Abstract
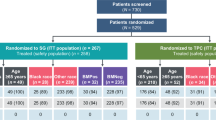
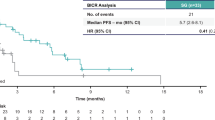
Chemotherapy remains a standard treatment option for metastatic triple-negative breast cancer (TNBC) but is associated with limited survival. Although some targeted antibody–drug conjugates have demonstrated clinical benefits and are considered standard therapy, persistent unmet medical needs remain due to varying accessibility. The OptiTROP-Breast01 phase 3 trial assessed sacituzumab tirumotecan (sac-TMT) versus chemotherapy in patients with locally recurrent or metastatic TNBC who had received two or more prior therapies, including at least one for metastatic disease. Patients were randomized to sac-TMT (n = 130) or chemotherapy (n = 133). The primary endpoint of progression-free survival (PFS) by blinded independent central review (BICR) was met based on the protocol-specified interim analysis. At final analysis, the median PFS by BICR was 6.7 (95% confidence interval (CI), 5.5–8.0) months with sac-TMT and 2.5 (95% CI, 1.7–2.7) months with chemotherapy (hazard ratio (HR), 0.32; 95% CI, 0.24–0.44; P < 0.00001). Concurrently, at the protocol-specified interim analysis for overall survival (OS), the median OS was not reached (95% CI, 11.2 months to not estimable (NE)) with sac-TMT and 9.4 (95% CI, 8.5–11.7) months with chemotherapy (HR, 0.53; 95% CI, 0.36–0.78; P = 0.0005). The percentage of patients with an objective response was 45.4% with sac-TMT and 12.0% with chemotherapy. The median duration of response was 7.1 (95% CI, 5.6–NE) months with sac-TMT and 3.0 (95% CI, 2.5–NE) months with chemotherapy. The most common treatment-related adverse event with sac-TMT was hematologic toxicity. Sac-TMT demonstrated statistically significant and clinically meaningful improvements in PFS compared to chemotherapy, with a manageable safety profile. The study findings support sac-TMT as an additional effective treatment option for pretreated metastatic TNBC. ClinicalTrials.gov identifier: NCT05347134.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
To protect the privacy of study participants and proprietary information, the sponsor shares anonymized individual patient data upon reasonable request or as required by law or regulation with qualified external researchers based on submitted curriculum vitae and reflecting non-conflict of interest. The data provided include demographic, efficacy and safety information. Approval of such requests is at the sponsor’s discretion and depends on the nature of the request, the merit of the research proposed, the availability of the data and the intended use of the data. Data requests should be sent to mict@kelun.com. In response to the inquiry, the timeframe for responding to requests is approximately 2 weeks. The redacted version of the trial protocol and statistical analysis plan is in the supplementary documents.
References
Cleator, S., Heller, W. & Coombes, R. C. Triple-negative breast cancer: therapeutic options. Lancet Oncol. 8, 235–244 (2007).
Stecklein, S. R. et al. ctDNA and residual cancer burden are prognostic in triple-negative breast cancer patients with residual disease. NPJ Breast Cancer 9, 10 (2023).
Leon-Ferre, R. A. & Goetz, M. P. Advances in systemic therapies for triple negative breast cancer. BMJ 381, e071674 (2023).
Foulkes, W. D., Smith, I. E. & Reis-Filho, J. S. Triple-negative breast cancer. N. Engl. J. Med. 363, 1938–1948 (2010).
Kazmi, S., Chatterjee, D., Raju, D., Hauser, R. & Kaufman, P. A. Overall survival analysis in patients with metastatic breast cancer and liver or lung metastases treated with eribulin, gemcitabine, or capecitabine. Breast Cancer Res. Treat. 184, 559–565 (2020).
O’Shaughnessy, J. et al. Assessment of sacituzumab govitecan (SG) versus treatment of physician’s choice (TPC) cohort by agent in the phase 3 ASCENT study of patients (pts) with metastatic triple-negative breast cancer (mTNBC). J. Clin. Oncol. 39, 1077-1077 (2021).
Modi, S. et al. Trastuzumab deruxtecan in previously treated HER2-low advanced breast cancer. N. Engl. J. Med. 387, 9–20 (2022).
Seligson, J. M., Patron, A. M., Berger, M. J., Harvey, R. D. & Seligson, N. D. Sacituzumab govitecan-hziy: an antibody-drug conjugate for the treatment of refractory, metastatic, triple-negative breast cancer. Ann. Pharmacother. 55, 921–931 (2021).
Bardia, A. et al. Sacituzumab govitecan in metastatic triple-negative breast cancer. N. Engl. J. Med. 384, 1529–1541 (2021).
Bardia, A. et al. Final results from the randomized phase III ASCENT clinical trial in metastatic triple-negative breast cancer and association of outcomes by human epidermal growth factor receptor 2 and trophoblast cell surface antigen 2 expression. J. Clin. Oncol. 42, 1738–1744 (2024).
Rodon, J. et al. 514O An open-label, global, first-in-human study of SKB264 in patients with locally advanced or metastatic solid tumors. Ann. Oncol. 32, S585 (2021).
Yin, Y. M. et al. Efficacy and safety of SKB264 for previously treated metastatic triple negative breast cancer in phase 2 study. Cancer Res. https://doi.org/10.1158/1538-7445.SABCS22-OT1-03-02 (2023).
Yin, Y. M. et al. Abstract PS08-08: Updated efficacy and safety of SKB264 (MK-2870) for previously treated metastatic triple negative breast cancer (mTNBC) in phase 2 study. Cancer Res. https://doi.org/10.1158/1538-7445.SABCS23-PS08-08 (2024).
Cheng, Y. et al. Preclinical profiles of SKB264, a novel anti-TROP2 antibody conjugated to topoisomerase inhibitor, demonstrated promising antitumor efficacy compared to IMMU-132. Front. Oncol. 12, 951589 (2022).
Ouyang, Q. et al. 380MO - SKB264 (MK-2870) in previously treated hormone receptor-positive (HR+)/HER2-negative metastatic breast cancer (mBC): results from a phase I/II, single-arm, basket trial. Ann. Oncol. 34, S337 (2023).
Fang, W. F. et al. SKB264 (TROP2-ADC) for the treatment of patients with advanced NSCLC: efficacy and safety data from a phase 2 study. J. Clin. Oncol. 41, 9114-9114 (2023).
Rodon, J. et al. Abstract CT038: Preliminary efficacy and safety results of anti-TROP2 ADC SKB264 (MK-2870) in patients (pts) with previously treated advanced gastric (G) or gastroesophageal junction (GEJ) cancer from a phase 2 study. Cancer Res. https://doi.org/10.1158/1538-7445.am2024-ct038 (2024).
Cortés, J. et al. KEYNOTE-119: phase III study of pembrolizumab (pembro) versus single-agent chemotherapy (chemo) for metastatic triple negative breast cancer (mTNBC). Ann. Oncol. 30, V859–V860 (2019).
Won, K. A. & Spruck, C. Triple‑negative breast cancer therapy: current and future perspectives (Review). Int. J. Oncol. 57, 1245–1261 (2020).
Mougalian, S. S., Kish, J. K., Zhang, J., Liassou, D. & Feinberg, B. A. Effectiveness of eribulin in metastatic breast cancer: 10 years of real-world clinical experience in the United States. Adv. Ther. 38, 2213–2225 (2021).
Schmid, P. et al. 166MO Datopotamab deruxtecan (Dato-DXd) + durvalumab (D) as first-line (1L) treatment for unresectable locally advanced/metastatic triple-negative breast cancer (a/mTNBC): initial results from BEGONIA, a phase Ib/II study. Ann. Oncol. 33, S199 (2022).
Schmid, P. et al. 181O Interim analysis (IA) of the atezolizumab (atezo) + sacituzumab govitecan (SG) arm in patients (pts) with triple-negative breast cancer (TNBC) in MORPHEUS-pan BC: a phase Ib/II study of multiple treatment (tx) combinations in pts with locally advanced/metastatic BC (LA/mBC). ESMO Open 9, 103203 (2024).
Bardia, A. et al. Biomarker analyses in the phase III ASCENT study of sacituzumab govitecan versus chemotherapy in patients with metastatic triple-negative breast cancer. Ann. Oncol. 32, 1148–1156 (2021).
Cortés, J. et al. Trastuzumab deruxtecan versus trastuzumab emtansine for breast cancer. N. Engl. J. Med. 386, 1143–1154 (2022).
André, F. et al. Trastuzumab deruxtecan versus treatment of physician’s choice in patients with HER2-positive metastatic breast cancer (DESTINY-Breast02): a randomised, open-label, multicentre, phase 3 trial. Lancet 401, 1773–1785 (2023).
Saura, C. et al. Trastuzumab deruxtecan in previously treated patients with HER2-positive metastatic breast cancer: updated survival results from a phase II trial (DESTINY-Breast01). Ann. Oncol. 35, 302–307 (2024).
Powles, T. et al. Enfortumab vedotin in previously treated advanced urothelial carcinoma. N. Engl. J. Med. 384, 1125–1135 (2021).
Xu, B. et al. A phase IIb, single arm, multicenter trial of sacituzumab govitecan in Chinese patients with metastatic triple-negative breast cancer who received at least two prior treatments. Int. J. Cancer 152, 2134–2144 (2023).
Acknowledgements
We thank the patients who volunteered to participate in the OptiTROP-Breast01 study and their families for their valuable contribution and commitment as well as the dedicated clinical trial investigators and their devoted team members for participating in the OptiTROP-Breast01 study. This study was funded by Sichuan Kelun-Biotech Biopharmaceutical Co., Ltd. and was partly supported by the CAMS Innovation Fund for Medical Sciences (CIFMS; 2021-I2M-1-014 received by B.X. and 2023-I2M-C & T-B-077 received by Y.F.) and by Major Project of Medical Oncology Key Foundation of Cancer Hospital Chinese Academy of Medical Sciences CICAMS-MOMP202203 (received by Y.F.). Sichuan Kelun-Biotech Biopharmaceutical Co., Ltd. participated in study conception and design, data analysis, data interpretation, drafting or revising the paper and approval of the final version of the paper.
Author information
Authors and Affiliations
Contributions
Conception and design: B.X., X.J. and Y.D. Provision of study materials or patients: B.X., Y.Y., Y.F., Q.O., L.S., X.W., W.L., M.L., X.Y., S.W., T.S., Y.T., X.T., Z.T. and Z.S. Collection and assembly of data: B.X., Y.Y., Y.F., Q.O., L.S., X.W., W.L., M.L., X.Y., S.W., T.S., Y.T., X.T., Z.T. and Z.S. Data analysis and interpretation: B.X., Y.Y., J.G., X.J., Y.D. and G.L. Writing, review and editing of the paper: all authors. Final approval of the paper: all authors. Accountable for all aspects of the work: all authors.
Corresponding author
Ethics declarations
Competing interests
B.X. has served as advisor or consultant for Novartis and AstraZeneca. J.G., X.J., Y.D. and G.L. are employees of Sichuan Kelun-Biotech Biopharmaceutical Co., Ltd. The other authors declare no competing interests.
Peer review
Peer review information
Nature Medicine thanks Min Yan and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Ulrike Harjes, in collaboration with the Nature Medicine team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Kaplan-Meier Curves for PFS by BICR Assessment (Interim Analysis).
Data cutoff: June 21, 2023; the protocol-specified interim analysis of PFS. The 95% CI of median PFS was determined using the Brookmeyer and Crowley method with log-log transformation. HR along with 95% CI was estimated by a stratified Cox proportional hazards model. The P value was calculated by a stratified log-rank test. The multiplicity-adjusted, one-sided alpha at this interim analysis (determined using the Lan-DeMets O’Brien-Fleming spending function) was 0.0104. The exact one-sided P value was 2.0 × 10−11. BICR, blinded independent central review; CI, confidence interval; HR, hazard ratio; PFS, progression-free survival; sac-TMT, sacituzumab tirumotecan.
Extended Data Fig. 2 Kaplan-Meier Curves for PFS by Investigator Assessment (Interim Analysis).
Data cutoff: June 21, 2023; the protocol-specified interim analysis of PFS. The 95% CI of median PFS was determined using the Brookmeyer and Crowley method with log-log transformation. HR along with 95% CI was estimated by a stratified Cox proportional hazards model. The P value was calculated by a stratified log-rank test. The exact one-sided P value was 4.0 × 10−11. CI, confidence interval; HR, hazard ratio; PFS, progression-free survival; sac-TMT, sacituzumab tirumotecan.
Extended Data Fig. 3 Kaplan-Meier Curves for PFS by Investigator Assessment (Final Analysis).
Data cutoff: November 30, 2023; the protocol-specified final analysis of PFS. The 95% CI of median PFS was determined using the Brookmeyer and Crowley method with log-log transformation. HR along with 95% CI was estimated by a stratified Cox proportional hazards model. The P value was calculated by a stratified log-rank test. The exact one-sided P value was 2.9 × 10−14. CI, confidence interval; HR, hazard ratio; PFS, progression-free survival; sac-TMT, sacituzumab tirumotecan.
Extended Data Fig. 4 Forest Plot of PFS (per Investigator Assessment) in Key Patient Subgroups.
Data cutoff: November 30, 2023; the protocol-specified final analysis of PFS. Statistical analysis was not performed when the number of patients in the subgroup was less than 10% of the total patients. HRs along with 95% CIs were estimated using an unstratified Cox proportional hazards model in each subgroup. CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; HER2, human epidermal growth factor receptor 2; HR, hazard ratio; NE, not estimable; PD-1: programmed death-1; PD-L1: programmed death-ligand 1; PFS, progression-free survival; sac-TMT, sacituzumab tirumotecan; TNBC, triple-negative breast cancer.
Extended Data Fig. 5 Forest Plot of OS in Key Patient Subgroups.
Data cutoff: November 30, 2023; the protocol-specified interim analysis of OS. Statistical analysis was not performed when the number of patients in the subgroup was less than 10% of the total patients. HRs along with 95% CIs were estimated using an unstratified Cox proportional hazards model in each subgroup. CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; HER2, human epidermal growth factor receptor 2; HR, hazard ratio; NE, not estimable; NR, not reached; OS, overall survival; PD-1: programmed death-1; PD-L1: programmed death-ligand 1; sac-TMT, sacituzumab tirumotecan; TNBC, triple-negative breast cancer.
Extended Data Fig. 6 Best Change in Target Lesions by BICR.
Data cutoff: November 30, 2023. BICR, blinded independent central review; IQR, interquartile range; sac-TMT, sacituzumab tirumotecan.
Extended Data Fig. 7 Overall Response (per BICR) by TROP2 Expression.
Data cutoff: November 30, 2023. BICR, blinded independent central review; Chemo, chemotherapy; ORR, objective response rate; sac-TMT, sacituzumab tirumotecan; TROP2, trophoblast cell surface antigen 2.
Supplementary information
Supplementary Information (download PDF )
Supplementary Tables 1–4, Figs. 1 and 2, study protocol and statistical analysis plan.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yin, Y., Fan, Y., Ouyang, Q. et al. Sacituzumab tirumotecan in previously treated metastatic triple-negative breast cancer: a randomized phase 3 trial. Nat Med 31, 1969–1975 (2025). https://doi.org/10.1038/s41591-025-03630-w
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41591-025-03630-w
This article is cited by
-
Phase II trial of anlotinib-chemotherapy combination in pretreated HER2-negative metastatic breast cancer: therapeutic efficacy and proteomic biomarker profiling
npj Breast Cancer (2026)
-
Cost-effectiveness of antibody-drug conjugates in previously treated metastatic triple-negative breast cancer in China
Health Economics Review (2025)
-
Technological advancements in antibody-based therapeutics for treatment of diseases
Journal of Biomedical Science (2025)
-
Antibody–drug conjugates in breast cancer: mechanisms of resistance and future therapeutic perspectives
npj Breast Cancer (2025)
-
Sacituzumab tirumotecan improves OS in mTNBC
Nature Reviews Clinical Oncology (2025)