Abstract
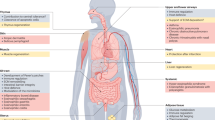
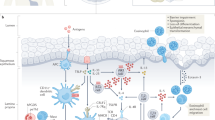
Eosinophils are multifunctional granulocytes involved in immune regulation, metabolism and tissue repair; however, the extent of their heterogeneity across tissues and the principles governing their specialization are not well defined. Here, we present a single-cell transcriptomic and proteomic atlas of mouse eosinophils spanning immune, barrier and metabolic sites. By integrating transcriptional profiling, high-dimensional surface proteomics and in vivo fate mapping, we show that eosinophil identity is shaped by both tissue-derived cues and the duration of local residency. Long-lived eosinophils in the small intestine diversify into transcriptionally and phenotypically distinct subsets, whereas short-lived and intermediate-lived populations in the lungs and colon, respectively, remain comparatively uniform. We identify trajectory-associated surface markers that stratify eosinophil maturation from bone-marrow progenitors to long-term tissue-resident subsets. This atlas establishes a unified framework in which tissue-specific residency time drives eosinophil maturation and diversification, providing a molecular toolkit for resolving eosinophil states in health and disease.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The scRNA-seq data reported in this paper have been deposited into the National Center for Biotechnology Information Sequence Read Archive under accession code PRJNA1301548. Processed scRNA-seq data can be viewed in figshare at https://doi.org/10.6084/m9.figshare.29816303.v1 (ref. 45). scRNA-seq datasets are available in an interactive portal: Cell-omics Data Coordinate Platform under dataset ID SCDS0000645. All other data supporting the findings of this study are available in the main text and supplementary files. Source data are provided with this paper.
Code availability
The code for scRNA-seq data analysis can be viewed in GitHub at https://github.com/liziyie/eosinophil_atlas.
Change history
16 January 2026
In the version of the article initially published, Fig. 6 erroneously contained the panel label “f” which has now been removed from the HTML and PDF versions of the article.
References
Klion, A. D., Ackerman, S. J. & Bochner, B. S. Contributions of eosinophils to human health and disease. Annu. Rev. Pathol. 15, 179–209 (2020).
Arnold, I. C. & Munitz, A. Spatial adaptation of eosinophils and their emerging roles in homeostasis, infection and disease. Nat. Rev. Immunol. 24, 858–877 (2024).
Jorssen, J. et al. Single-cell proteomics and transcriptomics capture eosinophil development and identify the role of IL-5 in their lineage transit amplification. Immunity 57, 1549–1566 e1548 (2024).
Rothenberg, M. E. & Hogan, S. P. The eosinophil. Annu. Rev. Immunol. 24, 147–174 (2006).
Nussbaum, J. C. et al. Type 2 innate lymphoid cells control eosinophil homeostasis. Nature 502, 245–248 (2013).
Loffredo, L. F. et al. Eosinophil accumulation in postnatal lung is specific to the primary septation phase of development. Sci. Rep. 10, 4425 (2020).
Heredia, J. E. et al. Type 2 innate signals stimulate fibro/adipogenic progenitors to facilitate muscle regeneration. Cell 153, 376–388 (2013).
Goh, Y. P. et al. Eosinophils secrete IL-4 to facilitate liver regeneration. Proc. Natl Acad. Sci. USA 110, 9914–9919 (2013).
Qiu, Y. et al. Eosinophils and type 2 cytokine signaling in macrophages orchestrate development of functional beige fat. Cell 157, 1292–1308 (2014).
Wu, D. et al. Eosinophils sustain adipose alternatively activated macrophages associated with glucose homeostasis. Science 332, 243–247 (2011).
Brigger, D. et al. Eosinophils regulate adipose tissue inflammation and sustain physical and immunological fitness in old age. Nat. Metab. 2, 688–702 (2020).
Hu, Y. & Chakarov, S. Eosinophils in obesity and obesity-associated disorders. Discov. Immunol. 2, kyad022 (2023).
Ignacio, A. et al. Small intestinal resident eosinophils maintain gut homeostasis following microbial colonization. Immunity 55, 1250–1267 e1212 (2022).
Chu, V. T. et al. Eosinophils promote generation and maintenance of immunoglobulin-A-expressing plasma cells and contribute to gut immune homeostasis. Immunity 40, 582–593 (2014).
Wang, W. L. et al. The aryl hydrocarbon receptor instructs the immunomodulatory profile of a subset of Clec4a4(+) eosinophils unique to the small intestine. Proc. Natl Acad. Sci. USA 119, e2204557119 (2022).
Schworer, S. A. et al. Notch 2 signaling contributes to intestinal eosinophil adaptations in steady state and tissue burden following oral allergen challenge. J. Leukoc. Biol. 116, 379–391 (2024).
Li, Y. et al. Neuromedin U programs eosinophils to promote mucosal immunity of the small intestine. Science 381, 1189–1196 (2023).
Diny, N. L. et al. The aryl hydrocarbon receptor contributes to tissue adaptation of intestinal eosinophils in mice. J. Exp. Med. https://doi.org/10.1084/jem.20210970 (2022).
Vassily, I. K., Korn, L. L. & Medzhitov, R. Nutrient-derived signals regulate eosinophil adaptation to the small intestine. Proc. Natl Acad. Sci. USA 121, e2316446121 (2024).
Gurtner, A. et al. Active eosinophils regulate host defence and immune responses in colitis. Nature 615, 151–157 (2023).
Liu, Z. et al. Fate mapping via Ms4a3-expression history traces monocyte-derived cells. Cell 178, 1509–1525 e1519 (2019).
Becht, E. et al. High-throughput single-cell quantification of hundreds of proteins using conventional flow cytometry and machine learning. Sci. Adv. 7, eabg0505 (2021).
Zhang, T., Warden, A. R., Li, Y. & Ding, X. Progress and applications of mass cytometry in sketching immune landscapes. Clin. Transl. Med. 10, e206 (2020).
Larsen, L. D., Dockstader, K., Olbrich, C. L., Cartwright, I. M. & Spencer, L. A. Modulation of surface CD11c expression tracks plasticity in murine intestinal tissue eosinophils. J. Leukoc. Biol. 111, 943–952 (2022).
Reis e Sousa, C., Yamasaki, S. & Brown, G. D. Myeloid C-type lectin receptors in innate immune recognition. Immunity 57, 700–717 (2024).
Trapnell, C. et al. The dynamics and regulators of cell fate decisions are revealed by pseudotemporal ordering of single cells. Nat. Biotechnol. 32, 381–386 (2014).
Adams, J. M. & Cory, S. The Bcl-2 protein family: arbiters of cell survival. Science 281, 1322–1326 (1998).
Carneiro, B. A. & El-Deiry, W. S. Targeting apoptosis in cancer therapy. Nat. Rev. Clin. Oncol. 17, 395–417 (2020).
Ng, M. S. F. et al. Deterministic reprogramming of neutrophils within tumors. Science 383, eadf6493 (2024).
Mesnil, C. et al. Lung-resident eosinophils represent a distinct regulatory eosinophil subset. J. Clin. Invest. 126, 3279–3295 (2016).
Dolitzky, A. et al. Mouse resident lung eosinophils are dependent on IL-5. Allergy 77, 2822–2825 (2022).
Evrard, M. et al. Developmental analysis of bone marrow neutrophils reveals populations specialized in expansion, trafficking, and effector functions. Immunity 48, 364–379.e368 (2018).
Kimura, I., Ichimura, A., Ohue-Kitano, R. & Igarashi, M. Free fatty acid receptors in health and disease. Physiol. Rev. 100, 171–210 (2020).
Wen, T. et al. The pan-B cell marker CD22 is expressed on gastrointestinal eosinophils and negatively regulates tissue eosinophilia. J. Immunol. 188, 1075–1082 (2012).
Ma, A. & Malynn, B. A. A20: linking a complex regulator of ubiquitylation to immunity and human disease. Nat. Rev. Immunol. 12, 774–785 (2012).
Coelho, A. L. et al. The chemokine CCL6 promotes innate immunity via immune cell activation and recruitment. J. Immunol. 179, 5474–5482 (2007).
Yuan, K. et al. Novel diagnostic biomarkers of oxidative stress, immune- infiltration characteristics and experimental validation of SERPINE1 in colon cancer. Discov. Oncol. 14, 206 (2023).
Lee, N. A. et al. Expression of IL-5 in thymocytes/T cells leads to the development of a massive eosinophilia, extramedullary eosinophilopoiesis, and unique histopathologies. J. Immunol. 158, 1332–1344 (1997).
Jiménez-Saiz, R. et al. Microbial regulation of enteric eosinophils and its impact on tissue remodeling and Th2 immunity. Front. Immunol. 11, 155 (2020).
Li, J. et al. Sexual dimorphism in obesity is governed by RELMα regulation of adipose macrophages and eosinophils. eLife https://doi.org/10.7554/elife.86001 (2023).
Karkout, R. et al. Female-specific enhancement of eosinophil recruitment and activation in a type 2 innate inflammation model in the lung. Clin. Exp. Immunol. 216, 13–24 (2024).
Artham, S., Chang, C. Y. & McDonnell, D. P. Eosinophilia in cancer and its regulation by sex hormones. Trends Endocrinol. Metab. 34, 5–20 (2023).
Finck, R. et al. Normalization of mass cytometry data with bead standards. Cytometry A 83, 483–494 (2013).
Newell, E. W., Sigal, N., Bendall, S. C., Nolan, G. P. & Davis, M. M. Cytometry by time-of-flight shows combinatorial cytokine expression and virus-specific cell niches within a continuum of CD8+ T cell phenotypes. Immunity 36, 142–152 (2012).
Z. Li. Temporal and spatial determinants of eosinophil specialization across tissues. Figshare https://doi.org/10.6084/m9.figshare.29816303 (2025).
Acknowledgements
This work was supported by the National Key R&D Program of China (no. 2023YFC2306300) to S.C., the National Natural Science Fund of China Research Fund for International Excellent Young Scientists (no. W2532025) to S.C., the National Natural Science Fund of China (no. 32470920) to S.C., The National Natural Science Fund for Excellent Young Scientists Fund Program (Overseas) to S.C. and the Shanghai Frontiers Science Center of Cellular Homeostasis and Human Diseases to S.C. L.G.N. is supported by the National Natural Science Foundation of China (grant no. 92374205) and Research Fund for International Senior Scientist (grant no. W2431020). We thank Shanghai Jiao Tong University School of Medicine and Shanghai Institute of Immunology for financial support. The sequencing data analysis was supported by the High-performance Computing Platform of the Suzhou Institute of Systems Medicine, Chinese Academy of Medical Sciences and Peking Union Medical College.
Author information
Authors and Affiliations
Contributions
Conception: S.C. Discussion: S.C., Y.H., Z. Li, F.G., C.B., Z. Liu, L.G.N., B.S. and L.L. Research design and experimentation: S.C., Y.H., Z. Li, S.Q., J.X., X.Y., J.H. and Z.X. Data analysis: Y.H., Z. Li, L.W., S.Q., W.T.K., C.W. and S.C. Writing draft and editing: S.C. and Y.H. Project administration: S.C.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Immunology thanks Ariel Munitz and the other anonymous reviewers for their contribution to the peer review of this work. Primary Handling Editor: Nick Bernard, in collaboration with the Nature Immunology team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Single-cell transcriptomics analysis reveals tissue-specific eosinophil states.
a, Schematic gating strategy for flow cytometric sorting of eosinophils across indicated tissues (BM, blood, lung, colon, SI, scWAT, eWAT and BAT). Skin eosinophils were isolated from ear skin. b, Quality control metrics for all cells of single-cell RNA sequencing (scRNA-seq), including gene counts, UMI counts, and mitochondrial read percentages. c, Proportional distribution of eosinophil clusters identified by scRNA-seq within each tissue compartment. d, Heatmap of DEGs across eosinophil clusters. e, Bubble plot of enriched pathway analysis showing significantly altered biological processes (dot size: gene ratio; color: adjusted p-value). f, Top: Hierarchical clustering of gene expression similarity between scRNA-seq clusters and Yu et al. dataset. Bottom: Heatmap of conserved signature genes shared between datasets.
Extended Data Fig. 2 High dimensional protein profiling reveals tissue-imprinted eosinophil identities.
a, Schematic workflow of the InfinityFlow antibody screening pipeline. b, Unified UMAP projection of eosinophils colored by predicted surface markers (left) and tissue origin (right). c, Differentially expressed surface proteins across eosinophil subclusters. d, Multi-tissue overlay plots of representative markers showing expression density distributions across Blood (blue), Lung (green), SI (purple) and eWAT (red).
Extended Data Fig. 3 High dimensional protein profiling reveals tissue-imprinted eosinophil identities.
a, Unified UMAP projection of eosinophils using the optimized panel. b, Marker validation heatmap: Z-score scaled expression of panel-derived signature markers. c, Tissue distribution on UMAP. d, Median fluorescence intensity (MFI) plots of key surface markers across tissues on gated eosinophils (n = 3 /group, skin samples in CLEC4a4 plot: n = 2).
Extended Data Fig. 4 Residency-dependent lifespan stratifies eosinophils across tissues.
a, Heatmap of developmentally regulated genes ordered by Monocle3-predicted trajectory (columns: single cells; rows: core latency genes). b, tdTomato labeling rates in eosinophils from tissue of Ms4a3Cre-Rosa26tdTomato mice (n = 2 /group). c, Ms4a3 expression (qPCR) in granulocyte-monocyte progenitors (GMP), bone marrow eosinophils (BME), and peripheral blood eosinophils (n = 3 /group). The data were normalized to Actb expression and are presented relative to the GMP group (set as 1). d, Fate-mapping principle: Schematic of tamoxifen-inducible Ms4a3CreERT2-Rosa26tdTomato system for eosinophil lineage tracing and pulse-chase design: Experimental timeline for inducible fate-mapping. e, Mathematical framework for estimating eosinophil half-life (t1/2) and lifespan (t5%) (shaded areas: 95% CI). f, Tissue residency kinetics: tdTomato+ decay curves fitted to model (e), (n = 6 for days 3, 7, 14, 21, and 28 in Lung and scWAT, and days 3, 7, and 14 in eWAT; n = 3 for remaining time points). g, Eosinophil frequency and absolute counts across BM, blood and tissue (n = 3 /group). h, in vivo anti-CD45 antibody pulse-chase system schematic for measuring cellular turnover rates. Each dot represents an individual animal; bars show mean ± SEM (****P ≤ 0.0001) by one-way ANOVA with Tukey’s correction (c).
Extended Data Fig. 5 Eosinophil heterogeneity emerges in the long-lived small intestine niche.
a, Heatmap of different subgroups of lung eosinophils based on GSEA pathway enrichment. b, Heatmap of conserved signature genes shared between datasets. c, Cd101 expression on lung eosinophil clusters. d, Flow cytometry gating strategy for Cd101tdTomato reporter (left) vs. anti-CD101 antibody staining (right) in eosinophils and neutrophils. e, Quantification of the Cd101tdTomato and anti-CD101 antibody staining. Each dot represents an individual animal; bars show mean ± SEM (****P ≤ 0.0001) f, Heatmap of different subgroups of colon eosinophils based on GSEA pathway enrichment. g, Heatmap of different subgroups of SIE based on GSEA pathway enrichment. See the Methods section for details of the enrichment procedure. h, Hierarchical clustering of gene expression similarity between our SI eosinophil clusters with public dataset (W. Wang, et al.).
Extended Data Fig. 6 Residency-associated trajectories in the small intestine resolve eosinophil maturation states.
a, Heatmap displaying latency-associated gene expression patterns across eosinophil subsets. Clec12a, Itgal, Fcgr4 and Cd22 were highlighted. b, Heatmap showing expression levels of key surface markers (CD371, CD11a, CD16.2, CD22) across eosinophil subpopulations. c, Schematic gating strategy for identifying five distinct eosinophil subsets. d, Flow cytometry analysis of CD117 (c-Kit) expression in BME (n = 3 /group). e, Representative flow plots supplementing the gating strategy for the five subsets. f, Quantitative distribution of the five subpopulations among different tissues (n = 3 /group in BM, Blood and Colon. n = 4/group in Lung and SI). g, Expression patterns of CLEC4a4 and immune checkpoint molecules (PD-L1 and CD80) across eosinophil subsets in different tissues (n = 3 /group). Values shown as mean ± SEM. Statistic tests: Two-tailed unpaired t-test (d), one-way ANOVA (f, g) with Tukey’s correction for multiple comparisons. ns = not significant; *P ≤ 0.05; **P ≤ 0.01; ***P ≤ 0.001; ****P ≤ 0.0001.
Extended Data Fig. 7 Spatially compartmentalized diversification of intestinal eosinophils.
a, Representative immunofluorescence image of eosinophils on Ms4a3CreERT2-Rosa26tdTomato lung slides. Scale bar=20μm. b, Representative immunofluorescence image of colocalization of blood vessels with eosinophils on wild-type (WT) lung slides. Scale bar=10μm. c, Schematic diagram of intestinal segment selection for analysis. Scale bar=500μm (colon). Scale bar=1 mm (SI). d, Representative immunofluorescence images of CD22 staining in different intestinal segments 3 weeks post-tamoxifen induction on Ms4a3CreERT2-Rosa26tdTomato intestine slides. Representative of 5 independent experiments. Scale bar=20μm. Duode, duodenum; Jeju, jejunum; Ile, ileum; Proxi-Colon, proximal colon; Dis-colon, distal colon.
Extended Data Fig. 8 Spatially compartmentalized diversification of intestinal eosinophils.
a, Representative immunofluorescence images of CD11a staining in different intestinal segments 3 weeks post-tamoxifen induction on Ms4a3CreERT2-Rosa26tdTomato intestine slides. Representative of 5 independent experiments. Scale bar=20μm. b, Quantification of tdTomato+ eosinophil proportions across colonic segments (proximal to distal) by immunofluorescence analysis (n = 3 /group). Each dot represents a region of interest in individual mice. Values shown as mean ± SEM. Statistic tests: Two-tailed unpaired t-test (b), *P ≤ 0.05. Jeju, jejunum; Ile, ileum; Proxi-Colon, proximal colon; Dis-colon, distal colon.
Supplementary information
Supplementary Information (download PDF )
Abstract Schematic and Methodological Tables.
Supplementary Table 1 (download XLSX )
Supplementary Table 1.
Supplementary Table 2 (download XLSX )
Supplementary Table 2.
Supplementary Table 3 (download XLSX )
Supplementary Table 3.
Supplementary Table 4 (download XLSX )
Supplementary Table 4.
Supplementary Table 5 (download XLSX )
Supplementary Table 5.
Supplementary Table 6 (download XLSX )
Supplementary Table 6.
Supplementary Table 7 (download XLSX )
Supplementary Table 7.
Source data
Source Data Fig. 1 (download XLSX )
Statistical Source Data.
Source Data Fig. 2 (download XLSX )
Statistical Source Data.
Source Data Fig. 3 (download XLSX )
Statistical Source Data.
Source Data Fig. 5 (download XLSX )
Statistical Source Data.
Source Data Fig. 6 (download XLSX )
Statistical Source Data.
Source Data Extended Data Fig. 3 (download XLSX )
Statistical Source Data.
Source Data Extended Data Fig. 4 (download XLSX )
Statistical Source Data.
Source Data Extended Data Fig. 5 (download XLSX )
Statistical Source Data.
Source Data Extended Data Fig. 6 (download XLSX )
Statistical Source Data.
Source Data Extended Data Fig. 8 (download XLSX )
Statistical Source Data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hu, Y., Wu, L., Qu, S. et al. Temporal and spatial atlas of eosinophil specialization across tissues. Nat Immunol 27, 364–375 (2026). https://doi.org/10.1038/s41590-025-02382-6
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41590-025-02382-6
This article is cited by
-
Mapping the spatiotemporal adaptation of eosinophils
Nature Immunology (2026)