Abstract
Avian H5N1 influenza viruses are circulating widely in cattle and other mammals and pose a risk for a human pandemic. Previous studies suggest that older humans are more resistant to H5N1 infections due to childhood imprinting with other group 1 viruses (H1N1 and H2N2); however, the immunological basis for this is incompletely understood. Here we measured H5N1 antibody responses in sera from 157 individuals born between 1927 and 2016. We show that antibody titers to historical and recent H5N1 strains are highest in older individuals and correlate more strongly with birth year than with age, consistent with immune imprinting. Young children, who were likely not yet exposed to seasonal influenza viruses, had low levels of H5-specific antibodies. We also measured H5N1 antibody responses in sera from 100 individuals before and after receiving an A/Vietnam/1203/2004 H5N1 vaccine. We found that both younger and older humans produced H5-reactive antibodies to the A/Vietnam/1203/2004 vaccine strain and to a contemporary clade 2.3.4.4b strain, with higher seroconversion rates in young children who had lower levels of antibodies before vaccination. These studies suggest that younger individuals might benefit more from vaccination than older individuals in the event of an H5N1 pandemic.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
All data are shown in the main figures and the Extended Data. Source data are provided with this paper.
Code availability
Code implementing the analyses is available at https://github.com/cobeylab/H5_titers_vs_imprinting.
References
Lewis, N. S. et al. Emergence and spread of novel H5N8, H5N5 and H5N1 clade 2.3.4.4 highly pathogenic avian influenza in 2020. Emerg. Microbes Infect. 10, 148–151 (2021).
Caliendo, V. et al. Transatlantic spread of highly pathogenic avian influenza H5N1 by wild birds from Europe to North America in 2021. Sci. Rep. 12, 11729 (2022).
King, J. et al. Highly pathogenic avian influenza virus incursions of subtype H5N8, H5N5, H5N1, H5N4, and H5N3 in Germany during 2020–21. Virus Evol. 8, veac035 (2022).
Adlhoch, C. et al. Avian influenza overview December 2022 – March 2023. EFSA J. 21, e07917 (2023).
Rijks, J. M. et al. Highly pathogenic avian influenza A(H5N1) virus in wild red foxes, the Netherlands, 2021. Emerg. Infect. Dis. 27, 2960–2962 (2021).
Puryear, W. et al. Outbreak of highly pathogenic avian influenza H5N1 in New England seals. Preprint at bioRxiv https://doi.org/10.1101/2022.07.29.501155 (2022).
Pyankova, O. G. et al. Isolation of clade 2.3.4.4b A(H5N8), a highly pathogenic avian influenza virus, from a worker during an outbreak on a poultry farm, Russia, December 2020. Euro Surveill. 26, 2100439 (2021).
Agüero, M. et al. Highly pathogenic avian influenza A(H5N1) virus infection in farmed minks, Spain, October 2022. Euro Surveill. 28, 2300001 (2023).
Nguyen, T. Q. et al. Emergence and interstate spread of highly pathogenic avian influenza A(H5N1) in dairy cattle. Preprint at bioRxiv https://doi.org/10.1101/2024.05.01.591751 (2024).
Burrough, E. R. et al. Highly pathogenic avian influenza A(H5N1) clade 2.3.4.4b virus infection in domestic dairy cattle and cats, United States, 2024. Emerg. Infect. Dis. 30, 1335–1343 (2024).
Santos, J. J. S. et al. Bovine H5N1 influenza virus binds poorly to human-type sialic acid receptors. Preprint at bioRxiv https://doi.org/10.1101/2024.08.01.606177 (2024).
Chopra, P. et al. Receptor binding specificity of a bovine A(H5N1) influenza virus. Preprint at bioRxiv https://doi.org/10.1101/2024.07.30.605893 (2024).
Neumann, G. & Kawaoka, Y. Highly pathogenic H5N1 avian influenza virus outbreak in cattle: the knowns and unknowns. Nat. Rev. Microbiol. 22, 525–526 (2024).
Moratorio, G. et al. H5N1 influenza: urgent questions and directions. Cell 187, 4546–4548 (2024).
Qin, Y. et al. Differences in the epidemiology of human cases of avian influenza A(H7N9) and A(H5N1) viruses infection. Clin. Infect. Dis. 61, 563–571 (2015).
Cowling, B. J. et al. Comparative epidemiology of human infections with avian influenza A H7N9 and H5N1 viruses in China: a population-based study of laboratory-confirmed cases. Lancet 382, 129–137 (2013).
Krammer, F. et al. Influenza. Nat. Rev. Dis. Primers 4, 3 (2018).
Gostic, K. M., Ambrose, M., Worobey, M. & Lloyd-Smith, J. O. Potent protection against H5N1 and H7N9 influenza via childhood hemagglutinin imprinting. Science 354, 722–726 (2016).
Nachbagauer, R. et al. Age dependence and isotype specificity of influenza virus hemagglutinin stalk-reactive antibodies in humans. mBio 7, e01996-15 (2016).
Yassine, H. M. et al. Use of hemagglutinin stem probes demonstrate prevalence of broadly reactive group 1 influenza antibodies in human sera. Sci. Rep. 8, 8628 (2018).
Sanchez-de Prada, L. et al. Group 1 and group 2 hemagglutinin stalk antibody response according to age. Front. Immunol. 14, 1194073 (2023).
Ledgerwood, J. E. et al. DNA priming and influenza vaccine immunogenicity: two phase 1 open label randomised clinical trials. Lancet Infect. Dis. 11, 916–924 (2011).
Ellebedy, A. H. et al. Induction of broadly cross-reactive antibody responses to the influenza HA stem region following H5N1 vaccination in humans. Proc. Natl Acad. Sci. USA 111, 13133–13138 (2014).
Treanor, J. J., Campbell, J. D., Zangwill, K. M., Rowe, T. & Wolff, M. Safety and immunogenicity of an inactivated subvirion influenza A(H5N1) vaccine. N. Engl. J. Med. 354, 1343–1351 (2006).
Khurana, S. et al. Licensed H5N1 vaccines generate cross-neutralizing antibodies against highly pathogenic H5N1 clade 2.3.4.4b influenza virus. Nat. Med. 30, 2771–2776 (2024).
DiLillo, D. J., Palese, P., Wilson, P. C. & Ravetch, J. V. Broadly neutralizing anti-influenza antibodies require Fc receptor engagement for in vivo protection. J. Clin. Invest. 126, 605–610 (2016).
Arevalo, C. P. et al. Original antigenic sin priming of influenza virus hemagglutinin stalk antibodies. Proc. Natl Acad. Sci. USA 117, 17221–17227 (2020).
Hobson, D., Curry, R. L., Beare, A. S. & Ward-Gardner, A. The role of serum haemagglutination-inhibiting antibody in protection against challenge infection with influenza A2 and B viruses. J. Hyg. 70, 767–777 (1972).
Christensen, S. R. et al. Assessing the protective potential of H1N1 influenza virus hemagglutinin head and stalk antibodies in humans. J. Virol. 93, e02134-18 (2019).
Hoy, G. et al. Humoral correlates of protection against influenza A/H3N2 virus infection. J. Infect. Dis. 230, 1319–1328 (2024).
Ng, S. et al. Novel correlates of protection against pandemic H1N1 influenza A virus infection. Nat. Med. 25, 962–967 (2019).
Webby, R. J. The practical longevity of stockpiled A(H5N1) influenza vaccine. Nat. Med. 30, 2729–2730 (2024).
Whittle, J. R. et al. Flow cytometry reveals that H5N1 vaccination elicits cross-reactive stem-directed antibodies from multiple Ig heavy-chain lineages. J. Virol. 88, 4047–4057 (2014).
Corbett, K. S. et al. Design of nanoparticulate group 2 influenza virus hemagglutinin stem antigens that activate unmutated ancestor B cell receptors of broadly neutralizing antibody lineages. mBio 10, e02810-18 (2019).
Bushell, K. M., Sollner, C., Schuster-Boeckler, B., Bateman, A. & Wright, G. J. Large-scale screening for novel low-affinity extracellular protein interactions. Genome Res. 18, 622–630 (2008).
Doud, M. B., Hensley, S. E. & Bloom, J. D. Complete mapping of viral escape from neutralizing antibodies. PLoS Pathog. 13, e1006271 (2017).
Lee, J. M. et al. Deep mutational scanning of hemagglutinin helps predict evolutionary fates of human H3N2 influenza variants. Proc. Natl Acad. Sci. USA 115, E8276–E8285 (2018).
Bloom, J. D., Gong, L. I. & Baltimore, D. Permissive secondary mutations enable the evolution of influenza oseltamivir resistance. Science 328, 1272–1275 (2010).
World Health Organization. Manual for the Laboratory Diagnosis and Virological Surveillance of Influenza (World Health Organization, 2011).
Gostic, K. imprinting: Calculate birth year-specific probabilities of immune imprinting to influenza. R package version 0.1.0. GitHub cobeylab.github.io/imprinting/ (2022).
Acknowledgements
We thank the Penn Medicine BioBank and Children’s Hospital of Philadelphia as a resource for obtaining serum from humans with different birth years. We thank the NIAID and the VTEU clinical study teams from DMID 04-063, DMID 04-076 and DMID04-0077 for providing serum samples from clinical studies. This project was funded in part with federal funds from the National Institute of Allergy and Infectious Diseases, National Institutes of Health, Department of Health and Human Services, under contract no. 75N93021C00015 (S.E.H., S. Cobey and S. Cherry) and grant number R01AI08686 (S.E.H.). S.E.H. holds an Investigators in the Pathogenesis of Infectious Disease Award from the Burroughs Wellcome Fund.
Author information
Authors and Affiliations
Contributions
S.E.H., T.A.G., J.L., S.H.L., G.S., C.F. and J.J.S.S. designed experiments. T.A.G., J.L., S.H.L., G.S., J.J.S.S., G.H., C.F., R.K.A., N.Y., J.T.O., K.A.H. and T.E. completed experiments and analyzed data. K.K., M.C.V. and S. Cobey performed modeling studies. S.E.H. wrote the paper, and all authors contributed to editing the paper. D.C.S., S. Cherry, S. Cobey and S.E.H. supervised experiments and data analyses. S.E.H. obtained funding for the study.
Corresponding author
Ethics declarations
Competing interests
S.E.H. is a co-inventor on patents that describe the use of nucleoside-modified mRNA as a vaccine platform. S.E.H. reports receiving consulting fees from Sanofi, Pfizer, Lumen, Novavax and Merck. T.A.G. was an employee of the University of Pennsylvania when the work was completed and is now an employee of GSK. The authors declare no other competing interests.
Peer review
Peer review information
Nature Medicine thanks the anonymous reviewers for their contribution to the peer review of this work. Primary Handling Editor: Saheli Sadanand, in collaboration with the Nature Medicine team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Imprinting probabilities of influenza subtypes by year of birth.
Probabilities that an individual was 1st exposed (that is ‘imprinted’) in childhood with an (a) H1N1 virus, an (b) H2N2 virus, (c) group 1 viruses (H1N1 or H2N2), or an (d) H3N2 virus based on their year of birth. Birth year-specific probabilities of immune imprinting to different influenza subtypes were generated using the methods described in Gostic et al.18 and the imprinting R package40.
Extended Data Fig. 2 Group 1 immune imprinting primes antibodies that cross-react to 2.3.4.4b H5N1 isolate currently circulating in cattle.
Sera samples were collected from healthy individuals (n = 157) at the Hospital of the University of Pennsylvania and Children’s Hospital of Philadelphia in 2017 and we quantified IgG binding to clade 2.3.4.4b A/Dairy Cattle/Texas/24-008749-002-v/2024 full length H5 HA. Vertical dashed lines mark years of the 1957 H2N2 and 1968 H3N2 pandemics and 1977 reemergence of H1N1. Each circle represents a geometric mean antibody titer in serum from a single individual from two independent replicates. Trend lines are locally estimated scatterplot smoothing (LOESS) curves (smoothing parameter = 0.4, degree = 2) with 95% CIs.
Extended Data Fig. 3 Most humans possess low levels of neutralizing antibodies against clade 1 and clade 2.3.4.4b H5N1.
Sera samples were collected from healthy individuals (n = 157) at the Hospital of the University of Pennsylvania and Children’s Hospital of Philadelphia in 2017 and we performed neutralization assays using conditionally replicative viruses expressing the HAs from (a) clade 1 A/Vietnam/1203/2004 and (b) clade 2.3.4.4b A/Pheasant/New York/22-009066-011/2022. Neutralizing titers are reported as reciprocal dilutions of serum that inhibit 90% of virus infection (NT90). Vertical dashed lines mark years of the 1957 H2N2 and 1968 H3N2 pandemics and 1977 reemergence of H1N1. Each circle represents a geometric mean antibody titer in serum from a single individual from two independent replicates. Trend lines are locally estimated scatterplot smoothing (LOESS) curves (smoothing parameter = 0.4, degree = 2) with 95% CIs.
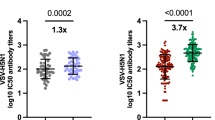
Extended Data Fig. 4 H5N1 vaccination elicits strong H5 stalk-reactive antibodies in children.
Sera samples were obtained from participants (n = 100) before (day 0) and 28 days after receiving a first and second dose of an unadjuvanted A/Vietnam/1203/2004 H5N1 vaccine in 2005-2006. We quantified IgG binding to a ‘headless’ A/Vietnam/1203/2004 H5 stalk (a-c), and we calculated titer fold change by dividing day 28 post-boost titers by day 0 titers (d). We also measured antibody binding and fold change to clade 1 A/Vietnam/1203/2004 full length H5 HA (e-h) and clade 2.3.4.4b A/Pheasant/New York/22-009066-011/2022 full length H5 HA (i-l). Vertical dashed lines mark years of the 1957 H2N2 and 1968 H3N2 pandemics and 1977 reemergence of H1N1. Each circle represents a geometric mean antibody titer in serum from a single individual from two independent replicates. Samples from adults are grey and children are blue. Trend lines are locally estimated scatterplot smoothing (LOESS) curves (smoothing parameter = 0.4, degree = 2) with 95% CIs, fitted to adult samples.
Extended Data Fig. 5 H5N1 vaccination elicits antibodies that efficiently neutralize vaccine virus and mediate ADCC to matched and mismatched H5N1 viruses.
Sera samples were obtained from participants (n = 100) before (day 0) and 28 days after receiving a first and second dose of an unadjuvanted A/Vietnam/1203/2004 H5N1 vaccine in 2005-2006. We quantified levels of antibodies that neutralized virus with a clade 1 A/Vietnam/1203/2004 H5 HA (a-d) and clade 2.3.4.4b A/Pheasant/New York/22-009066-011/2022 H5 HA (e-h). Neutralizing titers are reported as reciprocal dilutions of serum that inhibit 90% of virus infection (NT90). We quantified levels of antibodies that mediated antibody-dependent cellular cytotoxicity (ADCC) with HAs from a clade 1 A/Vietnam/1203/2004 H5N1 virus (i-l) and a clade 2.3.4.4b A/Pheasant/New York/22-009066-011/2022 H5N1 virus (m-p). Fold change in panels d, h, l, and p were calculated by dividing day 28 post-boost values by day 0 values. Samples that did not have neutralizing antibodies were assigned a value of 10. Each circle represents a geometric mean antibody titer in serum from a single individual from two independent replicates. Samples from adults are grey and children are blue. Trend lines are locally estimated scatterplot smoothing (LOESS) curves (smoothing parameter = 0.4, degree = 2) with 95% CIs, fitted to adult samples.
Supplementary information
Source data
Source Data Figs. 1 and 2, Extended Data Figs. 2–5 and Extended Data Tables 1–4 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Garretson, T.A., Liu, J., Li, S.H. et al. Immune history shapes human antibody responses to H5N1 influenza viruses. Nat Med 31, 1454–1458 (2025). https://doi.org/10.1038/s41591-025-03599-6
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41591-025-03599-6
This article is cited by
-
Adjuvanted influenza vaccination increases pre-existing H5N1 cross-reactive antibodies
Nature Communications (2026)
-
Immunological sin: how a person’s earliest flu infections dictate life-long immunity
Nature (2025)
-
Immunogenicity and safety of a rabies-based highly pathogenic influenza A virus H5 vaccine in cattle
npj Vaccines (2025)
-
Pre-existing cross-reactive immunity to highly pathogenic avian influenza 2.3.4.4b A(H5N1) virus in the United States
Nature Communications (2025)
-
Human infections with highly pathogenic avian influenza A(H5N1) viruses in the United States from March 2024 to May 2025
Nature Medicine (2025)