Abstract
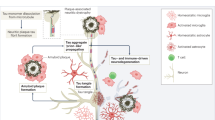
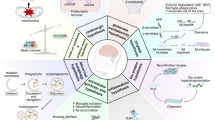
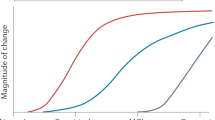
Prevention of Alzheimer disease (AD) is a medical challenge owing to its complex pathogenesis, which involves amyloid-β (Aβ) and tau aggregation, neuroinflammation and progressive neurodegeneration. Development of disease-specific biomarkers has transformed our ability to detect AD pathology early, enabling more accurate diagnosis, monitoring and the development of targeted disease-modifying therapies. Consequently, primary and secondary prevention of AD have become feasible goals. In this Perspective, we examine current and emerging pharmacological strategies for the prevention of AD, particularly the use of existing anti-Aβ therapies and emerging anti-tau approaches, among people at risk of AD or in the earliest, presymptomatic stages of the disease. We highlight the key challenges in implementing prevention trials, discuss ongoing prevention trials and their implications, and consider the potential and challenges of translation into clinical practice. Implementation of preventative strategies, supported by biomarker-guided patient selection and innovative trial designs, has the potential to substantially delay or prevent the cognitive decline caused by AD. Success would fundamentally transform the AD therapeutic landscape, reducing the socioeconomic burden of dementia and preserving cognitive function in ageing populations worldwide.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Jack, C. R. et al. Hypothetical model of dynamic biomarkers of the Alzheimer’s pathological cascade. Lancet Neurol. 9, 119–128 (2010).
McDade, E. M. Alzheimer disease. Continuum 28, 648–675 (2022).
Galasko, D. et al. High cerebrospinal fluid tau and low amyloid β42 levels in the clinical diagnosis of alzheimer disease and relation to apolipoprotein E genotype. Arch. Neurol. 55, 937–945 (1998).
Klunk, W. E. et al. Imaging brain amyloid in Alzheimer’s disease with Pittsburgh compound-B. Ann. Neurol. 55, 306–319 (2004).
Zhang, W. et al. A highly selective and specific PET tracer for imaging of tau pathologies. J. Alzheimer’s Dis. 31, 601–612 (2012).
Hampel, H. et al. Blood-based biomarkers for Alzheimer disease: mapping the road to the clinic. Nat. Rev. Neurol. 14, 639–652 (2018).
Dubois, B. et al. Alzheimer disease as a clinical-biological construct — an International Working Group recommendation. JAMA Neurol. 81, 1304–1311 (2024).
Jack, C. R. et al. Revised criteria for diagnosis and staging of Alzheimer’s disease: Alzheimer’s Association Workgroup. Alzheimers Dement. 20, 5143–5169 (2024).
Cummings, J. et al. Lecanemab: appropriate use recommendations. J. Prev. Alzheimers Dis. 10, 362–377 (2023).
Frisoni, G. B. et al. European intersocietal recommendations for the biomarker-based diagnosis of neurocognitive disorders. Lancet Neurol. 23, 302–312 (2024).
Dickerson, B. C. et al. The Alzheimer’s Association clinical practice guideline for the Diagnostic Evaluation, Testing, Counseling, and Disclosure of Suspected Alzheimer’s Disease and Related Disorders (DETeCD-ADRD): executive summary of recommendations for specialty care. Alzheimers Dement. 21, e14337 (2024).
Atri, A. et al. Alzheimer’s Association clinical practice guideline for the Diagnostic Evaluation, Testing, Counseling, and Disclosure of Suspected Alzheimer’s Disease and Related Disorders (DETeCD-ADRD): executive summary of recommendations for primary care. Alzheimer’s Dement. 21, e14333 (2024).
Mintun, M. A. et al. Donanemab in early Alzheimer’s disease. N. Engl. J. Med. 384, 1691–1704 (2021).
van Dyck, C. H. et al. Lecanemab in early Alzheimer’s disease. N. Engl. J. Med. 388, 142–143 (2023).
Whitmer, R., Sidney, S., Selby, J., Claiborne Johnston, S. & Yaffe, K. Midlife cardiovascular risk factors and risk of dementia in late life. Neurology 64, 277–281 (2005).
Borenstein, A. R., Copenhaver, C. I. & Mortimer, J. A. Early-life risk factors for Alzheimer disease. Alzheimer Dis. Assoc. Disord. 20, 63–72 (2006).
Kivipelto, M. et al. Midlife vascular risk factors and Alzheimer’s disease in later life: longitudinal, population based study. Br. Med. J. 322, 1447–1451 (2001).
Nianogo, R. A. et al. Risk factors associated with Alzheimer disease and related dementias by sex and race and ethnicity in the US. JAMA Neurol. 79, 584–591 (2022).
Weninger, S. et al. Collaboration for Alzheimer’s prevention: principles to guide data and sample sharing in preclinical Alzheimer’s disease trials. Alzheimers Dement. 12, 631–632 (2016).
Calandri, I. L. et al. LatAm FINGERS: overcoming barriers for a dementia prevention trial startup. Alzheimers Dement. 18, e065869 (2022).
Kivipelto, M. et al. World-wide FINGERS network: a global approach to risk reduction and prevention of dementia. Alzheimers Dement. 16, 1078–1094 (2020).
Baker, L. D. et al. U.S. POINTER (USA). Alzheimers Dement. 16, e046951 (2020).
Ngandu, T. et al. A 2 year multidomain intervention of diet, exercise, cognitive training, and vascular risk monitoring versus control to prevent cognitive decline in at-risk elderly people (FINGER): a randomised controlled trial. Lancet 385, 2255–2263 (2015).
Rabinovici, G. D. et al. Association of amyloid positron emission tomography with subsequent change in clinical management among medicare beneficiaries with mild cognitive impairment or dementia. JAMA 321, 1286–1294 (2019).
Palmqvist, S. et al. Blood biomarkers to detect Alzheimer disease in primary care and secondary care. JAMA 332, 1245–1257 (2024).
Beach, T. G., Monsell, S. E., Phillips, L. E. & Kukull, W. Accuracy of the clinical diagnosis of Alzheimer disease at National Institute on Aging Alzheimer Disease Centers, 2005–2010. J. Neuropathol. Exp. Neurol. 71, 266–273 (2012).
McKhann, G. M. et al. The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National Institute on Aging—Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 7, 263–269 (2011).
Albert, M. S. et al. The diagnosis of mild cognitive impairment due to Alzheimer’s disease: recommendations from the National Institute on Aging —Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 7, 270–279 (2011).
Andreasen, N. et al. Evaluation of CSF-tau and CSF-Aβ42 as diagnostic markers for Alzheimer disease in clinical practice. Arch. Neurol. 58, 373–379 (2001).
Brier, M. R. et al. Tau and Aβ imaging, CSF measures, and cognition in Alzheimer’s disease. Sci. Transl. Med. 8, 338ra66 (2016).
Johnson, K. A. et al. Appropriate use criteria for amyloid PET: a report of the Amyloid imaging task force, the society of nuclear medicine and molecular imaging, and the Alzheimer’s association. Alzheimers Dement. 9, E1–E16 (2013).
Shaw, L. M. et al. Appropriate use criteria for lumbar puncture and cerebrospinal fluid testing in the diagnosis of Alzheimer’s disease. Alzheimers Dement. 14, 1505–1521 (2018).
Schindler, S. E. & Atri, A. The role of cerebrospinal fluid and other biomarker modalities in the Alzheimer’s disease diagnostic revolution. Nature Aging 3, 460–462 (2023).
Schindler, S. E. et al. Head-to-head comparison of leading blood tests for Alzheimer’s disease pathology. Alzheimers Dement. 20, 8074–8096 (2024).
Hampel, H. et al. Blood-based biomarkers for Alzheimer’s disease: current state and future use in a transformed global healthcare landscape. Neuron 111, 2781–2799 (2023).
Schindler, S. E. et al. High-precision plasma β-amyloid 42/40 predicts current and future brain amyloidosis. Neurology 93, E1647–E1659 (2019).
Ovod, V. et al. Amyloid β concentrations and stable isotope labeling kinetics of human plasma specific to central nervous system amyloidosis. Alzheimers Dement. 13, 841–849 (2017).
Mielke, M. M. et al. Plasma phospho-tau181 increases with Alzheimer’s disease clinical severity and is associated with tau- and amyloid-positron emission tomography. Alzheimers Dement. 14, 989–997 (2018).
Karikari, T. K. et al. Blood phosphorylated tau 181 as a biomarker for Alzheimer’s disease: a diagnostic performance and prediction modelling study using data from four prospective cohorts. Lancet Neurol. 19, 422–433 (2020).
Preische, O. et al. Serum neurofilament dynamics predicts neurodegeneration and clinical progression in presymptomatic Alzheimer’s disease. Nat. Med. 25, 277–283 (2019).
Barthélemy, N. R. et al. Highly accurate blood test for Alzheimer’s disease is similar or superior to clinical cerebrospinal fluid tests. Nat. Med. 30, 1085–1095 (2024).
Warmenhoven, N. et al. A comprehensive head-to-head comparison of key plasma phosphorylated tau 217 biomarker tests. Brain 139, 16–17 (2012).
Barthélemy, N. R., Horie, K., Sato, C. & Bateman, R. J. Blood plasma phosphorylated-tau isoforms track CNS change in Alzheimer’s disease. J. Exp. Med. 217, e20200861 (2020).
Sperling, R., Mormino, E. & Johnson, K. The evolution of preclinical Alzheimer’s disease: implications for prevention trials. Neuron 84, 608–622 (2014).
Parnetti, L., Chipi, E., Salvadori, N., D’Andrea, K. & Eusebi, P. Prevalence and risk of progression of preclinical Alzheimer’s disease stages: a systematic review and meta-analysis. Alzheimers Res. Ther. 11, 7 (2019).
Ossenkoppele, R. et al. Amyloid and tau PET-positive cognitively unimpaired individuals are at high risk for future cognitive decline. Nat. Med. 28, 2381–2387 (2022).
Younes, K. et al. Amyloid PET predicts longitudinal functional and cognitive trajectories in a heterogeneous cohort. Alzheimers Dement. 21, e70075 (2025).
Cummings, J. et al. Alzheimer’s disease drug development pipeline: 2024. Alzheimers Dement. 10, e12465 (2024).
Rafii, M. S. et al. The AHEAD 3-45 study: design of a prevention trial for Alzheimer’s disease. Alzheimers Dement. 19, 1227–1233 (2023).
McDade, E. & Bateman, R. J. Stop Alzheimer’s before it starts. Nature 547, 153–155 (2017).
Sims, J. R. et al. Donanemab in early symptomatic Alzheimer disease: the TRAILBLAZER-ALZ 2 randomized clinical trial. JAMA 330, 512–527 (2023).
Mielke, M. M. et al. Recommendations for clinical implementation of blood-based biomarkers for Alzheimer’s disease. Alzheimers Dement. 20, 8216–8224 (2024).
Schindler, S. E. et al. Acceptable performance of blood biomarker tests of amyloid pathology — recommendations from the Global CEO initiative on Alzheimer’s disease. Nat. Rev. Neurol. 20, 426–439 (2024).
Erickson, C. M., Largent, E. A. & O’Brien, K. S. Paving the way for Alzheimer’s disease blood-based biomarkers in primary care. Alzheimers Dement. 21, e14203 (2025).
VandeVrede, L., Schindler, S. E. & Suzanne Schindler, C. E. Clinical use of biomarkers in the era of Alzheimer’s disease treatments. Alzheimers Dement. 21, e14201 (2025).
Shen, Y. et al. CSF proteomics identifies early changes in autosomal dominant Alzheimer’s disease. Cell 187, 6309–6326.e15 (2024).
Bornstein, R., Mulholland, M. T., Sedensky, M., Morgan, P. & Johnson, S. C. Glutamine metabolism in diseases associated with mitochondrial dysfunction. Mol. Cell Neurosci. 126, 103887 (2023).
Accelerated Approval Program. FDA https://www.fda.gov/drugs/nda-and-bla-approvals/accelerated-approval-program (2024).
Assunção, S. S. et al. Meaningful benefits: a framework to assess disease-modifying therapies in preclinical and early Alzheimer’s disease. Alzheimers Res. Ther. 14, 54 (2022).
Weintraub, W. S., Lüscher, T. F. & Pocock, S. The perils of surrogate endpoints. Eur. Heart J. 36, 2212–2218 (2015).
Bateman, R. J. et al. Autosomal-dominant Alzheimer’s disease: a review and proposal for the prevention of Alzheimer’s disease. Alzheimers Res. Ther. 3, 1 (2011).
Llibre-Guerra, J. J. et al. Association of longitudinal changes in cerebrospinal fluid total tau and phosphorylated tau 181 and brain atrophy with disease progression in patients with Alzheimer disease. JAMA Netw. Open. 2, e1917126 (2019).
Bateman, R. J. et al. CSF and blood plasma mass spectrometry measures of Aβ, tau, and NfL species and longitudinal relationship to preclinical and clinical staging of amyloid and tau aggregation and clinical stage of Alzheimer’s disease. Alzheimers Dement. 17, e050711 (2021).
Gordon, B. A. et al. Tau PET in autosomal dominant Alzheimer’s disease: relationship with cognition, dementia and other biomarkers. Brain 142, 1063 (2019).
McDade, E. et al. Longitudinal cognitive and biomarker changes in dominantly inherited Alzheimer disease. Neurology 91, e1295–e1306 (2018).
Fagan, A. M. et al. Comparison of CSF biomarkers in Down syndrome and autosomal dominant Alzheimer’s disease: a cross-sectional study. Lancet Neurol. 20, 615–626 (2021).
Hartley, S. L. et al. AT(N) biomarker profiles and Alzheimer’s disease symptomology in Down syndrome. Alzheimers Dement. 20, 366–375 (2024).
Fortea, J. et al. Clinical and biomarker changes of Alzheimer’s disease in adults with Down syndrome: a cross-sectional study. Lancet 395, 1988–1997 (2020).
Bateman, R. J. et al. Clinical and biomarker changes in dominantly inherited Alzheimer’s disease. N. Engl. J. Med. 367, 795–804 (2012).
Barthélemy, N. R. et al. A soluble phosphorylated tau signature links tau, amyloid and the evolution of stages of dominantly inherited Alzheimer’s disease. Nat. Med. 26, 398–407 (2020).
Brown, M. S. & Goldstein, J. L. A tribute to Akira Endo, discoverer of a “penicillin” for cholesterol. Atheroscler. Suppl. 5, 13–16 (2004).
Mawuenyega, K. G. et al. Decreased clearance of CNS beta-amyloid in Alzheimer’s disease. Science 330, 1774 (2010).
US National Library of Medicine. A study of potential disease modifying treatments in individuals at risk for or with a type of early onset AD caused by a genetic mutation (DIAN-TU). ClinicalTrials.gov https://clinicaltrials.gov/study/NCT05552157 (2025).
US National Library of Medicine. A study of a potential disease modifying treatment in individuals at risk for or with a type of early onset AD caused by a genetic mutation (DIAN-TU-002). ClinicalTrials.gov https://clinicaltrials.gov/study/NCT06647498 (2025).
US National Library of Medicine. A study of remternetug (LY3372993) in participants With Alzheimer’s disease (TRAILRUNNER-ALZ 1). ClinicalTrials.gov https://clinicaltrials.gov/study/NCT05463731 (2025).
US National Library of Medicine. A study of remternetug (LY3372993) in early Alzheimer’s disease (TRAILRUNNER-ALZ 3). ClinicalTrials.gov https://clinicaltrials.gov/study/NCT06653153 (2025).
Koyama, A. Does the current evidence base support lecanemab continued dosing for early Alzheimer’s disease? Eisai Medical Information https://www.eisaimedicalinformation.com/-/media/Files/EisaiMedicalInformation/Neurology/Congress-Materials/CTAD-2024/Symp-1_van-Dyck-et-al_Continued-Dosing-CTAD24---27OCT2024-FINAL.pdf (2024).
Shcherbinin, S. et al. Association of amyloid reduction after donanemab treatment with tau pathology and clinical outcomes: the TRAILBLAZER-ALZ randomized clinical trial. JAMA Neurol. 79, 1015 (2022).
Bateman, R. J. et al. Plasma biomarkers of AD emerging as essential tools for drug development: an EU/US CTAD task force report. J. Prev. Alzheimers Dis. 6, 169–173 (2019).
Mormino, E. C. & Papp, K. V. Amyloid accumulation and cognitive decline in clinically normal older individuals: implications for aging and early Alzheimer’s disease. J. Alzheimers Dis. 64, S633 (2018).
Aisen, P. et al. The case for regulatory approval of amyloid-lowering immunotherapies in Alzheimer’s disease based on clearcut biomarker evidence. Alzheimers Dement. 21, e14342 (2024).
Bateman, R. J. et al. Safety and efficacy of long-term gantenerumab treatment in dominantly inherited Alzheimer’s disease: an open-label extension of the phase 2/3 multicentre, randomised, double-blind, placebo-controlled platform DIAN-TU trial. Lancet Neurol. 24, 316–330 (2025).
US National Library of Medicine. DIAN-TU amyloid removal trial (ART) in dominantly inherited Alzheimer’s disease (DIAN-TU) ClinicalTrials.gov https://www.clinicaltrials.gov/study/NCT06384573 (2025).
US National Library of Medicine. AHEAD 3-45 study: a study to evaluate efficacy and safety of treatment with lecanemab in participants with preclinical Alzheimer’s disease and elevated amyloid and also in participants with early preclinical Alzheimer’s disease and intermediate amyloid. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT04468659 (2025).
US National Library of Medicine. A donanemab (LY3002813) study in participants with preclinical Alzheimer’s disease (TRAILBLAZER-ALZ 3). ClinicalTrials.gov https://clinicaltrials.gov/study/NCT05026866 (2025).
US National Library of Medicine. Dominantly inherited Alzheimer network trial: an opportunity to prevent dementia. a study of potential disease modifying treatments in individuals with a type of early onset Alzheimer’s disease caused by a genetic mutation (DIAN-TU). ClinicalTrials.gov https://clinicaltrials.gov/study/NCT05269394 (2025).
US National Library of Medicine. A study of donanemab, RG6289, or the combination of donanemab and RG6289 in presenilin 1 (PSEN1) E280A mutation carriers for the treatment of autosomal-dominant Alzheimer’s disease.ClinicalTrials.gov https://clinicaltrials.gov/study/NCT06996730 (2025).
Li, Y. et al. Timing of biomarker changes in sporadic Alzheimer’s disease in estimated years from symptom onset. Ann. Neurol. 95, 951–965 (2024).
Greenberg, S. M., Bax, F. & van Veluw, S. J. Amyloid-related imaging abnormalities: manifestations, metrics and mechanisms. Nat. Rev. Neurol. 21, 193–203 (2025).
Grimm, H. P. et al. Delivery of the BrainshuttleTM amyloid-beta antibody fusion trontinemab to non-human primate brain and projected efficacious dose regimens in humans. mAbs 15, 2261509 (2023).
Cummings, J. et al. Aducanumab: appropriate use recommendations. J. Prev. Alzheimers Dis. 8, 398–410 (2021).
Li, M. et al. Design and feasibility of an Alzheimer’s disease blood test study in a diverse community-based population. Alzheimers Dement. 19, 5387–5398 (2023).
Grill, J. D. et al. Disclosure of elevated amyloid status is not associated with long-term suicidality in a preclinical AD trial. Alzheimers Dement. 21, e14623 (2025).
Harkins, K. et al. Development of a process to disclose amyloid imaging results to cognitively normal older adult research participants. Alzheimers Res. Ther. 7, 26 (2015).
Tortelli, R. et al. A phase IIA study investigating a γ-secretase modulator in individuals at risk for or at the prodromal stage of Alzheimer’s disease. In Intl Conf. Alzheimer’s and Parkinson’s Diseases (AD/PD 2024) https://medically.roche.com/content/dam/pdmahub/restricted/neurology/adpd-2024/ADPD-2024-presentation-tortelli-a-phase-IIa-study.pdf (2024).
A study of JNJ-64042056 in participants with preclinical Alzheimer’s disease. ISRCTN https://www.isrctn.com/ISRCTN78730935 (2024).
Sperling, R. A. et al. Trial of solanezumab in preclinical Alzheimer’s disease. N. Engl. J. Med. 389, 1096–1107 (2023).
Honig, L. S. Trial of solanezumab for mild dementia due to Alzheimer’s disease. N. Engl. J. Med. 378, 321–330 (2018).
US National Library Of Medicine. A study of crenezumab versus placebo in preclinical presenilin1 (PSEN1) E280a mutation carriers to evaluate efficacy and safety in the treatment of autosomal-dominant Alzheimer’s Disease (AD), including a placebo-treated non-carrier cohort. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT01998841 (2024).
Ostrowitzki, S. et al. Evaluating the safety and efficacy of crenezumab vs placebo in adults with early Alzheimer disease. JAMA Neurol. 79, 1113–1121 (2022).
Bateman, R. J. et al. Two phase 3 trials of gantenerumab in early Alzheimer’s disease. N. Engl. J. Med. 389, 1862–1876 (2023).
McKhann, G. et al. Clinical diagnosis of Alzheimer’s disease — report of the NINCDS–ADRDA work group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s disease. Neurology 34, 939–944 (1984).
Davis, K. L. et al. A double-blind, placebo-controlled multicenter study of tacrine for Alzheimer’s disease. N. Engl. J. Med. 327, 1253–1259 (1992).
Goate, A. et al. Segregation of a missense mutation in the amyloid precursor protein gene with familial Alzheimer’s disease. Nature 349, 704–706 (1991).
Acknowledgements
The authors thank all study participants and their families, whose commitment makes prevention research in Alzheimer disease possible.
Author information
Authors and Affiliations
Contributions
J.J.L.-G. researched data for the article. All authors contributed substantially to discussion of the content, wrote the article, and reviewed and/or edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
J.J.L.-G.’s research is supported by the National Institutes of Health National Institute of Aging (K01AG073526), the Alzheimer’s Association (AARFD-21-851415, SG-20-690363, 24HPE-1287320), the Michael J. Fox Foundation (MJFF-020770) and The Foundation for Barnes–Jewish Hospital. E.M.McD. has received research funding from the Alzheimer’s Association, Eisai, Eli Lilly, GHR, Hoffman–La Roche, and the National Institutes of Health. E.M.McD. has served as a consultant or adviser for Alzamend, AstraZeneca, Hoffman–La Roche, Ionis and Sanofi. E.M.McD. has served as a data and safety monitoring board member for Alector and Alnylum, and is on the scientific advisory board for Foundation Alzheimer. S.E.S. has served on scientific advisory boards related to biomarker testing and education for companies including Eisai, Eli Lilly and Novo Nordisk. D.B.C. receives royalties from Wolters Kluwer. D.B.C. serves as scientific consultant to Atara Biotherapeutics, Cellevolve Bio, Excision BioTherapeutics, F. Hoffmann–La Roche/Genentech, ICON (Teva), Sanofi Genzyme, Seagen and Wave Life Sciences. D.B.C. has carried out legal consulting for Lewis, Thomason, King, Krieg and Waldrop and Loughren, Loughren, Loughren Powell Gilbert. D.B.C. serves on the data safety monitoring boards for Atara Biotherapeutics, Avidity, Cellevolve Bio, Excision Biotherapeutics, Morphic, Sanofi Genzyme, Teva and Wave Life Sciences. A.A. has received consulting fees from multiple pharmaceutical and biotechnology companies, including Eisai, Lundbeck, Merck, Novo Nordisk, Prothena and Roche/Genentech. A.A.’s institution receives grant and research support from the National Institutes of Health (NIH), the State of Arizona, Gates Ventures and several industry partners. R.J.B. receives laboratory research funding from the Alzheimer’s Association, Association for Frontotemporal Degeneration, Association for Frontotemporal Degeneration FTD Biomarkers Initiative, Avid Radiopharmaceuticals, BrightFocus Foundation, Centene Corporation, Coins for Alzheimer’s Research Trust Fund, the Cure Alzheimer’s Fund, DIAN-TU Pharma Consortium, Good Ventures Foundation, Janssen, the National Institutes of Health, the NfL Consortium (AbbVie, Biogen, Bristol Meyers Squibb, Hoffman La Roche), Novartis, Rainwater Foundation, Tau Consortium, Tau SILK Consortium (AbbVie, Biogen, Eli Lilly and Company and an anonymous organization), The Foundation for Barnes–Jewish Hospital and the Tracy Family SILQ Center. Washington University and R.J.B. have equity ownership interest in C2N Diagnostics and receive income based on technology (stable isotope labelling kinetics, blood plasma assay, and methods of diagnosing AD with phosphorylation changes) licensed by Washington University to C2N Diagnostics. C.S. declares no financial competing interests.
Peer review
Peer review information
Nature Reviews Neurology thanks S. Rios-Romenets, J. Therriault and W. van der Flier for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Llibre-Guerra, J.J., McDade, E.M., Schindler, S.E. et al. Towards pharmacological prevention of Alzheimer disease. Nat Rev Neurol 21, 721–733 (2025). https://doi.org/10.1038/s41582-025-01154-y
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41582-025-01154-y