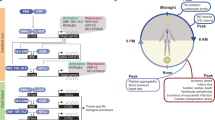
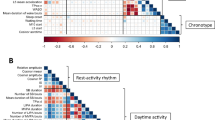
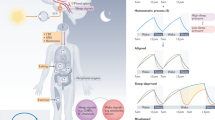
Abstract
The interaction between sleep, circadian rhythms and cardiovascular resilience is a crucial yet underexplored research area with important public health implications. Disruptions in sleep and circadian rhythms exacerbate hypertension, diabetes mellitus and obesity, conditions that are increasingly prevalent globally and increase the risk of cardiovascular disease. A National Heart, Lung, and Blood Institute workshop examined these connections, as well as the emerging concept of cardiovascular resilience as a dynamic and multifaceted concept spanning molecular, cellular and systemic levels across an individual’s lifespan. The workshop emphasized the need to expand the focus from solely understanding whether and how sleep and circadian rhythm disturbances contribute to disease, to also exploring how healthy sleep and aligned circadian rhythms can increase cardiovascular resilience. To develop a Roadmap towards this goal, workshop participants identified key knowledge gaps and research opportunities, including the need to integrate biological, behavioural, environmental and societal factors in sleep and circadian health with cardiovascular research to identify therapeutic targets. Proposed interventions encompass behavioural therapies, chronotherapy, lifestyle changes, organizational policies and public health initiatives aimed at improving sleep and circadian health for better cardiovascular outcomes. Future cross-disciplinary research and translation of discoveries into public health strategies and clinical practices could improve cardiovascular resilience across the lifespan in all populations.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Piber, D. et al. Sleep disturbance and activation of cellular and transcriptional mechanisms of inflammation in older adults. Brain Behav. Immun. 106, 67–75 (2022).
Leproult, R., Holmback, U. & Van Cauter, E. Circadian misalignment augments markers of insulin resistance and inflammation, independently of sleep loss. Diabetes 63, 1860–1869 (2014).
Copinschi, G., Leproult, R. & Spiegel, K. The important role of sleep in metabolism. Front. Horm. Res. 42, 59–72 (2014).
Irwin, M. R., Olmstead, R. & Carroll, J. E. Sleep disturbance, sleep duration, and inflammation: a systematic review and meta-analysis of cohort studies and experimental sleep deprivation. Biol. Psychiatry 80, 40–52 (2016).
Grandner, M. A. & Fernandez, F. X. The translational neuroscience of sleep: a contextual framework. Science 374, 568–573 (2021).
Cappuccio, F. P., Cooper, D., D’Elia, L., Strazzullo, P. & Miller, M. A. Sleep duration predicts cardiovascular outcomes: a systematic review and meta-analysis of prospective studies. Eur. Heart J. 32, 1484–1492 (2011).
Scheer, F. A., Hilton, M. F., Mantzoros, C. S. & Shea, S. A. Adverse metabolic and cardiovascular consequences of circadian misalignment. Proc. Natl Acad. Sci. USA 106, 4453–4458 (2009).
Aziz, M. et al. Association of subjective and objective sleep duration as well as sleep quality with non-invasive markers of sub-clinical cardiovascular disease (CVD): a systematic review. J. Atheroscler. Thromb. 24, 208–226 (2017).
Xiao, Q., Full, K. M., Rutter, M. K. & Lipworth, L. Long-term trajectories of sleep duration are associated with incident diabetes in middle-to-older-aged Black and White Americans. Diabetologia https://doi.org/10.1007/s00125-024-06202-8 (2024).
Chaput, J. P. et al. The role of insufficient sleep and circadian misalignment in obesity. Nat. Rev. Endocrinol. 19, 82–97 (2023).
Potter, G. D. et al. Circadian rhythm and sleep disruption: causes, metabolic consequences, and countermeasures. Endocr. Rev. 37, 584–608 (2016).
Tsao, C. W. et al. Heart disease and stroke statistics–2023 update: a report from the american heart association. Circulation 147, e93–e621 (2023).
Caraballo, C. et al. Evaluation of temporal trends in racial and ethnic disparities in sleep duration among US adults, 2004–2018. JAMA Netw. Open 5, e226385 (2022).
Tubbs, A. S. et al. Racial/ethnic minorities have greater declines in sleep duration with higher risk of cardiometabolic disease: an analysis of the U.S. National health interview survey. Sleep Epidemiol. 2, 100022 (2022).
Johnson, D. A., Jackson, C. L., Williams, N. J. & Alcantara, C. Are sleep patterns influenced by race/ethnicity - a marker of relative advantage or disadvantage? Evidence to date. Nat. Sci. Sleep. 11, 79–95 (2019).
Tan, S. C. W. et al. Global burden of cardiovascular diseases and its risk factors, 1990–2021: a systematic analysis for the global burden of disease study 2021. QJM https://doi.org/10.1093/qjmed/hcaf022 (2025).
Lim, D. C. et al. The need to promote sleep health in public health agendas across the globe. Lancet Public Health 8, e820–e826 (2023).
Smith, B. W. et al. The brief resilience scale: assessing the ability to bounce back. Int. J. Behav. Med. 15, 194–200 (2008).
National Institutes of Health. Trans-NIH Resilience Working Group (NIH). ods.od.nih.gov https://ods.od.nih.gov/Research/resilience.aspx (2024).
Taylor, H. A. et al. Scientific opportunities in resilience research for cardiovascular health and wellness. Report from a National Heart, Lung, and Blood Institute workshop. FASEB J. 36, e22639 (2022).
Guida, J. L. et al. Integrating sleep health into resilience research. Stress Health 39, 22–27 (2023).
Javaheri, S. et al. Sleep apnea: types, mechanisms, and clinical cardiovascular consequences. J. Am. Coll. Cardiol. 69, 841–858 (2017).
National Heart, Lung, and Blood Institute. NHLBI workshop on sleep and circadian rhythms in cardiovascular resilience (NHLBI). videocast.nih.gov https://videocast.nih.gov/watch=54632 (2024).
Southwick, S. M., Bonanno, G. A., Masten, A. S., Panter-Brick, C. & Yehuda, R. Resilience definitions, theory, and challenges: interdisciplinary perspectives. Eur. J. Psychotraumatol. https://doi.org/10.3402/ejpt.v5.25338 (2014).
Brown, L., Cohen, B., Costello, R., Brazhnik, O. & Galis, Z. Conceptualizing a resilience research framework at the National Institutes of Health. Stress Health 39, 4–9 (2023).
Sartor, F. et al. Circadian clock and hypoxia. Circ. Res. 134, 618–634 (2024).
Lloyd-Jones, D. M. et al. Defining and setting national goals for cardiovascular health promotion and disease reduction: the American Heart Association’s strategic impact goal through 2020 and beyond. Circulation 121, 586–613 (2010).
Lloyd-Jones, D. M. et al. Life’s Essential 8: updating and enhancing the American Heart Association’s construct of cardiovascular health: a presidential advisory from the American Heart Association. Circulation 146, e18–e43 (2022).
Sun, J. et al. Association of the American Heart Association’s new “Life’s Essential 8” with all-cause and cardiovascular disease-specific mortality: prospective cohort study. BMC Med. 21, 116 (2023).
Yi, J., Wang, L., Guo, X. & Ren, X. Association of Life’s Essential 8 with all-cause and cardiovascular mortality among US adults: a prospective cohort study from the NHANES 2005–2014. Nutr. Metab. Cardiovasc. Dis. 33, 1134–1143 (2023).
Herraiz-Adillo, A. et al. Life’s Essential 8 in relation to self-rated health and health-related quality of life in a large population-based sample: the SCAPIS project. Qual. Life Res. 33, 1003–1014 (2024).
Vanhoutte, P. M., Shimokawa, H., Feletou, M. & Tang, E. H. Endothelial dysfunction and vascular disease—a 30th anniversary update. Acta Physiol. 219, 22–96 (2017).
Gao, Y. & Galis, Z. S. Exploring the role of endothelial cell resilience in cardiovascular health and disease. Arterioscler. Thromb. Vasc. Biol. 41, 179–185 (2021).
Baeyens, N. et al. Vascular remodeling is governed by a VEGFR3-dependent fluid shear stress set point. eLife 4, e04645 (2015).
Astone, M., Oberkersch, R. E., Tosi, G., Biscontin, A. & Santoro, M. M. The circadian protein BMAL1 supports endothelial cell cycle during angiogenesis. Cardiovasc. Res. 119, 1952–1968 (2023).
Trimm, E. & Red-Horse, K. Vascular endothelial cell development and diversity. Nat. Rev. Cardiol. 20, 197–210 (2023).
Buysse, D. J. Sleep health: can we define it? Does it matter? Sleep 37, 9–17 (2014).
Mieda, M. The central circadian clock of the suprachiasmatic nucleus as an ensemble of multiple oscillatory neurons. Neurosci. Res. 156, 24–31 (2020).
Starnes, A. N. & Jones, J. R. Inputs and outputs of the mammalian circadian clock. Biology 12, 508 (2023).
Van Drunen, R. & Eckel-Mahan, K. Circadian rhythms of the hypothalamus: from function to physiology. Clocks Sleep 3, 189–226 (2021).
Abe, Y. O. et al. Rhythmic transcription of Bmal1 stabilizes the circadian timekeeping system in mammals. Nat. Commun. 13, 4652 (2022).
Xu, H. et al. Cryptochrome 1 regulates the circadian clock through dynamic interactions with the BMAL1 C terminus. Nat. Struct. Mol. Biol. 22, 476–484 (2015).
Koike, N. et al. Transcriptional architecture and chromatin landscape of the core circadian clock in mammals. Science 338, 349–354 (2012).
Duong, H. A. et al. Environmental circadian disruption re-writes liver circadian proteomes. Nat. Commun. 15, 5537 (2024).
Narasimamurthy, R. & Virshup, D. M. The phosphorylation switch that regulates ticking of the circadian clock. Mol. Cell 81, 1133–1146 (2021).
Marcheva, B. et al. in Circadian Clocks and Metabolism (eds Kramer, A. & Merrow, M.) 127–155 (Springer, 2013).
Reilly, D. F., Westgate, E. J. & FitzGerald, G. A. Peripheral circadian clocks in the vasculature. Arterioscler. Thromb. Vasc. Biol. 27, 1694–1705 (2007).
Acosta-Rodriguez, V. et al. Circadian alignment of early onset caloric restriction promotes longevity in male C57BL/6J mice. Science 376, 1192–1202 (2022).
Klarsfeld, A. & Rouyer, F. Effects of circadian mutations and LD periodicity on the life span of Drosophila melanogaster. J. Biol. Rhythm. 13, 471–478 (1998).
Libert, S., Bonkowski, M. S., Pointer, K., Pletcher, S. D. & Guarente, L. Deviation of innate circadian period from 24 h reduces longevity in mice. Aging Cell 11, 794–800 (2012).
Lananna, B. V. & Musiek, E. S. The wrinkling of time: aging, inflammation, oxidative stress, and the circadian clock in neurodegeneration. Neurobiol. Dis. 139, 104832 (2020).
Chellappa, S. L., Vujovic, N., Williams, J. S. & Scheer, F. Impact of circadian disruption on cardiovascular function and disease. Trends Endocrinol. Metab. 30, 767–779 (2019).
Ruger, M. & Scheer, F. A. Effects of circadian disruption on the cardiometabolic system. Rev. Endocr. Metab. Disord. 10, 245–260 (2009).
Sletten, T. L. et al. Health consequences of circadian disruption. Sleep 43, zsz194 (2020).
Carrington, M. J. et al. Changes in cardiovascular function during the sleep onset period in young adults. J. Appl. Physiol. 98, 468–476 (2005).
Trinder, J., Waloszek, J., Woods, M. J. & Jordan, A. S. Sleep and cardiovascular regulation. Pflugers Arch. 463, 161–168 (2012).
Ruben, M. D. et al. A database of tissue-specific rhythmically expressed human genes has potential applications in circadian medicine. Sci. Transl. Med. 10, eaat8806 (2018).
Anafi, R. C. et al. Sleep is not just for the brain: transcriptional responses to sleep in peripheral tissues. BMC Genomics 14, 362 (2013).
Shah, R. et al. Sleep deprivation impairs vascular function in healthy women: a clinical trial. Ann. Am. Thorac. Soc. 19, 2097–2100 (2022).
Gumz, M. L. et al. Toward precision medicine: circadian rhythm of blood pressure and chronotherapy for hypertension—2021 NHLBI workshop report. Hypertension 80, 503–522 (2023).
St-Onge, M. P. et al. Mild sleep restriction increases 24-hour ambulatory blood pressure in premenopausal women with no indication of mediation by psychological effects. Am. Heart J. 223, 12–22 (2020).
Lempiainen, P. A., Ylitalo, A., Huikuri, H., Kesaniemi, Y. A. & Ukkola, O. H. Non-dipping blood pressure pattern is associated with cardiovascular events in a 21-year follow-up study. J. Hum. Hypertens. 38, 444–451 (2024).
Bowman, M. A., Buysse, D. J., Foust, J. E., Oyefusi, V. & Hall, M. H. Disturbed sleep as a mechanism of race differences in nocturnal blood pressure non-dipping. Curr. Hypertens. Rep. 21, 51 (2019).
Ucak, S. et al. Heart rate variability analysis in obstructive sleep apnea patients with daytime sleepiness. Sleep 47, zsae075 (2024).
Ma, Y., Mullington, J. M., Wayne, P. M. & Yeh, G. Y. Heart rate variability during sleep onset in patients with insomnia with or without comorbid sleep apnea. Sleep Med. 122, 92–98 (2024).
Tsuji, H. et al. Impact of reduced heart rate variability on risk for cardiac events. The Framingham Heart Study. Circulation 94, 2850–2855 (1996).
Yugar, L. B. T. et al. The role of heart rate variability (HRV) in different hypertensive syndromes. Diagnostics 13, 785 (2023).
Zhong, X. et al. Increased sympathetic and decreased parasympathetic cardiovascular modulation in normal humans with acute sleep deprivation. J. Appl. Physiol. 98, 2024–2032 (2005).
Tall, A. R. & Jelic, S. How broken sleep promotes cardiovascular disease. Nature 566, 329–330 (2019).
Shah, R. et al. Mild sleep restriction increases endothelial oxidative stress in female persons. Sci. Rep. 13, 15360 (2023).
Morris, C. J., Purvis, T. E., Hu, K. & Scheer, F. A. Circadian misalignment increases cardiovascular disease risk factors in humans. Proc. Natl Acad. Sci. USA 113, E1402–E1411 (2016).
Poggiogalle, E., Jamshed, H. & Peterson, C. M. Circadian regulation of glucose, lipid, and energy metabolism in humans. Metabolism 84, 11–27 (2018).
Paschos, G. K. & FitzGerald, G. A. Circadian clocks and vascular function. Circ. Res. 106, 833–841 (2010).
Kumar, A. et al. Brain–muscle communication prevents muscle aging by maintaining daily physiology. Science 384, 563–572 (2024).
de Assis, L. V. M. & Oster, H. The circadian clock and metabolic homeostasis: entangled networks. Cell Mol. Life Sci. 78, 4563–4587 (2021).
Bass, J. Interorgan rhythmicity as a feature of healthful metabolism. Cell Metab. 36, 655–669 (2024).
Mortimer, T. et al. The epidermal circadian clock integrates and subverts brain signals to guarantee skin homeostasis. Cell Stem Cell 31, 834–849 e834 (2024).
Wang, H. et al. Time-restricted feeding shifts the skin circadian clock and alters UVB-induced DNA damage. Cell Rep. 20, 1061–1072 (2017).
Acosta-Rodriguez, V. A. et al. Misaligned feeding uncouples daily rhythms within brown adipose tissue and between peripheral clocks. Cell Rep. 43, 114523 (2024).
Franken, P. & Dijk, D. J. Sleep and circadian rhythmicity as entangled processes serving homeostasis. Nat. Rev. Neurosci. 25, 43–59 (2024).
Morf, J. et al. Cold-inducible RNA-binding protein modulates circadian gene expression posttranscriptionally. Science 338, 379–383 (2012).
Windred, D. P. et al. Higher central circadian temperature amplitude is associated with greater metabolite rhythmicity in humans. Sci. Rep. 14, 16796 (2024).
Archer, S. N. et al. Mistimed sleep disrupts circadian regulation of the human transcriptome. Proc. Natl Acad. Sci. USA 111, E682–E691 (2014).
Aggarwal, B. et al. Effects of inadequate sleep on blood pressure and endothelial inflammation in women: findings from the American Heart Association Go Red for Women Strategically Focused Research Network. J. Am. Heart Assoc. 7, e008590 (2018).
Makarem, N., St-Onge, M. P., Liao, M., Lloyd-Jones, D. M. & Aggarwal, B. Association of sleep characteristics with cardiovascular health among women and differences by race/ethnicity and menopausal status: findings from the American Heart Association Go Red for Women Strategically Focused Research Network. Sleep Health 5, 501–508 (2019).
St-Onge, M. P. et al. Sleep duration and quality: impact on lifestyle behaviors and cardiometabolic health: a scientific statement from the American Heart Association. Circulation 134, e367–e386 (2016).
Zuraikat, F. M. et al. Evening chronotype is associated with poorer habitual diet in US women, with dietary energy density mediating a relation of chronotype with cardiovascular health. J. Nutr. 151, 1150–1158 (2021).
Kanagasabai, T. & Chaput, J. P. Sleep duration and the associated cardiometabolic risk scores in adults. Sleep Health 3, 195–203 (2017).
Torquati, L., Mielke, G. I., Brown, W. J. & Kolbe-Alexander, T. Shift work and the risk of cardiovascular disease. A systematic review and meta-analysis including dose–response relationship. Scand. J. Work Environ. Health 44, 229–238 (2018).
Schettini, M. A. S., Passos, R. & Koike, B. D. V. Shift work and metabolic syndrome updates: a systematic review. Sleep Sci. 16, 237–247 (2023).
Garbarino, S., Lanteri, P., Bragazzi, N. L., Magnavita, N. & Scoditti, E. Role of sleep deprivation in immune-related disease risk and outcomes. Commun. Biol. 4, 1304 (2021).
Barragan, R. et al. Paradoxical effects of prolonged insufficient sleep on lipid profile: a pooled analysis of 2 randomized trials. J. Am. Heart Assoc. 12, e032078 (2023).
Humer, E., Pieh, C. & Brandmayr, G. Metabolomics in sleep, insomnia and sleep apnea. Int. J. Mol. Sci. 21, 7244 (2020).
Panda, S. Circadian physiology of metabolism. Science 354, 1008–1015 (2016).
Zuraikat, F. M. et al. Chronic insufficient sleep in women impairs insulin sensitivity independent of adiposity changes: results of a randomized trial. Diabetes Care 47, 117–125 (2024).
St-Onge, M. P. et al. Impact of change in bedtime variability on body composition and inflammation: secondary findings from the Go Red for Women Strategically Focused Research Network. Int. J. Obes. 44, 1803–1806 (2020).
Mullington, J. M., Haack, M., Toth, M., Serrador, J. M. & Meier-Ewert, H. K. Cardiovascular, inflammatory, and metabolic consequences of sleep deprivation. Prog. Cardiovasc. Dis. 51, 294–302 (2009).
Blake, M. J., Trinder, J. A. & Allen, N. B. Mechanisms underlying the association between insomnia, anxiety, and depression in adolescence: implications for behavioral sleep interventions. Clin. Psychol. Rev. 63, 25–40 (2018).
Gao, X., Huang, N., Guo, X. & Huang, T. Role of sleep quality in the acceleration of biological aging and its potential for preventive interaction on air pollution insults: findings from the UK Biobank cohort. Aging Cell 21, e13610 (2022).
Lisan, Q. et al. Sleep apnea is associated with accelerated vascular aging: results from 2 European community-based cohort studies. J. Am. Heart Assoc. 10, e021318 (2021).
Gaspar, L. S., Alvaro, A. R., Moita, J. & Cavadas, C. Obstructive sleep apnea and hallmarks of aging. Trends Mol. Med. 23, 675–692 (2017).
Cortese, R. Epigenetics and aging: relevance for sleep medicine. Curr. Opin. Pulm. Med. https://doi.org/10.1097/MCP.0000000000001109 (2024).
Vaccaro, A. et al. Sleep loss can cause death through accumulation of reactive oxygen species in the gut. Cell 181, 1307–1328 e1315 (2020).
Williams, M. J. et al. Recurrent sleep fragmentation induces insulin and neuroprotective mechanisms in middle-aged flies. Front. Aging Neurosci. 8, 180 (2016).
Silva-Islas, C. A. & Maldonado, P. D. Canonical and non-canonical mechanisms of Nrf2 activation. Pharmacol. Res. 134, 92–99 (2018).
Zhang, D. D. Mechanistic studies of the Nrf2–Keap1 signaling pathway. Drug Metab. Rev. 38, 769–789 (2006).
Mayat, Z., Giardina, E. V., Liao, M. & Aggarwal, B. Associations of inflammation, physical activity, and sleep in a diverse population of women. J. Women Health 29, 1007–1016 (2020).
Wright, K. P. Jr. et al. Influence of sleep deprivation and circadian misalignment on cortisol, inflammatory markers, and cytokine balance. Brain Behav. Immun. 47, 24–34 (2015).
Ferreira, J. P., Vasques-Novoa, F., Neves, J. S., Zannad, F. & Leite-Moreira, A. Comparison of interleukin-6 and high-sensitivity C-reactive protein for cardiovascular risk assessment: findings from the MESA study. Atherosclerosis 390, 117461 (2024).
Janssen, H., Koekkoek, L. L. & Swirski, F. K. Effects of lifestyle factors on leukocytes in cardiovascular health and disease. Nat. Rev. Cardiol. 21, 157–169 (2024).
McAlpine, C. S. et al. Sleep exerts lasting effects on hematopoietic stem cell function and diversity. J. Exp. Med. 219, e20220081 (2022).
McAlpine, C. S. et al. Sleep modulates haematopoiesis and protects against atherosclerosis. Nature 566, 383–387 (2019).
Vallat, R., Shah, V. D., Redline, S., Attia, P. & Walker, M. P. Broken sleep predicts hardened blood vessels. PLoS Biol. 18, e3000726 (2020).
Scheiermann, C., Gibbs, J., Ince, L. & Loudon, A. Clocking in to immunity. Nat. Rev. Immunol. 18, 423–437 (2018).
Angeles-Castellanos, M., Salgado-Delgado, R., Rodriguez, K., Buijs, R. M. & Escobar, C. The suprachiasmatic nucleus participates in food entrainment: a lesion study. Neuroscience 165, 1115–1126 (2010).
Dumbell, R., Matveeva, O. & Oster, H. Circadian clocks, stress, and immunity. Front. Endocrinol. 7, 37 (2016).
Alexander, R. K. et al. Bmal1 integrates mitochondrial metabolism and macrophage activation. eLife 9, e54090 (2020).
Nakabo, S. et al. The circadian clock gene BMAL1 modulates autoimmunity features in lupus. Front. Immunol. 15, 1465185 (2024).
Benasi, G., Cheng, B., Aggarwal, B. & St-Onge, M. P. The effects of sustained mild sleep restriction on stress and distress among healthy adults: findings from two randomized crossover studies. Sleep Med. 115, 83–87 (2024).
Demichelis, O. P. et al. Sleep, stress and aggression: meta-analyses investigating associations and causality. Neurosci. Biobehav. Rev. 139, 104732 (2022).
Guyon, A. et al. Adverse effects of two nights of sleep restriction on the hypothalamic–pituitary–adrenal axis in healthy men. J. Clin. Endocrinol. Metab. 99, 2861–2868 (2014).
Vgontzas, A. N. et al. Adverse effects of modest sleep restriction on sleepiness, performance, and inflammatory cytokines. J. Clin. Endocrinol. Metab. 89, 2119–2126 (2004).
van Leeuwen, W. M. A. et al. Physiological and autonomic stress responses after prolonged sleep restriction and subsequent recovery sleep in healthy young men. Sleep Biol. Rhythm. 16, 45–54 (2018).
Messa, R. M. et al. The effect of total sleep deprivation on autonomic nervous system and cortisol responses to acute stressors in healthy individuals: a systematic review. Psychoneuroendocrinology 168, 107114 (2024).
Morin, C. M. & Jarrin, D. C. Epidemiology of insomnia: prevalence, course, risk factors, and public health burden. Sleep Med. Clin. 17, 173–191 (2022).
Dressle, R. J. et al. HPA axis activity in patients with chronic insomnia: a systematic review and meta-analysis of case–control studies. Sleep Med. Rev. 62, 101588 (2022).
Dressle, R. J. & Riemann, D. Hyperarousal in insomnia disorder: current evidence and potential mechanisms. J. Sleep Res. 32, e13928 (2023).
Dai, Y. et al. A meta-analysis of the association between insomnia with objective short sleep duration and risk of hypertension. Sleep Med. Rev. 75, 101914 (2024).
Pejovic, S. et al. Insomnia with objective but not subjective short sleep duration is associated with incident cardiovascular and/or cerebrovascular disease. J. Clin. Sleep Med. 20, 1049–1057 (2024).
Li, X. L., Wei, J., Zhang, X., Meng, Z. & Zhu, W. Relationship between night-sleep duration and risk for depression among middle-aged and older people: a dose–response meta-analysis. Front. Physiol. 14, 1085091 (2023).
Mac Giollabhui, N., Ng, T. H., Ellman, L. M. & Alloy, L. B. The longitudinal associations of inflammatory biomarkers and depression revisited: systematic review, meta-analysis, and meta-regression. Mol. Psychiatry 26, 3302–3314 (2021).
Koch, C., Wilhelm, M., Salzmann, S., Rief, W. & Euteneuer, F. A meta-analysis of heart rate variability in major depression. Psychol. Med. 49, 1948–1957 (2019).
Zajkowska, Z. et al. Cortisol and development of depression in adolescence and young adulthood—a systematic review and meta-analysis. Psychoneuroendocrinology 136, 105625 (2022).
Chen, M., Li, B., Fan, G. & Zhou, Y. Restricted linear association between night sleep duration and diabetes risk in middle-aged and older adults: a 7-year follow-up analysis from the China health and retirement longitudinal study. Front. Endocrinol. 15, 1364106 (2024).
Barragan, R., Zuraikat, F. M., Tam, V., RoyChoudhury, A. & St-Onge, M. P. Changes in eating patterns in response to chronic insufficient sleep and their associations with diet quality: a randomized trial. J. Clin. Sleep Med. 19, 1867–1875 (2023).
St-Onge, M. P. et al. The interrelationship between sleep, diet, and glucose metabolism. Sleep Med. Rev. 69, 101788 (2023).
Shimizu, I., Yoshida, Y. & Minamino, T. A role for circadian clock in metabolic disease. Hypertens. Res. 39, 483–491 (2016).
Tasali, E., Leproult, R. & Spiegel, K. Reduced sleep duration or quality: relationships with insulin resistance and type 2 diabetes. Prog. Cardiovasc. Dis. 51, 381–391 (2009).
Mullington, J. M., Simpson, N. S., Meier-Ewert, H. K. & Haack, M. Sleep loss and inflammation. Best Pract. Res. Clin. Endocrinol. Metab. 24, 775–784 (2010).
Hurtado-Alvarado, G. et al. Sleep loss as a factor to induce cellular and molecular inflammatory variations. Clin. Dev. Immunol. 2013, 801341 (2013).
Chua, E. C., Shui, G., Cazenave-Gassiot, A., Wenk, M. R. & Gooley, J. J. Changes in plasma lipids during exposure to total sleep deprivation. Sleep 38, 1683–1691 (2015).
Arnett, D. K., Khera, A. & Blumenthal, R. S. 2019 ACC/AHA guideline on the primary prevention of cardiovascular disease: part 1, lifestyle and behavioral factors. JAMA Cardiol. 4, 1043–1044 (2019).
Tempaku, P. F., D’Almeida, V., Andersen, M. L. & Tufik, S. Sleep is associated with telomere shortening: a population-based longitudinal study. J. Sleep Res. 34, e14274 (2024).
Li, X. et al. Association between sleep disordered breathing and epigenetic age acceleration: evidence from the multi-ethnic study of atherosclerosis. EBioMedicine 50, 387–394 (2019).
Cortese, R., Sanz-Rubio, D., Kheirandish-Gozal, L., Marin, J. M. & Gozal, D. Epigenetic age acceleration in obstructive sleep apnoea is reversible with adherent treatment. Eur. Respir. J. 59, 2103042 (2022).
Besedovsky, L., Lange, T. & Haack, M. The sleep–immune crosstalk in health and disease. Physiol. Rev. 99, 1325–1380 (2019).
Liu, X. et al. Effects of poor sleep on the immune cell landscape as assessed by single-cell analysis. Commun. Biol. 4, 1325 (2021).
Carroll, J. E. & Prather, A. A. Sleep and biological aging: a short review. Curr. Opin. Endocr. Metab. Res. 18, 159–164 (2021).
D’Agostino, R. B. Sr. et al. General cardiovascular risk profile for use in primary care: the Framingham Heart Study. Circulation 117, 743–753 (2008).
Li, J., Vitiello, M. V. & Gooneratne, N. S. Sleep in normal aging. Sleep Med. Clin. 13, 1–11 (2018).
Miner, B. & Kryger, M. H. Sleep in the aging population. Sleep Med. Clin. 12, 31–38 (2017).
Jaqua, E. E., Hanna, M., Labib, W., Moore, C. & Matossian, V. Common sleep disorders affecting older adults. Perm. J. 27, 122–132 (2023).
Cohen, Z. L., Eigenberger, P. M., Sharkey, K. M., Conroy, M. L. & Wilkins, K. M. Insomnia and other sleep disorders in older adults. Psychiatr. Clin. North Am. 45, 717–734 (2022).
Carroll, J. E. et al. Obstructive sleep apnea, nighttime arousals, and leukocyte telomere length: the multi-ethnic study of atherosclerosis. Sleep 42, zsz089 (2019).
Carroll, J. E. et al. Epigenetic aging and immune senescence in women with insomnia symptoms: findings from the Women’s Health Initiative Study. Biol. Psychiatry 81, 136–144 (2017).
Carvalhas-Almeida, C., Cavadas, C. & Alvaro, A. R. The impact of insomnia on frailty and the hallmarks of aging. Aging Clin. Exp. Res. 35, 253–269 (2023).
Hood, S. & Amir, S. The aging clock: circadian rhythms and later life. J. Clin. Invest. 127, 437–446 (2017).
Cai, R. et al. Circadian disturbances and frailty risk in older adults. Nat. Commun. 14, 7219 (2023).
Badran, M. et al. Senolytic-facilitated reversal of end-organ dysfunction in a murine model of obstructive sleep apnea. Am. J. Respir. Crit. Care Med. 209, 1001–1012 (2024).
Rasmussen-Torvik, L. J. et al. The mediation of racial differences in hypertension by sleep characteristics: Chicago area sleep study. Am. J. Hypertens. 29, 1353–1357 (2016).
Kim, I. B., Lee, J. H. & Park, S. C. The relationship between stress, inflammation, and depression. Biomedicines 10, 1929 (2022).
Back, L. & Solomos, J. Theories of Race and Racism: a Reader 2nd edn (Routledge, 2020).
Harrell, S. P. A multidimensional conceptualization of racism‐related stress: implications for the well‐being of people of color. Am. J. Orthopsychiatry 70, 42–57 (2000).
Grandner, M. A., Williams, N. J., Knutson, K. L., Roberts, D. & Jean-Louis, G. Sleep disparity, race/ethnicity, and socioeconomic position. Sleep Med. 18, 7–18 (2016).
Grandner, M. A., Rattanaumpawan, P., Jackson, N. J. & Patel, N. P. Sleep disparity, race/ethnicity, and socioeconomic position. Sleep 35, A348–A348 (2012).
Jean-Louis, G., Grandner, M. A. & Seixas, A. A. Social determinants and health disparities affecting sleep. Lancet Neurol. 21, 864–865 (2022).
Laposky, A. D., Van Cauter, E. & Diez-Roux, A. V. Reducing health disparities: the role of sleep deficiency and sleep disorders. Sleep Med. 18, 3–6 (2016).
Lee, S. et al. Sleep health composites are associated with the risk of heart disease across sex and race. Sci. Rep. https://doi.org/10.1038/s41598-022-05203-0 (2022).
Johnson, D. A. et al. Association between sleep apnea and blood pressure control among blacks. Circulation 139, 1275–1284 (2019).
Scott, H. et al. Sleep irregularity is associated with hypertension: findings from over 2 million nights with a large global population sample. Hypertension 80, 1117–1126 (2023).
Lao, X. Q. et al. Sleep quality, sleep duration, and the risk of coronary heart disease: a prospective cohort study with 60,586 adults. J. Clin. Sleep Med. 14, 109–117 (2018).
Hughes, A. J. et al. Eliminating sleep health disparities and achieving health equity: seven areas for action in the behavioral sleep medicine community. Behav. Sleep Med. 21, 633–645 (2023).
Dietch, J. R. et al. Gender and racial/ethnic differences in sleep duration in the North Texas heart study. Sleep Health 3, 324–327 (2017).
Cheng, P. et al. Racial discrimination as a mediator of racial disparities in insomnia disorder. Sleep Health 6, 543–549 (2020).
Saelee, R., Haardorfer, R., Johnson, D. A., Gazmararian, J. A. & Suglia, S. F. Neighborhood and household environment as contributors to racial disparities in sleep duration among U.S. adolescents. Sleep Epidemiol. 3, 100065 (2023).
Billings, M. E. et al. Disparities in sleep health and potential intervention models: a focused review. Chest 159, 1232–1240 (2021).
Johnson, D. A. et al. The neighborhood social environment and objective measures of sleep in the multi-ethnic study of atherosclerosis. Sleep 40, zsw016 (2017).
Bowleg, L. Evolving intersectionality within public health: from analysis to action. Am. J. Public Health 111, 88–90 (2021).
Festus, I. D. et al. Pioneering new frontiers in circadian medicine chronotherapies for cardiovascular health. Trends Endocrinol. Metab. 35, 607–623 (2024).
McCarthy, C. E. et al. Behavioral sleep interventions and cardiovascular risk factors: systematic review and meta-analysis of randomized controlled trials. J. Cardiovasc. Nurs. 39, E158–E171 (2024).
Savin, K. L. et al. The effect of cognitive behavioral therapy for insomnia (CBT-I) on cardiometabolic health biomarkers: a systematic review of randomized controlled trials. Behav. Sleep Med. 21, 671–694 (2023).
Goldstein, C. A. et al. Artificial intelligence in sleep medicine: an American academy of sleep medicine position statement. J. Clin. Sleep Med. 16, 605–607 (2020).
Lujan, M. R., Perez-Pozuelo, I. & Grandner, M. A. Past, present, and future of multisensory wearable technology to monitor sleep and circadian rhythms. Front. Digit. Health 3, 721919 (2021).
Baron, K. G. et al. Sleep technology intervention to target cardiometabolic health (STITCH): a randomized controlled study of a behavioral sleep extension intervention compared to an education control to improve sleep duration, blood pressure, and cardiometabolic health among adults with elevated blood pressure/hypertension. Trials 24, 658 (2023).
Sulli, G., Manoogian, E. N. C., Taub, P. R. & Panda, S. Training the circadian clock, clocking the drugs, and drugging the clock to prevent, manage, and treat chronic diseases. Trends Pharmacol. Sci. 39, 812–827 (2018).
Gunal, A. M. Sleep, activity, and diet in harmony: unveiling the relationships of chronotype, sleep quality, physical activity, and dietary intake. Front. Nutr. 10, 1301818 (2023).
Raji, O. E., Kyeremah, E. B., Sears, D. D., St-Onge, M. P. & Makarem, N. Chrononutrition and cardiometabolic health: an overview of epidemiological evidence and key future research directions. Nutrients 16, 2332 (2024).
Franzago, M., Alessandrelli, E., Notarangelo, S., Stuppia, L. & Vitacolonna, E. Chrono-nutrition: circadian rhythm and personalized nutrition. Int. J. Mol. Sci. 24, 2571 (2023).
Yan, L., Rust, B. M. & Palmer, D. G. Time-restricted feeding restores metabolic flexibility in adult mice with excess adiposity. Front. Nutr. 11, 1340735 (2024).
McAllister, M. J., Pigg, B. L., Renteria, L. I. & Waldman, H. S. Time-restricted feeding improves markers of cardiometabolic health in physically active college-age men: a 4-week randomized pre–post pilot study. Nutr. Res. 75, 32–43 (2020).
Moro, T. et al. Twelve months of time-restricted eating and resistance training improves inflammatory markers and cardiometabolic risk factors. Med. Sci. Sports Exerc. 53, 2577–2585 (2021).
Stratton, M. T. et al. Four weeks of time-restricted feeding combined with resistance training does not differentially influence measures of body composition, muscle performance, resting energy expenditure, and blood biomarkers. Nutrients 12, 1126 (2020).
Farshchi, H. R., Taylor, M. A. & Macdonald, I. A. Deleterious effects of omitting breakfast on insulin sensitivity and fasting lipid profiles in healthy lean women. Am. J. Clin. Nutr. 81, 388–396 (2005).
Carlson, O. et al. Impact of reduced meal frequency without caloric restriction on glucose regulation in healthy, normal-weight middle-aged men and women. Metabolism 56, 1729–1734 (2007).
Petersen, M. C. et al. Complex physiology and clinical implications of time-restricted eating. Physiol. Rev. 102, 1991–2034 (2022).
Dote-Montero, M. et al. Effects of early, late and self-selected time-restricted eating on visceral adipose tissue and cardiometabolic health in participants with overweight or obesity: a randomized controlled trial. Nat. Med. https://doi.org/10.1038/s41591-024-03375-y (2025).
Fishbein, A. B., Knutson, K. L. & Zee, P. C. Circadian disruption and human health. J. Clin. Invest. 131, e148286 (2021).
Kansagara, D., Wilt, T. J., Starkey, M. & Qaseem, A. Management of chronic insomnia disorder in adults. Ann. Intern. Med. 165, 892 (2016).
Qaseem, A. et al. Management of chronic insomnia disorder in adults: a clinical practice guideline from the American College of Physicians. Ann. Intern. Med. 165, 125–133 (2016).
Edinger, J. D. et al. Behavioral and psychological treatments for chronic insomnia disorder in adults: an American Academy of Sleep Medicine clinical practice guideline. J. Clin. Sleep Med. 17, 255–262 (2021).
Trauer, J. M., Qian, M. Y., Doyle, J. S., Rajaratnam, S. M. & Cunnington, D. Cognitive behavioral therapy for chronic insomnia: a systematic review and meta-analysis. Ann. Intern. Med. 163, 191–204 (2015).
Edinger, J. D. et al. Behavioral and psychological treatments for chronic insomnia disorder in adults: an American Academy of Sleep Medicine systematic review, meta-analysis, and GRADE assessment. J. Clin. Sleep Med. 17, 263–298 (2021).
Ong, J. C. & Kalmbach, D. A. Mindfulness as an adjunct or alternative to CBT-I. Sleep Med. Clin. 18, 59–71 (2023).
Ong, J. C., Shapiro, S. L. & Manber, R. Combining mindfulness meditation with cognitive-behavior therapy for insomnia: a treatment-development study. Behav. Ther. 39, 171–182 (2008).
Alcantara, C. et al. Cultural adaptations of psychological interventions for prevalent sleep disorders and sleep disturbances: a systematic review of randomized controlled trials in the United States. Sleep Med. Rev. 56, 101455 (2021).
O’Bryan, S. E. et al. Community based participatory research as a promising practice for addressing vaccine hesitancy, rebuilding trust and addressing health disparities among racial and ethnic minority communities. Hum. Vaccin. Immunother. 20, 2326781 (2024).
Berkman, L. F., Glass, T., Brissette, I. & Seeman, T. E. From social integration to health: durkheim in the new millennium. Soc. Sci. Med. 51, 843–857 (2000).
Lu, W. Z., Lin, H. A., Hou, S. K. & Lin, S. F. ABCD2-I score predicts unplanned emergency department revisits within 72 hours due to recurrent acute ischemic stroke. Diagnostics 14, 1118 (2024).
Moretti Anfossi, C. et al. Work exposures and development of cardiovascular diseases: a systematic review. Ann. Work Expo. Health 66, 698–713 (2022).
Virtanen, M. & Kivimaki, M. Long working hours and risk of cardiovascular disease. Curr. Cardiol. Rep. 20, 123 (2018).
Rabiei, H., Ramezanifar, S., Hassanipour, S. & Gharari, N. Investigating the effects of occupational and environmental noise on cardiovascular diseases: a systematic review and meta-analysis. Environ. Sci. Pollut. Res. Int. 28, 62012–62029 (2021).
Yang, B. et al. Association between insomnia and job stress: a meta-analysis. Sleep Breath. 22, 1221–1231 (2018).
Harden, A., Peersman, G., Oliver, S., Mauthner, M. & Oakley, A. A systematic review of the effectiveness of health promotion interventions in the workplace. Occup. Med. 49, 540–548 (1999).
Grossmeier, J. et al. Linking workplace health promotion best practices and organizational financial performance: tracking market performance of companies with highest scores on the HERO scorecard. J. Occup. Environ. Med. 58, 16–23 (2016).
Redeker, N. S. et al. Workplace interventions to promote sleep health and an alert, healthy workforce. J. Clin. Sleep Med. 15, 649–657 (2019).
Proper, K. I. & van Oostrom, S. H. The effectiveness of workplace health promotion interventions on physical and mental health outcomes—a systematic review of reviews. Scand. J. Work Environ. Health 45, 546–559 (2019).
Arena, R. et al. Promoting health and wellness in the workplace: a unique opportunity to establish primary and extended secondary cardiovascular risk reduction programs. Mayo Clin. Proc. 88, 605–617 (2013).
Ajslev, J. Z. N., Moller, J. L., Andersen, M. F., Pirzadeh, P. & Lingard, H. The hierarchy of controls as an approach to visualize the impact of occupational safety and health coordination. Int. J. Environ. Res. Public Health 19, 2731 (2022).
Vargas, I., Egeler, M., Walker, J. & Benitez, D. D. Examining the barriers and recommendations for integrating more equitable insomnia treatment options in primary care. Front. Sleep https://doi.org/10.3389/frsle.2023.1279903 (2023).
Hermida, R. C., Ayala, D. E., Calvo, C. & Lopez, J. E. Aspirin administered at bedtime, but not on awakening, has an effect on ambulatory blood pressure in hypertensive patients. J. Am. Coll. Cardiol. 46, 975–983 (2005).
Hermida, R. C., Ayala, D. E., Mojon, A. & Fernandez, J. R. Chronotherapy with nifedipine GITS in hypertensive patients: improved efficacy and safety with bedtime dosing. Am. J. Hypertens. 21, 948–954 (2008).
Millar-Craig, M. W., Bishop, C. N. & Raftery, E. B. Circadian variation of blood-pressure. Lancet 1, 795–797 (1978).
Hermida, R. C., Ayala, D. E., Mojon, A. & Fernandez, J. R. Influence of circadian time of hypertension treatment on cardiovascular risk: results of the MAPEC study. Chronobiol. Int. 27, 1629–1651 (2010).
Hermida, R. C. et al. Bedtime hypertension treatment improves cardiovascular risk reduction: the Hygia chronotherapy trial. Eur. Heart J. 41, 4565–4576 (2020).
Brunstrom, M. et al. Missing verification of source data in hypertension research: the Hygia project in perspective. Hypertension 78, 555–558 (2021).
Guthrie, G. et al. Chronotherapy in hypertension: the devil is in the details. Eur. Heart J. 41, 1606–1607 (2020).
Kreutz, R. et al. Disregard the reported data from the Hygia project: blood pressure medication not to be routinely dosed at bedtime. J. Hypertens. 38, 2144–2145 (2020).
Garrison, S. R. et al. Bedtime versus morning use of antihypertensives in frail continuing care residents (BedMed-Frail): protocol for a prospective, randomised, open-label, blinded end-point pragmatic trial. BMJ Open 13, e074777 (2023).
Mackenzie, I. S. et al. Cardiovascular outcomes in adults with hypertension with evening versus morning dosing of usual antihypertensives in the UK (TIME study): a prospective, randomised, open-label, blinded-endpoint clinical trial. Lancet 400, 1417–1425 (2022).
Muller, J. E. et al. Circadian variation in the frequency of onset of acute myocardial infarction. N. Engl. J. Med. 313, 1315–1322 (1985).
Muller, J. E. et al. Circadian variation in the frequency of sudden cardiac death. Circulation 75, 131–138 (1987).
Willich, S. N. et al. Circadian variation in the incidence of sudden cardiac death in the Framingham Heart Study population. Am. J. Cardiol. 60, 801–806 (1987).
White, W. B. et al. Comparison of effects of controlled onset extended release verapamil at bedtime and nifedipine gastrointestinal therapeutic system on arising on early morning blood pressure, heart rate, and the heart rate-blood pressure product. Am. J. Cardiol. 81, 424–431 (1998).
White, W. B., Elliott, W. J., Johnson, M. F. & Black, H. R. Chronotherapeutic delivery of verapamil in obese versus non-obese patients with essential hypertension. J. Hum. Hypertens. 15, 135–141 (2001).
Prisant, L. M., Black, H. R., Messerli, F. & Weber, M. Chrono: a community-based hypertension trial of a chronotherapeutic formulation of verapamil. Am. J. Ther. 9, 476–483 (2002).
Petrenko, V., Sinturel, F., Riezman, H. & Dibner, C. Lipid metabolism around the body clocks. Prog. Lipid Res. 91, 101235 (2023).
Petsouki, E., Cabrera, S. N. S. & Heiss, E. H. AMPK and NRF2: interactive players in the same team for cellular homeostasis? Free Radic. Biol. Med. 190, 75–93 (2022).
Cuadrado, A. et al. Therapeutic targeting of the NRF2 and KEAP1 partnership in chronic diseases. Nat. Rev. Drug Discov. 18, 295–317 (2019).
Bayo Jimenez, M. T. et al. Protective actions of nuclear factor erythroid 2-related factor 2 (NRF2) and downstream pathways against environmental stressors. Free Radic. Biol. Med. 187, 72–91 (2022).
Ungvari, Z. et al. Nrf2 dysfunction and impaired cellular resilience to oxidative stressors in the aged vasculature: from increased cellular senescence to the pathogenesis of age-related vascular diseases. Geroscience 41, 727–738 (2019).
Rana, U. et al. Nogo-B receptor deficiency causes cerebral vasculature defects during embryonic development in mice. Dev. Biol. 410, 190–201 (2016).
Fang, Z. et al. NOGOB receptor deficiency increases cerebrovascular permeability and hemorrhage via impairing histone acetylation-mediated CCM1/2 expression. J. Clin. Invest. 132, e151382 (2022).
He, L. et al. Activation of Nrf2 inhibits atherosclerosis in ApoE−/− mice through suppressing endothelial cell inflammation and lipid peroxidation. Redox Biol. 74, 103229 (2024).
Lim, J. L. et al. Protective effects of monomethyl fumarate at the inflamed blood–brain barrier. Microvasc. Res. 105, 61–69 (2016).
Oh, C. J. et al. Dimethylfumarate attenuates restenosis after acute vascular injury by cell-specific and Nrf2-dependent mechanisms. Redox Biol. 2, 855–864 (2014).
Fledderus, J. O. et al. KLF2 primes the antioxidant transcription factor Nrf2 for activation in endothelial cells. Arterioscler. Thromb. Vasc. Biol. 28, 1339–1346 (2008).
Dabravolski, S. A. et al. The role of KLF2 in the regulation of atherosclerosis development and potential use of KLF2-targeted therapy. Biomedicines 10, 254 (2022).
Mezhnina, V., Ebeigbe, O. P., Poe, A. & Kondratov, R. V. Circadian control of mitochondria in reactive oxygen species homeostasis. Antioxid. Redox Signal. 37, 647–663 (2022).
Xie, M. et al. BMAL1-downregulation aggravates porphyromonas gingivalis-induced atherosclerosis by encouraging oxidative stress. Circ. Res. 126, e15–e29 (2020).
Barone, I. et al. Synaptic BMAL1 phosphorylation controls circadian hippocampal plasticity. Sci. Adv. 9, eadj1010 (2023).
Li, H. et al. ISX-9 potentiates CaMKIIδ-mediated BMAL1 activation to enhance circadian amplitude. Commun. Biol. 5, 750 (2022).
Young, M. E. The cardiac circadian clock: implications for cardiovascular disease and its treatment. JACC Basic Transl. Sci. 8, 1613–1628 (2023).
Ludwig, M., Wienke, C., Betts, M. J., Zaehle, T. & Hammerer, D. Current challenges in reliably targeting the noradrenergic locus coeruleus using transcutaneous auricular vagus nerve stimulation (taVNS). Auton. Neurosci. 236, 102900 (2021).
He, J. K. et al. Transcutaneous auricular vagus nerve stimulation modulates the prefrontal cortex in chronic insomnia patients: fMRI study in the first session. Front. Neurol. 13, 827749 (2022).
Englot, D. J., Chang, E. F. & Auguste, K. I. Vagus nerve stimulation for epilepsy: a meta-analysis of efficacy and predictors of response. J. Neurosurg. 115, 1248–1255 (2011).
Lim, M. J. R. et al. Vagus nerve stimulation for treatment of drug-resistant epilepsy: a systematic review and meta-analysis. Neurosurg. Rev. 45, 2361–2373 (2022).
Bottomley, J. M., LeReun, C., Diamantopoulos, A., Mitchell, S. & Gaynes, B. N. Vagus nerve stimulation (VNS) therapy in patients with treatment resistant depression: a systematic review and meta-analysis. Compr. Psychiatry 98, 152156 (2019).
Zhang, L. et al. Transcutaneous vagus nerve stimulation for insomnia in people living in places or cities with high altitudes: a randomized controlled trial. Brain Sci. 13, 985 (2023).
Jackowska, M., Koenig, J., Vasendova, V. & Jandackova, V. K. A two-week course of transcutaneous vagal nerve stimulation improves global sleep: findings from a randomised trial in community-dwelling adults. Auton. Neurosci. 240, 102972 (2022).
Besedovsky, L. et al. Auditory closed-loop stimulation of EEG slow oscillations strengthens sleep and signs of its immune-supportive function. Nat. Commun. 8, 1984 (2017).
Ong, J. L. et al. Effects of phase-locked acoustic stimulation during a nap on EEG spectra and declarative memory consolidation. Sleep Med. 20, 88–97 (2016).
Santostasi, G. et al. Phase-locked loop for precisely timed acoustic stimulation during sleep. J. Neurosci. Methods 259, 101–114 (2016).
Brunoni, A. R. et al. Repetitive transcranial magnetic stimulation for the acute treatment of major depressive episodes: a systematic review with network meta-analysis. JAMA Psychiatry 74, 143–152 (2017).
Jiang, B., He, D., Guo, Z., Mu, Q. & Zhang, L. Efficacy and placebo response of repetitive transcranial magnetic stimulation for primary insomnia. Sleep Med. 63, 9–13 (2019).
Duraccio, K. M., Kamhout, S., Baron, K. G., Reutrakul, S. & Depner, C. M. Sleep extension and cardiometabolic health: what it is, possible mechanisms and real-world applications. J. Physiol. https://doi.org/10.1113/JP284911 (2024).
Baron, K. G. et al. Behavioral interventions to extend sleep duration: a systematic review and meta-analysis. Sleep Med. Rev. 60, 101532 (2021).
Al Khatib, H. K. et al. Sleep extension is a feasible lifestyle intervention in free-living adults who are habitually short sleepers: a potential strategy for decreasing intake of free sugars? A randomized controlled pilot study. Am. J. Clin. Nutr. 107, 43–53 (2018).
Tasali, E., Chapotot, F., Wroblewski, K. & Schoeller, D. The effects of extended bedtimes on sleep duration and food desire in overweight young adults: a home-based intervention. Appetite 80, 220–224 (2014).
Dutil, C. et al. Sleep and insulin sensitivity in adolescents at risk of type 2 diabetes: the sleep manipulation in adolescents at risk of type 2 diabetes randomized crossover study. Sleep 47, zsad313 (2024).
Tasali, E., Wroblewski, K., Kahn, E., Kilkus, J. & Schoeller, D. A. Effect of sleep extension on objectively assessed energy intake among adults with overweight in real-life settings: a randomized clinical trial. JAMA Intern. Med. 182, 365–374 (2022).
de Zambotti, M., Cellini, N., Goldstone, A., Colrain, I. M. & Baker, F. C. Wearable sleep technology in clinical and research settings. Med. Sci. Sports Exerc. 51, 1538–1557 (2019).
Tasali, E., Leproult, R., Ehrmann, D. A. & Van Cauter, E. Slow-wave sleep and the risk of type 2 diabetes in humans. Proc. Natl Acad. Sci. USA 105, 1044–1049 (2008).
Yeghiazarians, Y. et al. Obstructive sleep apnea and cardiovascular disease: a scientific statement from the American Heart Association. Circulation 144, e56–e67 (2021).
Bailly, S., Mendelson, M., Baillieul, S., Tamisier, R. & Pepin, J. L. The future of telemedicine for obstructive sleep apnea treatment: a narrative review. J. Clin. Med. 13, 2700 (2024).
Pizinger, T. M., Aggarwal, B. & St-Onge, M. P. Sleep extension in short sleepers: an evaluation of feasibility and effectiveness for weight management and cardiometabolic disease prevention. Front. Endocrinol. 9, 392 (2018).
Broussard, J. L. et al. Elevated ghrelin predicts food intake during experimental sleep restriction. Obesity 24, 132–138 (2016).
Azarbarzin, A., Labarca, G., Kwon, Y. & Wellman, A. Physiological consequences of upper airway obstruction in sleep apnea. Chest https://doi.org/10.1016/j.chest.2024.05.028 (2024).
Azarbarzin, A. et al. The hypoxic burden of sleep apnoea predicts cardiovascular disease-related mortality: the osteoporotic fractures in men study and the sleep heart health study. Eur. Heart J. 40, 1149–1157 (2019).
Solelhac, G. et al. Pulse wave amplitude drops index: a biomarker of cardiovascular risk in obstructive sleep apnea. Am. J. Respir. Crit. Care Med. 207, 1620–1632 (2023).
Redline, S., Azarbarzin, A. & Peker, Y. Obstructive sleep apnoea heterogeneity and cardiovascular disease. Nat. Rev. Cardiol. 20, 560–573 (2023).
Pfammatter, A. F. et al. The development of a novel mhealth tool for obstructive sleep apnea: tracking continuous positive airway pressure adherence as a percentage of time in bed. J. Med. Internet Res. 24, e39489 (2022).
Caceres, B. A. et al. Assessing and addressing cardiovascular health in LGBTQ adults: a scientific statement from the American Heart Association. Circulation 142, e321–e332 (2020).
Acknowledgements
The workshop that set the foundation for this article was supported by the NHLBI and the NIH. The authors thank M. Tirumalasetty (New York University Grossman Long Island School of Medicine, USA) for his assistance with Fig. 4 for initial submission, and D. Goff (NHLBI, USA) for his critical review and suggestions on the manuscript. The authors acknowledge the following funding support: B.A.: NIH HL169991; A. Azarbarzin: NIH HL153874, HL161766 and American Academy of Sleep Medicine SR-2217; R.C.A.: NIH AG068577; K.G.B.: NIH NR018891; V.L.B.: NIH HL139950; N.B.: NIH HL151745 and National Institute for Occupational Safety and Health U19OH010154; J.L.B.: NIH HL168081 and DK125653; P.C.: NIH HL159180, HL160870 HL, HL163783 and American Academy of Sleep Medicine 245-SR-21; S.H.C.: NIH HL169503, HL173882, OD037655 and MH134051; R.C.: NIH HL169266; S.J.: NIH HL106041 and HL137234; J.O.L.: NIH HL151368 and NS126547; P.L.L.: NIH HL159246; Q.M.: NIH HL141733 and HL137234; A.A.P.: HL142051; F.K.S.: NIH HL131478; E.T.: NIH DK136214, DK120312, HL146127 and HL146127; I.V.: NIH HL141581; and M.A.G.: NIH MD011600 and DA051321.
Author information
Authors and Affiliations
Contributions
B.A. and Y.G. researched data for the article. B.A., Y.G., A. Azarbarzin, R.C.A., K.G.B., V.L.B., N.B., J.L.B., P.C., S.H.C., R.C., F.-X.F., D.A.J., S.J., J.O.L., P.L.L., Q.M., J.M.O., A.A.P., F.K.S., E.T., I.V., M.A.G. and D.L.-J. wrote the article. All the authors contributed substantially to the discussion of content, and reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The views expressed in this article are those of the authors and reflect those of the workshop, and do not necessarily represent those of the NHLBI, the NIH, or the US Department of Health and Human Services. No industry support was used for this project. A. Azarbarzin is a consultant for Apnimed, Cerebra, Eli Lilly, Inspire and Respicardia. D.A.J. has received consulting fees from Idorsia Pharmaceuticals. J.O.L. has received consulting fees from Synchronicity Pharma. E.T. is an adviser to NeuroGeneCES and Vitaliti. The other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Cardiology thanks Linda Van Laake, who co-reviewed with Yvonne Koop; and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Aggarwal, B., Gao, Y., Alfini, A. et al. Sleep and circadian rhythms in cardiovascular resilience: mechanisms, implications, and a Roadmap for research and interventions. Nat Rev Cardiol 23, 116–130 (2026). https://doi.org/10.1038/s41569-025-01188-1
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41569-025-01188-1