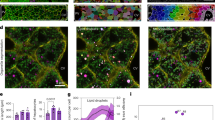
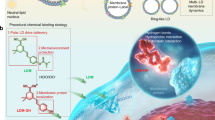
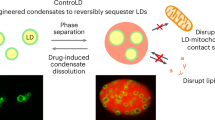
Abstract
Lipid droplets (LDs) dynamically interact with other organelles, such as mitochondria, in surveillance of cellular metabolic homeostasis. The transient nature of LDs, however, poses technical challenges to snapshot molecular information underlying these interactions. Herein, we present a small-molecule-based photocatalytic protein proximity labeling method (named LipoID) to enable in situ labeling, capturing and profiling of the LD-interacting proteome. This method is enabled by a set of LD-targeting probes designed to catalyze protein modifications nearby LDs using nucleophilic substrates. Profiled by liquid chromatography–tandem mass spectrometry, LipoID identifies tethered interorganellar interactions, particularly with mitochondria, in addition to reliable capture of validated LD biomarkers (for example, perilipins (PLINs)). Coupled with comparative proteomics, LipoID discovers mitochondrial voltage-dependent anion channel 3 as a potential regulator of LD–mitochondria proximity through interacting with PLIN3 on LDs. Further metabolomics analysis suggested remodeled lipid metabolism in line with the LD–mitochondria interaction. Together, LipoID enables in situ profiling of the LD interactome and reveals interorganellar regulation.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All supporting data in this study are fully provided within this article and its Supplementary Information. Data are available from the corresponding authors upon request. Source data are provided with this paper.
Code availability
The Python code for calculating the distance between organelles used in this study was deposited to Zenodo (https://doi.org/10.5281/zenodo.17547536)58.
References
Mathiowetz, A. J. & Olzmann, J. A. Lipid droplets and cellular lipid flux. Nat. Cell Biol. 26, 331–345 (2024).
Olzmann, J. A. & Carvalho, P. Dynamics and functions of lipid droplets. Nat. Rev. Mol. Cell Biol. 20, 137–155 (2018).
Benador, I. Y., Veliova, M., Liesa, M. & Shirihai, O. S. Mitochondria bound to lipid droplets: where mitochondrial dynamics regulate lipid storage and utilization. Cell Metab. 29, 827–835 (2019).
Lodhi, I. J. & Semenkovich, C. F. Peroxisomes: a nexus for lipid metabolism and cellular signaling. Cell Metab. 19, 380–392 (2014).
Zadoorian, A., Du, X. & Yang, H. Lipid droplet biogenesis and functions in health and disease. Nat. Rev. Endocrinol. 19, 443–459 (2023).
Lumaquin-Yin, D. et al. Lipid droplets are a metabolic vulnerability in melanoma. Nat. Commun. 14, 3192 (2023).
Liu, P. et al. Chinese hamster ovary K2 cell lipid droplets appear to be metabolic organelles involved in membrane traffic. J. Biol. Chem. 279, 3787–3792 (2004).
Zhang, S. et al. Morphologically and functionally distinct lipid droplet subpopulations. Sci. Rep. 6, 29539 (2016).
Cui, L., Mirza, A. H., Zhang, S., Liang, B. & Liu, P. Lipid droplets and mitochondria are anchored during brown adipocyte differentiation. Protein Cell 10, 921–926 (2019).
Ding, Y. et al. Isolating lipid droplets from multiple species. Nat. Protoc. 8, 43–51 (2012).
Bersuker, K. et al. A proximity labeling strategy provides insights into the composition and dynamics of lipid droplet proteomes. Dev. Cell 44, 97–112 (2018).
Martell, J. effreyD. et al. Engineered ascorbate peroxidase as a genetically encoded reporter for electron microscopy. Nat. Biotechnol. 30, 1143–1148 (2012).
Lam, S. S. et al. Directed evolution of APEX2 for electron microscopy and proximity labeling. Nat. Methods 12, 51–54 (2015).
James, C. et al. Proteomic mapping by rapamycin-dependent targeting of APEX2 identifies binding partners of VAPB at the inner nuclear membrane. J. Biol. Chem. 294, 16241–16254 (2019).
Benhalevy, D., Anastasakis, D. G. & Hafner, M. Proximity-CLIP provides a snapshot of protein-occupied RNA elements in subcellular compartments. Nat. Methods 15, 1074–1082 (2018).
Zhou, Y. et al. Expanding APEX2 substrates for proximity-dependent labeling of nucleic acids and proteins in living cells. Angew. Chem. Int. Ed. Engl. 58, 11763–11767 (2019).
Liu, G. et al. Mechanism of adrenergic CaV1.2 stimulation revealed by proximity proteomics. Nature 577, 695–700 (2020).
Qin, W., Cho, K. F., Cavanagh, P. E. & Ting, A. Y. Deciphering molecular interactions by proximity labeling. Nat. Methods 18, 133–143 (2021).
Shu, X. et al. A genetically encoded tag for correlated light and electron microscopy of intact cells, tissues, and organisms. PLoS Biol. 9, e1001041 (2011).
To, T.-L. et al. Photoactivatable protein labeling by singlet oxygen mediated reactions. Bioorg. Med. Chem. Lett. 26, 3359–3363 (2016).
Wang, P. et al. Mapping spatial transcriptome with light-activated proximity-dependent RNA labeling. Nat. Chem. Biol. 15, 1110–1119 (2019).
Zhai, Y. et al. Spatiotemporal-resolved protein networks profiling with photoactivation dependent proximity labeling. Nat. Commun. 13, 4906 (2022).
Branon, T. C. et al. Efficient proximity labeling in living cells and organisms with TurboID. Nat. Biotechnol. 36, 880–887 (2018).
Cho, K. F. et al. Split-TurboID enables contact-dependent proximity labeling in cells. Proc. Natl Acad. Sci. USA 117, 12143–12154 (2020).
Cho, K. F. et al. Proximity labeling in mammalian cells with TurboID and split-TurboID. Nat. Protoc. 15, 3971–3999 (2020).
Tamura, T., Takato, M., Shiono, K. & Hamachi, I. Development of a photoactivatable proximity labeling method for the identification of nuclear proteins. Chem. Lett. 49, 145–148 (2020).
Huang, Z. et al. Bioorthogonal photocatalytic decaging-enabled mitochondrial proteomics. J. Am. Chem. Soc. 143, 18714–18720 (2021).
Geri, J. B. O. J. et al. Microenvironment mapping via Dexter energy transfer on immune cells. Science 367, 1091–1097 (2020).
Pan, C. R., Knutson, S. D., Huth, S. W. & MacMillan, D. W. C. microMap proximity labeling in living cells reveals stress granule disassembly mechanisms. Nat. Chem. Biol. 21, 490–500 (2025).
Buksh, B. F. et al. μMap-red: proximity labeling by red light photocatalysis. J. Am. Chem. Soc. 144, 6154–6162 (2022).
Collot, M. et al. Probing polarity and heterogeneity of lipid droplets in live cells using a push-pull fluorophore. Anal. Chem. 91, 1928–1935 (2019).
Liu, S. et al. Strategies to enhance the photosensitization: polymerization and the donor–acceptor even–odd effect. Angew. Chem. Int. Ed. Engl. 57, 15189–15193 (2018).
Niu, N. et al. A cell membrane-targeting AIE photosensitizer as a necroptosis inducer for boosting cancer theranostics. Chem. Sci. 13, 5929–5937 (2022).
Pham, T. C., Nguyen, V. N., Choi, Y., Lee, S. & Yoon, J. Recent strategies to develop innovative photosensitizers for enhanced photodynamic therapy. Chem. Rev. 121, 13454–13619 (2021).
Chanderbhan, R., Noland, B. J., Scallen, T. J. & Vahouny, G. V. Sterol carrier protein2. Delivery of cholesterol from adrenal lipid droplets to mitochondria for pregnenolone synthesis. J. Biol. Chem. 257, 8928–8934 (1982).
Herms, A. et al. AMPK activation promotes lipid droplet dispersion on detyrosinated microtubules to increase mitochondrial fatty acid oxidation. Nat. Commun. 6, 7176 (2015).
Nguyen, T. B. et al. DGAT1-dependent lipid droplet biogenesis protects mitochondrial function during starvation-induced autophagy. Dev. Cell 42, 9–21 (2017).
Rambold, A. S., Cohen, S. & Lippincott-Schwartz, J. Fatty acid trafficking in starved cells: regulation by lipid droplet lipolysis, autophagy, and mitochondrial fusion dynamics. Dev. Cell 32, 678–692 (2015).
Ye, Z. et al. Burst of hopping trafficking correlated reversible dynamic interactions between lipid droplets and mitochondria under starvation. Exploration 3, 20230002 (2023).
Shoshan-Barmatz, V. et al. VDAC, a multi-functional mitochondrial protein regulating cell life and death. Mol. Aspects Med. 31, 227–285 (2010).
Colombini, M. VDAC: the channel at the interface between mitochondria and the cytosol. Mol. Cell. Biochem. 256–257, 107–115 (2004).
Yang, Y. et al. Nedd4 ubiquitylates VDAC2/3 to suppress erastin-induced ferroptosis in melanoma. Nat. Commun. 11, 433 (2020).
Lee, K., Kerner, J. & Hoppel, C. L. Mitochondrial carnitine palmitoyltransferase 1a (CPT1a) is part of an outer membrane fatty acid transfer complex. J. Biol. Chem. 286, 25655–25662 (2011).
Bian, X. et al. Lipid metabolism and cancer. J. Exp. Med. 218, e20201606 (2021).
Yoon, H., Shaw, J. L., Haigis, M. C. & Greka, A. Lipid metabolism in sickness and in health: Emerging regulators of lipotoxicity. Mol. Cell 81, 3708–3730 (2021).
Brasaemle, D. L., Dolios, G., Shapiro, L. & Wang, R. Proteomic analysis of proteins associated with lipid droplets of basal and lipolytically stimulated 3T3-L1 adipocytes. J. Biol. Chem. 279, 46835–46842 (2004).
MacAskill, A. F. & Kittler, J. T. Control of mitochondrial transport and localization in neurons. Trends Cell Biol. 20, 102–112 (2010).
Jin, Y., Ren, Z., Tan, Y., Zhao, P. & Wu, J. Motility plays an important role in the lifetime of mammalian lipid droplets. Int. J. Mol. Sci. 22, 3802 (2021).
Feng, H. et al. Enabling photo-crosslinking and photo-sensitizing properties for synthetic fluorescent protein chromophores. Angew. Chem. Int. Ed. Engl. 62, e202215215 (2022).
Teng, K. X. et al. BODIPY-based photodynamic agents for exclusively generating superoxide radical over singlet oxygen. Angew. Chem. Int. Ed. Engl. 60, 19912–19920 (2021).
Hanser, F. et al. Nile red-based GPCR ligands as ultrasensitive probes of the local lipid microenvironment of the receptor. ACS Chem. Biol. 16, 651–660 (2021).
Wang, Y. et al. Construction of nanodroplet/adiposome and artificial lipid droplets. ACS Nano 10, 3312–3322 (2016).
Bai, Y. et al. Carbon dots with absorption red-shifting for two-photon fluorescence imaging of tumor tissue pH and synergistic phototherapy. ACS Appl. Mater. Interfaces 13, 35365–35375 (2021).
Chen, T. et al. iProX in 2021: connecting proteomics data sharing with big data. Nucleic Acids Res. 50, D1522–D1527 (2022).
Ma, J. et al. iProX: an integrated proteome resource. Nucleic Acids Res. 47, D1211–D1217 (2019).
Bligh, E. G. & Dyer, W. J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 37, 911–917 (1959).
Lam, S. M. et al. A multi-omics investigation of the composition and function of extracellular vesicles along the temporal trajectory of COVID-19. Nat. Metab. 3, 909–922 (2021).
Guo, H. LipoID code for distance between organelles: code for LipoID (v1.0.0). Zenodo https://doi.org/10.5281/zenodo.17547536 (2025).
Acknowledgements
This work was supported, in part, by funds from National Natural Science Foundation of China (22222410 to Y.L.; 22494700 to X.Z. and Q. Z.; 22322411 to Q.Z.; 22374148 to Y.L.; 22207106 to D.S.) and the Innovation Program of Science and Research from the Dalian Institute of Chemical Physics, Chinese Academy of Sciences (DICP I202245 to Y.L.; DICP I202458 to W.W). We thank the 'Pioneer' and 'Leading Goose' R&D Program of Zhejiang (Grant 2025SDXHDX000*) for partially supporting this work.
Author information
Authors and Affiliations
Contributions
H.G. and W.W. designed and synthesized LD-targeted probes, screened candidates and identified the lead probe B6. H.G. and Y.L. established the LipoID workflow. R.S., H.F., D.S. and W.W. elucidated the photochemical mechanism of B6. H.G., N.Z., C.W. and B.Z. performed proteomic profiling (LC–MS/MS) and analyzed the data, demonstrating that LipoID captures LD-interacting proteins and identifying VDAC3 as a regulatory factor. H.G., Y.H. and J.Y. carried out the confocal imaging. X.D. provided the tissue sections. Q.Z., X.Z., L.Z. and Y.L. conceptualized the experiments, supervised the work and revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemical Biology thanks Douglas G. Mashek and the other, anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Chemical structures and fluorescence characterizations of 28 probes.
a, These 28 probes were designed with the X-furan-Y scaffold by varying the structures of electron-donating groups (X: A to D) and electron-accepting groups (Y: 0 to 6) on both ends. b-f, These probes exhibited fluorescence characteristics with notably emission wavelength and brightness variations in dioxane.
Extended Data Fig. 2 Examination of photocatalytic protein proximity labeling conditions using in vitro artificial liposome-lipoprotein model.
a, Co-localization fluorescence imaging between 28 probes and commercial LDs sensors. BODIPY or Nile red was chosen to match the em. wavelengths of probes. b, Labeling efficiency was dependent on the probe concentrations. 25 µM B6 reached to labeling plateau. c, B6 selectively catalyzed photo-induced labeling (red dashed box) and crosslinking (blue dashed box) of apolipoprotein APOA4 surrounding artificial liposomes. Propargylamine (PA) was used as the labeling substrate. d, Labeling efficiency was dependent on the illumination time. e, White and green light sources both catalyzed the labeling. f, Labeling efficiency was dependent on the concentration of PA substrate. 5 mM provided favorable signal-to-noise ratio. g, Labeling efficiency was dependent on the APOA4/Liposomes ratio. 50 µM: 250 AU was the optimal protein condition. FL: fluorescence gel detected by TMR fluorescence after clicking the labeled proteins with TMR-N3. CBB: Coomassie brilliant blue gel. These experiments were repeated twice showing similar results.(To editor: Please replace the "ration" to "ratio" in panel g, We have provided the updated version.)
Extended Data Fig. 3 Supporting experiments for LDs-targeted proximity labeling by B6 probe in the cell.
a, Cell viability assay in AML-12 cells. 1 µM of B6 was used in our work. Data were presented as mean ± SD (n = 6 wells). b, Evidence of ROS production by B6 upon illumination in cells. The ROS generated in cells by B6 (1 µM) were detected by DCF-DA (10 µM) under the white light illumination for 1 min. c-d, Immunofluorescence imaging showed labeled proteins were spatially in proximity to LDs. B6 (1 µM) catalyzed proteins labeling (green fluorescence from antibody or FAM-N3) nearby LDs (TMR-SNAP or mCherry). e, B6 (1 μM) selectively labeled and crosslinked LDs interacting proteome in cells shown by SDS-PAGE gel and dots assay. The fluorescence signal of proximity labeling was from conjugation with TMR-N3. f, Evidence to show selective proteome enrichment in photo-illuminated samples. The labeled proteome was conjugated with biotin-PEG4-N3, enriched with streptavidin beads, and then visualized using silver staining. g, Negative controls demonstrated the efficiency and background of LipoID labeling in live cells. h, Western blotting showed PLIN3 and PLIN2 enrichment in labeled and captured samples. i, KEGG analysis of enriched mitochondrial proteins revealed their involvement in the lipid metabolism pathway. The pathways associated with mitochondrial functions were highlighted. The one-sided Fisher’s exact test was applied. p values were corrected for multiple comparisons using the Benjamini–Hochberg procedure (FDR). Confocal Images are representative of 5-7 fields of view across 2 cultures in one experiment (c,d). Gel images are representative of n = 3 (e-g) independent experiments. Blot images are representative of twice independent experiments (h).
Extended Data Fig. 4 Short-term photo-illumination has negligible interference toward cell viability and functions of LDs.
a, The AML-12 cell incubated with B6 (1 μM) for 15 min then illuminated under white light (25 mW·cm−2) for 5 min. The cell viability was evaluated by CCK8 assay, and no significant difference was observed. Data were presented as mean ± SD (n = 12 wells). b, Image analysis revealed light exposure did not significantly alter LDs parameters including area (μm²), mean diameter (μm), or shape circularity (4π[area/perimeter²]). Data were presented as mean ± SD (n = 945, 1268, 941, 1272, 727, and 936 LDs, respectively). The data pooled from 7 fields of view across 2 cultures in one experiment. Statistical significance was assessed using a two-tailed Welch’s t-test. c, The AML-12 cell was incubated with B6 (1 μM) for 15 min and then illuminated under white light for 0, 5, 30 min. Western-blot gel of lysed cell indicated no significant change on 4-HNE (metabolic product of fatty acid peroxidation) or HO-1 (biomarker of oxidative stress) production. Data were presented as mean ± SD (n = 3 independent biological replicates). d-e, Free fatty acid content and fatty acid oxidation ability display subtle variation within 5 min photo-illumination. For elongated illumination period, both of them increased significantly. Data were presented as mean ± SD (n = 3 cultures). Statistical significance was analyzed using one-way ANOVA followed by Dunnett’s many-to-one comparisons with hv 0 min as the reference (two-sided).
Extended Data Fig. 5 Comparison of two LDs’ interactome datasets independently collected by using 2 different control probes.
a, Structures of B6, and control probes Rhodamine 6 G (Rh6G) and C6. B6 and C6 are LD-targeted, whereas the Rh6G non-specifically stains and labels the entire proteome, serving as background controls. Images are representative of 4-5 fields of view across 2 cultures in one experiment. b, Fluorescent electrophoresis gel showed B6 and Rh6G were capable of labeling proteins in AML-12 cell lysate, whereas C6 probe displayed almost no photocatalytic labeling function. Images are representative of twice independent experiments with three technical replicates. c, Volcano plot of the significantly differentially enriched proteins identified by LipoID in AML-12 cells using Rh6G as the background probe. LDs-related proteins were highlighted in red. n = 3 biological replicates. Statistical significance was assessed using a two-tailed Student’s t-test. Note: One should notice that proteomic background subtraction can be influenced by the labeling bias caused by different labeling mechanisms of different photocatalytic probes producing different types of ROS. d, Venn’s diagram showed strong overlap between two sets of proteomics by using C6 and Rh6G as background controls. e, Suborganellar distribution of LipoID captured proteins using C6 and Rh6G as background controls. Both datasets display similarity in suborganellar distribution, highlighting the reproducibility and specificity of the LipoID probe, regardless of control probes. f, Comparing LDs interactome profiles upon altering LDs-Mito interacting distance by erastin. g, Upon erastin inhibition of LDs-Mito interaction, we observed large number of down-regulated mitochondrial proteins captured by LipoID, suggesting its labeling was contact-based. n = 3 biological replicates. Statistical significance was assessed using a two-tailed Student’s t-test.
Extended Data Fig. 6 Comparison of LipoID and previous PLIN2-APEX2-based LD proteomics results (Olzmann et al., Dev. Cell. 2018, 44, 97-112).
a, Differences in experimental protocols. The PLIN2-APEX2 approach included an additional centrifugation based biochemical fractionation enrichment step to harvest/enrich the buoyant LD fractions prior to LC-MS/MS analysis. Our LipoID method in-situ labeled, captured and enriched directly from live cells. b, Comparison of LD biomarker proteins coverage under different p value cut-off between LipoID and previous APEX2 strategy. PLIN2-APEX2 dataset only averaged two repeated results and did not cut off proteomics hits by controlling p values, leading to better coverage of LDs biomarkers from Uniprot. In contrast, our LipoID proteomics analysis all employed P < 0.05 and fold of change > 1.5 cut-off strategy to ensure the reproducibility of protein hits. n = 3 biological replicates. Statistical significance was assessed using a two-tailed Student’s t-test. c-d, Both PLIN2-APEX2 and LipoID results shared similarity in interactome suborganellar distribution, and both PLIN2-APEX2 and LipoID captured mitochondrial proteome, including inner membrane and matrix proteins. PLIN2-APEX2 tended to capture more ER proteome due to its off-targeted localization to ER, even after an additional buoyant LDs fractionation step. e, PLIN2 was not specific to LD in AML12 cell line shown by the diffuse pattern around LDs in the confocal image. f, PLIN2 showed partial co-localization with the endoplasmic reticulum (ER) in the confocal image. Images are representative of twice independent experiments (e-f).
Extended Data Fig. 7 Effects of VDAC3 knock-down (VDAC3i) and overexpression (VDAC3OE) on LDs-Mito interacting distance, LD functions and morphologies, and mitochondrial fitness.
a, Western-blot of VDAC3 upon knock-down of VDAC3 (VDAC3i). Quantification of gel images corresponding to Fig. 4e. b, JC-1 assay evaluated mitochondrial membrane potential after VDAC3i treatment. The red/green fluorescence ratio reduced upon VDAC3i treatment, confirming decreased membrane potential. c, VDAC3i treatment reduced ATP level. d-e, Evaluations of LDs’ function upon VDAC3i treatment. The Amplex Red free fatty acid (FFA) assay and fatty acid oxidation (FAO) colorimetric assay were used to evaluate the content of free fatty acid and fatty acid oxidation ability, respectively. In VDAC3i-treated cells, the fatty acid was accumulated, and the capacity of fatty acid oxidation was suppressed due to mitochondrial dysfunction. f, Western-blot gels quantification of VDAC3 expression level at indicated conditions. g, JC-1 assay showed recovered mitochondrial membrane potential upon VDAC3OE, which was quantified by red/green fluorescence ratio. h-j, VDAC3OE partially recovered lost-of-function of mitochondria caused by erastin inhibition of VDAC3, shown by ATP level, FFA content, and FAO ability. Data were presented as mean ± SD (n = 3 cultures). Statistical significance was assessed using a two-tailed Student’s t-test (a-g) or one-way ANOVA followed by Dunnett’s many-to-one comparisons (two-sided) (h-j). Images are representative of twice independent experiments (b and g).
Extended Data Fig. 8 Mini-sized TC tagged VDAC3 ensures its localization on mitochondrial surface for imaging experiments.
a, 4 plasmid constructs of VDAC3. N- or C-termini were fused with TC tag or APEX2 protein. b, Mini-sized TC tag and its self-labeling mechanism by FlAsH-EDT2 fluorescent reporter for imaging experiments. c, N-terminal fusion or C-terminal fusion of VDAC3 with mini-sized TC tag ensured its localization to mitochondrial surface (3#, 4# lanes). The large-sized APEX2 protein fusion with VDAC3 mis-localized VDAC3 to nucleus or formed aggregates in the cell (1#, 2# lanes), impeding its application for interaction assay. Green: FlAsH signal. Red: mitochondria stained by Mito-TrackerTM Deep Red. d, Co-localization of VDAC3 labeled by FlAsH and LDs stained by B6, indicating close contact between LDs and VDAC3. e, Co-localization of VDAC3 labeled by FlAsH and PLIN3 tagged by mCherry, indicating close proximity between PLIN3 and VDAC3. Images are representative of twice independent experiments (c-e). f, Schematic of split GFP complementation assay.
Extended Data Fig. 9 APEX2 fingerprinting proximity labeling assay reveals the interaction between PLIN3 and VDAC3.
a, Schematic illustration of APEX2-mediated proximity labeling to assess the spatial interaction between PLIN3 and VDAC3. AML-12 cells co-expressing 3×FLAG-tagged PLIN3-APEX2 and 3×FLAG-VDAC3 were treated with biotin-phenol and H2O2 to initiate labeling. In the presence of proximity (upper panel), VDAC3 is biotinylated by APEX2 and captured by streptavidin enrichment, then detected by anti-FLAG antibody. In the absence of interaction (lower panel), no biotinylation or enrichment of VDAC3 occurs. b, Validation of the PLIN3-VDAC3 interaction using APEX2 fingerprinting proximity labeling assay and streptavidin enrichment. Left: Coomassie brilliant blue staining of total protein input. Middle: streptavidin-HRP blot showing biotinylated proteins in the input lysate. Right: anti-FLAG immunoblots after streptavidin-based enrichment. Both 3× FLAG-PLIN3-APEX2 and 3× FLAG-VDAC3 were pulled down in the presence of H2O2, indicating successful biotinylation and enrichment of VDAC3 via APEX2 fingerprinting proximity labeling assay. Blots are representative of twice independent experiments.
Extended Data Fig. 10 VDAC3 inhibition remodeled lipid metabolism.
a, Schematic of the HPLC-MS/MS based lipidomic analysis using density gradient centrifugation to enrich the LDs fraction. b-c, Heatmap (b) and its quantification (c) revealed significant orchestrated regulations of triacylglycerol (TAG), total cholesteryl esters (CE), free fatty acids (FFA) and diacylglycerols (DAG) upon VDAC3 inhibition. Box plots show the median (center line), 25th and 75th percentiles (box limits) and whiskers extending 1.5× the interquartile range. n = 4 independent biological replicates. Statistical significance was assessed using a two-tailed Welch’s t-test. d-g, Heat maps of lipid subtypes with different fatty acid chain lengths, total number of carbons and double bonds in the attached fatty acyl chains. (d) for TAG, (e) for CE, (f) for FFA, and (g) for DAG. Data were constructed using z-scores values. Data were presented as mean (n = 4 biological replicates). (h) The plot of significantly differential metabolites displayed the fold of change and p values of the top 10 metabolites with the smallest P values. The x-axis of the bar plot represents log2(fold of change), where positive values indicate upregulation and negative values indicate downregulation.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–14, Tables 1–4, Notes 1 and 2, synthesis and characterizations.
Supplementary Data 1 (download XLSX )
LC–MS/MS analysis of probe-derived oxidation intermediates and labeling sites.
Supplementary Data 2 (download XLSX )
Proteomic profiling of the LD interactome.
Supplementary Data 3-5 (download XLSX )
GO analysis of the LD interactome.
Supplementary Data 6 (download XLSX )
Proteomic profiling of the LD interactome under starvation stress.
Supplementary Data 7 (download XLSX )
Erastin inhibition proteomics data.
Supplementary Data 8 (download XLSX )
VDAC3-regulated LD–Mito interaction affects cellular lipidomic profile.
Supplementary Data 9 (download XLSX )
VDAC3-regulated LD–Mito interaction affects LD-specific lipidomic profile.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Fig. 6 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 7 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 10 (download XLSX )
Statistical source data.
Source Data Figs. 1 and 4 and Extended Data Figs. 2–5 and 9 (download PDF )
Unprocessed western blots and/or gels.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guo, H., Wan, W., Huang, Y. et al. LipoID profiles lipid droplet interactions and identifies interorganelle regulators. Nat Chem Biol (2026). https://doi.org/10.1038/s41589-025-02127-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41589-025-02127-4