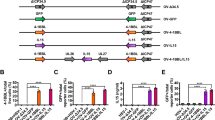
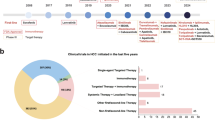
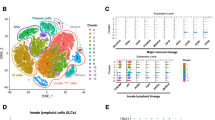
Abstract
Hepatocellular carcinoma remains a life-threatening malignancy with limited therapeutic options following the failure of second-line treatments1,2. Oncolytic viruses selectively replicate in and lyse cancer cells, releasing neoantigens and stimulating systemic antitumour immunity3, offering a potential therapeutic option. Here we present the results of a multicentre phase 1 clinical trial evaluating VG161, an engineered oncolytic herpes simplex virus that expresses IL-12, IL-15, IL-15Rα and a PD-1–PD-L1-blocking fusion protein4, for safety and efficacy in patients with advanced liver cancer. VG161 was well tolerated, with no dose-limiting toxicities observed, and it demonstrated promising efficacy by reshaping the tumour immune microenvironment and re-sensitizing tumours that were previously resistant to systemic treatments. Notably, we also found that patients who had previously been sensitive to checkpoint inhibitor therapy showed enhanced efficacy with VG161 treatment. Furthermore, we developed an efficacy-prediction model based on differentially expressed genes, which successfully identified patients who were likely to benefit from VG161 and predicted prolonged overall survival. These findings position VG161 as a promising third-line therapeutic option for refractory hepatocellular carcinoma. This provides a new avenue for treatment and advances the field of oncolytic virus-based immunotherapies. ClinicalTrials.gov registration: NCT04806464.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The study protocol is available in the Supplementary Information. The datasets from the clinical trial can be requested 12 months after the publication of this article. Researchers wishing to access raw and analysed data should email the corresponding author, clearly stating the research purpose. Requests will be reviewed by the institutional review board, considering the risk of patient reidentification, and a response can be expected within 14 days. Individual deidentified data of the participants will be available to approved eligible applicants and investigators after signing a data access agreement. The raw sequence data reported in this study have been deposited in the Genome Sequence Archive (GSA-Human: HRA007839 (RNA sequence); GSA-Human: HRA007880 (scRNA-seq, scTCR-seq, scBCR-seq and spatial transcriptome)) and are publicly accessible at https://ngdc.cncb.ac.cn/gsa-human. Source data are provided with this paper.
Change history
23 April 2025
A Correction to this paper has been published: https://doi.org/10.1038/s41586-025-09022-x
References
Vogel, A., Meyer, T., Sapisochin, G., Salem, R. & Saborowski, A. Hepatocellular carcinoma. Lancet 400, 1345–1362 (2022).
Siegel, R. L., Miller, K. D., Wagle, N. S. & Jemal, A. Cancer statistics, 2023. CA 73, 17–48 (2023).
Lin, D., Shen, Y. & Liang, T. Oncolytic virotherapy: basic principles, recent advances and future directions. Signal Transduct. Target. Ther. 8, 156 (2023).
Chouljenko, D. V. et al. Induction of durable antitumor response by a novel oncolytic herpesvirus expressing multiple immunomodulatory transgenes. Biomedicines 8, 484 (2020).
Llovet, J. M. et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 359, 378–390 (2008).
Kudo, M. et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: a randomised phase 3 non-inferiority trial. Lancet 391, 1163–1173 (2018).
Abou-Alfa, G. K. et al. Cabozantinib in patients with advanced and progressing hepatocellular carcinoma. N. Engl. J. Med. 379, 54–63 (2018).
Bruix, J. et al. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 389, 56–66 (2017).
Lencioni, R. New data supporting modified RECIST (mRECIST) for hepatocellular carcinoma. Clin. Cancer Res. 19, 1312–1314 (2013).
Lencioni, R. et al. GIDEON (Global Investigation of therapeutic DEcisions in hepatocellular carcinoma and Of its treatment with sorafeNib): second interim analysis. Int. J. Clin. Pract. 68, 609–617 (2014).
Cheng, A. L. et al. Updated efficacy and safety data from IMbrave150: atezolizumab plus bevacizumab vs. sorafenib for unresectable hepatocellular carcinoma. J. Hepatol. 76, 862–873 (2022).
Finn, R. S. et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N. Engl. J. Med. 382, 1894–1905 (2020).
Kelley, R. K. et al. Safety, efficacy, and pharmacodynamics of tremelimumab plus durvalumab for patients with unresectable hepatocellular carcinoma: randomized expansion of a phase I/II study. J. Clin. Oncol. 39, 2991–3001 (2021).
Heo, J. et al. Randomized dose-finding clinical trial of oncolytic immunotherapeutic vaccinia JX-594 in liver cancer. Nat. Med. 19, 329–336 (2013).
Moehler, M. et al. Vaccinia-based oncolytic immunotherapy pexastimogene devacirepvec in patients with advanced hepatocellular carcinoma after sorafenib failure: a randomized multicenter phase IIb trial (TRAVERSE). Oncoimmunology 8, 1615817 (2019).
Kaufman, H. L. et al. Local and distant immunity induced by intralesional vaccination with an oncolytic herpes virus encoding GM-CSF in patients with stage IIIc and IV melanoma. Ann. Surg. Oncol. 17, 718–730 (2010).
Kim, J. H. et al. Systemic armed oncolytic and immunologic therapy for cancer with JX-594, a targeted poxvirus expressing GM-CSF. Mol. Ther. 14, 361–370 (2006).
Dranoff, G. et al. Vaccination with irradiated tumor cells engineered to secrete murine granulocyte-macrophage colony-stimulating factor stimulates potent, specific, and long-lasting anti-tumor immunity. Proc. Natl Acad. Sci. USA 90, 3539–3543 (1993).
Tamadaho, R. S. E., Hoerauf, A. & Layland, L. E. Immunomodulatory effects of myeloid-derived suppressor cells in diseases: role in cancer and infections. Immunobiology 223, 432–442 (2018).
Shen, Y. et al. VG161 activates systemic antitumor immunity in pancreatic cancer models as a novel oncolytic herpesvirus expressing multiple immunomodulatory transgenes. J. Med. Virol. 95, e28108 (2022).
Todo, T. et al. Intratumoral oncolytic herpes virus G47∆ for residual or recurrent glioblastoma: a phase 2 trial. Nat. Med. 28, 1630–1639 (2022).
Lencioni, R. & Llovet, J. M. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin. Liver Dis. 30, 52–60 (2010).
Llovet, J. M. & Lencioni, R. mRECIST for HCC: performance and novel refinements. J. Hepatol. 72, 288–306 (2020).
Arulanandam, R. et al. VEGF-mediated induction of PRD1-BF1/Blimp1 expression sensitizes tumor vasculature to oncolytic virus infection. Cancer Cell 28, 210–224 (2015).
Kurozumi, K. et al. Oncolytic HSV-1 infection of tumors induces angiogenesis and upregulates CYR61. Mol. Ther. 16, 1382–1391 (2008).
Finn, R. S. et al. Pembrolizumab as second-line therapy in patients with advanced hepatocellular carcinoma in KEYNOTE-240: a randomized, double-blind, phase III trial. J. Clin. Oncol. 38, 193–202 (2020).
Lin, Z. Z. et al. A multicenter phase II study of second-line axitinib for patients with advanced hepatocellular carcinoma failing first-line sorafenib monotherapy. Oncologist 25, e1280–e1285 (2020).
Qin, S. et al. Apatinib as second-line or later therapy in patients with advanced hepatocellular carcinoma (AHELP): a multicentre, double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Gastroenterol. Hepatol. 6, 559–568 (2021).
Qin, S. et al. Camrelizumab in patients with previously treated advanced hepatocellular carcinoma: a multicentre, open-label, parallel-group, randomised, phase 2 trial. Lancet Oncol. 21, 571–580 (2020).
Chaurasiya, S. et al. Oncolytic poxvirus CF33-hNIS-DeltaF14.5 favorably modulates tumor immune microenvironment and works synergistically with anti-PD-L1 antibody in a triple-negative breast cancer model. Oncoimmunology 9, 1729300 (2020).
Noonan, A. M. et al. Randomized phase 2 trial of the oncolytic virus pelareorep (Reolysin) in upfront treatment of metastatic pancreatic adenocarcinoma. Mol. Ther. 24, 1150–1158 (2016).
Konyn, P., Ahmed, A. & Kim, D. Current epidemiology in hepatocellular carcinoma. Expert Rev. Gastroenterol. Hepatol. 15, 1295–1307 (2021).
McGlynn, K. A., Petrick, J. L. & El-Serag, H. B. Epidemiology of hepatocellular carcinoma. Hepatology 73, 4–13 (2021).
De Clercq, E. A cutting-edge view on the current state of antiviral drug development. Med. Res. Rev. 33, 1249–1277 (2013).
Wiedemeyer, W. R. et al. ABBV-011, a novel, calicheamicin-based antibody–drug conjugate, targets SEZ6 to eradicate small cell lung cancer tumors. Mol. Cancer Ther. 21, 986–998 (2022).
Wang, X., Yang, B., Li, Y., Luo, J. & Wang, Y. AKR1C1 alleviates LPS-induced ALI in mice by activating the JAK2/STAT3 signaling pathway. Mol. Med. Rep. 24, 833 (2021); retraction 31, 84 (2025).
Truong, A. et al. Molecular identification and characterisation of a novel chicken leukocyte immunoglobulin-like receptor A5. Br. Poult. Sci. 62, 68–80 (2020).
Chu, X. et al. Overview of human 20 alpha-hydroxysteroid dehydrogenase (AKR1C1): Functions, regulation, and structural insights of inhibitors. Chem. Biol. Interact. 351, 109746 (2021).
Liu, Z.-X. et al. Whole-exome sequencing among Chinese patients with hereditary diffuse gastric cancer. JAMA Netw. Open 5, e2245836 (2022).
Liu, W. et al. Circ-ZEB1 promotes PIK3CA expression by silencing miR-199a-3p and affects the proliferation and apoptosis of hepatocellular carcinoma. Mol. Cancer 21, 72 (2022).
Scheiter, A. et al. RASSF1A independence and early galectin‐1 upregulation in PIK3CA‐induced hepatocarcinogenesis: new therapeutic venues. Mol. Oncol. 16, 1091–1118 (2022).
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30, 2114–2120 (2014).
Kim, D., Langmead, B. & Salzberg, S. L. HISAT: a fast spliced aligner with low memory requirements. Nat. Methods 12, 357–360 (2015).
Roberts, A., Trapnell, C., Donaghey, J., Rinn, J. L. & Pachter, L. Improving RNA-seq expression estimates by correcting for fragment bias. Genome Biol. 12, R22 (2011).
Trapnell, C. et al. Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat. Biotechnol. 28, 511–515 (2010).
Anders, S., Pyl, P. T. & Huber, W. HTSeq-a Python framework to work with high-throughput sequencing data. Bioinformatics 31, 166–169 (2015).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Kong, L. et al. CPC: assess the protein-coding potential of transcripts using sequence features and support vector machine. Nucleic Acids Res. 35, W345–W349 (2007).
Li, A., Zhang, J. & Zhou, Z. PLEK: a tool for predicting long non-coding RNAs and messenger RNAs based on an improved k-mer scheme. BMC Bioinformatics 15, 311 (2014).
Sun, L. et al. Utilizing sequence intrinsic composition to classify protein-coding and long non-coding transcripts. Nucleic Acids Res. 41, e166 (2013).
Finn, R. D. et al. Pfam: the protein families database. Nucleic Acids Res. 42, D222–D230 (2014).
Gao, Y., Wang, J. & Zhao, F. CIRI: an efficient and unbiased algorithm for de novo circular RNA identification. Genome Biol. 16, 4 (2015).
Zhang, X. O. et al. Complementary sequence-mediated exon circularization. Cell 159, 134–147 (2014).
Hao, Y. et al. Integrated analysis of multimodal single-cell data. Cell 184, 3573–3587.e3529 (2021).
McGinnis, C. S., Murrow, L. M. & Gartner, Z. J. DoubletFinder: doublet detection in single-cell RNA sequencing data using artificial nearest neighbors. Cell Syst. 8, 329–337.e324 (2019).
Stuart, T. et al. Comprehensive integration of single-cell data. Cell 177, 1888–1902.e1821 (2019).
Hafemeister, C. & Satija, R. Normalization and variance stabilization of single-cell RNA-seq data using regularized negative binomial regression. Genome Biol. 20, 296 (2019).
Cooper, J. K. et al. Species identification in cell culture: a two-pronged molecular approach. In Vitro Cell Dev. Biol. Anim. 43, 344–351 (2007).
Acknowledgements
This work was supported by National Key Research and Development Program of China (2024YFA1306400 to T.L. and 2020YFA0804300 to Q.Z.); CNBG-Virogin Biotech (Shanghai); Virogin Biotech; the Joint Fund for Regional Innovation and Development of National Natural Science Foundation of China (U23A20462 to X.B.); National Natural Science Foundation of China (82071867 to X.B., 82103044 to Y.S., 92359304 to Q.Z., 32321002 to Q.Z., 82473461 to Q.Z., 82203699 to T.F. and 81902597 to L.W.); “Ling Yan” Research and Development Program of Department of Zhejiang Province Science and Technology (2024C03167 to X.B.); Natural Science Foundation of Zhejiang Province/Exploration Project (LY21H160037 to W.C. and LZ22H030003 to Y.Y.); and Medical Health Science and Technology Project of Zhejiang Province (2024KY958 to W.C.). The authors thank OE Biotech for providing technical support with RNA-seq, single-cell sequencing and spatial transcriptomics.
Author information
Authors and Affiliations
Contributions
Y.S., Q.T., R.Z. and T.L. designed the clinical trial. Y.S., X.B., Q.Z., X. Liang, S. Gu, G.S., Y.W., T.F., Y.L., L.W., X.S., W.C., Y.Y., Y.C. and X. Li were responsible for patient treatment and management. X.J. and G.W. performed ultrasound-guided puncture administration. Z. Zheng, J.S. and M.C. recruited patients and collected data. Y.S., A.Q., L.X., Y.Q., W.B., S.R., R.Z. and M.H. conducted statistical analysis. Z. Zhao, W.J., Z.W., Z.Y., S. Guo, D.L., F.W. and J.D. were responsible for the basic research work. W.S., M.S. and Y.M. performed bioinformatics analysis. Y.S. and T.L. wrote the manuscript. All authors read and approved the final draft of the paper.
Corresponding author
Ethics declarations
Competing interests
CNBG-Virogin Biotech (Shanghai) owns the patent rights for VG161 in mainland China. Virogin Biotech owns the patent rights for VG161 outside mainland China. The authors declare no competing interests.
Peer review
Peer review information
Nature thanks Howard Kaufman, Lars Zender and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Patient enrolment.
Of 92 patients who provided informed consent, 44 patients who matched the eligibility criteria were enrolled.
Extended Data Fig. 2 Exclusion of the possible effect of anti-HBV agents on VG161 replication in vitro and in vivo.
a, b, Comparison of the blood VG161 viral DNA concentration between patients who received (n = 6 biologically independent patients) or did not receive (n = 5 biologically independent patients) oral administration of anti-HBV agents within 24 h. c, Female NCG mice were implanted subcutaneously with Hep3B cells and gavaged with entecavir (0.1 mg/kg/day) or PBS (n = 3 biologically independent animals). Intratumoral injection of 1 × 107 PFU VG161 was performed 3 days after gavage. Hep3B tumors were excised at indicated time (0, 0.5, 1, 2, 3, and 7 days). The virus titers of tumor lysate were measured by plaque assay on Vero cells. d, The in vitro cytotoxicity of different anti-HBV agents on the tumor cells. Cells were treated with anti-HBV agents, and cell viability was measured using the CCK-8 assay at 65 h (6.7 × 104 cells examined over 4 independent experiments). e, Hep3B cells were treated with or without anti-HBV agents for 12 h, followed by replacement with fresh medium and infection with VG161. The cell viability upon different MOI and pretreatments was measured using the CCK-8 assay at 65 h (n = 6.7 × 104 cells examined over 4 independent experiments). f, The effect of anti-HBV agent treatment on VG161 replication ability in Hep3B cells. Hep3B cells were treated with or without anti-HBV agents for 12 h, followed by replacement with fresh medium and infection with VG161 for 24, 48, 72 h. The culture supernatant were collected, mixed, and lysed, then titrated by plaque assay (n = 0.8 × 106 cells examined over 3 independent experiments). For b, Fisher’s exact test was used for analysis. For d and e, No statistical tests were performed to compare differences. For a, c and f, Data are presented as the mean ± standard error and were analyzed using the unpaired two-sided Student’s t-test (*p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001 and NS, not significant).
Extended Data Fig. 3 Immunological profiles in patients’ blood samples after VG161 administration.
a, Lymphocyte subset trends in peripheral blood (n = 9). b, Plasma cytokine changes in peripheral blood (n = 11). c, Changes of typical T cell markers representing immune state in peripheral blood (n = 11). The dosing regimen for each patient was as follows: Patient A1-101 received a single dose of 1.0 × 108 PFU. Patient A2-102 received daily doses of 1.0 × 108 PFU for 2 consecutive days, resulting in a total dose of 2.0 × 108 PFU. Patients A3-104, A3-105, and A3-108 each received daily doses of 1.0 × 108 PFU for 3 consecutive days, resulting in a total dose of 3.0 × 108 PFU. Patients A4-109, A4-110, and A4-202 were administered daily doses of 1.3 × 108 PFU for 3 consecutive days, resulting in a total dose of 4.0 × 108 PFU. Finally, patients A5-119, A5-120, and A5-205 received daily doses of 1.7 × 108 PFU for 3 consecutive days, resulting in a total dose of 5.0 × 108 PFU. The 1.3 × 108 PFU and 1.7 × 108 PFU dose represents a rounded value to one decimal place.
Extended Data Fig. 4 Pathological examination of patient no. A4-109 from Fig. 2.
a, Pathological results from biopsy samples taken before the patient received VG161. b, A puncture biopsy was performed at the injection site after the VG161 treatment, and the pathological results demonstrated typical tumor cell necrosis. Please note that these biopsies were obtained from a single clinical patient and could not be repeated due to ethical and procedural considerations.
Extended Data Fig. 5 Efficacy prediction model for VG161 in advanced HCC.
a, Graphic overview of the experimental strategy. Created in BioRender. Lin, D. (2025) https://biorender.com/m07m206. b, KM survival curves between low- and high-survival groups. c, Volcano plot for DEGs of low- (n = 10 biologically independent patients) and high-survival groups (n = 11 biologically independent patients), P values are calculated using two-sided Student’s t-test. Benjamini-Hochberg adjusted p value < 0.05 is calculated as a significance cutoff. d, GO and KEGG analysis of low- (n = 10 biologically independent patients) and high-survival groups (n = 11 biologically independent patients), p values are calculated using two-sided hypergeometric test. Benjamini-Hochberg adjusted p value < 0.05 is calculated as a significance cutoff. e, Univariate Cox regression analysis identification of the prognosis-associated DEGs. f and g, LASSO Cox regression analysis assessment of the prognostic and screen risk genes, data is the cross validation mean (cvm) ± cross validation standard error (cvsd) (Low-survival group, n = 10 biologically independent patients and high-survival group, n = 11 biologically independent patients). h, Multivariate Cox regression analysis of risk genes, data is the hazard ratio (HR), 95% confidence intervals (CIs), p values are calculated using two-sided log rank tests. i, KM survival curves based on the risk model. j, ROC curves based on the risk model. k, KM survival curves based on the risk model in the TCGA-LIHC cohort. For b, i and k, p values are calculated using two-sided log rank tests, the shadows on either side of the survival curves depict 95% CIs.
Extended Data Fig. 6 The contrast of KM curves between the VG161-C102 group vs. control group.
a, Overall OS in the VG161-C102 group vs. control group. b, Comparison of the OS between the VG161-C102 and control groups in the >3 m subgroup. c, OS of the non-PST and control groups in the VG161-C102 group. d, Comparison of the OS between the non-PST+PreCPI > 3 m patients in the VG161-C102 group and the PreCPI > 3 m patients in the control group. Subgroups were compared using the log-rank test.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shen, Y., Bai, X., Zhang, Q. et al. Oncolytic virus VG161 in refractory hepatocellular carcinoma. Nature 641, 503–511 (2025). https://doi.org/10.1038/s41586-025-08717-5
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41586-025-08717-5
This article is cited by
-
Tumor microenvironment dynamics in gastric cancer pathogenesis and therapeutic resistance
Molecular Cancer (2026)
-
Rewiring tumor cytokine networks to enhance immune checkpoint blockade: mechanisms, engineering, and clinical translation
Journal of Experimental & Clinical Cancer Research (2026)
-
Oncolytic viruses: advanced strategies in cancer therapy
Signal Transduction and Targeted Therapy (2026)
-
Single-cell RNA sequencing combined with single-cell genome-wide association study identifies SF3B4 as a hub gene in hepatocellular carcinoma progression
Discover Oncology (2026)
-
Oncolytic human herpesvirus for cancer therapy
Molecular Biology Reports (2026)