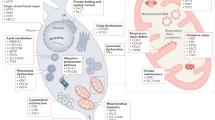
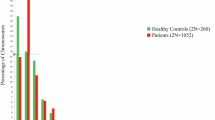
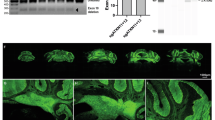
Abstract
Sporadic late-onset cerebellar ataxia (SLOCA) is a syndrome defined by subacute or chronic and progressive ataxia occurring after the age of 40 years in individuals without a family history of ataxia. The 2022 publication of revised consensus diagnostic criteria for multiple system atrophy and the emergence of promising biomarkers provides a thorough diagnostic framework that now enables the diagnosis of numerous acquired causes of SLOCA, including autoimmune disorders and neurodegenerative diseases. The ongoing development and increased availability of DNA sequencing technology have uncovered several molecular causes of SLOCA besides spastic paraplegia type 7 and very late-onset Friedreich ataxia. These additional causes include sporadic genetic disorders, such as spinocerebellar atrophy type 27B, caused by GAA expansion in the FGF14 gene, and cerebellar ataxia with neuropathy and vestibular areflexia syndrome (CANVAS), caused by biallelic expansions in the RFC1 gene. This Review presents an updated clinical approach to the diagnosis and management of SLOCA that focuses on the most important developments in this field. Future challenges are also discussed, including the identification of additional missing genetic causes of SLOCA, especially via the use of long-read genome sequencing, improvements in SLOCA prognostication and the implementation of clinical trials of neuroprotective interventions.
Key points
-
Sporadic late-onset cerebellar ataxia (SLOCA) is a syndrome defined by subacute or chronic, progressive ataxia occurring after the age of 40 years in individuals with no family history of ataxia.
-
The most frequent cause of SLOCA is multiple system atrophy, a rapidly progressive neurodegenerative condition that combines cerebellar ataxia, dysautonomia (typically genitourinary impairment or orthostatic hypotension) and parkinsonism.
-
Autoimmune cerebellar ataxias are rare causes of non-genetic, usually rapidly progressive SLOCA that are frequently responsive to immunosuppressive treatment, which can improve the disease course.
-
The ongoing development of DNA sequencing technologies has revealed that a substantial proportion of SLOCA can be explained by specific pathogenic genetic variants.
-
The most frequent genetic causes of SLOCA are heterozygous GAA expansions in FGF14 (spinocerebellar atrophy type 27B) and biallelic expansions in RFC1 (cerebellar ataxia with neuropathy and vestibular areflexia (CANVAS)).
-
Long-read genome sequencing could facilitate the discovery of additional SLOCA-causing gene variants, and validation of surrogate biomarkers would promote the identification of disease-modifying drugs.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Klockgether, T. Sporadic ataxia with adult onset: classification and diagnostic criteria. Lancet Neurol. 9, 94–104 (2010).
Teive, H. A. G. & Ashizawa, T. Primary and secondary ataxias. Curr. Opin. Neurol. 28, 413–422 (2015).
Coarelli, G. et al. The inherited cerebellar ataxias: an update. J. Neurol. 270, 208–222 (2023).
Divya, K. P. & Kishore, A. Treatable cerebellar ataxias. Clin. Park. Relat. Disord. 3, 100053 (2020).
Muñiz-Castrillo, S. et al. Novelties in autoimmune and paraneoplastic cerebellar ataxias: twenty years of progresses. Cerebellum 21, 573–591 (2022).
Mitoma, H., Hadjivassiliou, M. & Honnorat, J. Guidelines for treatment of immune-mediated cerebellar ataxias. Cerebellum Ataxias 2, 14 (2015).
Joubert, B. & Honnorat, J. Nonparaneoplastic autoimmune cerebellar ataxias. Curr. Opin. Neurol. 32, 484–492 (2019).
Hadjivassiliou, M. et al. Diagnostic criteria for primary autoimmune cerebellar ataxia — guidelines from an international task force on immune-mediated cerebellar ataxias. Cerebellum 19, 605–610 (2020).
Narayan, R. N., McKeon, A. & Fife, T. D. Autoimmune vestibulocerebellar syndromes. Semin. Neurol. 40, 97–115 (2020).
Simard, C. et al. Clinical spectrum and diagnostic pitfalls of neurologic syndromes with Ri antibodies. Neurol. Neuroimmunol. Neuroinflamm. 7, e699 (2020).
Irani, S. R. et al. Antibodies to Kv1 potassium channel-complex proteins leucine-rich, glioma inactivated 1 protein and contactin-associated protein-2 in limbic encephalitis, Morvan’s syndrome and acquired neuromyotonia. Brain 133, 2734–2748 (2010).
Lancaster, E. et al. Investigations of CASPR2, an autoantigen of encephalitis and neuromyotonia. Ann. Neurol. 69, 303–311 (2011).
Joubert, B. et al. Autoimmune episodic ataxia in patients with anti-CASPR2 antibody-associated encephalitis. Neurol. Neuroimmunol. Neuroinflamm. 4, e371 (2017).
Becker, E. B. et al. Contactin-associated protein-2 antibodies in non-paraneoplastic cerebellar ataxia. J. Neurol. Neurosurg. Psychiatry 83, 437–440 (2012).
Benoit, J. et al. Early-stage contactin-associated protein-like 2 limbic encephalitis: clues for diagnosis. Neurol. Neuroimmunol. Neuroinflamm. 10, e200041 (2023).
Mandel-Brehm, C. et al. Kelch-like protein 11 antibodies in seminoma-associated paraneoplastic encephalitis. N. Engl. J. Med. 381, 47–54 (2019).
Hadjivassiliou, M., Sanders, D. S., Woodroofe, N., Williamson, C. & Grünewald, R. A. Gluten ataxia. Cerebellum 7, 494–498 (2008).
Gaig, C. et al. Clinical manifestations of the anti-IgLON5 disease. Neurology 88, 1736–1743 (2017).
Honorat, J. A. et al. IgLON5 antibody: neurological accompaniments and outcomes in 20 patients. Neurol. Neuroimmunol. Neuroinflamm. 4, e385 (2017).
Gaig, C. et al. Frequency and characterization of movement disorders in anti-IgLON5 disease. Neurology 97, e1367–e1381 (2021).
Mitoma, H., Manto, M. & Hadjivassiliou, M. Immune-mediated cerebellar ataxias: clinical diagnosis and treatment based on immunological and physiological mechanisms. J. Mov. Disord. 14, 10–28 (2021).
Chan, J. L., Murphy, K. A. & Sarna, J. R. Myoclonus and cerebellar ataxia associated with COVID-19: a case report and systematic review. J. Neurol. 268, 3517–3548 (2021).
Thompson, A. J. et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol. 17, 162–173 (2018).
Montalban, X. et al. Ocrelizumab versus placebo in primary progressive multiple sclerosis. N. Engl. J. Med. 376, 209–220 (2017).
Pittock, S. J. et al. Chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids (CLIPPERS). Brain 133, 2626–2634 (2010).
Tobin, W. O. et al. Diagnostic criteria for chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids (CLIPPERS). Brain 140, 2415–2425 (2017).
Mitoma, H. & Manto, M. Recent advances in diagnosis of immune-mediated cerebellar ataxias: novel concepts and fundamental questions on autoimmune mechanisms. J. Neurol. 271, 7046–7053 (2024).
Krismer, F., Fanciulli, A., Meissner, W. G., Coon, E. A. & Wenning, G. K. Multiple system atrophy: advances in pathophysiology, diagnosis, and treatment. Lancet Neurol. 23, 1252–1266 (2024).
Bogdan, T. et al. Unravelling the etiology of sporadic late-onset cerebellar ataxia in a cohort of 205 patients: a prospective study. J. Neurol. 269, 6354–6365 (2022).
Oender, D. et al. Evolution of clinical outcome measures and biomarkers in sporadic adult-onset degenerative ataxia. Mov. Disord. 38, 654–664 (2023).
Wenning, G. K. et al. The Movement Disorder Society criteria for the diagnosis of multiple system atrophy. Mov. Disord. 37, 1131–1148 (2022).
Virameteekul, S., Revesz, T., Jaunmuktane, Z., Warner, T. T. & De Pablo-Fernández, E. Pathological validation of the MDS criteria for the diagnosis of multiple system atrophy. Mov. Disord. 38, 444–452 (2023).
Sekiya, H. et al. Validation study of the MDS criteria for the diagnosis of multiple system atrophy in the Mayo Clinic Brain Bank. Neurology 101, e2460–e2471 (2023).
Teive, H. A. G., Arruda, W. O., Moro, A., Moscovich, M. & Munhoz, R. P. Differential diagnosis of sporadic adult-onset ataxia: the role of REM sleep behavior disorder. Parkinsonism Relat. Disord. 21, 640–643 (2015).
Kadodwala, V. H., Hadjivassiliou, M., Currie, S., Skipper, N. & Hoggard, N. Is 1H-MR spectroscopy useful as a diagnostic aid in MSA-C? Cerebellum Ataxias 6, 7 (2019).
Krismer, F. et al. Progressive brain atrophy in multiple system atrophy: a longitudinal, multicenter, magnetic resonance imaging study. Mov. Disord. 39, 119–129 (2024).
Spillantini, M. G. et al. Filamentous α-synuclein inclusions link multiple system atrophy with Parkinson’s disease and dementia with Lewy bodies. Neurosci. Lett. 251, 205–208 (1998).
Kim, H. Y. et al. A novel brain PET radiotracer for imaging α-synuclein fibrils in multiple system atrophy. J. Med. Chem. 66, 12185–12202 (2023).
Smith, R. et al. The α-synuclein PET tracer [18F] ACI-12589 distinguishes multiple system atrophy from other neurodegenerative diseases. Nat. Commun. 14, 6750 (2023).
Wirth, T. et al. Progression of nigrostriatal denervation in cerebellar multiple system atrophy: a prospective study. Neurology 98, 232–236 (2021).
Cong, S., Xiang, C., Wang, H. & Cong, S. Diagnostic utility of fluid biomarkers in multiple system atrophy: a systematic review and meta-analysis. J. Neurol. 268, 2703–2712 (2021).
Shen, X.-N. et al. Systematic assessment of plasma biomarkers in spinocerebellar ataxia. Neurobiol. Dis. 181, 106112 (2023).
Okuzumi, A. et al. Propagative α-synuclein seeds as serum biomarkers for synucleinopathies. Nat. Med. 29, 1448–1455 (2023).
Poggiolini, I. et al. Diagnostic value of cerebrospinal fluid α-synuclein seed quantification in synucleinopathies. Brain 145, 584–595 (2022).
Rossi, M. et al. Ultrasensitive RT-QuIC assay with high sensitivity and specificity for Lewy body-associated synucleinopathies. Acta Neuropathol. 140, 49–62 (2020).
Shahnawaz, M. et al. Discriminating α-synuclein strains in Parkinson’s disease and multiple system atrophy. Nature 578, 273–277 (2020).
Ma, Y. et al. Sensitivity and specificity of a seed amplification assay for diagnosis of multiple system atrophy: a multicentre cohort study. Lancet Neurol. 23, 1225–1237 (2024).
Donadio, V. et al. Phosphorylated α-synuclein in skin schwann cells: a new biomarker for multiple system atrophy. Brain 146, 1065–1074 (2023).
Gibbons, C. H. et al. Skin biopsy detection of phosphorylated α-synuclein in patients with synucleinopathies. JAMA 331, 1298–1306 (2024).
Höglinger, G. U. et al. Clinical diagnosis of progressive supranuclear palsy: the movement disorder society criteria. Mov. Disord. 32, 853–864 (2017).
Gao, A. F. et al. Progressive ataxia and palatal tremor: two autopsy cases of a novel tauopathy. Mov. Disord. 32, 1465–1473 (2017).
Foutz, A. et al. Diagnostic and prognostic value of human prion detection in cerebrospinal fluid. Ann. Neurol. 81, 79–92 (2017).
Hermann, P. et al. Biomarkers and diagnostic guidelines for sporadic Creutzfeldt–Jakob disease. Lancet Neurol. 20, 235–246 (2021).
Zerr, I. et al. Updated clinical diagnostic criteria for sporadic Creutzfeldt–Jakob disease. Brain 132, 2659–2668 (2009).
Pedroso, J. L. et al. Cerebellar degeneration and progressive ataxia associated with HIV-virus infection. Parkinsonism Relat. Disord. 54, 95–98 (2018).
Koralnik, I. J. et al. JC virus granule cell neuronopathy: a novel clinical syndrome distinct from progressive multifocal leukoencephalopathy. Ann. Neurol. 57, 576–580 (2005).
Bernard-Valnet, R., Koralnik, I. J. & Du Pasquier, R. Advances in treatment of progressive multifocal leukoencephalopathy. Ann. Neurol. 90, 865–873 (2021).
Klockgether, T., Döller, G., Wüllner, U., Petersen, D. & Dichgans, J. Cerebellar encephalitis in adults. J. Neurol. 240, 17–20 (1993).
Mitoma, H., Manto, M. & Shaikh, A. G. Mechanisms of ethanol-induced cerebellar ataxia: underpinnings of neuronal death in the cerebellum. Int. J. Environ. Res. Publ. Health 18, 8678 (2021).
van Gaalen, J., Kerstens, F. G., Maas, R. P. P. W. M., Härmark, L. & van de Warrenburg, B. P. C. Drug-induced cerebellar ataxia: a systematic review. CNS Drugs 28, 1139–1153 (2014).
Natarajan, U., Onyechi, A. & Ohemeng-Dapaah, J. 5-Fluorouracil neurotoxicity in a patient with normal dihydropyrimidine dehydrogenase activity. Cureus 15, e49898 (2023).
Dinoto, A., Mantovani, E., Ferrari, S., Mariotto, S. & Tamburin, S. Cerebellar involvement associated with immune checkpoint inhibitors: a systematic review. Eur. J. Neurol. 30, 774–781 (2023).
Kumar, N. Superficial siderosis: a clinical review. Ann. Neurol. 89, 1068–1079 (2021).
Flores Martin, A., Shanmugarajah, P., Hoggard, N. & Hadjivassiliou, M. Treatment response of deferiprone in infratentorial superficial siderosis: a systematic review. Cerebellum 20, 454–461 (2021).
McClugage, S. G. & Oakes, W. J. The Chiari I malformation. J. Neurosurg. Pediatr. 24, 217–226 (2019).
Barrie, U. et al. Basilar impression: a systematic review and meta-analysis of clinical features, operative strategies, and outcomes. World Neurosurg. 189, 323.e25–338.e25 (2024).
Bal, B. S., Finelli, F. C., Shope, T. R. & Koch, T. R. Nutritional deficiencies after bariatric surgery. Nat. Rev. Endocrinol. 8, 544–556 (2012).
Green, R. et al. Vitamin B12 deficiency. Nat. Rev. Dis. Prim. 3, 17040 (2017).
Mariotti, C. et al. Ataxia with isolated vitamin E deficiency: neurological phenotype, clinical follow-up and novel mutations in TTPA gene in Italian families. Neurol. Sci. 25, 130–137 (2004).
Guerreiro Stucklin, A. S. & Grotzer, M. A. Cerebellar tumors. Handb. Clin. Neurol. 155, 289–299 (2018).
McClain, K. L. et al. Histiocytic disorders. Nat. Rev. Dis. Prim. 7, 73 (2021).
Salardaine, Q., Desjardins, C., Baille, G., Roze, E. & Nardin, C. Is ethyl chloride the new nitrous oxide? A case report. BMC Neurol. 24, 186 (2024).
Manto, M. Toxic agents causing cerebellar ataxias. Handb. Clin. Neurol. 103, 201–213 (2012).
Olgiati, S., Quadri, M. & Bonifati, V. Genetics of movement disorders in the next-generation sequencing era. Mov. Disord. 31, 458–470 (2016).
Cortese, A. et al. Biallelic expansion of an intronic repeat in RFC1 is a common cause of late-onset ataxia. Nat. Genet. 51, 649–658 (2019).
Traschütz, A. et al. Natural history, phenotypic spectrum, and discriminative features of multisystemic RFC1 disease. Neurology 96, e1369–e1382 (2021).
Hadjivassiliou, M. et al. Can CANVAS due to RFC1 biallelic expansions present with pure ataxia? J. Neurol. Neurosurg. Psychiatry 95, 171–174 (2024).
Sullivan, R. et al. RFC1-related ataxia is a mimic of early multiple system atrophy. J. Neurol. Neurosurg. Psychiatry 92, 444–446 (2021).
Pellerin, D. et al. Deep intronic FGF14 GAA repeat expansion in late-onset cerebellar ataxia. N. Engl. J. Med. 388, 128–141 (2023).
Rafehi, H. et al. An intronic GAA repeat expansion in FGF14 causes the autosomal-dominant adult-onset ataxia SCA50/ATX-FGF14. Am. J. Hum. Genet. 110, 105–119 (2023).
Wirth, T. et al. Natural history and phenotypic spectrum of GAA-FGF14 sporadic late-onset cerebellar ataxia (SCA27B). Mov. Disord. 38, 1950–1956 (2023).
Méreaux, J.-L. et al. Clinical and genetic keys to cerebellar ataxia due to FGF14 GAA expansions. eBioMedicine 99, 104931 (2023).
Mohren, L. et al. Identification and characterisation of pathogenic and non-pathogenic FGF14 repeat expansions. Nat. Commun. 15, 7665 (2024).
Pellerin, D. et al. Somatic instability of the FGF14-SCA27B GAA•TTC repeat reveals a marked expansion bias in the cerebellum. Brain 148, 1258–1270 (2024).
Pfeffer, G. et al. SPG7 mutations are a common cause of undiagnosed ataxia. Neurology 84, 1174–1176 (2015).
Iruzubieta, P. et al. Frequency and phenotypic spectrum of spinocerebellar ataxia 27B and other genetic ataxias in a Spanish cohort of late-onset cerebellar ataxia. Eur. J. Neurol. 30, 3828–3833 (2023).
Beijer, D. et al. The genetic landscape of sporadic adult-onset degenerative ataxia: a multi-modal genetic study of 377 consecutive patients from the longitudinal multi-centre SPORTAX cohort. EBioMedicine 115, 105715 (2025).
Pfeffer, G. et al. Mutations in the SPG7 gene cause chronic progressive external ophthalmoplegia through disordered mitochondrial DNA maintenance. Brain 137, 1323–1336 (2014).
Hewamadduma, C. A. et al. Novel genotype–phenotype and MRI correlations in a large cohort of patients with SPG7 mutations. Neurol. Genet. 4, e279 (2018).
Lecocq, C. et al. Delayed-onset Friedreich’s ataxia revisited. Mov. Disord. 31, 62–69 (2016).
Lynch, D. R. et al. Safety and efficacy of omaveloxolone in Friedreich ataxia (MOXIe Study). Ann. Neurol. 89, 212–225 (2021).
Lynch, D. R. et al. Efficacy of omaveloxolone in Friedreich’s ataxia: delayed-start analysis of the MOXIe extension. Mov. Disord. 38, 313–320 (2023).
Apartis, E. et al. FXTAS: new insights and the need for revised diagnostic criteria. Neurology 79, 1898–1907 (2012).
Renaud, M. et al. Relevance of corpus callosum splenium versus middle cerebellar peduncle hyperintensity for FXTAS diagnosis in clinical practice. J. Neurol. 262, 435–442 (2015).
Durr, A. Autosomal dominant cerebellar ataxias: polyglutamine expansions and beyond. Lancet Neurol. 9, 885–894 (2010).
Coarelli, G., Coutelier, M. & Durr, A. Autosomal dominant cerebellar ataxias: new genes and progress towards treatments. Lancet Neurol. 22, 735–749 (2023).
Anheim, M., Tranchant, C. & Koenig, M. The autosomal recessive cerebellar ataxias. N. Engl. J. Med. 366, 636–646 (2012).
Gorman, G. S. et al. Mitochondrial diseases. Nat. Rev. Dis. Prim. 2, 16080 (2016).
Arnett, A. B., Wang, T., Eichler, E. E. & Bernier, R. A. Reflections on the genetics-first approach to advancements in molecular genetic and neurobiological research on neurodevelopmental disorders. J. Neurodev. Disord. 13, 24 (2021).
Jobanputra, V. et al. Advancing access to genome sequencing for rare genetic disorders: recent progress and call to action. NPJ Genom. Med. 9, 23 (2024).
Delude, C. M. Deep phenotyping: the details of disease. Nature 527, S14–S15 (2015).
Gebus, O. et al. Deciphering the causes of sporadic late-onset cerebellar ataxias: a prospective study with implications for diagnostic work. J. Neurol. 264, 1118–1126 (2017).
Carré, G. et al. Brain MRI of multiple system atrophy of cerebellar type: a prospective study with implications for diagnosis criteria. J. Neurol. 267, 1269–1277 (2020).
Federoff, M. et al. Genome-wide estimate of the heritability of multiple system atrophy. Parkinsonism Relat. Disord. 22, 35–41 (2016).
Wirth, T. et al. Does spinocerebellar ataxia 27B mimic cerebellar multiple system atrophy? J. Neurol. 271, 2078–2085 (2024).
Ibañez, K. et al. Whole genome sequencing for the diagnosis of neurological repeat expansion disorders in the UK: a retrospective diagnostic accuracy and prospective clinical validation study. Lancet Neurol. 21, 234–245 (2022).
Sun, Y. et al. Next-generation diagnostics: gene panel, exome, or whole genome? Hum. Mutat. 36, 648–655 (2015).
Dolzhenko, E. et al. ExpansionHunter: a sequence-graph-based tool to analyze variation in short tandem repeat regions. Bioinformatics 35, 4754–4756 (2019).
Gardner, E. J. et al. The Mobile Element Locator Tool (MELT): population-scale mobile element discovery and biology. Genome Res. 27, 1916–1929 (2017).
Logsdon, G. A., Vollger, M. R. & Eichler, E. E. Long-read human genome sequencing and its applications. Nat. Rev. Genet. 21, 597–614 (2020).
Maestri, S. et al. A long-read sequencing approach for direct haplotype phasing in clinical settings. Int. J. Mol. Sci. 21, 9177 (2020).
Jiang, T., Liu, S., Cao, S. & Wang, Y. Structural variant detection from long-read sequencing data with cuteSV. Methods Mol. Biol. 2493, 137–151 (2022).
Sakamoto, Y. et al. Long-read whole-genome methylation patterning using enzymatic base conversion and nanopore sequencing. Nucleic Acids Res. 49, e81 (2021).
Karczewski, K. J. et al. The mutational constraint spectrum quantified from variation in 141,456 humans. Nature 581, 434–443 (2020).
Lee, S., Abecasis, G. R., Boehnke, M. & Lin, X. Rare-variant association analysis: study designs and statistical tests. Am. J. Hum. Genet. 95, 5–23 (2014).
Walsh, N., Cooper, A., Dockery, A. & O’Byrne, J. J. Variant reclassification and clinical implications. J. Med. Genet. 61, 207–211 (2024).
Lunke, S. et al. Integrated multi-omics for rapid rare disease diagnosis on a national scale. Nat. Med. 29, 1681–1691 (2023).
Lee, S. et al. Comparison of methylation episignatures in KMT2B- and KMT2D-related human disorders. Epigenomics 14, 537–547 (2022).
Yépez, V. A. et al. Clinical implementation of RNA sequencing for Mendelian disease diagnostics. Genome Med. 14, 38 (2022).
Koga, S. et al. Cerebellar ataxia in progressive supranuclear palsy: an autopsy study of PSP-C. Mov. Disord. 31, 653–662 (2016).
Basile, A. O. & Ritchie, M. D. Informatics and machine learning to define the phenotype. Expert Rev. Mol. Diagn. 18, 219–226 (2018).
Faber, J. et al. Prominent white matter involvement in multiple system atrophy of cerebellar type. Mov. Disord. 35, 816–824 (2020).
Tezenas du Montcel, S. et al. Factors influencing disease progression in autosomal dominant cerebellar ataxia and spastic paraplegia. Arch. Neurol. 69, 500–508 (2012).
Currò, R. et al. Role of the repeat expansion size in predicting age of onset and severity in RFC1 disease. Brain 147, 1887–1898 (2024).
Chelban, V. et al. Neurofilament light levels predict clinical progression and death in multiple system atrophy. Brain 145, 4398–4408 (2022).
Tan, A. H. et al. Altered gut microbiome and metabolome in patients with multiple system atrophy. Mov. Disord. 33, 174–146 (2018).
Öz, G. et al. MR imaging in ataxias: consensus recommendations by the Ataxia Global Initiative Working Group on MRI biomarkers. Cerebellum 23, 931–945 (2023).
Stephen, C. D., Vangel, M., Gupta, A. S., MacMore, J. P. & Schmahmann, J. D. Rates of change of pons and middle cerebellar peduncle diameters are diagnostic of multiple system atrophy of the cerebellar type. Brain Commun. 6, fcae019 (2024).
Wegner, P. et al. Sensor-free motion registration and automated movement evaluation: leveraging machine learning for clinical gait analysis in ataxia disorders. Preprint at medRxiv https://doi.org/10.1101/2024.05.29.24308057 (2024).
Hohenfeld, C. et al. Application of quantitative motor assessments in Friedreich ataxia and evaluation of their relation to clinical measures. Cerebellum 18, 896–909 (2019).
Grobe-Einsler, M. et al. SARAspeech — feasibility of automated assessment of ataxic speech disturbance. NPJ Digit. Med. 6, 43 (2023).
Devaux, A., Helmer, C., Genuer, R. & Proust-Lima, C. Random survival forests with multivariate longitudinal endogenous covariates. Stat. Methods Med. Res. 32, 2331–2346 (2023).
Ilg, W. et al. Intensive coordinative training improves motor performance in degenerative cerebellar disease. Neurology 73, 1823–1830 (2009).
Miyai, I. et al. Cerebellar ataxia rehabilitation trial in degenerative cerebellar diseases. Neurorehabil. Neural Repair. 26, 515–522 (2012).
Marquer, A., Barbieri, G. & Pérennou, D. The assessment and treatment of postural disorders in cerebellar ataxia: a systematic review. Ann. Phys. Rehabil. Med. 57, 67–78 (2014).
He, M., Zhang, H.-N., Tang, Z.-C. & Gao, S.-G. Balance and coordination training for patients with genetic degenerative ataxia: a systematic review. J. Neurol. 268, 3690–3705 (2021).
Vogel, A. P., Folker, J. & Poole, M. L. Treatment for speech disorder in Friedreich ataxia and other hereditary ataxia syndromes. Cochrane Database Syst. Rev. 2014, CD008953 (2014).
Egger, K. et al. Voxel based morphometry reveals specific gray matter changes in primary dystonia. Mov. Disord. 22, 1538–1542 (2007).
Vogel, A. P. et al. Home-based biofeedback speech treatment improves dysarthria in repeat-expansion SCAs. Ann. Clin. Transl. Neurol. 9, 1310–1315 (2022).
Assadi, M. et al. Treatment of spinocerebellar ataxia with buspirone. J. Neurol. Sci. 260, 143–146 (2007).
Feil, K. et al. Safety and efficacy of acetyl-DL-leucine in certain types of cerebellar ataxia: the ALCAT randomized clinical crossover trial. JAMA Netw. Open 4, e2135841 (2021).
Claassen, J. et al. A randomised double-blind, cross-over trial of 4-aminopyridine for downbeat nystagmus — effects on slowphase eye velocity, postural stability, locomotion and symptoms. J. Neurol. Neurosurg. Psychiatry 84, 1392–1399 (2013).
Wilke, C. et al. GAA-FGF14 ataxia (SCA27B): phenotypic profile, natural history progression and 4-aminopyridine treatment response. Brain 146, 4144–4157 (2023).
Ristori, G. et al. Riluzole in cerebellar ataxia: a randomized, double-blind, placebo-controlled pilot trial. Neurology 74, 839–845 (2010).
Romano, S. et al. Riluzole in patients with hereditary cerebellar ataxia: a randomised, double-blind, placebo-controlled trial. Lancet Neurol. 14, 985–991 (2015).
Coarelli, G. et al. Safety and efficacy of riluzole in spinocerebellar ataxia type 2 in France (ATRIL): a multicentre, randomised, double-blind, placebo-controlled trial. Lancet Neurol. 21, 225–233 (2022).
Nishizawa, M. et al. Effect of rovatirelin in patients with cerebellar ataxia: two randomised double-blind placebo-controlled phase 3 trials. J. Neurol. Neurosurg. Psychiatry 91, 254–262 (2020).
Qiu, M., Wang, R., Shen, Y., Hu, Z. & Zhang, Y. Efficacy and safety of repetitive transcranial magnetic stimulation in spinocerebellar ataxia type 3: a systematic review and meta-analysis of randomized controlled trials. Cerebellum 23, 1604–1613 (2024).
Benussi, A., Pascual-Leone, A. & Borroni, B. Non-invasive cerebellar stimulation in neurodegenerative ataxia: a literature review. Int. J. Mol. Sci. 21, 1948 (2020).
Pilloni, G. et al. Tolerability and feasibility of at-home remotely supervised transcranial direct current stimulation (RS-tDCS): single-center evidence from 6,779 sessions. Brain Stimul. 15, 707–716 (2022).
Pandey, S. et al. A retrospective study on outcomes following posterior fossa decompression with dural splitting surgery in patients with Chiari type I malformation. Clin. Neurol. Neurosurg. 196, 106035 (2020).
Peter, E. et al. Cerebellar ataxia with anti-DNER antibodies: outcomes and immunologic features. Neurol. Neuroimmunol. Neuroinflamm. 9, e200018 (2022).
Spatola, M. et al. Clinical features, prognostic factors, and antibody effects in anti-mGluR1 encephalitis. Neurology 95, e3012–e3025 (2020).
Shambrook, P. et al. Delayed benefit from aggressive immunotherapy in waxing and waning anti-IgLON5 disease. Neurol. Neuroimmunol. Neuroinflamm. 8, e1009 (2021).
Kaufmann, H. Droxidopa for symptomatic neurogenic orthostatic hypotension: what can we learn? Clin. Auton. Res. 27 (Suppl. 1), 1–3 (2017).
Squair, J. W. et al. Implanted system for orthostatic hypotension in multiple-system atrophy. N. Engl. J. Med. 386, 1339–1344 (2022).
Bendetowicz, D. et al. Recent advances in clinical trials in multiple system atrophy. Curr. Neurol. Neurosci. Rep. 24, 95–112 (2024).
Mitsui, J. et al. High-dose ubiquinol supplementation in multiple-system atrophy: a multicentre, randomised, double-blinded, placebo-controlled phase 2 trial. EClinicalMedicine 59, 101920 (2023).
Brunet de Courssou, J.-B., Durr, A., Adams, D., Corvol, J.-C. & Mariani, L.-L. Antisense therapies in neurological diseases. Brain 145, 816–831 (2022).
Mitoma, H., Manto, M. & Gandini, J. Recent advances in the treatment of cerebellar disorders. Brain Sci. 10, 11 (2019).
Vázquez-Mojena, Y., León-Arcia, K., González-Zaldivar, Y., Rodríguez-Labrada, R. & Velázquez-Pérez, L. Gene therapy for polyglutamine spinocerebellar ataxias: advances, challenges, and perspectives. Mov. Disord. 36, 2731–2744 (2021).
Acknowledgements
The authors thank Thibault Willaume from the Radiology Department of Strasbourg University Hospital for providing the imaging scan shown in Fig. 3o. J.H. declares that he is supported by a public grant overseen by the Agence Nationale de la Recherche (French Research Agency) as part of the Investissements d’Avenir programme (ANR-18-RHUS-0012) and by the European Reference Network Rare Immunodeficiency, Autoinflammatory and Autoimmune diseases network (RITA).
Author information
Authors and Affiliations
Contributions
The authors contributed to all aspects of the article. Specifically, T.W. and M.A. jointly conceptualized the manuscript, which was coordinated by M.A. T.W. wrote the Abstract, the Introduction, the sections ‘Molecular causes of SLOCA’ and ‘Hidden genetic causes of idiopathic SLOCA’ (with C.D. and P.G.) and the Conclusion. T.W. prepared Figs. 1–4, Table 1 (with C.T.), Table 3, and Supplementary Fig. 1 (with T.K.). J.F. wrote the section ‘Biomarkers of disease trajectory’. J.H. wrote the section ‘Autoimmune and inflammatory causes’ and prepared Table 2. W.G.M. wrote the section ‘Multiple system atrophy’. C.T. wrote the section ‘Acquired causes of SLOCA’. T.K. wote the section ‘Towards disease-modifying treatments’. M.A. and E.R. synthesized all co-author drafts into the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
T.W. declares that he has received honoraria from Abbvie, Edimark and Ipsen; research grants from APTES, Fondation Planiol, the France Parkinson organizations and Revue Neurologique; prize money from the Société Française de Neurologie; and travel funding from Homeperf, Lübeck, LVL Medical and the Movement Disorder Society. C.T. declares that she has received honoraria from Abbvie, Biogen, Ipsen and Lynde. M.A. declares that he has received honoraria from Abbvie, Aguettant, Asdia Biogen, Ever Pharma, Ipsen, Merz, Orkyn, Reata Pharmaceuticals and Teva. The other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Neurology thanks Hélio Teive, Ludger Schöls and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wirth, T., Faber, J., Depienne, C. et al. Progress and challenges in sporadic late-onset cerebellar ataxias. Nat Rev Neurol 21, 687–705 (2025). https://doi.org/10.1038/s41582-025-01136-0
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41582-025-01136-0