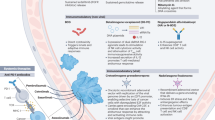
Abstract
Radical cystectomy is the most commonly used definitive local treatment for muscle-invasive bladder cancer (MIBC), yet it carries substantial perioperative complication risk, alongside major changes in urinary and sexual function. Chemoradiotherapy (CRT) is used as a bladder-sparing alternative but is usually reserved for small, solitary tumors. Highly active systemic induction therapy could enable bladder preservation for patients with more advanced tumors and reduce recurrence risk. We previously demonstrated high activity of preoperative ipilimumab (3 mg kg−1) plus nivolumab in patients with stage III MIBC. Given this high activity, the single-arm, multicenter phase 2 INDIBLADE trial aimed to provide effective bladder-sparing treatment to patients with stage II/III (cT2-4aN0-2, n = 50) MIBC, using induction ipilimumab (3 mg kg−1) plus nivolumab followed by CRT. After a median follow-up of 28.7 months, the primary endpoint of estimated 2-year bladder-intact event-free survival (BI-EFS) was met at 78% (95% confidence interval (CI): 0.67−0.9, P < 0.001). Secondary endpoints included overall survival, safety and the predictive value of circulating tumor DNA (ctDNA). Two-year overall survival was 96% (95% CI: 0.91−1). Grade 3−4 immune-related adverse events occurred in 24% of patients; grade 3−4 CRT-related adverse events occurred in 7% of patients. Absence of detectable ctDNA after induction immunotherapy was associated with BI-EFS (hazard ratio 8.3, 95% CI: 1.38−50.36, P = 0.02). In conclusion, our results show that induction combination immunotherapy followed by CRT is an effective bladder-sparing treatment in MIBC, and response can be monitored by ctDNA. ClinicalTrials.gov identifier: NCT05200988.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
To minimize the risk of patient reidentification, anonymized patient-level clinical data will be made available upon scientifically sound request for academic use only and within the confinements of the patient informed consent and European Data Protection Regulation. Data access can be obtained via the Netherlands Cancer Institute’s scientific repository at repository@nki.nl, which will contact the corresponding author (M.S.v.d.H.). Requests will be answered within 4 weeks. Requests will subsequently be reviewed by the institutional review board of the Netherlands Cancer Institute. A data access agreement must be signed between the researcher and the Netherlands Cancer Institute upon approval.
Code availability
No custom code or mathematical algorithm was specifically developed for this study. Software and packages used in the analyses can be found in the Methods or in the Reporting Summary.
References
Stein, J. P. et al. Radical cystectomy in the treatment of invasive bladder cancer: long-term results in 1,054 patients. J. Clin. Oncol. 19, 666–675 (2001).
Maibom, S. L. et al. Short-term morbidity and mortality following radical cystectomy: a systematic review. BMJ Open 11, e043266 (2021).
Marqueen, K. E. et al. Early mortality in patients with muscle-invasive bladder cancer undergoing cystectomy in the United States. JNCI Cancer Spectr. 2, pky075 (2018).
Mak, K. S. et al. Quality of life in long-term survivors of muscle-invasive bladder cancer. Int. J. Radiat. Oncol. Biol. Phys. 96, 1028–1036 (2016).
Zlotta, A. R. et al. Radical cystectomy versus trimodality therapy for muscle-invasive bladder cancer: a multi-institutional propensity score matched and weighted analysis. Lancet Oncol. 24, 669–681 (2023).
Brück, K. et al. Disease-free survival of patients with muscle-invasive bladder cancer treated with radical cystectomy versus bladder-preserving therapy: a nationwide study. Int. J. Radiat. Oncol. Biol. Phys. 118, 41–49 (2024).
van der Heijden, A. G. et al. European Association of Urology guidelines on muscle-invasive and metastatic bladder cancer: summary of the 2025 guidelines. Eur. Urol. 87, 582–600 (2025).
Powles, T. et al. Bladder cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann. Oncol. 33, 244–258 (2022).
Fahmy, O. et al. A systematic review and meta-analysis on the oncological long-term outcomes after trimodality therapy and radical cystectomy with or without neoadjuvant chemotherapy for muscle-invasive bladder cancer. Urol. Oncol. 36, 43–53 (2018).
Kool, R. et al. Benefit of neoadjuvant cisplatin-based chemotherapy for invasive bladder cancer patients treated with radiation-based therapy in a real-world setting: an inverse probability treatment weighted analysis. Eur. Urol. Oncol. 7, 1350–1357 (2024).
Shipley, W. U. et al. Phase III trial of neoadjuvant chemotherapy in patients with invasive bladder cancer treated with selective bladder preservation by combined radiation therapy and chemotherapy: initial results of Radiation Therapy Oncology Group 89-03. J. Clin. Oncol. 16, 3576–3583 (1998).
van Dijk, N. et al. Preoperative ipilimumab plus nivolumab in locoregionally advanced urothelial cancer: the NABUCCO trial. Nat. Med. 26, 1839–1844 (2020).
van Dorp, J. et al. High- or low-dose preoperative ipilimumab plus nivolumab in stage III urothelial cancer: the phase 1B NABUCCO trial. Nat. Med. 29, 588–592 (2023).
Stockem, C. F. et al. Final clinical analysis of pre-operative ipilimumab and nivolumab in locally advanced urothelial cancer and exploration of tumor-draining lymph node composition: the NABUCCO trial. Eur. J. Cancer 229, 115731 (2025).
James, N. D. et al. Radiotherapy with or without chemotherapy in muscle-invasive bladder cancer. N. Engl. J. Med. 366, 1477–1488 (2012).
de Haar-Holleman, A. et al. Chemoradiation for muscle-invasive bladder cancer using 5-fluorouracil versus capecitabine: a nationwide cohort study. Radiother. Oncol. 183, 109584 (2023).
Powles, T. et al. ctDNA-guided adjuvant atezolizumab in muscle-invasive bladder cancer. N. Engl. J. Med. 393, 2395−2408 (2025).
Powles, T. et al. Enfortumab vedotin and pembrolizumab in untreated advanced urothelial cancer. N. Engl. J. Med. 390, 875–888 (2024).
Vulsteke, C. et al. LBA2 Perioperative (periop) enfortumab vedotin (EV) plus pembrolizumab (pembro) in participants (pts) with muscle-invasive bladder cancer (MIBC) who are cisplatin-ineligible: the phase III KEYNOTE-905 study. Ann. Oncol. 36, S1648 (2025).
Powles, T. et al. Circulating tumor DNA (ctDNA) in patients with muscle-invasive bladder cancer (MIBC) who received perioperative durvalumab (D) in NIAGARA. J. Clin. Oncol. 43, 4503 (2025).
van der Heijden, M. S. et al. Nivolumab plus ipilimumab (NIVO+IPI) vs gemcitabine-carboplatin (gem-carbo) chemotherapy for previously untreated unresectable or metastatic urothelial carcinoma (mUC): final results for cisplatin-ineligible patients from the CheckMate 901 trial. J. Clin. Oncol. 43, 4500 (2025).
Powles, T. et al. Perioperative durvalumab with neoadjuvant chemotherapy in operable bladder cancer. N. Engl. J. Med. 391, 1773–1786 (2024).
Galsky, M. D. et al. Gemcitabine and cisplatin plus nivolumab as organ-sparing treatment for muscle-invasive bladder cancer: a phase 2 trial. Nat. Med. 29, 2825–2834 (2023).
Geynisman, D. M. et al. Phase II trial of risk-enabled therapy after neoadjuvant chemotherapy for muscle-invasive bladder cancer (RETAIN 1). J. Clin. Oncol. 43, 1113–1122 (2025).
Reinert, T. et al. Analysis of plasma cell-free DNA by ultradeep sequencing in patients with stages I to III colorectal cancer. JAMA Oncol. 5, 1124–1131 (2019).
Acknowledgements
We thank, first and foremost, all patients and their families who participated in the INDIBLADE trial and the medical and supporting staff of the University Medical Center Utrecht, the Erasmus Medical Center and the Netherlands Cancer Institute involved in the care of study patients. We thank H. Schrijver for trial management; Y. Wijnands and K. Snapper for data management; H. Hoogenhout for trial support; and J. P. E. Lagendijk on behalf of the patients advisory committee. We thank the Core Facility of Molecular Pathology and Biobanking at the Netherlands Cancer Institute (A. Broeks, S. Cornelissen and L. Braafs) and the Genomics Core Facility (C. van Steenis and W. Brugman). The INDIBLADE trial was supported by a grant from the KWF Dutch Cancer Society (grant 13226; M.S.v.d.H.). Research at the Netherlands Cancer Institute is supported by institutional grants from the Dutch Ministry of Health, Welfare and Sport and the KWF Dutch Cancer Society. Bristol Myers Squibb provided study drugs and funding for the trial (CA209-6JG; M.S.v.d.H.). The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
M.S.v.d.H. conceived the INDIBLADE study. M.S.v.d.H., E.E.S., B.W.G.v.R., B.B.M.S., D.G.J.R., R.P.M., J.L.B., M.F., J.L.N., T.N.B. and S.B. contributed to the study design. C.F.S., J.J.J.M. and M.S.v.d.H. wrote and amended the protocol. C.F.S., J.J.J.M. and M.S.v.d.H. were responsible for coordination and logistics of the trial. D.G.J.R. was the principal investigator for the Erasmus Medical Center. B.B.M.S. was the principal investigator for the University Medical Center Utrecht. M.S.v.d.H. was the principal investigator for the study overall and for the Netherlands Cancer Institute. J.J.J.M., C.F.S., B.W.G.v.R., L.S.M., T.N.B., J.M.d.F., J.C.K.v.d.M., J.L.B., M.F., R.P.M., J.L.N., E.E.S., D.G.J.R., B.B.M.S. and M.S.v.d.H. were involved in recruitment and management of study patients. M.L.v.M. was involved in collection, storage and processing of clinical samples. S.B. carried out the clinical statistical analyses and wrote the statistical analysis plan. C.H., S.K.C., S.S. and A.C.E. carried out plasma sample and WES data processing and subsequent ctDNA analyses. J.J.J.M. and M.S.v.d.H. drafted the manuscript. L.S.M., B.W.G.v.R., J.M.d.F., T.N.B., R.P.M., J.L.B., E.E.S., B.B.M.S., D.G.J.R., C.H., S.S. and C.F.S. made important contributions to the final manuscript. All authors reviewed, amended and approved the final manuscript. All authors personally vouch for the adherence to protocol and the accuracy of the reported data.
Corresponding author
Ethics declarations
Competing interests
M.S.v.d.H. has received institutional funding from Bristol Myers Squibb to finance the INDIBLADE trial. C.H., S.K.C., S.S. and A.C.E. are all full-time employee at Natera, Inc. with stocks or options to own stocks in the company. No other author received financial support for the work reported in this paper. No medical writers were involved in drafting the manuscript. B.W.G.v.R. has received consulting fees from Cepheid and Astellas (institutional). L.S.M. received consulting fees from Johnson & Johnson and Merck (institutional). T.N.B. makes use of free-to-use server and software for research purposes from Siemens. J.M.d.F. has an advisory role for Merck and Janssen (institutional). J.C.K.v.d.M. received research support from Bayer (institutional) and received consultancy fees from Merck/Merck Sharp & Dohme, IPSEN, Jansen, Bayer, Novartis and AstraZeneca (institutional). J.L.B. received consulting fees from Bristol Myers Squibb, Janssen, AstraZeneca, Merck AG/Pfizer, Merck Sharp & Dohme, Bayer and Ismar Healthcare (institutional) and received speaker’s fees from Springer Media, Congress Care and NLC Health (personal). R.P.M. has an advisory role for Astellas, Merck and Bristol Myers Squibb (institutional) and received research support from Merck, Janssen-Cilag, Astellas and Gilead Sciences Netherlands (institutional). D.G.J.R. has an advisory role for MSD Oncology, Pfizer, Astellas, AstraZeneca, Johnson & Johnson, Merck and Gilead Sciences Netherlands (institutional) and has received research support from Merck (institutional). B.B.M.S. has an advisory role for AstraZeneca, Bristol Myers Squibb, MDS, IPSEN, Merck, Astellas and Pfizer (institutional) and has received research support from AstraZeneca, Bristol Myers Squibb, MDS, IPSEN, Merck, Astellas and Pfizer (institutional). M.S.v.d.H. has received consulting fees from Astellas, AstraZeneca, Bristol Myers Squibb, MSD Oncology, Pfizer/Seagen, Johnson & Johnson and Daiichi-Sankyo (institutional); has received research support from Bristol Myers Squibb, Roche, AstraZeneca, Seagen, 4SC and MSD Oncology (institutional); and has received travel expenses paid by Bristol Myers Squibb (institutional). All other authors have nothing to disclose.
Peer review
Peer review information
Nature Medicine thanks the anonymous reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available. Primary Handling Editor: Ulrike Harjes, in collaboration with the Nature Medicine team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Longitudinal ctDNA results.
Representation of plasma ctDNA measurements longitudinally collected at various time points (Extended Data Table 1). Scoring of ctDNA is binary, with samples classified as positive or negative. Follow up was cut-off at 24 months to improve readability.
Supplementary information
Supplementary Information (download PDF )
Supplementary Tables 1 and 2, study protocol and statistical analysis plan.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mellema, JJ.J., Stockem, C.F., Herberts, C. et al. Ipilimumab and nivolumab followed by chemoradiotherapy as bladder-sparing treatment in muscle-invasive bladder cancer: a phase 2 trial. Nat Med (2026). https://doi.org/10.1038/s41591-026-04271-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41591-026-04271-3