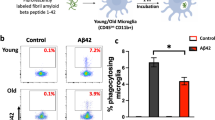
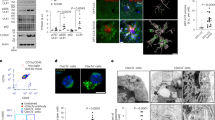
Abstract
Accumulating evidence indicates that Alzheimer disease (AD) is caused by dysregulated microglial phagocytosis. The main risk factor for AD is age, and ageing reduces microglial phagocytosis of amyloid-β (Aβ) plaques, while increasing microglial phagocytosis of synapses and neurons. Most of the known genetic risk for AD can be linked to microglial phagocytosis, including ABCA1, ABI3, ACE, ADAM17, APOE, APP, BIN1, BLNK, CD2AP, CD33, CLU, CR1, CTSB, CTSH, EED, GRN, INPP5D, LILRB2, PICALM, PLCG2, PSEN1, PTK2B, SIGLEC11, SORL1, SPI1, TMEM106B and TREM2. Moreover, the only disease-modifying treatments for AD — anti-Aβ antibodies — work by increasing microglial phagocytosis of Aβ aggregates. Microglial phagocytosis of Aβ via TREM2, LRP1, CD33, TAM receptors and anti-Aβ antibodies appears to reduce AD pathology by pruning and compacting plaques, restricting subsequent tau pathology, whereas microglial phagocytosis of synapses and neurons seems detrimental in the later stages of AD, via complement, P2Y6 receptor and TREM2. However, the roles of microglial phagocytosis in AD are complex and multifaceted, and improved treatments are likely to require a deeper understanding of these roles.
Key points
-
Insufficient microglial phagocytosis of amyloid-β (Aβ) and/or excessive phagocytosis of synapses and neurons might underly Alzheimer disease (AD) pathology.
-
Most of the known genetic risk for AD is via genes that affect microglial phagocytosis.
-
Increase of microglial phagocytosis of Aβ aggregates using anti-Aβ antibodies clears amyloid plaques and slows AD progression in people with the disease.
-
Microglial phagocytosis of Aβ via antibodies, TREM2, LRP1 and TAM receptors is protective in AD animal models by pruning and compacting plaques.
-
Microglial phagocytosis of synapses and neurons, via complement, P2Y6 receptor and TREM2, is detrimental in AD animal models.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Knopman, D. S. et al. Alzheimer disease. Nat. Rev. Dis. Primers 7, 33 (2021).
Heneka, M. T. et al. Neuroinflammation in Alzheimer disease. Nat. Rev. Immunol. 25, 321–352 (2025).
Prinz, M., Jung, S. & Priller, J. Microglia biology: one century of evolving concepts. Cell 179, 292–311 (2019).
Ginhoux, F. et al. Fate mapping analysis reveals that adult microglia derive from primitive macrophages. Science 330, 841–845 (2010).
Paolicelli, R. C. et al. Microglia states and nomenclature: a field at its crossroads. Neuron 110, 3458–3483 (2022).
Cockram, T. O. J., Dundee, J. M., Popescu, A. S. & Brown, G. C. The phagocytic code regulating phagocytosis of mammalian cells. Front. Immunol. 12, 629979 (2021).
Butler, C. A. et al. Microglial phagocytosis of neurons in neurodegeneration, and its regulation. J. Neurochem. 158, 621–639 (2021).
Yoo, C. J. et al. Complement receptor 4 mediates the clearance of extracellular tau fibrils by microglia. FEBS J. 291, 3499–3520 (2024).
Greven, J. A. et al. Biophysical mapping of TREM2-ligand interactions reveals shared surfaces for engagement of multiple Alzheimer’s disease ligands. Mol. Neurodegener. 20, 3 (2025).
Koizumi, S. et al. UDP acting at P2Y6 receptors is a mediator of microglial phagocytosis. Nature 446, 1091–1095 (2007).
Hu, W. et al. Neuronal CD47 induces behavioral alterations and ameliorates microglial synaptic pruning in wild-type and Alzheimer’s mouse models. Cell Biosci. 15, 38 (2025).
Gonzalez-Gil, A. et al. Human brain sialoglycan ligand for CD33, a microglial inhibitory siglec implicated in Alzheimer’s disease. J. Biol. Chem. 298, 101960 (2022).
Dodd, R. B. et al. CD33 and clusterin interact biophysically and genetically to modulate Alzheimer risk. Preprint at bioRxiv https://doi.org/10.1101/2025.07.29.667318 (2025).
Lv, J. et al. DW14006 as a direct AMPKalpha1 activator improves pathology of AD model mice by regulating microglial phagocytosis and neuroinflammation. Brain Behav. Immun. 90, 55–69 (2020).
Zhang, Y. et al. Activation of AMPK by GLP-1R agonists mitigates Alzheimer-related phenotypes in transgenic mice. Nat. Aging 5, 1097–1113 (2025).
Wang, K. et al. The role of microglial/macrophagic salt-inducible kinase 3 on normal and excessive phagocytosis after transient focal cerebral ischemia. Cell Mol. Life Sci. 79, 439 (2022).
Shi, Q., Chang, C., Saliba, A. & Bhat, M. A. Microglial mTOR activation upregulates trem2 and enhances beta-amyloid plaque clearance in the 5XFAD Alzheimer’s disease model. J. Neurosci. 42, 5294–5313 (2022).
Zhu, B. et al. G-protein-coupled receptor ADGRG1 drives a protective microglial state in Alzheimer’s disease through MYC activation. Neuron 113, 3224–3242.e7 (2025).
Das, R. & Chinnathambi, S. Microglial remodeling of actin network by tau oligomers, via G protein-coupled purinergic receptor, P2Y12R-driven chemotaxis. Traffic 22, 153–170 (2021).
Sun, N. et al. Human microglial state dynamics in Alzheimer’s disease progression. Cell 186, 4386–4403.e29 (2023).
Keren-Shaul, H. et al. A unique microglia type associated with restricting development of Alzheimer’s disease. Cell 169, 1276–1290.e17 (2017).
Chen, Y. & Colonna, M. Microglia in Alzheimer’s disease at single-cell level. Are there common patterns in humans and mice? J. Exp. Med. 218, e20202717 (2021).
Li, Q. et al. Developmental heterogeneity of microglia and brain myeloid cells revealed by deep single-cell RNA sequencing. Neuron 101, 207–223.e10 (2019).
Hammond, T. R. et al. Single-Cell RNA sequencing of microglia throughout the mouse lifespan and in the injured brain reveals complex cell-state changes. Immunity 50, 253–271.e6 (2019).
Huang, Y. et al. Microglia use TAM receptors to detect and engulf amyloid beta plaques. Nat. Immunol. 22, 586–594 (2021).
Olah, M. et al. Single cell RNA sequencing of human microglia uncovers a subset associated with Alzheimer’s disease. Nat. Commun. 11, 6129 (2020).
Grubman, A. et al. Transcriptional signature in microglia associated with Aβ plaque phagocytosis. Nat. Commun. 12, 3015 (2021).
Dolan, M. J. et al. Exposure of iPSC-derived human microglia to brain substrates enables the generation and manipulation of diverse transcriptional states in vitro. Nat. Immunol. 24, 1382–1390 (2023).
Wang, S. et al. TREM2 drives microglia response to amyloid-β via SYK-dependent and -independent pathways. Cell 185, 4153–4169.e19 (2022).
Krasemann, S. et al. The TREM2–APOE pathway drives the transcriptional phenotype of dysfunctional microglia in neurodegenerative diseases. Immunity 47, 566–581.e9 (2017).
Diaz-Aparicio, I. et al. Microglia actively remodel adult hippocampal neurogenesis through the phagocytosis secretome. J. Neurosci. 40, 1453–1482 (2020).
Marschallinger, J. et al. Lipid-droplet-accumulating microglia represent a dysfunctional and proinflammatory state in the aging brain. Nat. Neurosci. 23, 194–208 (2020).
Wu, X., Miller, J. A., Lee, B. T. K., Wang, Y. & Ruedl, C. Reducing microglial lipid load enhances beta amyloid phagocytosis in an Alzheimer’s disease mouse model. Sci. Adv. 11, eadq6038 (2025).
Gerrits, E. et al. Distinct amyloid-beta and tau-associated microglia profiles in Alzheimer’s disease. Acta Neuropathol. 141, 681–696 (2021).
Terry, R. D. et al. Physical basis of cognitive alterations in Alzheimer’s disease: synapse loss is the major correlate of cognitive impairment. Ann. Neurol. 30, 572–580 (1991).
Mecca, A. P. et al. Synaptic density and cognitive performance in Alzheimer’s disease: a PET imaging study with [(11) C]UCB-J. Alzheimers Dement. 18, 2527–2536 (2022).
Tzioras, M., McGeachan, R. I., Durrant, C. S. & Spires-Jones, T. L. Synaptic degeneration in Alzheimer disease. Nat. Rev. Neurol. 19, 19–38 (2023).
Spangenberg, E. E. et al. Eliminating microglia in Alzheimer’s mice prevents neuronal loss without modulating amyloid-beta pathology. Brain 139, 1265–1281 (2016).
Olmos-Alonso, A. et al. Pharmacological targeting of CSF1R inhibits microglial proliferation and prevents the progression of Alzheimer’s-like pathology. Brain 139, 891–907 (2016).
Tzioras, M. et al. Human astrocytes and microglia show augmented ingestion of synapses in Alzheimer’s disease via MFG-E8. Cell Rep. Med. 4, 101175 (2023).
Paolicelli, R. C. et al. Synaptic pruning by microglia is necessary for normal brain development. Science 333, 1456–1458 (2011).
Favuzzi, E. et al. GABA-receptive microglia selectively sculpt developing inhibitory circuits. Cell 184, 4048–4063.e32 (2021).
Andoh, M. et al. Nonapoptotic caspase-3 guides C1q-dependent synaptic phagocytosis by microglia. Nat. Commun. 16, 918 (2025).
Weinhard, L. et al. Microglia remodel synapses by presynaptic trogocytosis and spine head filopodia induction. Nat. Commun. 9, 1228 (2018).
Kurematsu, C. et al. Synaptic pruning of murine adult-born neurons by microglia depends on phosphatidylserine. J. Exp. Med. 219, e20202304 (2022).
Cangalaya, C. et al. Real-time mechanisms of exacerbated synaptic remodeling by microglia in acute models of systemic inflammation and tauopathy. Brain Behav. Immun. 110, 245–259 (2023).
Schafer, D. P. et al. Microglia sculpt postnatal neural circuits in an activity and complement-dependent manner. Neuron 74, 691–705 (2012).
Sipe, G. O. et al. Microglial P2Y12 is necessary for synaptic plasticity in mouse visual cortex. Nat. Commun. 7, 10905 (2016).
Liu, Y. J. et al. Microglia elimination increases neural circuit connectivity and activity in adult mouse cortex. J. Neurosci. 41, 1274–1287 (2021).
Hong, S. et al. Complement and microglia mediate early synapse loss in Alzheimer mouse models. Science 352, 712–716 (2016).
Lv, Z. et al. Clearance of β-amyloid and synapses by the optogenetic depolarization of microglia is complement selective. Neuron 112, 740–754.e7 (2024).
Dejanovic, B. et al. Complement C1q-dependent excitatory and inhibitory synapse elimination by astrocytes and microglia in Alzheimer’s disease mouse models. Nat. Aging 2, 837–850 (2022).
Gomez-Arboledas, A. et al. C5aR1 signaling promotes region- and age-dependent synaptic pruning in models of Alzheimer’s disease. Alzheimers Dement. 20, 2173–2190 (2024).
Sanfilippo, C. et al. Synaptic pruning genes networks in Alzheimer’s disease: correlations with neuropathology and cognitive decline. Geroscience https://doi.org/10.1007/s11357-025-01740-4 (2025).
Gouras, G. K. et al. Intraneuronal Aβ42 accumulation in human brain. Am. J. Pathol. 156, 15–20 (2000).
Capetillo-Zarate, E. et al. High-resolution 3D reconstruction reveals intra-synaptic amyloid fibrils. Am. J. Pathol. 179, 2551–2558 (2011).
De Schepper, S. et al. Perivascular cells induce microglial phagocytic states and synaptic engulfment via SPP1 in mouse models of Alzheimer’s disease. Nat. Neurosci. 26, 406–415 (2023).
Brown, G. C. & Neher, J. J. Microglial phagocytosis of live neurons. Nat. Rev. Neurosci. 15, 209–216 (2014).
Scott-Hewitt, N. et al. Local externalization of phosphatidylserine mediates developmental synaptic pruning by microglia. EMBO J. 39, e105380 (2020).
Djannatian, M. et al. Myelination generates aberrant ultrastructure that is resolved by microglia. J. Cell Biol. 222, e202204010 (2023).
Rueda-Carrasco, J. et al. Microglia-synapse engulfment via PtdSer-TREM2 ameliorates neuronal hyperactivity in Alzheimer’s disease models. EMBO J. 42, e113246 (2023).
Bader Lange, M. L. et al. Loss of phospholipid asymmetry and elevated brain apoptotic protein levels in subjects with amnestic mild cognitive impairment and Alzheimer disease. Neurobiol. Dis. 29, 456–464 (2008).
Puigdellivol, M. et al. The microglial P2Y6 receptor mediates neuronal loss and memory deficits in neurodegeneration. Cell Rep. 37, 110148 (2021).
Kanekiyo, T. et al. Neuronal clearance of amyloid-beta by endocytic receptor LRP1. J. Neurosci. 33, 19276–19283 (2013).
Taddei, R. N. et al. Tau oligomer-containing synapse elimination by microglia and astrocytes in Alzheimer disease. JAMA Neurol. 80, 1209–1221 (2023).
Brelstaff, J. H. et al. Microglia become hypofunctional and release metalloproteases and tau seeds when phagocytosing live neurons with P301S tau aggregates. Sci. Adv. 7, eabg4980 (2021).
Asai, H. et al. Depletion of microglia and inhibition of exosome synthesis halt tau propagation. Nat. Neurosci. 18, 1584–1593 (2015).
Wang, C. et al. Selective removal of astrocytic APOE4 strongly protects against tau-mediated neurodegeneration and decreases synaptic phagocytosis by microglia. Neuron 109, 1657–1674.e7 (2021).
Lish, A. M. et al. CLU alleviates Alzheimer’s disease-relevant processes by modulating astrocyte reactivity and microglia-dependent synaptic density. Neuron 113, 1925–1946.e11 (2025).
Matera, A. et al. Microglial lipid phosphatase SHIP1 limits complement-mediated synaptic pruning in the healthy developing hippocampus. Immunity 58, 197–217.e13 (2025).
Andrade-Moraes, C. H. et al. Cell number changes in Alzheimer’s disease relate to dementia, not to plaques and tangles. Brain 136, 3738–3752 (2013).
Brown, G. C. Cell death by phagocytosis. Nat. Rev. Immunol. 24, 91–102 (2024).
Neher, J. J. et al. Inhibition of microglial phagocytosis is sufficient to prevent inflammatory neuronal death. J. Immunol. 186, 4973–4983 (2011).
Neniskyte, U., Neher, J. J. & Brown, G. C. Neuronal death induced by nanomolar amyloid beta is mediated by primary phagocytosis of neurons by microglia. J. Biol. Chem. 286, 39904–39913 (2011).
Neher, J. J., Neniskyte, U., Hornik, T. & Brown, G. C. Inhibition of UDP/P2Y6 purinergic signaling prevents phagocytosis of viable neurons by activated microglia in vitro and in vivo. Glia 62, 1463–1475 (2014).
Fricker, M. et al. MFG-E8 mediates primary phagocytosis of viable neurons during neuroinflammation. J. Neurosci. 32, 2657–2666 (2012).
Pampuscenko, K. et al. Extracellular tau induces microglial phagocytosis of living neurons in cell cultures. J. Neurochem. 154, 316–329 (2020).
Pampuscenko, K. et al. Extracellular tau stimulates phagocytosis of living neurons by activated microglia via Toll-like 4 receptor-NLRP3 inflammasome-caspase-1 signalling axis. Sci. Rep. 13, 10813 (2023).
Brelstaff, J., Tolkovsky, A. M., Ghetti, B., Goedert, M. & Spillantini, M. G. Living neurons with tau filaments aberrantly expose phosphatidylserine and are phagocytosed by microglia. Cell Rep. 24, 1939–1948.e4 (2018).
Milde, S. et al. Inflammatory neuronal loss in the substantia nigra induced by systemic lipopolysaccharide is prevented by knockout of the P2Y6 receptor in mice. J. Neuroinflamm. 18, 225 (2021).
Fuhrmann, M. et al. Microglial Cx3cr1 knockout prevents neuron loss in a mouse model of Alzheimer’s disease. Nat. Neurosci. 13, 411–413 (2010).
Huang, Y. & Lemke, G. Early death in a mouse model of Alzheimer’s disease exacerbated by microglial loss of TAM receptor signaling. Proc. Natl Acad. Sci. USA 119, e2204306119 (2022).
Umpierre, A. D. et al. Microglial P2Y6 calcium signaling promotes phagocytosis and shapes neuroimmune responses in epileptogenesis. Neuron 112, 1959–1977.e10 (2024).
Dundee, J. M., Puigdellivol, M., Butler, R., Cockram, T. O. J. & Brown, G. C. P2Y6 receptor-dependent microglial phagocytosis of synapses mediates synaptic and memory loss in aging. Aging Cell 22, e13761 (2023).
Chen, Y. et al. Microglia efferocytosis: an emerging mechanism for the resolution of neuroinflammation in Alzheimer’s disease. J. Neuroinflamm. 22, 96 (2025).
Romero-Molina, C., Garretti, F., Andrews, S. J., Marcora, E. & Goate, A. M. Microglial efferocytosis: diving into the Alzheimer’s disease gene pool. Neuron 110, 3513–3533 (2022).
Meyer, C. et al. Early Locus Coeruleus noradrenergic axon loss drives olfactory dysfunction in Alzheimer’s disease. Nat. Commun. 16, 7338 (2025).
Pantazopoulou, M. et al. Differential intracellular trafficking of extracellular vesicles in microglia and astrocytes. Cell Mol. Life Sci. 80, 193 (2023).
Mandrekar, S. et al. Microglia mediate the clearance of soluble Aβ through fluid phase macropinocytosis. J. Neurosci. 29, 4252–4262 (2009).
Laporte, V. et al. Uptake of Aβ 1-40- and Aβ 1-42-coated yeast by microglial cells: a role for LRP. J. Leukoc. Biol. 76, 451–461 (2004).
Rushworth, J. V., Griffiths, H. H., Watt, N. T. & Hooper, N. M. Prion protein-mediated toxicity of amyloid-beta oligomers requires lipid rafts and the transmembrane LRP1. J. Biol. Chem. 288, 8935–8951 (2013).
Kanekiyo, T. et al. Heparan sulphate proteoglycan and the low-density lipoprotein receptor-related protein 1 constitute major pathways for neuronal amyloid-beta uptake. J. Neurosci. 31, 1644–1651 (2011).
Paresce, D. M., Ghosh, R. N. & Maxfield, F. R. Microglial cells internalize aggregates of the Alzheimer’s disease amyloid beta-protein via a scavenger receptor. Neuron 17, 553–565 (1996).
Husemann, J., Loike, J. D., Kodama, T. & Silverstein, S. C. Scavenger receptor class B type I (SR-BI) mediates adhesion of neonatal murine microglia to fibrillar beta-amyloid. J. Neuroimmunol. 114, 142–150 (2001).
Yeh, F. L., Wang, Y., Tom, I., Gonzalez, L. C. & Sheng, M. TREM2 binds to apolipoproteins, including APOE and CLU/APOJ, and thereby facilitates uptake of amyloid-beta by microglia. Neuron 91, 328–340 (2016).
Fu, H. et al. Complement component C3 and complement receptor type 3 contribute to the phagocytosis and clearance of fibrillar Aβ by microglia. Glia 60, 993–1003 (2012).
Lemke, G. Biology of the TAM receptors. Cold Spring Harb. Perspect. Biol. 5, a009076 (2013).
Gotkiewicz, M., Capra, J., Miettinen, P. O., Natunen, T. & Tanila, H. Three-dimensional view of microglia-amyloid plaque interactions. Glia 73, 196–209 (2025).
Casali, B. T., MacPherson, K. P., Reed-Geaghan, E. G. & Landreth, G. E. Microglia depletion rapidly and reversibly alters amyloid pathology by modification of plaque compaction and morphologies. Neurobiol. Dis. 142, 104956 (2020).
Grathwohl, S. A. et al. Formation and maintenance of Alzheimer’s disease beta-amyloid plaques in the absence of microglia. Nat. Neurosci. 12, 1361–1363 (2009).
Spangenberg, E. et al. Sustained microglial depletion with CSF1R inhibitor impairs parenchymal plaque development in an Alzheimer’s disease model. Nat. Commun. 10, 3758 (2019).
Liu, C. C. et al. ApoE4 accelerates early seeding of amyloid pathology. Neuron 96, 1024–1032.e3 (2017).
Kaji, S. et al. Apolipoprotein E aggregation in microglia initiates Alzheimer’s disease pathology by seeding β-amyloidosis. Immunity 57, 2651–2668.e12 (2024).
Fourgeaud, L. et al. TAM receptors regulate multiple features of microglial physiology. Nature 532, 240–244 (2016).
Su, Y. & Chang, P. T. Acidic pH promotes the formation of toxic fibrils from beta-amyloid peptide. Brain Res. 893, 287–291 (2001).
Jacquet, R. G. et al. Microglia degrade Alzheimer’s amyloid-beta deposits extracellularly via digestive exophagy. Cell Rep. 43, 115052 (2024).
Lemke, G. & Huang, Y. The dense-core plaques of Alzheimer’s disease are granulomas. J. Exp. Med. 219, e20212477 (2022).
Fruhwurth, S., Zetterberg, H. & Paludan, S. R. Microglia and amyloid plaque formation in Alzheimer’s disease - Evidence, possible mechanisms, and future challenges. J. Neuroimmunol. 390, 578342 (2024).
Guerreiro, R. et al. TREM2 variants in Alzheimer’s disease. N. Engl. J. Med. 368, 117–127 (2013).
Song, W. M. et al. Humanized TREM2 mice reveal microglia-intrinsic and -extrinsic effects of R47H polymorphism. J. Exp. Med. 215, 745–760 (2018).
Zhou, Y. et al. Human and mouse single-nucleus transcriptomics reveal TREM2-dependent and TREM2-independent cellular responses in Alzheimer’s disease. Nat. Med. 26, 131–142 (2020).
Jay, T. R. et al. Disease progression-dependent effects of TREM2 deficiency in a mouse model of Alzheimer’s disease. J. Neurosci. 37, 637–647 (2017).
Griciuc, A. et al. TREM2 acts downstream of CD33 in modulating microglial pathology in Alzheimer’s disease. Neuron 103, 820–835.e7 (2019).
Wang, Y. et al. TREM2 lipid sensing sustains the microglial response in an Alzheimer’s disease model. Cell 160, 1061–1071 (2015).
Condello, C., Yuan, P., Schain, A. & Grutzendler, J. Microglia constitute a barrier that prevents neurotoxic protofibrillar Aβ42 hotspots around plaques. Nat. Commun. 6, 6176 (2015).
Parhizkar, S. et al. Author correction: loss of TREM2 function increases amyloid seeding but reduces plaque-associated ApoE. Nat. Neurosci. 27, 1858–1859 (2024).
van Dyck, C. H. et al. Lecanemab in early Alzheimer’s disease. N. Engl. J. Med. 388, 9–21 (2023).
Sims, J. R. et al. Donanemab in early symptomatic Alzheimer disease: The TRAILBLAZER-ALZ 2 randomized clinical trial. JAMA 330, 512–527 (2023).
Loeffler, D. A. Antibody-mediated clearance of brain amyloid-beta: mechanisms of action, effects of natural and monoclonal anti-Aβ antibodies, and downstream effects. J. Alzheimers Dis. Rep. 7, 873–899 (2023).
van Olst, L. et al. Microglial mechanisms drive amyloid-beta clearance in immunized patients with Alzheimer’s disease. Nat. Med. 31, 1604–1616 (2025).
Lue, L. F. & Walker, D. G. Modeling Alzheimer’s disease immune therapy mechanisms: interactions of human postmortem microglia with antibody-opsonized amyloid beta peptide. J. Neurosci. Res. 70, 599–610 (2002).
Wilcock, D. M. et al. Microglial activation facilitates Aβ plaque removal following intracranial anti-Aβ antibody administration. Neurobiol. Dis. 15, 11–20 (2004).
Bacskai, B. J. et al. Non-Fc-mediated mechanisms are involved in clearance of amyloid-beta in vivo by immunotherapy. J. Neurosci. 22, 7873–7878 (2002).
Garcia-Alloza, M. et al. A limited role for microglia in antibody mediated plaque clearance in APP mice. Neurobiol. Dis. 28, 286–292 (2007).
Webster, S. D. et al. Antibody-mediated phagocytosis of the amyloid beta-peptide in microglia is differentially modulated by C1q. J. Immunol. 166, 7496–7503 (2001).
Rogers, J. et al. Complement activation by beta-amyloid in Alzheimer disease. Proc. Natl Acad. Sci. USA 89, 10016–10020 (1992).
Batista, A. F., Khan, K. A., Papavergi, M. T. & Lemere, C. A. The importance of complement-mediated immune signaling in Alzheimer’s disease pathogenesis. Int. J. Mol. Sci. 25, 817 (2024).
Bradt, B. M., Kolb, W. P. & Cooper, N. R. Complement-dependent proinflammatory properties of the Alzheimer’s disease beta-peptide. J. Exp. Med. 188, 431–438 (1998).
Jucker, M. & Walker, L. C. Alzheimer’s disease: from immunotherapy to immunoprevention. Cell 186, 4260–4270 (2023).
Fastenau, C. et al. Distinct patterns of plaque and microglia glycosylation in Alzheimer’s disease. Brain Pathol. 34, e13267 (2024).
Jung, H. et al. Anti-inflammatory clearance of amyloid-β by a chimeric Gas6 fusion protein. Nat. Med. 28, 1802–1812 (2022).
Sheng, L. et al. Microglial Trem2 induces synaptic impairment at early stage and prevents amyloidosis at late stage in APP/PS1 mice. FASEB J. 33, 10425–10442 (2019).
Hornik, T. C., Neniskyte, U. & Brown, G. C. Inflammation induces multinucleation of microglia via PKC inhibition of cytokinesis, generating highly phagocytic multinucleated giant cells. J. Neurochem. 128, 650–661 (2014).
Neniskyte, U. & Brown, G. C. Lactadherin/MFG-E8 is essential for microglia-mediated neuronal loss and phagoptosis induced by amyloid beta. J. Neurochem. 126, 312–317 (2013).
Sokolova, D. et al. Astrocyte-derived MFG-E8 facilitates microglial synapse elimination in Alzheimer’s disease mouse models. Preprint at bioRxiv https://doi.org/10.1101/2024.08.31.606944 (2024).
Condic, M. et al. N-truncation and pyroglutaminylation enhances the opsonizing capacity of Aβ-peptides and facilitates phagocytosis by macrophages and microglia. Brain Behav. Immun. 41, 116–125 (2014).
Schulz, B. et al. Beta-amyloid (Aβ40, Aβ42) binding to modified LDL accelerates macrophage foam cell formation. Biochim. Biophys. Acta 1771, 1335–1344 (2007).
Velasco, P. T. et al. Synapse-binding subpopulations of Aβ oligomers sensitive to peptide assembly blockers and scFv antibodies. ACS Chem. Neurosci. 3, 972–981 (2012).
Simakova, O. & Arispe, N. J. The cell-selective neurotoxicity of the Alzheimer’s Aβ peptide is determined by surface phosphatidylserine and cytosolic ATP levels. Membrane binding is required for Aβ toxicity. J. Neurosci. 27, 13719–13729 (2007).
Falkon, K. F. et al. Microglia internalize tau monomers and fibrils using distinct receptors but similar mechanisms. Alzheimers Dement. 21, e14418 (2025).
Chidambaram, H., Das, R. & Chinnathambi, S. G-protein coupled purinergic P2Y12 receptor interacts and internalizes tau(RD)-mediated by membrane-associated actin cytoskeleton remodeling in microglia. Eur. J. Cell Biol. 101, 151201 (2022).
Chidambaram, H., Desale, S. E., Qureshi, T. & Chinnathambi, S. Microglial uptake of extracellular tau by actin-mediated phagocytosis. Methods Mol. Biol. 2761, 231–243 (2024).
Hopp, S. C. et al. The role of microglia in processing and spreading of bioactive tau seeds in Alzheimer’s disease. J. Neuroinflamm. 15, 269 (2018).
Jankeviciute, S. et al. Cerebrospinal fluids from Alzheimer’s disease patients exhibit neurotoxic effects on neuronal cell cultures. Eur. J. Neurosci. 50, 1994–2006 (2019).
Pampuscenko, K. et al. Distinct neurotoxic effects of extracellular tau species in primary neuronal-glial cultures. Mol. Neurobiol. 58, 658–667 (2021).
Bolos, M. et al. Direct evidence of internalization of tau by microglia in vitro and in vivo. J. Alzheimers Dis. 50, 77–87 (2016).
Luo, W. et al. Microglial internalization and degradation of pathological tau is enhanced by an anti-tau monoclonal antibody. Sci. Rep. 5, 11161 (2015).
Andersson, C. R. et al. Antibody-mediated clearance of tau in primary mouse microglial cultures requires Fcgamma-receptor binding and functional lysosomes. Sci. Rep. 9, 4658 (2019).
Thomas, A. L., Lehn, M. A., Janssen, E. M., Hildeman, D. A. & Chougnet, C. A. Naturally-aged microglia exhibit phagocytic dysfunction accompanied by gene expression changes reflective of underlying neurologic disease. Sci. Rep. 12, 19471 (2022).
Pluvinage, J. V. et al. CD22 blockade restores homeostatic microglial phagocytosis in ageing brains. Nature 568, 187–192 (2019).
Roy, N. et al. Elevated expression of the retrotransposon LINE-1 drives Alzheimer’s disease-associated microglial dysfunction. Acta Neuropathol. 148, 75 (2024).
Ritzel, R. M. et al. Age- and location-related changes in microglial function. Neurobiol. Aging 36, 2153–2163 (2015).
Millet, A., Ledo, J. H. & Tavazoie, S. F. An exhausted-like microglial population accumulates in aged and APOE4 genotype Alzheimer’s brains. Immunity 57, 153–170.e6 (2024).
Yanguas-Casas, N., Crespo-Castrillo, A., Arevalo, M. A. & Garcia-Segura, L. M. Aging and sex: impact on microglia phagocytosis. Aging Cell 19, e13182 (2020).
Shi, Q. et al. Complement C3-deficient mice fail to display age-related hippocampal decline. J. Neurosci. 35, 13029–13042 (2015).
Linnartz-Gerlach, B. et al. TREM2 triggers microglial density and age-related neuronal loss. Glia 67, 539–550 (2019).
Brown, G. C. The endotoxin hypothesis of neurodegeneration. J. Neuroinflamm. 16, 180 (2019).
Brown, G. C. & Heneka, M. T. The endotoxin hypothesis of Alzheimer’s disease. Mol. Neurodegener. 19, 30 (2024).
Bellenguez, C. et al. New insights into the genetic etiology of Alzheimer’s disease and related dementias. Nat. Genet. 54, 412–436 (2022).
Podlesny-Drabiniok, A., Marcora, E. & Goate, A. M. Microglial phagocytosis: a disease-associated process emerging from Alzheimer’s disease genetics. Trends Neurosci. 43, 965–979 (2020).
Saunders, A. M. et al. Association of apolipoprotein E allele epsilon 4 with late-onset familial and sporadic Alzheimer’s disease. Neurology 43, 1467–1472 (1993).
Atagi, Y. et al. Apolipoprotein E is a ligand for triggering receptor expressed on myeloid cells 2 (TREM2). J. Biol. Chem. 290, 26043–26050 (2015).
Fitz, N. F. et al. Phospholipids of APOE lipoproteins activate microglia in an isoform-specific manner in preclinical models of Alzheimer’s disease. Nat. Commun. 12, 3416 (2021).
Lin, Y. T. et al. APOE4 causes widespread molecular and cellular alterations associated with Alzheimer’s disease phenotypes in human iPSC-derived brain cell types. Neuron 98, 1141–1154.e7 (2018).
Verghese, P. B. et al. ApoE influences amyloid-beta (Aβ) clearance despite minimal apoE/Aβ association in physiological conditions. Proc. Natl Acad. Sci. USA 110, E1807–E1816 (2013).
Yin, C. et al. ApoE attenuates unresolvable inflammation by complex formation with activated C1q. Nat. Med. 25, 496–506 (2019).
Chung, W. S. et al. Novel allele-dependent role for APOE in controlling the rate of synapse pruning by astrocytes. Proc. Natl Acad. Sci. USA 113, 10186–10191 (2016).
Narayan, P. et al. The extracellular chaperone clusterin sequesters oligomeric forms of the amyloid-β1-40 peptide. Nat. Struct. Mol. Biol. 19, 79–83 (2011).
Narayan, P. et al. Amyloid-beta oligomers are sequestered by both intracellular and extracellular chaperones. Biochemistry 51, 9270–9276 (2012).
Howlett, D. R., Hortobagyi, T. & Francis, P. T. Clusterin associates specifically with Aβ40 in Alzheimer’s disease brain tissue. Brain Pathol. 23, 623–632 (2013).
Bartl, M. M., Luckenbach, T., Bergner, O., Ullrich, O. & Koch-Brandt, C. Multiple receptors mediate apoJ-dependent clearance of cellular debris into nonprofessional phagocytes. Exp. Cell Res. 271, 130–141 (2001).
Cunin, P. et al. Clusterin facilitates apoptotic cell clearance and prevents apoptotic cell-induced autoimmune responses. Cell Death Dis. 7, e2215 (2016).
Kounnas, M. Z. et al. Identification of glycoprotein 330 as an endocytic receptor for apolipoprotein J/clusterin. J. Biol. Chem. 270, 13070–13075 (1995).
Colonna, M. The biology of TREM receptors. Nat. Rev. Immunol. 23, 580–594 (2023).
Kober, D. L. et al. Functional insights from biophysical study of TREM2 interactions with apoE and Aβ1-42. Alzheimers Dement. 17, 475–488 (2021).
Jendresen, C., Arskog, V., Daws, M. R. & Nilsson, L. N. The Alzheimer’s disease risk factors apolipoprotein E and TREM2 are linked in a receptor signaling pathway. J. Neuroinflamm. 14, 59 (2017).
Vilalta, A. et al. Wild-type sTREM2 blocks Aβ aggregation and neurotoxicity, but the Alzheimer’s R47H mutant increases Aβ aggregation. J. Biol. Chem. 296, 100631 (2021).
Boza-Serrano, A. et al. Galectin-3, a novel endogenous TREM2 ligand, detrimentally regulates inflammatory response in Alzheimer’s disease. Acta Neuropathol. 138, 251–273 (2019).
Sudom, A. et al. Molecular basis for the loss-of-function effects of the Alzheimer’s disease-associated R47H variant of the immune receptor TREM2. J. Biol. Chem. 293, 12634–12646 (2018).
Swain, P. S., Panda, S., Pati, S. & Dehury, B. Computational saturation mutagenesis to explore the effect of pathogenic mutations on extra-cellular domains of TREM2 associated with Alzheimer’s and Nasu-Hakola disease. J. Mol. Model. 29, 360 (2023).
Tran, K. M. et al. A Trem2R47H mouse model without cryptic splicing drives age- and disease-dependent tissue damage and synaptic loss in response to plaques. Mol. Neurodegener. 18, 12 (2023).
Bemiller, S. M. et al. TREM2 deficiency exacerbates tau pathology through dysregulated kinase signaling in a mouse model of tauopathy. Mol. Neurodegener. 12, 74 (2017).
Leyns, C. E. G. et al. TREM2 function impedes tau seeding in neuritic plaques. Nat. Neurosci. 22, 1217–1222 (2019).
Lee, S. H. et al. Trem2 restrains the enhancement of tau accumulation and neurodegeneration by β-amyloid pathology. Neuron 109, 1283–1301.e6 (2021).
Lee, C. Y. D. et al. Elevated TREM2 gene dosage reprograms microglia responsivity and ameliorates pathological phenotypes in Alzheimer’s disease models. Neuron 97, 1032–1048.e5 (2018).
Brown, G. C. & St George-Hyslop, P. Does soluble TREM2 protect against Alzheimer’s disease? Front. Aging Neurosci. 13, 834697 (2021).
Cheng-Hathaway, P. J. et al. The Trem2 R47H variant confers loss-of-function-like phenotypes in Alzheimer’s disease. Mol. Neurodegener. 13, 29 (2018).
Korvatska, O. et al. Triggering receptor expressed on myeloid cell 2 R47H exacerbates immune response in Alzheimer’s disease brain. Front. Immunol. 11, 559342 (2020).
Popescu, A. S. et al. Alzheimer’s disease-associated R47H TREM2 increases, but wild-type TREM2 decreases, microglial phagocytosis of synaptosomes and neuronal loss. Glia 71, 974–990 (2023).
Penney, J. et al. iPSC-derived microglia carrying the TREM2 R47H/+ mutation are proinflammatory and promote synapse loss. Glia 72, 452–469 (2024).
Ramakrishnan, G. S. et al. SHIP inhibition mediates select TREM2-induced microglial functions. Mol. Immunol. 170, 35–45 (2024).
Sayed, F. A. et al. AD-linked R47H-TREM2 mutation induces disease-enhancing microglial states via AKT hyperactivation. Sci. Transl. Med. 13, eabe3947 (2021).
Boudesco, C. et al. Novel potent liposome agonists of triggering receptor expressed on myeloid cells 2 phenocopy antibody treatment in cells. Glia 70, 2290–2308 (2022).
Luis, E. O. et al. Frontobasal gray matter loss is associated with the TREM2 p.R47H variant. Neurobiol. Aging 35, 2681–2690 (2014).
Cousins, O. et al. Microglial activation, tau and amyloid deposition in TREM2 p.R47H carriers and mild cognitive impairment patients: a multi-modal/multi-tracer PET/MRI imaging study with influenza vaccine immune challenge. J. Neuroinflamm. 20, 272 (2023).
Jain, N., Lewis, C. A., Ulrich, J. D. & Holtzman, D. M. Chronic TREM2 activation exacerbates Aβ-associated tau seeding and spreading. J. Exp. Med. 220, e20220654 (2023).
Tsai, A. P. et al. PLCG2 is associated with the inflammatory response and is induced by amyloid plaques in Alzheimer’s disease. Genome Med. 14, 17 (2022).
Andreone, B. J. et al. Alzheimer’s-associated PLCgamma2 is a signaling node required for both TREM2 function and the inflammatory response in human microglia. Nat. Neurosci. 23, 927–938 (2020).
Sims, R. et al. Rare coding variants in PLCG2, ABI3, and TREM2 implicate microglial-mediated innate immunity in Alzheimer’s disease. Nat. Genet. 49, 1373–1384 (2017).
Kleineidam, L. et al. PLCG2 protective variant p.P522R modulates tau pathology and disease progression in patients with mild cognitive impairment. Acta Neuropathol. 139, 1025–1044 (2020).
Bunney, T. D., Kampyli, C., Gregory, A. & Katan, M. Characterisation of molecular mechanisms for PLCgamma2 disease-linked variants. Adv. Biol. Regul. 94, 101053 (2024).
Tsai, A. P. et al. Genetic variants of phospholipase C-gamma2 alter the phenotype and function of microglia and confer differential risk for Alzheimer’s disease. Immunity 56, 2121–2136.e6 (2023).
Bradshaw, E. M. et al. CD33 Alzheimer’s disease locus: altered monocyte function and amyloid biology. Nat. Neurosci. 16, 848–850 (2013).
Butler, C. A., Thornton, P. & Brown, G. C. CD33M inhibits microglial phagocytosis, migration and proliferation, but the Alzheimer’s disease-protective variant CD33m stimulates phagocytosis and proliferation, and inhibits adhesion. J. Neurochem. 158, 297–310 (2021).
Eskandari-Sedighi, G. et al. Alzheimer’s disease associated isoforms of human CD33 distinctively modulate microglial cell responses in 5XFAD mice. Mol. Neurodegener. 19, 42 (2024).
Chan, G. et al. CD33 modulates TREM2: convergence of Alzheimer loci. Nat. Neurosci. 18, 1556–1558 (2015).
Haure-Mirande, J. V., Audrain, M., Ehrlich, M. E. & Gandy, S. Microglial TYROBP/DAP12 in Alzheimer’s disease: Transduction of physiological and pathological signals across TREM2. Mol. Neurodegener. 17, 55 (2022).
Puntambekar, S. S. et al. CX3CR1 deficiency aggravates amyloid driven neuronal pathology and cognitive decline in Alzheimer’s disease. Mol. Neurodegener. 17, 47 (2022).
Gomez-Arboledas, A. et al. C5aR1 antagonism alters microglial polarization and mitigates disease progression in a mouse model of Alzheimer’s disease. Acta Neuropathol. Commun. 10, 116 (2022).
Zhao, Y. et al. TREM2 is a receptor for β-amyloid that mediates microglial function. Neuron 97, 1023–1031.e7 (2018).
Paidassi, H. et al. C1q binds phosphatidylserine and likely acts as a multiligand-bridging molecule in apoptotic cell recognition. J. Immunol. 180, 2329–2338 (2008).
Nomura, K., Vilalta, A., Allendorf, D. H., Hornik, T. C. & Brown, G. C. Activated microglia desialylate and phagocytose cells via neuraminidase, galectin-3, and mer tyrosine kinase. J. Immunol. 198, 4792–4801 (2017).
Allendorf, D. H., Puigdellivol, M. & Brown, G. C. Activated microglia desialylate their surface, stimulating complement receptor 3-mediated phagocytosis of neurons. Glia 68, 989–998 (2020).
Allendorf, D. H. & Brown, G. C. Neu1 is released from activated microglia, stimulating microglial phagocytosis and sensitizing neurons to glutamate. Front. Cell Neurosci. 16, 917884 (2022).
Klaus, C., Liao, H., Allendorf, D. H., Brown, G. C. & Neumann, H. Sialylation acts as a checkpoint for innate immune responses in the central nervous system. Glia 69, 1619–1636 (2021).
Fricker, M., Oliva-Martin, M. J. & Brown, G. C. Primary phagocytosis of viable neurons by microglia activated with LPS or Aβ is dependent on calreticulin/LRP phagocytic signalling. J. Neuroinflamm. 9, 196 (2012).
Dundee, J. M. & Brown, G. C. The microglial P2Y(6) receptor as a therapeutic target for neurodegenerative diseases. Transl. Neurodegener. 13, 47 (2024).
Tacnet-Delorme, P., Chevallier, S. & Arlaud, G. J. Beta-amyloid fibrils activate the C1 complex of complement under physiological conditions: evidence for a binding site for A beta on the C1q globular regions. J. Immunol. 167, 6374–6381 (2001).
Hamon, Y. et al. ABC1 promotes engulfment of apoptotic cells and transbilayer redistribution of phosphatidylserine. Nat. Cell Biol. 2, 399–406 (2000).
Hamon, Y. et al. Cooperation between engulfment receptors: the case of ABCA1 and MEGF10. PLoS ONE 1, e120 (2006).
Karasinska, J. M. et al. ABCA1 influences neuroinflammation and neuronal death. Neurobiol. Dis. 54, 445–455 (2013).
Morizawa, Y. M. et al. Author correction: reactive astrocytes function as phagocytes after brain ischemia via ABCA1-mediated pathway. Nat. Commun. 8, 1598 (2017).
Jehle, A. W. et al. ATP-binding cassette transporter A7 enhances phagocytosis of apoptotic cells and associated ERK signaling in macrophages. J. Cell Biol. 174, 547–556 (2006).
Fu, Y., Hsiao, J. H., Paxinos, G., Halliday, G. M. & Kim, W. S. ABCA7 mediates phagocytic clearance of amyloid-beta in the brain. J. Alzheimers Dis. 54, 569–584 (2016).
Butler, C. A. et al. The Abca7V1613M variant reduces Aβ generation, plaque load, and neuronal damage. Alzheimers Dement. 20, 4914–4934 (2024).
Karahan, H. et al. Deletion of Abi3 gene locus exacerbates neuropathological features of Alzheimer’s disease in a mouse model of Aβ amyloidosis. Sci. Adv. 7, eabe3954 (2021).
Gomez, A. R. et al. Boosting angiotensin-converting enzyme (ACE) in microglia protects against Alzheimer’s disease in 5xFAD mice. Nat. Aging 5, 1280–1294 (2025).
Sommer, D. et al. ADAM17-deficiency on microglia but not on macrophages promotes phagocytosis and functional recovery after spinal cord injury. Brain Behav. Immun. 80, 129–145 (2019).
Zheng, Y. et al. The autism spectrum disorder-associated bacterial metabolite p-cresol derails the neuroimmune response of microglial cells partially via reduction of ADAM17 and ADAM10. Int. J. Mol. Sci. 23, 11013 (2022).
Machlovi, S. I. et al. APOE4 confers transcriptomic and functional alterations to primary mouse microglia. Neurobiol. Dis. 164, 105615 (2022).
Ma, C. et al. Therapeutic modulation of APP–CD74 axis can activate phagocytosis of TAMs in GBM. Biochim. Biophys. Acta Mol. Basis Dis. 1870, 167449 (2024).
Nott, A. et al. Brain cell type-specific enhancer–promoter interactome maps and disease-risk association. Science 366, 1134–1139 (2019).
Sudwarts, A. et al. BIN1 is a key regulator of proinflammatory and neurodegeneration-related activation in microglia. Mol. Neurodegener. 17, 33 (2022).
Bonilla, F. A., Fujita, R. M., Pivniouk, V. I., Chan, A. C. & Geha, R. S. Adapter proteins SLP-76 and BLNK both are expressed by murine macrophages and are linked to signaling via Fcgamma receptors I and II/III. Proc. Natl Acad. Sci. USA 97, 1725–1730 (2000).
Zhang, L. et al. Microglial CD2AP deficiency exerts protection in an Alzheimer’s disease model of amyloidosis. Mol. Neurodegener. 19, 95 (2024).
Newman, S. L., Devery-Pocius, J. E., Ross, G. D. & Henson, P. M. Phagocytosis by human monocyte-derived macrophages. Independent function of receptors for C3b (CR1) and iC3b (CR3). Complement 1, 213–227 (1984).
Crehan, H., Hardy, J. & Pocock, J. Blockage of CR1 prevents activation of rodent microglia. Neurobiol. Dis. 54, 139–149 (2013).
Daskoulidou, N. et al. Complement receptor 1 is expressed on brain cells and in the human brain. Glia 71, 1522–1535 (2023).
Jiang, M. et al. Cathepsin B modulates microglial migration and phagocytosis of amyloid β in Alzheimer’s disease through PI3K-Akt signaling. Neuropsychopharmacology 50, 640–650 (2025).
Li, Y. et al. Functional genomics identify causal variant underlying the protective CTSH locus for Alzheimer’s disease. Neuropsychopharmacology 48, 1555–1566 (2023).
Wang, Y. Y. et al. Loss of microglial EED impairs synapse density, learning, and memory. Mol. Psychiatry 27, 2999–3009 (2022).
Gockley, J. et al. Multi-tissue neocortical transcriptome-wide association study implicates 8 genes across 6 genomic loci in Alzheimer’s disease. Genome Med. 13, 76 (2021).
Guan, Z., Chen, Z., Fu, S., Dai, L. & Shen, Y. Progranulin administration attenuates beta-amyloid deposition in the hippocampus of 5xFAD mice through modulating BACE1 expression and microglial phagocytosis. Front. Cell Neurosci. 14, 260 (2020).
Minami, S. S. et al. Progranulin protects against amyloid beta deposition and toxicity in Alzheimer’s disease mouse models. Nat. Med. 20, 1157–1164 (2014).
Takahashi, H. et al. Opposing effects of progranulin deficiency on amyloid and tau pathologies via microglial TYROBP network. Acta Neuropathol. 133, 785–807 (2017).
Uesaka, N. et al. Retrograde signaling from progranulin to sort1 counteracts synapse elimination in the developing cerebellum. Neuron 97, 796–805.e5 (2018).
Lui, H. et al. Progranulin deficiency promotes circuit-specific synaptic pruning by microglia via complement activation. Cell 165, 921–935 (2016).
Kimura, K. et al. Immune checkpoint TIM-3 regulates microglia and Alzheimer’s disease. Nature 641, 718–731 (2025).
Viana, G. M., Buri, M. V., Paredes-Gamero, E. J., Martins, A. M. & D’Almeida, V. Impaired hematopoiesis and disrupted monocyte/macrophage homeostasis in mucopolysaccharidosis type i mice. J. Cell Physiol. 231, 698–707 (2016).
Devlin, B. A. et al. Excitatory-neuron-derived interleukin-34 supports cortical developmental microglia function. Immunity 58, 1948–1965.e6 (2025).
Samuels, J. D. et al. The Alzheimer’s disease risk factor INPP5D restricts neuroprotective microglial responses in amyloid beta-mediated pathology. Alzheimers Dement. 19, 4908–4921 (2023).
Pils, S. et al. The adaptor molecule Nck localizes the WAVE complex to promote actin polymerization during CEACAM3-mediated phagocytosis of bacteria. PLoS ONE 7, e32808 (2012).
Zhao, P. et al. LILRB2-mediated TREM2 signaling inhibition suppresses microglia functions. Mol. Neurodegener. 17, 44 (2022).
Iyer, A. K. et al. Cell autonomous microglia defects in a stem cell model of frontotemporal dementia tau. Mol. Psychiatry 30, 4776–4791 (2025).
Kozlova, A. et al. PICALM Alzheimer’s risk allele causes aberrant lipid droplets in microglia. Nature 646, 1178–1186 (2025).
Rathore, N. et al. Paired immunoglobulin-like type 2 receptor alpha G78R variant alters ligand binding and confers protection to Alzheimer’s disease. PLoS Genet. 14, e1007427 (2018).
Miller, J. B. et al. Ramp sequence may explain synonymous variant association with Alzheimer’s disease in the paired immunoglobulin-like type 2 receptor alpha (PILRA). Biomedicines 13, 739 (2025).
Sherrington, R. et al. Cloning of a gene bearing missense mutations in early-onset familial Alzheimer’s disease. Nature 375, 754–760 (1995).
Ledo, J. H. et al. Presenilin 1 phosphorylation regulates amyloid-beta degradation by microglia. Mol. Psychiatry 26, 5620–5635 (2021).
Lee, J. W. et al. Enhanced phagocytosis associated with multinucleated microglia via Pyk2 inhibition in an acute beta-amyloid infusion model. J. Neuroinflamm. 21, 196 (2024).
Krishnan, D., Menon, R. N., Mathuranath, P. S. & Gopala, S. A novel role for SHARPIN in amyloid-beta phagocytosis and inflammation by peripheral blood-derived macrophages in Alzheimer’s disease. Neurobiol. Aging 93, 131–141 (2020).
Wang, Y. & Neumann, H. Alleviation of neurotoxicity by microglial human siglec-11. J. Neurosci. 30, 3482–3488 (2010).
Mishra, S. et al. The Alzheimer’s disease gene SORL1 regulates lysosome function in human microglia. Glia 73, 1329–1348 (2025).
Liu, T. et al. Multi-omic comparison of Alzheimer’s variants in human ESC-derived microglia reveals convergence at APOE. J. Exp. Med. 217, e20200474 (2020).
Martin, S., Vincent, J. P. & Mazella, J. Involvement of the neurotensin receptor-3 in the neurotensin-induced migration of human microglia. J. Neurosci. 23, 1198–1205 (2003).
Pimenova, A. A. et al. Alzheimer’s-associated PU.1 expression levels regulate microglial inflammatory response. Neurobiol. Dis. 148, 105217 (2021).
Zhang, T. et al. TMEM106B regulates microglial proliferation and survival in response to demyelination. Sci. Adv. 9, eadd2676 (2023).
Feng, T. et al. TMEM106B deficiency impairs cerebellar myelination and synaptic integrity with Purkinje cell loss. Acta Neuropathol. Commun. 10, 33 (2022).
Davis, L. C., Morgan, A. J. & Galione, A. NAADP-regulated two-pore channels drive phagocytosis through endo-lysosomal Ca2+ nanodomains, calcineurin and dynamin. EMBO J. 39, e104058 (2020).
Zhang, S. et al. Single-cell polygenic risk scores dissect cellular and molecular heterogeneity of complex human diseases. Nat. Biotechnol. https://doi.org/10.1038/s41587-025-02725-6 (2025).
Yoo, Y., Neumayer, G., Shibuya, Y., Mader, M. M. & Wernig, M. A cell therapy approach to restore microglial Trem2 function in a mouse model of Alzheimer’s disease. Cell Stem Cell 30, 1043–1053.e6 (2023).
Chadarevian, J. P. et al. Harnessing human iPSC-microglia for CNS-wide delivery of disease-modifying proteins. Cell Stem Cell 32, 914–934.e8 (2025).
Acknowledgements
P.St.G.-H. is supported by grants from the Canadian Institutes of Health Research (406915 Foundation Grant and Canadian Consortium on Neurodegeneration in Aging Grant), National Institute of Aging (grant nos. U01AG072572 and R01AG070864 to P.St.G.-H.). G.L. was supported by a grant from the National Institute of Aging (grant no. RF1AG060748). G.C.B. was funded by the Medical Research Council UK (grant no. MR/L010593), Alzheimer’s Research UK (Dementia Consortium Grant ARUK-DC2017-4, Network Grant G-102212) and the Wellcome Trust (Wellcome Institutional Partnership Award 222062/Z/20/Z). R.C.P. is supported by the Dementia Research Synapsis Foundation, by grants from the Swiss National Science Foundation (grant no. 310030_197940), the European Union Joint Programme — Neurodegenerative Disease Research (grant no. 31ND3O-2L3462), the European Research Area Network (grant no. 31NE30_219096) and funding from the University of Lausanne.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Neurology thanks Subashchandrabose Chinnathambi, Renzo Mancuso and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Brown, G.C., St George-Hyslop, P., Paolicelli, R.C. et al. Microglial phagocytosis in Alzheimer disease. Nat Rev Neurol 22, 54–69 (2026). https://doi.org/10.1038/s41582-025-01162-y
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41582-025-01162-y