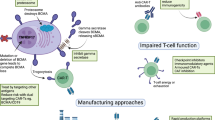
Abstract
In vivo chimeric antigen receptor (CAR)-T cell generation can bypass ex vivo manufacturing and lymphodepletion, potentially simplifying and accelerating access to cellular therapy; preliminary clinical experience supports feasibility and suggests preliminary efficacy. This phase 1, single-arm, open-label trial evaluated the safety and tolerability of ESO-T01, a nanobody-directed, immune-shielded lentiviral vector encoding a humanized anti-B cell maturation antigen (BCMA) CAR, in adults with relapsed or refractory multiple myeloma. ESO-T01 was administered as a single intravenous infusion of 0.2 × 109 transduction units without leukapheresis, ex vivo manufacturing or lymphodepleting chemotherapy. Five heavily pretreated male patients (median three prior lines) were consecutively enrolled and followed for a median of 6.0 months. The trial was stopped early in 2025, and no further enrollment was performed. The primary endpoint was safety and tolerability, and secondary endpoints included efficacy, pharmacokinetics and pharmacodynamics of ESO-T01. No dose-limiting toxicities occurred. All patients developed grade 3 or higher adverse events. Cytokine release syndrome occurred in four patients (three grade 3 and one grade 2) and was managed with corticosteroids, tocilizumab, or supportive care. The most frequent toxicities were transient cytopenias and reversible hepatic enzyme elevations, and three patients experienced grade 2 infections. One patient developed grade 1 immune effector cell-associated neurotoxicity and died from extramedullary lesion-related spinal cord compression. Preliminary antimyeloma activity was observed: four of five patients achieved objective responses, including three stringent complete remissions, with minimal residual disease negativity (10−5) in all evaluable responders (4/4) by day 60. These findings provide preliminary evidence on the feasibility and safety of in vivo CAR-T generation using an immune-shielded vector. ClinicalTrials.gov registration: NCT06791681.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All primary data supporting the findings of this study are included in this article and its supplementary files. Extended data tables and figures, together with the supplementary information, provide patient-level data for safety, response, pharmacokinetic and immunophenotyping analyses as well as vector construct, integration site analyses and gating strategies.
The clinical datasets generated and analyzed during this study are not publicly available due to proprietary restrictions and to protect patient privacy and sensitive information. All individual-level clinical data have been deidentified and are stored on the secure data server of Tongji Hospital. Interested researchers may contact the corresponding author to request access to the datasets. Access requires completion of a data access agreement describing the intended research use; commercial or for-profit use is prohibited. After approval by the applicant’s institution and our institutional data access committee, deidentified individual participant-level data will be provided within 6 months after publication and will remain accessible for at least 5 years. The full trial protocol and the statistical analysis plan are provided in the supplementary materials.
All requests for data access should be directed to the corresponding author (cunrui5650@hust.edu.cn), and responses will be provided within 10 working days.
References
Kumar, S. K. et al. Multiple myeloma. Nat. Rev. Dis. Primers 3, 17046 (2017).
Kumar, S. K. et al. Risk of progression and survival in multiple myeloma relapsing after therapy with IMiDs and bortezomib: a multicenter international myeloma working group study. Leukemia 26, 149–157 (2012).
Cappell, K. M. & Kochenderfer, J. N. Long-term outcomes following CAR T cell therapy: what we know so far. Nat. Rev. Clin. Oncol. 20, 359–371 (2023).
Yu, B., Jiang, T. & Liu, D. BCMA-targeted immunotherapy for multiple myeloma. J. Hematol. Oncol. 13, 125 (2020).
Munshi, N. C. et al. Idecabtagene vicleucel in relapsed and refractory multiple myeloma. N. Engl. J. Med. 384, 705–716 (2021).
Berdeja, J. G. et al. Ciltacabtagene autoleucel, a B-cell maturation antigen-directed chimeric antigen receptor T-cell therapy in patients with relapsed or refractory multiple myeloma (CARTITUDE-1): a phase 1b/2 open-label study. Lancet 398, 314–324 (2021).
Wang, D. et al. A phase 1 study of a novel fully human BCMA-targeting CAR (CT103A) in patients with relapsed/refractory multiple myeloma. Blood 137, 2890–2901 (2021).
Chen, W. et al. Phase II study of fully human BCMA-targeting CAR-T cells (zevorcabtagene autoleucel) in patients with relapsed/refractory multiple myeloma. Exp. Hematol. Oncol.14, 119 (2025).
Locke, F. L. et al. Impact of vein-to-vein time in patients with R/R LBCL treated with axicabtagene ciloleucel. Blood Adv. 9, 2663–2676 (2025).
Amini, L. et al. Preparing for CAR T cell therapy: patient selection, bridging therapies and lymphodepletion. Nat. Rev. Clin. Oncol. 19, 342–355 (2022).
Ayala Ceja, M., Khericha, M., Harris, C. M., Puig-Saus, C. & Chen, Y. Y. CAR-T cell manufacturing: major process parameters and next-generation strategies. J. Exp. Med. 221, e20230903 (2024).
Mailankody, S. et al. Allogeneic BCMA-targeting CAR T cells in relapsed/refractory multiple myeloma: phase 1 UNIVERSAL trial interim results. Nat. Med. 29, 422–429 (2023).
Marin, D. et al. Safety, efficacy and determinants of response of allogeneic CD19-specific CAR-NK cells in CD19+ B cell tumors: a phase 1/2 trial. Nat. Med. 30, 772–784 (2024).
Heczey, A. et al. Anti-GD2 CAR-NKT cells in relapsed or refractory neuroblastoma: updated phase 1 trial interim results. Nat. Med. 29, 1379–1388 (2023).
Diorio, C., Teachey, D. T. & Grupp, S. A. Allogeneic chimeric antigen receptor cell therapies for cancer: progress made and remaining roadblocks. Nat. Rev. Clin. Oncol. 22, 10–27 (2025).
Labanieh, L. & Mackall, C. L. CAR immune cells: design principles, resistance and the next generation. Nature 614, 635–648 (2023).
Hunter, T. L. et al. In vivo CAR T cell generation to treat cancer and autoimmune disease. Science 388, 1311–1317 (2025).
Nicolai, C. J. et al. In vivo CAR T-cell generation in nonhuman primates using lentiviral vectors displaying a multidomain fusion ligand. Blood 144, 977–987 (2024).
Nawaz, W. et al. AAV-mediated in vivo CAR gene therapy for targeting human T-cell leukemia. Blood Cancer J. 11, 119 (2021).
Bozza, M. et al. A nonviral, nonintegrating DNA nanovector platform for the safe, rapid, and persistent manufacture of recombinant T cells. Sci. Adv. 7, eabf1333 (2021).
Rurik, J. G. et al. CAR T cells produced in vivo to treat cardiac injury. Science 375, 91–96 (2022).
Xu, J. et al. In-vivo B-cell maturation antigen CAR T-cell therapy for relapsed or refractory multiple myeloma. Lancet 406, 228−231 (2025).
Yakoub-Agha, I. et al. Management of adults and children undergoing chimeric antigen receptor T-cell therapy: best practice recommendations of the European Society for Blood and Marrow Transplantation (EBMT) and the Joint Accreditation Committee of ISCT and EBMT (JACIE). Haematologica 105, 297–316 (2020).
Fan, H. et al. Current treatment paradigm and survival outcomes among patients with newly diagnosed multiple myeloma in China: a retrospective multicenter study. Cancer Biol. Med. 20, 77–87 (2023).
Tang, W. et al. Treatment attrition rates and relevant risk factors in multiple myeloma: a real-world study in China. Front. Pharmacol. 14, 979111 (2023).
Cowan, A. J. et al. The global state of hematopoietic cell transplantation for multiple myeloma: an analysis of the Worldwide Network of Blood and Marrow Transplantation Database and the Global Burden of Disease Study. Biol. Blood Marrow Transplant. 26, 2372–2377 (2020).
Milani, M. et al. Phagocytosis-shielded lentiviral vectors improve liver gene therapy in nonhuman primates. Sci. Transl. Med. 11, eaav7325 (2019).
Milani, M. et al. Genome editing for scalable production of alloantigen-free lentiviral vectors for in vivo gene therapy. EMBO Mol. Med. 9, 1558–1573 (2017).
Wang, Y. & Shao, W. Innate immune response to viral vectors in gene therapy. Viruses 15, 1801 (2023).
Kim, J. T. et al. Dendritic cell−targeted lentiviral vector immunization uses pseudotransduction and DNA-mediated STING and cGAS activation. Sci. Immunol. 2, eaal1329 (2017).
Ganaie, S. S., Wang, Z., Su, B. & Mir, S. Editorial: Virus-induced innate immune response and inflammation. Front. Microbiol. 14, 1213270 (2023).
Huang, J., Rong, L., Wang, E. & Fang, Y. Pseudoprogression of extramedullary disease in relapsed acute lymphoblastic leukemia after CAR T-cell therapy. Immunotherapy 13, 5–10 (2021).
Danylesko, I. et al. Immune imitation of tumor progression after anti-CD19 chimeric antigen receptor T cells treatment in aggressive B-cell lymphoma. Bone Marrow Transplant. 56, 1134–1143 (2021).
Ivashkiv, L. B. & Donlin, L. T. Regulation of type I interferon responses. Nat. Rev. Immunol. 14, 36–49 (2014).
Xiao, X. et al. Mechanisms of cytokine release syndrome and neurotoxicity of CAR T-cell therapy and associated prevention and management strategies. J. Exp. Clin. Cancer Res. 40, 367 (2021).
Butman, J. et al. Steroid prophylaxis in patients receiving CAR T-cell therapy for relapsed and refractory non-Hodgkin lymphoma. Blood 144, 4507–4507 (2024).
Costa, B. A. et al. Prognostic impact of corticosteroid and tocilizumab use following chimeric antigen receptor T-cell therapy for multiple myeloma. Blood Cancer J. 14, 84 (2024).
Li, C. et al. Equecabtagene autoleucel in patients with relapsed or refractory multiple myeloma: the FUMANBA-1 nonrandomized clinical trial. JAMA Oncol. 10, 1681–1688 (2024).
Dogan, A. et al. B-cell maturation antigen expression across hematologic cancers: a systematic literature review. Blood Cancer J. 10, 73 (2020).
Kim, I., Wu, G., Chai, N., Jordan, S. & Klein, A. Dynamic BCMA expression by alloreactive B cells coupled with donor specific antibody production during de novo alloantibody responses. J. Heart Lung Transplant. 38, S249 (2019).
Martin, J. et al. B-cell maturation antigen (BCMA) as a biomarker and potential treatment target in systemic lupus erythematosus. Int. J. Mol. Sci. 25, 10845 (2024).
Maeda, K. et al. Interleukin-6 aborts lymphopoiesis and elevates production of myeloid cells in systemic lupus erythematosus-prone B6.Sle1.Yaa animals. Blood 113, 4534–4540 (2009).
Sun, Y. et al. Early B lymphocyte subsets in blood predict prognosis in sepsis. Front. Immunol. 15, 1437864 (2024).
Wang, X. et al. Dysregulated hematopoiesis in bone marrow marks severe COVID-19. Cell Discov. 7, 60 (2021).
Raje, N. et al. Anti-BCMA CAR T-cell therapy bb2121 in relapsed or refractory multiple myeloma. N. Engl. J. Med. 380, 1726–1737 (2019).
Martin, N. et al. Idecabtagene vicleucel (ide-cel, bb2121) responses are characterized by early and temporally consistent activation and expansion of CAR T cells with a T effector phenotype. Blood 136, 17–18 (2020).
Wright, J. F. Quantification of CpG motifs in rAAV genomes: avoiding the Toll. Mol. Ther. 28, 1756–1758 (2020).
Paremskaia, A. I. et al. Codon-optimization in gene therapy: promises, prospects and challenges. Front. Bioeng. Biotechnol. 12, 1371596 (2024).
Yoshida, T. et al. Introduction of sugar-modified nucleotides into CpG-containing antisense oligonucleotides inhibits TLR9 activation. Sci. Rep. 14, 11540 (2024).
Kumar, S. et al. International Myeloma Working Group consensus criteria for response and minimal residual disease assessment in multiple myeloma. Lancet Oncol. 17, e328–e346 (2016).
Lee, D. W. et al. ASTCT consensus grading for cytokine release syndrome and neurologic toxicity associated with immune effector cells. Biol. Blood Marrow Transplant. 25, 625–638 (2019).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (82170223 to C.L.) and the Natural Science Foundation of Hubei Province (2024AFD421 to C.L.). We thank H. Mei for helpful discussions. We thank all clinical investigators, research nurses and study coordinators for their contributions. We also acknowledge the members of the independent data monitoring committee for their oversight. The study was supported in part by EsoBiotec, which provided the ESO-T01 vector and technical input. The funder had no role in study design, data collection, data analysis, data interpretation or writing of the report.
Author information
Authors and Affiliations
Contributions
C.L., N.A., D.W. and P.Z. contributed to the analyses and interpretation of the clinical data. J.Z. and P.P. contributed to the development of the ESO-T01 construct. C.L., D.W., P.Z. and J.Z. designed the clinical protocol. C.L., D.W., P.Z., L.X., H.R. and Y.W. contributed to the clinical treatments. N.A., J.H., X.W. and Y.G. collected and assembled data. N.A., C.L, P.Z. and Y.B. wrote the paper. C.L. directed the study and had final responsibility to submit for publication. All authors read and approved the final paper.
Corresponding author
Ethics declarations
Competing interests
P.P. is employed by and owns stocks in EsoBiotec. J.Z. is employed by and owns stocks in Shenzhen Pregene Biopharma Company. The other authors declare no competing interests.
Peer review
Peer review information
Nature Medicine thanks Leo Rasche, Eric Smith and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Anna Ranzoni, in collaboration with the Nature Medicine team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Clinical timelines, toxicity profiles and interventions in patients treated with ESO-T01.
a, Timelines illustrating infusion, CRS onset and duration, ICANS, administration of corticosteroids or tocilizumab, and the time of peak CAR-T expansion (Cmax). b, Maximum CRS and ICANS grades according to ASTCT criteria, together with the duration of CRS (days) in each patient. c, Summary of adverse events in the cohort, including infections, elevated AST/ALT and electrolyte abnormalities. Heatmap intensity indicates the maximum recorded grade for each event. Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; Cmax, maximum CAR-T cell concentration; CRS, cytokine release syndrome; ICANS, immune effector cell-associated neurotoxicity syndrome.
Extended Data Fig. 2 Longitudinal changes in peripheral blood counts after ESO-T01 infusion.
a, White blood cells declined within 24 hours post-infusion, followed by variable recovery profiles. b, Lymphocyte counts decreased rapidly and recovered to near-baseline levels over time. c, Neutrophil counts showed early depletion with delayed reconstitution. d, Monocyte counts declined acutely but demonstrated partial recovery. e, Hemoglobin levels reached nadirs around Days 10–15 and gradually recovered. f, Platelet counts similarly dropped around Days 10–15, followed by hematologic recovery over 60–120 days. Values represent absolute counts in peripheral blood.
Extended Data Fig. 3 Clinical trajectory, inflammatory markers, and CAR-T expansion in patient I001 following ESO-T01 infusion.
a, Temporal changes in body temperature and blood pressure with concurrent clinical interventions. The patient developed grade 3 CRS within 2 hours of infusion, presenting with fever, hypotension, hypoxemia, and tachycardia. Management consisted of high-dose corticosteroids, tocilizumab, renal replacement therapy, dopamine, and high-flow oxygen delivered in a hematology ward equipped for ICU-level monitoring. Oxygen flow rates (L/min) are annotated along the horizontal bar. b, Serum cytokines (IL-6, IL-8, IL-10) and ferritin showed a hyperacute surge coinciding with CRS onset, followed by a decline after supportive therapy. c, Circulating CAR transgene copies became detectable after Day 4, while serum IgG-κ M protein showed a transient decline, interpreted as an immune-related fluctuation rather than tumor lysis.Abbreviations: CAR, chimeric antigen receptor; CRS, cytokine release syndrome; IL-6, interleukin-6; IL-8, interleukin-8; IL-10, interleukin-10; LOD, limit of detection.
Extended Data Fig. 4 Clinical and immunologic profile of patient I003 following ESO-T01 infusion.
a, Clinical timeline from Day –1 to Day 19, showing neurologic symptoms and disease progression. The patient developed grade 3 CRS and grade 1 ICANS, ultimately progressing to cardiac arrest and death on Day 19. b, Daily monitoring of temperature and blood pressure, with annotations for corticosteroids (red), tocilizumab (black), norepinephrine (orange), and oxygen support (green; including bi-level positive airway pressure [BIPAP]). c, Serum cytokines (IL-6, IL-8, IL-10) and ferritin showed a biphasic inflammatory profile, with peaks corresponding to CRS episodes. d, Hemoglobin and platelet counts declined, necessitating repeated red blood cell (dark red) and platelet (dark blue) transfusions. e, CAR transgene copies expanded transiently in peripheral blood, accompanied by declines in M protein and FLC λ. These changes may represent transient antitumor activity or immune-related fluctuations, but no durable clinical remission was achieved. Abbreviations: BIPAP, bi-level positive airway pressure; BP, blood pressure; CAR, chimeric antigen receptor; CRS, cytokine release syndrome; FLC, free light chain; ICANS, immune effector cell-associated neurotoxicity syndrome; IL-6, interleukin-6; IL-8, interleukin-8; IL-10, interleukin-10; LOD, limit of detection.
Extended Data Fig. 5 Flow cytometry analysis of CAR-T cell expansion in peripheral blood.
Representative dot plots show CAR-T cell frequencies at baseline (pre-infusion) and on days 4, 14, and 28 after infusion for each patient (I001–I005). CAR-T cells were defined as CD3⁺ T cells co-expressing the CAR construct, detected using a PE-labeled anti-CAR antibody. Frequencies are expressed as percentages of total CD3⁺ T cells. Expansion peaked around day 14 in most patients and declined by day 28, consistent with transient in vivo CAR-T kinetics. Abbreviations: CAR-T, chimeric antigen receptor T cell.
Extended Data Fig. 6 Immune reconstitution following ESO-T01 infusion.
a, Absolute counts of NK cells over time. b, Absolute counts of T cells over time. c–e, Serum immunoglobulin levels, including IgA (c), IgG (d), and IgM (e), declined after infusion and remained suppressed during follow-up. Together, these findings illustrate treatment-related immune reconstitution after ESO-T01 infusion. Abbreviations: Ig, immunoglobulin; NK, natural killer.
Extended Data Fig. 7 Flow cytometry analysis of off-target transduction in patient peripheral blood at the peak of CAR-T expansion.
a-c, Representative dot plots from patients I001–I005 show CAR expression within CD3⁻ lymphocyte subsets (a), monocyte subsets (b) and neutrophil subsets (c) at the time of maximal CAR-T expansion. Across all patients, the frequency of CAR⁺ non-T cells remained below 1%, with CAR signal undetectable in most samples, indicating minimal off-target transduction. These data support the in vivo T-cell specificity of the ESO-T01 vector. Abbreviations: CAR, chimeric antigen receptor; CD3, cluster of differentiation 3; Lym, lymphocyte; Mon, monocyte; Neu, neutrophils.
Extended Data Fig. 8 Functional activation and phenotypic dynamics of CAR-T cells after ESO-T01 infusion.
a, Temporal changes in the percentage of CD25 and CD69 CAR-T cells in peripheral blood between Days 4 and 28 post-infusion. CD25 (IL-2Rα) and CD69 are markers of early T-cell activation. b, Longitudinal expression of HLA-DR on CAR-T cells, associated with immune activation, with some patients showing sustained expression. c, PD-1 expression on CAR-T cells across multiple timepoints. A transient increase was observed shortly after infusion, which declined by Day 30, suggesting activation-related dynamics but not sufficient alone to define exhaustion. d, Phenotypic distribution of CD4⁺ CAR-T cells at Days 4, 8, 14, 17, and 28, classified into naïve, effector, central memory (TCM), and effector memory (TEM) subsets. A progressive shift toward memory phenotypes was observed. Each bar at a given time point represents data from an individual patient. e, Distribution of CD8⁺ CAR-T subsets across the same timepoints, showing a similar effector-to-memory transition with early expansion followed by phenotypic diversification. Each bar at a given time point represents data from an individual patient.Grey bars (NA) indicate unavailable data. Abbreviations: CD25, interleukin-2 receptor α; CD69, early T-cell activation marker; HLA-DR, human leukocyte antigen–DR; PD-1, programmed cell death protein 1; TCM, central memory T cell; TEM, effector memory T cell; NA, not available.
Extended Data Fig. 9 Vector design and CAR construct of ESO-T01.
a, Vector design features of ESO-T01. An anti-TCR nanobody (VHH) targets the TCR/CD3 complex to confer T-cell specificity. A mutant VSVG (ENV) envelope reduces broad tropism and lowers immunogenicity. High CD47 expression decreases clearance by the mononuclear phagocyte system, enhancing persistence and transduction. Knockout of MHC class I reduces immunogenicity and susceptibility to complement- or antibody-mediated inactivation. A synthetic T cell–specific promoter (SYN) restricts expression to T cells, while the BCMA CAR backbone is derived from the clinically validated PRG1801 second-generation VHH CAR. b, CAR construct of ESO-T01. The CAR contains an anti-BCMA VHH for antigen recognition, a CD8 hinge and transmembrane domain (TMD) for structural stability, the 4-1BB costimulatory domain, and the CD3ζ signaling domain for T-cell activation. Abbreviations: CAR, chimeric antigen receptor; BCMA, B Cell Maturation Antigen; MHC, Major Histocompatibility Complex; SYN, Synthetic T cell-specific Promoter; TCR, T Cell Receptor; VHH, Variable Domain of Heavy-chain Antibodies; VSVG, Vesicular Stomatitis Virus Glycoprotein.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1−6 and Tables 1−7, clinical study project protocol, study protocol amendment summary and statistical analysis plan.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
An, N., Wang, D., Zhang, P. et al. In vivo generation of anti-BCMA CAR-T cells in relapsed or refractory multiple myeloma: a phase 1 study. Nat Med (2026). https://doi.org/10.1038/s41591-026-04244-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41591-026-04244-6