Abstract
Endothelial cells adopt tissue-specific characteristics to instruct organ development and regeneration1,2. This adaptability is lost in cultured adult endothelial cells, which do not vascularize tissues in an organotypic manner. Here, we show that transient reactivation of the embryonic-restricted ETS variant transcription factor 2 (ETV2)3 in mature human endothelial cells cultured in a serum-free three-dimensional matrix composed of a mixture of laminin, entactin and type-IV collagen (LEC matrix) ‘resets’ these endothelial cells to adaptable, vasculogenic cells, which form perfusable and plastic vascular plexi. Through chromatin remodelling, ETV2 induces tubulogenic pathways, including the activation of RAP1, which promotes the formation of durable lumens4,5. In three-dimensional matrices—which do not have the constraints of bioprinted scaffolds—the ‘reset’ vascular endothelial cells (R-VECs) self-assemble into stable, multilayered and branching vascular networks within scalable microfluidic chambers, which are capable of transporting human blood. In vivo, R-VECs implanted subcutaneously in mice self-organize into durable pericyte-coated vessels that functionally anastomose to the host circulation and exhibit long-lasting patterning, with no evidence of malformations or angiomas. R-VECs directly interact with cells within three-dimensional co-cultured organoids, removing the need for the restrictive synthetic semipermeable membranes that are required for organ-on-chip systems, therefore providing a physiological platform for vascularization, which we call ‘Organ-On-VascularNet’. R-VECs enable perfusion of glucose-responsive insulin-secreting human pancreatic islets, vascularize decellularized rat intestines and arborize healthy or cancerous human colon organoids. Using single-cell RNA sequencing and epigenetic profiling, we demonstrate that R-VECs establish an adaptive vascular niche that differentially adjusts and conforms to organoids and tumoroids in a tissue-specific manner. Our Organ-On-VascularNet model will permit metabolic, immunological and physiochemical studies and screens to decipher the crosstalk between organotypic endothelial cells and parenchymal cells for identification of determinants of endothelial cell heterogeneity, and could lead to advances in therapeutic organ repair and tumour targeting.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Source ChIP–seq data are provided in Supplementary Table 1 and source scRNA-seq data are provided in Supplementary Table 2. The RNA-seq data can be viewed at the Gene Expression Omnibus (GEO) under accession number GSE131039. The ChIP–seq data and scRNA-seq data can be viewed at the GEO under accession numbers GSE147746 and GSE148996, respectively. Source data are provided with this paper.
Code availability
All of the code used in this paper is available from the authors on request.
References
Augustin, H. G. & Koh, G. Y. Organotypic vasculature: from descriptive heterogeneity to functional pathophysiology. Science 357, eaal2379 (2017).
Rafii, S., Butler, J. M. & Ding, B. S. Angiocrine functions of organ-specific endothelial cells. Nature 529, 316–325 (2016).
Lee, D. et al. ER71 acts downstream of BMP, Notch, and Wnt signaling in blood and vessel progenitor specification. Cell Stem Cell 2, 497–507 (2008).
Barry, D. M. et al. Rasip1-mediated Rho GTPase signaling regulates blood vessel tubulogenesis via nonmuscle myosin II. Circ. Res. 119, 810–826 (2016).
Strilić, B. et al. The molecular basis of vascular lumen formation in the developing mouse aorta. Dev. Cell 17, 505–515 (2009).
Carmeliet, P. & Jain, R. K. Molecular mechanisms and clinical applications of angiogenesis. Nature 473, 298–307 (2011).
Cao, Z. et al. Molecular checkpoint decisions made by subverted vascular niche transform indolent tumor cells into chemoresistant cancer stem cells. Cancer Cell 31, 110–126 (2017).
Nolan, D. J. et al. Molecular signatures of tissue-specific microvascular endothelial cell heterogeneity in organ maintenance and regeneration. Dev. Cell 26, 204–219 (2013).
Pellegata, A. F., Tedeschi, A. M. & De Coppi, P. Whole organ tissue vascularization: engineering the tree to develop the fruits. Front. Bioeng. Biotechnol. 6, 56 (2018).
Giobbe, G. G. et al. Extracellular matrix hydrogel derived from decellularized tissues enables endodermal organoid culture. Nat. Commun. 10, 5658 (2019).
Ronaldson-Bouchard, K. & Vunjak-Novakovic, G. Organs-on-a-chip: a fast track for engineered human tissues in drug development. Cell Stem Cell 22, 310–324 (2018).
Bhatia, S. N. & Ingber, D. E. Microfluidic organs-on-chips. Nat. Biotechnol. 32, 760–772 (2014).
Lancaster, M. A. & Knoblich, J. A. Organogenesis in a dish: modeling development and disease using organoid technologies. Science 345, 1247125 (2014).
Tuveson, D. & Clevers, H. Cancer modeling meets human organoid technology. Science 364, 952–955 (2019).
Koyano-Nakagawa, N. & Garry, D. J. Etv2 as an essential regulator of mesodermal lineage development. Cardiovasc. Res. 113, 1294–1306 (2017).
Ginsberg, M. et al. Efficient direct reprogramming of mature amniotic cells into endothelial cells by ETS factors and TGFβ suppression. Cell 151, 559–575 (2012).
Nguyen, D. H. et al. Biomimetic model to reconstitute angiogenic sprouting morphogenesis in vitro. Proc. Natl Acad. Sci. USA 110, 6712–6717 (2013).
Eberhard, D., Kragl, M. & Lammert, E. ‘Giving and taking’: endothelial and beta-cells in the islets of Langerhans. Trends Endocrinol. Metab. 21, 457–463 (2010).
Sato, T. et al. Long-term expansion of epithelial organoids from human colon, adenoma, adenocarcinoma, and Barrett’s epithelium. Gastroenterology 141, 1762–1772 (2011).
Miyoshi, H. & Stappenbeck, T. S. In vitro expansion and genetic modification of gastrointestinal stem cells in spheroid culture. Nat. Protocols 8, 2471–2482 (2013).
Stan, R. V. et al. The diaphragms of fenestrated endothelia: gatekeepers of vascular permeability and blood composition. Dev. Cell 23, 1203–1218 (2012).
Lyden, D. et al. Id1 and Id3 are required for neurogenesis, angiogenesis and vascularization of tumour xenografts. Nature 401, 670–677 (1999).
Li, S. et al. Plasma mesothelin as a novel diagnostic and prognostic biomarker in colorectal cancer. J. Cancer 8, 1355–1361 (2017).
Si, M. & Lang, J. The roles of metallothioneins in carcinogenesis. J. Hematol. Oncol. 11, 107 (2018).
Baudin, B., Bruneel, A., Bosselut, N. & Vaubourdolle, M. A protocol for isolation and culture of human umbilical vein endothelial cells. Nat. Protocols 2, 481–485 (2007).
Seandel, M. et al. Generation of a functional and durable vascular niche by the adenoviral E4ORF1 gene. Proc. Natl Acad. Sci. USA 105, 19288–19293 (2008).
Ginsberg, M., Schachterle, W., Shido, K. & Rafii, S. Direct conversion of human amniotic cells into endothelial cells without transitioning through a pluripotent state. Nat. Protocols 10, 1975–1985 (2015).
Schachterle, W. et al. Sox17 drives functional engraftment of endothelium converted from non-vascular cells. Nat. Commun. 8, 13963 (2017).
Wareing, S., Eliades, A., Lacaud, G. & Kouskoff, V. ETV2 expression marks blood and endothelium precursors, including hemogenic endothelium, at the onset of blood development. Dev. Dyn. 241, 1454–1464 (2012).
Zudaire, E., Gambardella, L., Kurcz, C. & Vermeren, S. A computational tool for quantitative analysis of vascular networks. PLoS One 6, e27385 (2011).
Sato, T. et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459, 262–265 (2009).
Sugimoto, S. & Sato, T. Establishment of 3D intestinal organoid cultures from intestinal stem cells. Methods Mol. Biol. 1612, 97–105 (2017).
Dame, M. K. et al. Identification, isolation and characterization of human LGR5-positive colon adenoma cells. Development 145, dev153049 (2018).
Tsai, Y. H. et al. A method for cryogenic preservation of human biopsy specimens and subsequent organoid culture. Cell. Mol. Gastroenterol. Hepatol. 6, 218–222.e7 (2018).
Puca, L. et al. Patient derived organoids to model rare prostate cancer phenotypes. Nat. Commun. 9, 2404 (2018).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Liao, Y., Smyth, G. K. & Shi, W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30, 923–930 (2014).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Ritchie, M. E. et al. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 43, e47 (2015).
Huang, D. W., Sherman, B. T. & Lempicki, R. A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protocols 4, 44–57 (2009).
Liu, Y. et al. Epigenetic profiles signify cell fate plasticity in unipotent spermatogonial stem and progenitor cells. Nat. Commun. 7, 11275 (2016).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Thorvaldsdóttir, H., Robinson, J. T. & Mesirov, J. P. Integrative Genomics Viewer (IGV): high-performance genomics data visualization and exploration. Brief. Bioinform. 14, 178–192 (2013).
Zhang, Y. et al. Model-based analysis of ChIP–seq (MACS). Genome Biol. 9, R137 (2008).
Zang, C. et al. A clustering approach for identification of enriched domains from histone modification ChIP–seq data. Bioinformatics 25, 1952–1958 (2009).
Heinz, S. et al. Simple combinations of lineage-determining transcription factors prime cis-regulatory elements required for macrophage and B cell identities. Mol. Cell 38, 576–589 (2010).
Butler, A., Hoffman, P., Smibert, P., Papalexi, E. & Satija, R. Integrating single-cell transcriptomic data across different conditions, technologies, and species. Nat. Biotechnol. 36, 411–420 (2018).
Acknowledgements
S.R. is supported by the Ansary Stem Cell Institute; grants from the National Institutes of Health (NIH) (R35 HL150809, R01s DK095039, HL119872, HL128158, HL115128, HL139056, RC2 DK114777 and U01AI138329); the Empire State Stem Cell Board; grants from New York State Stem Cell Science (NYSTEM) (C026878, C028117, C029156, C030160); the Daedalus Fund for Innovation from Weill Cornell Medicine; and the Starr Foundation stem cell core project; and Qatar National Priorities Research Program (NPRP 8-1898-3-392); and the Tri-Institutional Stem Cell Initiatives (TRI-SCI 2013-032, 2014-023, 2016-013 and 2019-029). R.E.S. is supported by NIH grants R01CA234614, 2R01AI107301, R01DK121072,1R21AI117213, RO3DK117252, R01AA027327 and K08DK101754 and Department of Defense CA170574. R.E.S. is a Irma Hirschl Trust Research Award Scholars. R.S. is supported by NYSTEM contract C32596GG; P.D.C. by NIHR Research (NIHR-RP-2014-04-046); P.D.C., A.F.P., A.M.T. and F.S. by the OAK Foundation (W1095/OCAY-14-191), H2020 grant INTENS 668294 and the NIHR Biomedical Research Centre at Great Ormond Street Hospital for Children NHS Foundation Trust; A.M.T. by the BBSRC ICASE studentship 167881; Y. Liu and J.M.G.-S. by a New York Stem Cell Foundation Druckenmiller fellowship; B.K. by a T32 fellowship; O.E. by NIH UL1TR002384, R01CA194547, LLS SCOR 180078-02, 7021-20 and Janssen and Eli Lilly research grants; J.S. by HL119215; and Q.J.Z. by NIH R01DK106253, UC4DK116280, and the Neuroendocrine Tumor Research Foundation grant NTRF192269; J.R.S. by NIH R01HL119215. We thank R. Chavez for technical assistance and animal maintenance, the Visual Function Core at Weill Cornell Medicine for live-imaging resources, J. Shieh for HUVEC isolations, C. Wasserstein, M. Mishaan, M. Bell, C. Cheung and W. Gu for support with organoid cultures and A. Gjinovci for surgical help with the isolation of mouse small bowels.
Author information
Authors and Affiliations
Contributions
B.P. and S.R. conceived the study and wrote the manuscript. B.P., D.-H.T.N., R.S., K.S., R.E.S., S.Y.R. and S.R. discussed and analysed data. R.S. provided microscopy expertise. B.P., D.-H.T.N., G.L., R.S., Y. Liu, F.G., Y. Lin, J.M.G.-S., M.Y., S.Y.R. and S.R. performed experiments and analysed data. A.F.P., A.M.T., F.S. and P.D.C. carried out and analysed experiments on the decellularized intestines. Y. Liu, D.R., P.Z., T.Z., B.K., O.E. and J.Z.X. analysed ChIP–seq, RNA-seq and single-cell sequencing. M.W., T.H., S.L., L.D., J.S. and Q.J.Z. assisted with organoid cultures. All the authors read and provided feedback on the figures and manuscript.
Corresponding author
Ethics declarations
Competing interests
S.R. is the founder and a non-paid consultant to Angiocrine Bioscience. O.E. is a scientific advisor and equity holder in Freenome, Owkin, Volastra Therapeutics and One Three Biotech. R.E.S. is a scientific advisor and member of the SAB for Miromatrix Inc.
Additional information
Peer review information Nature thanks Hans Clevers, Harald Ott and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 ETV2 confers mature human ECs with the ability to autonomously self-assemble into lumenized, durable, branching and patterned vessels in vitro without the constraints of bioprinted scaffolds.
a, Overview of experimental set-up for vessel formation in vitro for screen of different media, extracellular matrix components and tissue-specific ECs. b, The proliferation of GFP-transduced R-VECs and control ECs (CTRL-ECs) at each stage of vessel formation. EdU+ cells were quantified after a 16-hour EdU pulse. c, Time course of vessel formation on Matrigel for GFP+ CTRL-EC and R-VECs over 8 weeks. d, Vessel formation using R-VEC or CTRL-EC in three different enriched pro-angiogenic media (Supplementary Data 2): Serum-free StemSpan with Knockout serum replacement and Cytokines, EGM-2 and complete EC media on Matrigel. R-VEC formed the most robust lumenized vessels in serum-free StemSpan with knockout serum replacement medium and cytokines, as compared to other media with serum. CTRL-EC failed to form durable stable vessels. e, f, Time course (e) and quantification (f) of tube formation for GFP+ human Adipose CTRL-EC and human Adipose R-VEC on Matrigel. g, Representative images of tissue-specific GFP+ R-VEC and CTRL-EC isolated from adult human heart (cardiac EC), aorta (aortic EC) and skin (dermal EC) demonstrated robust and stable vessels at 4 weeks on Matrigel. h, Representative images of GFP+ R-VEC vessels formed on Matrigel or a pre-defined matrix of laminin/entactin and collagen IV (LEC). i, Immunostaining of R-VEC-tubes displayed apicobasal proper polarity with podocalyxin, apical (in red) and laminin, basal (in green). The right image is an orthogonal projection. j, Stiffness measurements by atomic force microscopy (AFM) of adult Adipose and HUVEC ECs with and without ETV2. In both cases, ETV2-transduced ECs are significantly less stiff than their counterparts. The abbreviated box plots indicate the interquartile range and median for each condition. k, HUVECs were transduced with either an empty lentiviral vector or lentiviral vectors with ETV2, myrAKT or ETS1 constructs, and used in a vessel formation assay. Western Blot analysis for expression of ETV2, p-AKT, total AKT and ETS1 in those cells. l, Representative images for ETS1 or myrAKT1 transduced GFP+ HUVECs in a vessel formation assay on Matrigel. m, Quantification of vessel area for ETS1, myrAKT1 and ETV2 (R-VEC) cells indicated that ETS1-EC and myrAKT1-EC fail to form robust vessel formation as compared to R-VEC. Data are mean ± s.e.m. NS, not significant; *P < 0.05, **P < 0.01, ***P < 0.001. For statistics, see Supplementary Data 1. For medium formulations, see Supplementary Data 2.
Extended Data Fig. 2 Transient ETV2 expression in adult human ECs is sufficient for the generation and maintenance of durable long-lasting R-VEC vessels in vitro.
a, Schematic for ETV2 mRNA and protein levels assessment at each of the three stages of R-VEC vessel formation. b, Quantification of ETV2 mRNA levels at each stage of vessel formation. c, d, Western blot analysis (c) and densitometric quantification (d) of ETV2 protein levels at each stage of vessel formation. GAPDH was used as a loading control. e, A proteasome inhibitor (MG132) restored ETV2 levels by ~sixfold when added to R-VECs during the stabilization stage. f, Densitometric quantification of western blots in e. g, qRT–PCR (g) and western blot (h) assessment of ETV2 levels after doxycycline removal. i, Representative images of GFP+ iR-VECs on Matrigel with inducible ETV2 expression at 2 months. ETV2 was turned off at day 0, day 7 and at 4-weeks post start of the remodelling stage 2. j, Quantification of iR-VEC vessels at 2 months. k, Electron microscopy pictures of a lumen present both in vessels in which doxycycline was continuously present and in vessels in which doxycycline was removed after 1 month. Data are mean ± s.e.m. NS, not significant; *P < 0.05, **P < 0.01, ***P < 0.001. For statistics, see Supplementary Data 1. For medium formulations, see Supplementary Data 2.
Extended Data Fig. 3 R-VEC vessels are functionally anastomosed to host vessels and not leaky in vivo.
a, Fluorescently labelled R-VEC or CTRL-EC cells in LEC matrix were subcutaneously injected in the flank of SCID beige mice and retrieved at 2 months. Human-specific VEcad antibody (hVEcad) was injected intravitally right before euthanasia. Sections of the plugs were stained for mouse ECs with an anti-mouse endomucin antibody (mEndomucin), identifying properly organized human R-VECs anastomosing with mouse vessels (thickness = 50 μm). Sections were also stained with the nuclear stain DAPI. b, c, Plugs in a were post-stained with hVEcad and a mouse PDGFRβ antibody (b) or mouse SMA antibody (c) (thickness = 50 μm). d, In vivo plug assay, in which mice were subcutaneously injected with either control ECs (HUVECs transduced only with rtTA lentivirus) or stage 1 doxycycline-inducible-ETV2 ECs (iR-VECs: HUVECs transduced with both rtTA and inducible ETV2 lentivirus) in LEC matrix. One group of mice was on doxycycline (ETV2 continuously on) and another group of mice was on doxycycline food diet for 1 week (ETV2 on) and then switched to regular food (ETV2 off). All mice were euthanized 2 months post-implantation. Red indicates the GFP labelled human ECs, white: Anti-VEcad antibody that was retro-orbitally injected before euthanizing the mice. e, Quantification of vessel area for rtTA only plugs, mice on doxycycline for 1 week, and mice continuously on doxycycline diet (ETV2 on). All mice were euthanized 2 months post-implantation. f, 70 kDa fluorescent dextran (in blue) and human VEcad (in white) were injected in mice implanted with fluorescently labelled R-VECs (in red, 5-months post-implantation), iR-VECs (in red, 1 week on doxycycline food and euthanized at 2 months) or K-RAS-HUVECs (K-RAS-EC) (in red, 2-weeks post-implantation) to assess anastomosis and leakiness of vessels. K-RAS-EC vessels showed dextran leakiness, whereas R-VEC and iR-VEC vessels exhibited patency and non-leakiness. Green arrows point at perfused mouse vessels that were also perfused with dextran. Data are mean ± s.e.m. NS, not significant; *P < 0.05, **P < 0.01, ***P < 0.001. For statistics, see Supplementary Data 1. For medium formulations, see Supplementary Data 2.
Extended Data Fig. 4 Implanted R-VECs form stable, patterned, branching and durable vessels in vivo without features of vascular malformations, cysts, adenomas, haemangiomas or metastasis.
a, Representative images of non-haemorrhagic R-VEC plugs at 10 months. b, Whole-mount microscopy of R-VEC plugs at 10-months post perfusion with anti-human VEcad antibody (hVEcad). c, d, Representative H&E and Masson staining of R-VEC plugs at 10 months (c). There were no features of cysts or haemangiomas present, in contrast to KRAS-EC plugs (at 4 weeks) that formed an EC tumour (d). e, There was no metastasis of R-VECs to other tissues 10 months after plug implantation and the tissues were assessed to be normal without fibrosis and architectural disruption or tumorigenesis as evaluated by H&E, Masson and picrosirius staining.
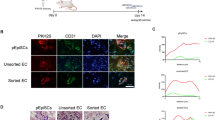
Extended Data Fig. 5 Decellularized intestinal scaffolds re-endothelialized with R-VECs engraft in vivo after omental implantation.
a, Schematic of experimental procedure for heterotopic implantation of decellularized intestinal scaffold vascularized using R-VECs. b, Rat intestines were cannulated through lumen, mesenteric artery and mesenteric vein. c, Decellularized intestine preserves native vasculature (green = GFP+ R-VECs). d, Seeded GFP labelled R-VECs spread evenly and reach distal capillaries. e, Heterotopic implantation of re-endothelialized intestines in immunodeficient mice omentum shows engraftment after 1 and 4 weeks of GFP+ R-VECs and anastomosis to the host vasculature as indicated by intravital intravenous injection of anti-human VEcad antibody (hVEcad). Representative H&E stainings show anatomical normal perfused vessels. f, Quantification of the area covered by R-VEC compared to CTRL-EC in implanted re-endothelialized intestines at 1 week and 4 weeks. g, Quantification of R-VEC and CTRL-EC proliferation and apoptosis in implanted re-endothelialized intestines at 1 and 4 weeks. Data are mean ± s.e.m. NS, not significant; *P < 0.05, **P < 0.01, ***P < 0.001. For statistics, see Supplementary Data 1.
Extended Data Fig. 6 ETV2, by directly binding to promoters and enhancers of target genes, regulates differentially expressed genes in R-VECs.
a, Schematic of RNA-sequencing performed on R-VECs and CTRL-ECs derived from different tissue-specific ECs during stage 1 induction phase (2D monolayers). b, R-VECs or CRTL-ECs were analysed by RNA sequencing. Heat maps of selected genes within top enriched GO categories. Values are log2-normalized CPM, centred and scaled by row. ETV2 binding from ChIP–seq at the promoter of each differentially expressed gene is shown in the yellow-green heat map. c, R-VECs retain essential EC fate genes at stage 1 induction phase across all tissue-specific ECs. The data are presented as log2(CPM) with no scaling by row or column. d, PCA plot based on the top 1,000 most variable genes across ECs with and without ETV2 from different tissues during stage 1 induction, using log2-normalized CPM after subtracting tissue-specific effects using limma’s removeBatchEffect function. e, ETV2 ChIP was performed on R-VECs using an anti-flag antibody at the induction stage 1 (2D), along with histone modification ChIP for H3K4me3 and H3K27ac. Enriched regions were analysed by ChIP–seq. f, Genomic distribution of ETV2 peaks in R-VEC (Stage 1). The number in brackets is the number of ETV2 peaks in each region. g, Promoters bound by ETV2 have an increase in both K4me3 and K27ac. h, GO enrichment in genes with ETV2 binding at promoters. For statistics, see Supplementary Data 1.
Extended Data Fig. 7 ETV2 in R-VECs endows ECs with transcriptional adaptability and plasticity.
a, Diagram of EC sample preparation from ETV2 Venus reporter mice by FACS sorting. ETV2+ and ETV2− ECs were sorted at day E9.5. ECs were sorted as non-haematopoietic CD31+CD45neg cells. b, Heat map of overlap of differentially expressed genes in ETV2+ vs. ETV2− ECs at E9.5 and R-VECs (stage 1) vs. CTRL-EC from different tissues, using tissue-adjusted log2(CPM), centred and scaled by row. c, Knockdown of RASGRP3 by two different shRNAs in R-VECs, shRNA against Luciferase was used as control. Vessel quantification upon RASGRP3 knockdown. d, Heat map displaying overlapping differentially expressed genes from R-VEC at stabilization stage 3 (4 weeks) vs. R-VEC at induction stage 1, R-VECs in vitro pre-plug (stage 1 induction stage) vs. R-VECs in vivo in plugs (1 month), and freshly isolated vs. cultured HUVECs. Values represent tissue-adjusted log2(CPM), centred and scaled by row. e, ChIP–seq depicting genes that are differentially expressed in the stabilization stage 3 phase, but that are already directly bound by ETV2 and epigenetically primed for expression at induction stage 1 (2D monolayers). ETV2 ChIP–sequencing was performed on R-VECs using an anti-flag antibody. Mouse IgG was used as a control for ETV2 ChIP. Histone modification ChIP for H3K4me3, H3K27ac and H3K27me3 was performed on both CTRL-EC and R-VEC at the induction stage 1 (2D monolayers). Enriched regions were analysed by ChIP–sequencing. Black bar, ETV2 enriched regions. Green bar, the region with increased K4me3 modification. Blue bar, the region with increased K27ac modification. Promoter regions bound by ETV2 are highlighted in cream. Track range ETV2/K27me3/K27ac/, 0–0.3; K4me3/input/IgG, 0–1. For statistics, see Supplementary Data 1. For medium formulations, see Supplementary Data 2.
Extended Data Fig. 8 R-VECs physiologically arborize human pancreatic islet explants and organoids.
a, Human islet explants were cultured in Matrigel droplets (volume 50 μl) either with GFP labelled CTRL-EC or R-VEC (day 4). b, Insulin secretion fold change after glucose stimulation at 16.7 mM vs. 2mM glucose (2-week time point). c, Vessel area of ECs directly interacting with islets at week 2. d, EpCAM and VEcad staining of islets co-cultured in a Matrigel droplets at 2 weeks. e, Orthogonal projections of R-VECs in co-culture with human islets at two weeks, demonstrating strong interaction of the sprouting R-VEC vessels with islets. f, Human COs were derived from isolated crypts from colon biopsies of healthy human donors. Colon organoids were confirmed to express proper markers by quantitative RT–PCR. g, Quantitative RT–PCR of various colon markers for human COs, co-cultured with CTRL-EC or co-cultured with R-VEC for 8 days. Epithelial cells were sorted out as live CD31neg non-vascular cells. h, Mouse small intestine organoids were cultured alone, or in the presence of CTRL-EC or R-VEC (day 8). Confocal representative images of EdU+ (proliferating cells), KRT20+ (differentiated epithelial cells in blue) and ECs (mCherry - red) of co-culture experiment with mouse intestinal organoids. i, Quantification of vessel area over the course of 7 days in co-cultures of mouse intestine organoids with CTRL-EC or R-VEC. j, Vessel arborization quantified as EC sprouts in direct contact/organoid in CTRL-EC versus R-VEC wells. k, Time-lapse representative images show the progression of interacting ECs with CRCOs. CTRL-EC (in green) did not interact with CRCOs (in red) (top panel), whereas R-VEC (in green) form robust EC tubes to tap and wrap CRCOs (in red) (bottom panel). l, Orthogonal projections of CRCOs co-cultured with R-VECs (day 8). Data are mean ± s.e.m. NS, not significant; *P < 0.05, **P < 0.01, ***P < 0.001. For statistics, see Supplementary Data 1. For medium formulations, see Supplementary Data 2.
Extended Data Fig. 9 Endothelial and epithelial cell identification by scRNA-seq from co-cultures of normal COs with R-VECs.
a, Schematic of 10X Chromium scRNA-seq experiments of R-VECs alone, R-VECs co-cultured with human COs, or COs alone. Samples were analysed 7 days post co-culture. The same compatible medium was used across all three conditions. b, UMAP of cells from each condition alone and the three conditions merged. c, Endothelial cells were identified as cells expressing either VEcad, CD31 or VEGFR2 and negative for the epithelial marker EPCAM. Epithelial cells were defined as positive for EPCAM and negative for any of the EC markers VEcad, CD31 or VEGFR2. d, UMAP of the 9 unique clusters identified in the merged samples. e, Endothelial and epithelial cell specific markers were used to confirm the EC clusters (clusters 1 to 7) vs. epithelial cell clusters (clusters 8 and 9). f, The identity of epithelial cells in clusters 8 and 9 was confirmed as colon-specific by expression of marker genes including SATB2, CA4, CA2 and others. For statistics, see Supplementary Data 1. For medium formulations, see Supplementary Data 2.
Extended Data Fig. 10 Endothelial and epithelial cell identification by scRNA-seq from co-cultures of CRCOs with R-VECs.
a, Schematic of 10X Chromium scRNA-seq experiments of R-VECs alone, R-VECs co-cultured with human CRCOs or CRCOs alone. Samples were analysed 7 days after co-culture. The same compatible medium was used across all three conditions. b, UMAP of cells from each condition alone and the three conditions merged. c, Endothelial cells were identified as cells expressing either VEcad, CD31 or VEGFR2 and negative for the epithelial marker EPCAM. Epithelial cells were defined as positive for EPCAM and negative for any EC markers VEcad, CD31 or VEGFR2. d, UMAP of the 9 unique clusters identified in the merged samples. e, Endothelial and epithelial cell-specific markers were used to confirm the endothelial cell clusters (clusters 6, 7, 8) vs. epithelial cell clusters (clusters 1, 2, 3, 4, 5, 9). f, UMAP of merged epithelial cell fractions from hCRCO cultured alone or co-cultured with R-VECs. Six unique clusters were identified. g, h, Heat map (g) and dot plot (h) of differentially expressed genes in tumour epithelial cells in cluster 2 and cluster 5 that are enriched in co-culture with R-VECs. Differential expression was performed using the Wilcoxon rank-sum test; FDR-adjusted P < 0.05. For statistics, see Supplementary Data 1. For medium formulations, see Supplementary Data 2.
Supplementary information
Supplementary Data (download PDF )
Supplementary Data 1: All statistics and replicates for Figures 1–4 and Extended Figures 1–10.
Supplementary Data (download PDF )
Supplementary Data 2: Summary of all medium components.
Supplementary Data (download PDF )
Supplementary Data 3: MATLAB code for calculation of EC area interacting with organoids (pertaining to Figure 4d and Figure 4g).
Supplementary Data (download PDF )
Supplementary Data 4: Summary of quantitative PCR primers.
Supplementary Information (download PDF )
This file contains Supplementary Video Legends.
Supplementary Table (download XLSX )
Supplementary Table 1: Summary and source data for ChIP analysis.
Supplementary Table (download XLSX )
Supplementary Table 2: Summary and source data for single-cell RNA-sequencing experiments.
Supplementary Figure (download TIF )
Supplementary Figure 1a: Original Western Blots.
Supplementary Figure (download TIF )
Supplementary Figure 1b: Original Western Blots.
Video 1 (download MP4 )
CTRL-EC and R-VEC in 3D gel matrices and and in large-volume perfusable in microfluidic devices.
Video 2 (download MP4 )
Interaction of R-VECs with human pancreatic islets.
Video 3 (download MP4 )
Time-lapse videos of R-VEC or CTRL-EC in co-culture with either normal human colon organoids (hCO) or human colorectal cancer organoids (hCRCO) in 3D matrix.
Source data
Rights and permissions
About this article
Cite this article
Palikuqi, B., Nguyen, DH.T., Li, G. et al. Adaptable haemodynamic endothelial cells for organogenesis and tumorigenesis. Nature 585, 426–432 (2020). https://doi.org/10.1038/s41586-020-2712-z
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41586-020-2712-z
This article is cited by
-
Lactate-lactylation in tumor angiogenesis and progression: mechanisms, biomarker potential, and therapeutic implications
Biomarker Research (2026)
-
miR126-mediated alteration of vascular integrity in Rett syndrome
Molecular Psychiatry (2026)
-
Heart-on-a-chip and vasculature-on-a-chip platforms as models of cardiovascular disease
Nature Reviews Cardiology (2026)
-
Exploring the hepatic-ophthalmic axis through immune modulation and cellular dynamics in diabetic retinopathy and non-alcoholic fatty liver disease
Human Genomics (2025)
-
Dissecting endothelial cell heterogeneity with new tools
Cell Regeneration (2025)