed in this study is available via Figshare at https://doi.org/10.6084/m9.figshare.29825972 (ref. 40), and the identical code can also be found on GitHub at https://github.com/MOSTYuan/DaMo (ref. 41). Additional implementation details and related code are available from the corresponding author upon reasonable request.
References
Demmerle, J. et al. Strategic and practical guidelines for successful structured illumination microscopy. Nat. Protoc. 12, 988–1010 (2017).
Lal, A., Shan, C. & Xi, P. Structured illumination microscopy image reconstruction algorithm. IEEE J. Sel. Top. Quantum Electron. 22, 50–63 (2016).
Gustafsson, M. G. L. Surpassing the lateral resolution limit by a factor of two using structured illumination microscopy. J. Microsc. 198, 82–87 (2000).
Schermelleh, L. et al. Super-resolution microscopy demystified. Nat. Cell Biol. 21, 72–84 (2019).
Deng, H., Ma, Y. & Zhang, Y.-H. Super-resolution microscopy: shedding new light on blood cell imaging. J. Innov. Opt. Health Sci. 18, 2430006 (2024).
Zhao, T. et al. Super-resolution imaging reveals changes in Escherichia coli SSB localization in response to DNA damage. Genes Cells 24, 814–826 (2019).
Huang, X. et al. Fast, long-term, super-resolution imaging with Hessian structured illumination microscopy. Nat. Biotechnol. 36, 451–459 (2018).
Wu, H. et al. Single-frame structured illumination microscopy for fast live-cell imaging. APL Photonics 9, 036102 (2024).
Zhao, T. & Lei, M. Fast, faster, and the fastest structured illumination microscopy. Light Sci. Appl. 13, 186 (2024).
Wen, G. et al. High-fidelity structured illumination microscopy by point-spread-function engineering. Light Sci. Appl. 10, 70 (2021).
Zhao, W. et al. Sparse deconvolution improves the resolution of live-cell super-resolution fluorescence microscopy. Nat. Biotechnol. 40, 606–617 (2022).
Qiao, C. et al. Rationalized deep learning super-resolution microscopy for sustained live imaging of rapid subcellular processes. Nat. Biotechnol. 41, 367–377 (2023).
Qu, L. et al. Self-inspired learning for denoising live-cell super-resolution microscopy. Nat. Methods 21, 1895–1908 (2024).
Prakash, K. et al. Resolution in super-resolution microscopy — definition, trade-offs and perspectives. Nat. Rev. Mol. Cell Biol. 25, 677–682 (2024).
Wang, L. et al. Direct measurement and optimization of the polarization-dependent modulation depth in super-resolution structured illumination microscopy. J. Innov. Opt. Health Sci. 18, 2550005 (2024).
Markwirth, A. et al. Video-rate multicolor structured illumination microscopy with simultaneous real-time reconstruction. Nat. Commun. 10, 4315 (2019).
Wang, Z. et al. High-speed image reconstruction for optically sectioned, super-resolution structured illumination microscopy. Adv. Photonics 4, 026003 (2022).
Tu, S. et al. Fast reconstruction algorithm for structured illumination microscopy. Opt. Lett. 45, 1567–1570 (2020).
Dan, D. et al. Rapid image reconstruction of structured illumination microscopy directly in the spatial domain. IEEE Photonics J. 13, 1–11 (2021).
Tu, S. et al. High-speed spatially re-modulated structured illumination microscopy. Opt. Lett. 48, 2535–2538 (2023).
Wen, G. et al. Spectrum-optimized direct image reconstruction of super-resolution structured illumination microscopy. PhotoniX 4, 19 (2023).
Schermelleh, L. et al. Subdiffraction multicolor imaging of the nuclear periphery with 3D structured illumination microscopy. Science 320, 1332–1336 (2008).
Gustafsson, M. G. L. et al. Three-dimensional resolution doubling in wide-field fluorescence microscopy by structured illumination. Biophys. J. 94, 4957–4970 (2008).
Shao, L., Kner, P., Rego, E. H. & Gustafsson, M. G. L. Super-resolution 3D microscopy of live whole cells using structured illumination. Nat. Methods 8, 1044–1046 (2011).
Zhong, Q. et al. High-definition imaging using line-illumination modulation microscopy. Nat. Methods 18, 309–315 (2021).
Deng, L. et al. Cryo-fluorescence micro-optical sectioning tomography for volumetric imaging of various whole organs with subcellular resolution. iScience 25, 104805 (2022).
Qiao, W. et al. Single-scan HiLo with line-illumination strategy for optical section imaging of thick tissues. Biomed. Opt. Express 12, 2373–2383 (2021).
Qiao, W. et al. Differential synthetic illumination based on multi-line detection for resolution and contrast enhancement of line confocal microscopy. Opt. Express 31, 16093–16106 (2023).
Zhong, Q. et al. High-throughput optical sectioning via line-scanning imaging with digital structured modulation. Opt. Lett. 46, 504–507 (2021).
Zhang, J. et al. Optical sectioning methods in three-dimensional bioimaging. Light Sci. Appl. 14, 11 (2025).
Hou, Y. et al. Multi-resolution analysis enables fidelity-ensured deconvolution for fluorescence microscopy. eLight 4, 14 (2024).
Cao, R. et al. Dark-based optical sectioning assists background removal in fluorescence microscopy. Nat. Methods 22, 1299–1310 (2025).
Mo, Y. et al. Quantitative structured illumination microscopy via a physical model-based background filtering algorithm reveals actin dynamics. Nat. Commun. 14, 3089 (2023).
Lucy, L. B. An iterative technique for the rectification of observed distributions. Astron. J. 79, 745 (1974).
Richardson, W. H. Bayesian-based iterative method of image restoration. J. Opt. Soc. Am. 62, 55–59 (1972).
Wicker, K., Mandula, O., Best, G., Fiolka, R. & Heintzmann, R. Phase optimisation for structured illumination microscopy. Opt. Express 21, 2032–2049 (2013).
Seal, S. et al. Cell Painting: a decade of discovery and innovation in cellular imaging. Nat. Methods 22, 254–268 (2025).
Zhao, J. et al. Simultaneous multicolor imaging using off-axis spectral encoding in a single camera without sacrificing frame rate. Photonics Res. 13, 1925–1935 (2025).
Ershov, D. et al. TrackMate 7: integrating state-of-the-art segmentation algorithms into tracking pipelines. Nat. Methods 19, 829–832 (2022).
Li, S. et al. Example dataset for ‘Three-dimensional super-resolution imaging with suppressed background via digital array modulation microscopy’. Figshare https://doi.org/10.6084/m9.figshare.29825972 (2026).
Li, S. et al. Example dataset for ‘Three-dimensional super-resolution imaging with suppressedbackground via digital array modulation microscopy’, Github, https://github.com/MOSTYuan/DaMo (2026).
Acknowledgements
We thank L. Schermelleh and S. Zeng for constructive comments, T. Yu, S. Qi and Y. Su for providing Thy1-YFP mouse samples, and B. Lu for technical support for video production. This work was financially supported by the National Natural Science Foundation of China grant nos. 62325502 (J.Y.), 81827901 (Q.L.), 92354305 and 32271428 (Y.-H.Z.), and 82203968 (N.Z.), and the National Key R&D Program of China grant no. 2022YFC3401100 (Y.-H.Z.). We also thank the Optical Bioimaging Core Facility of WNLO-HUST for supporting data acquisition and the innovation fund of WNLO.
Author information
Authors and Affiliations
Contributions
J.Y. and Q.L. conceived and designed the study. S.L., R.J., M.L., S.G. and J.Y. derived the theory, constructed the microscope and performed data acquisition. M.Z., Y.W. and Y.-H.Z. prepared cell samples. N.Z. and F.W. prepared tissue samples. S.L., M.L., Z.Z. and J.Y. analysed the data and performed visualization. J.Y., H.G. and Q.L. administered the project. S.L. and J.Y. wrote and modified the manuscript with the input from all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Photonics thanks the anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Simulation results of WF and the other 7 SR-SIM reconstruction algorithms.
Extended images from Fig. 1b. The full‑field (256×256‑pixel) ground truth (GT) image is shown on the left, with a yellow rectangle marking the region of interest (ROI) that is enlarged in the adjacent panel and used consistently in Fig. 1b. To the right, the top row displays the wide‑field (WF) image and reconstructions of HiFi‑SIM and Hessian‑SIM for the same ROI, with their corresponding magenta‑green merged images with the GT shown directly below in the second row. The bottom two rows present reconstructions of RL‑SIM, SecMRA‑SIM, BF‑SIM, SRM‑SIM and Direct‑SIM (third row) and their respective merged images with the GT (fourth row). In all merged images, the reconstruction result is shown in magenta and the GT in green. The experiment was repeated three times with similar results.
Extended Data Fig. 2 Co-localization scatter plot between each SR reconstruction algorithm and GT.
Extended images from Fig. 1c. The experiment was repeated three times with similar results.
Extended Data Fig. 3 Algorithm comparison on the same mitotic cell.
Extended images from Fig. 4e. Mitochondria (TOMM20, green), microtubules (Anti-β-tubulin, hot red), and chromatin (Hoechst 33342, cyan).
Extended Data Fig. 4 DaMo images of different cellular states in the U2OS cell smear.
Extended images from Fig. 4a. a, Tunneling nanotubes and transported mitochondria (pink arrows). b, Tunneling nanotubes (pink arrows) and Micronucleus (white arrows). c, Cytokinetic bridge (pink arrow). d, Cytoplasmic vacuolization (pink arrow). e, Binucleation from abnormal cell division (pink arrow). f, Micronucleus from abnormal cell division (pink arrow). Mitochondria (TOMM20, green), microtubules (Anti-β-tubulin, hot red), and chromatin (Hoechst 33342, cyan).
Extended Data Fig. 5 Comparative DaMo and Sparse-SIM imaging of mitochondria, membranes, and nuclei in intestinal villi architecture.
Extended images from Fig. 5c. DaMo imaging of mitochondria, cell membranes, and nuclei across intestinal villi subregions (villus base, mid-villus, villus tip). White rectangles indicate corresponding Sparse-SIM reconstruction using HIS-SIM imaging.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–17, Tables 1–4 and Note.
Supplementary Video 1 (download MP4 )
Image stacks of Sparse-SIM and DaMo of the same U2OS cell as in Fig. 2a.
Supplementary Video 2 (download MP4 )
Long-term observation of the same living U2OS cell as in Fig. 3c.
Supplementary Video 3 (download MP4 )
Long-term observation of another living U2OS cell.
Supplementary Video 4 (download MP4 )
Whole-smear tri-colour SR image for the asynchronous population of 10,905 U2OS cells.
Supplementary Video 5 (download MP4 )
Muscle layers from a mouse small intestinal tissue slice.
Supplementary Video 6 (download MP4 )
Tri-colour SR imaging of a mouse small intestinal tissue slice.
Supplementary Video 7 (download MP4 )
Image stacks of confocal and DaMo of the same tissue slice of a mouse brain.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, S., Jin, R., Lu, M. et al. Three-dimensional super-resolution imaging with suppressed background via digital array modulation microscopy. Nat. Photon. (2026). https://doi.org/10.1038/s41566-026-01869-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41566-026-01869-4