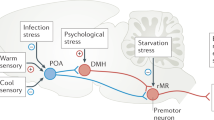
Abstract
Animal groups often collectively coordinate their behavior to withstand environmental challenges, yet the neural circuitry underlying such collective social dynamics remains unclear. Here we show that groups of mice self-organize into huddles under cold stress. We quantified the thermoregulatory benefits of huddling using thermal imaging and internal temperature loggers, which revealed that it stabilized core body temperature by increasing thermal contact points and reducing heat loss. We next characterized decision-making processes that govern huddling dynamics and found that mice employed both active (self-initiated) and passive (partner-initiated) strategies to enter or exit a huddle. Microendoscopic calcium imaging revealed that active and passive decisions are encoded in distinct neuronal ensembles within the dorsomedial prefrontal cortex. Chemogenetic silencing of dorsomedial prefrontal cortex activity selectively reduced active decisions in targeted mice but elicited compensatory increases in non-manipulated partners, preserving overall group-level huddle time. These findings uncover a cortical mechanism by which social groups collectively adapt to maintain homeostasis under environmental challenge.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data and analyses necessary to understand the conclusions of the article are presented in the main text and in Extended Data Figs. 1–10. Source data are provided with this paper.
Code availability
Code for behavior analysis toolboxes (https://github.com/pdollar/toolbox and https://github.com/hongw-lab/behavior_annotator), multi-animal pose tracking (SLEAP v1.1.5, https://github.com/talmolab/sleap), microendoscopic imaging data analysis (https://github.com/etterguillaume/miniscopeanalysis, https://github.com/zhoupc/cnmf_e, https://github.com/flatironinstitute/normcorre, https://github.com/hongw-lab/cscreener, https://github.com/hongw-lab/1p_preprocessing) and additional analyses supporting the conclusions of this paper (https://github.com/hongw-lab/Code_for_2025_Raam) is available on GitHub.
References
Krause, J. & Ruxton, G. D. Living in Groups (Oxford Univ. Press, 2002).
Silk, J. B. The adaptive value of sociality in mammalian groups. Philos. Trans. R. Soc. B 362, 539 (2007).
Shah, S. S. & Rubenstein, D. R. Group augmentation underlies the evolution of complex sociality in the face of environmental instability. Proc. Natl Acad. Sci. USA 120, e2212211120 (2023).
Polyakov, A. Y., Quinn, T. P., Myers, K. W. & Berdahl, A. M. Group size affects predation risk and foraging success in Pacific salmon at sea. Sci. Adv. 8, 7548 (2022).
Markham, A. C., Gesquiere, L. R., Alberts, S. C. & Altmann, J. Optimal group size in a highly social mammal. Proc. Natl Acad. Sci. USA 112, 14882–14887 (2015).
Testard, C. et al. Ecological disturbance alters the adaptive benefits of social ties. Science 384, 1330–1335 (2024).
Fisher, D. N. et al. Anticipated effects of abiotic environmental change on intraspecific social interactions. Biol. Rev. 96, 2661–2693 (2021).
Blumstein, D. T., Hayes, L. D. & Pinter-Wollman, N. Social consequences of rapid environmental change. Trends Ecol. Evol. 38, 337–345 (2023).
Sliwa, J. Toward collective animal neuroscience. Science 374, 397–398 (2021).
Yu, J. H., Napoli, J. L. & Lovett-Barron, M. Understanding collective behavior through neurobiology. Curr. Opin. Neurobiol. 86, 102866 (2024).
Pereira, T. D. et al. SLEAP: a deep learning system for multi-animal pose tracking. Nat. Methods 19, 486–495 (2022).
Chen, Z. et al. AlphaTracker: a multi-animal tracking and behavioral analysis tool. Front Behav. Neurosci. 17, 1111908 (2023).
Lauer, J. et al. Multi-animal pose estimation, identification and tracking with DeepLabCut. Nat. Methods 19, 496–504 (2022).
Kappel, J. M. et al. Visual recognition of social signals by a tectothalamic neural circuit. Nature 608, 146–152 (2022).
Zada, D. et al. Development of neural circuits for social motion perception in schooling fish. Curr. Biol. 34, 3380–3391.e5 (2024).
Forli, A. & Yartsev, M. M. Hippocampal representation during collective spatial behaviour in bats. Nature 621, 796–803 (2023).
Rose, M. C., Styr, B., Schmid, T. A., Elie, J. E. & Yartsev, M. M. Cortical representation of group social communication in bats. Science 374, eaba9584 (2021).
Ramdya, P. et al. Mechanosensory interactions drive collective behaviour in Drosophila. Nature 519, 233–236 (2014).
Ferreira, C. H. & Moita, M. A. Behavioral and neuronal underpinnings of safety in numbers in fruit flies. Nat. Commun. 11, 1–10 (2020).
Harshaw, C., Blumberg, M. S. & Alberts, J. R. in APA Handbook of Comparative Psychology, Vol. 1 (eds Call, J. et al.) Ch. 45 (APA, 2017).
Young, A. J., Sawka, M. N. & Pandolf, K. B. in Nutritional Needs in Cold and in High-Altitude Environments: Applications for Military Personnel in Field Operations (eds Marriott, B. M. & Carlson, S. J.) Ch. 7 (National Academies, 1996).
LeBlanc, J. Mechanisms of adaptation to cold. Int. J. Sports Med. 13 (Suppl. 1), S169–S172 (1992).
El Marzouki, H., Aboussaleh, Y., Najimi, M., Chigr, F. & Ahami, A. Effect of cold stress on neurobehavioral and physiological parameters in rats. Front. Physiol. 12, 660124 (2021).
Hu, Y., Liu, Y. & Li, S. Effect of acute cold stress on neuroethology in mice and establishment of its model. Animals (Basel) 12, 2671 (2022).
Han, A. et al. Temperature-dependent differences in mouse gut motility are mediated by stress. Lab. Anim. 53, 148–159 (2024).
Kokolus, K. M. et al. Baseline tumor growth and immune control in laboratory mice are significantly influenced by subthermoneutral housing temperature. Proc. Natl Acad. Sci. USA 110, 20176–20181 (2013).
Jung, S. et al. A forebrain neural substrate for behavioral thermoregulation. Neuron 110, 266–279.e9 (2022).
Lal, N. K. et al. Xiphoid nucleus of the midline thalamus controls cold-induced food seeking. Nature 621, 138–145 (2023).
Yang, S. et al. An mPOA-ARCAgRP pathway modulates cold-evoked eating behavior. Cell Rep. 36, 109502 (2021).
Glancy, J., Groß, R., Stone, J. V. & Wilson, S. P. A self-organising model of thermoregulatory huddling. PLoS Comput. Biol. 11, 1004283 (2015).
Alberts, J. R. Huddling by rat pups: ontogeny of individual and group behavior. Dev. Psychobiol. 49, 22–32 (2007).
Harshaw, C., Culligan, J. J. & Alberts, J. R. Sex differences in thermogenesis structure behavior and contact within huddles of infant mice. PLoS ONE 9, e87405 (2014).
Harshaw, C. & Alberts, J. R. Group and individual regulation of physiology and behavior: a behavioral, thermographic, and acoustic study of mouse development. Physiol. Behav. 106, 670–682 (2012).
Bautista, A., García-Torres, E., Martínez-Gómez, M. & Hudson, R. Do newborn domestic rabbits Oryctolagus cuniculus compete for thermally advantageous positions in the litter huddle? Behav. Ecol. Sociobiol. 62, 331–339 (2008).
Harshaw, C., Leffel, J. K. & Alberts, J. R. Oxytocin and the warm outer glow: thermoregulatory deficits cause huddling abnormalities in oxytocin-deficient mouse pups. Horm. Behav. 98, 145–158 (2018).
Endo, N. et al. Multiple animal positioning system shows that socially-reared mice influence the social proximity of isolation-reared cagemates. Commun. Biol. 1, 1–13 (2018).
Campbell, L. A. D., Tkaczynski, P. J., Lehmann, J., Mouna, M. & Majolo, B. Social thermoregulation as a potential mechanism linking sociality and fitness: Barbary macaques with more social partners form larger huddles. Sci. Rep. 8, 1–8 (2018).
Ishizuka, S. Do dominant monkeys gain more warmth? Number of physical contacts and spatial positions in huddles for male Japanese macaques in relation to dominance rank. Behav. Processes 185, 104317 (2021).
Zitterbart, D. P., Wienecke, B., Butler, J. P. & Fabry, B. Coordinated movements prevent jamming in an emperor penguin huddle. PLoS ONE 6, 20260 (2011).
Waters, A., Blanchette, F. & Kim, A. D. Modeling huddling penguins. PLoS ONE 7, e50277 (2012).
Canals, M. & Bozinovic, F. Huddling behavior as critical phase transition triggered by low temperatures. Complexity 17, 35–43 (2011).
Schank, J. C. & Alberts, J. R. Self-organized huddles of rat pups modeled by simple rules of individual behavior. J. Theor. Biol. 189, 11–25 (1997).
Báez-Mendoza, R., Vázquez, Y., Mastrobattista, E. P. & Williams, Z. M. Neuronal circuits for social decision-making and their clinical implications. Front Neurosci. 15, 720294 (2021).
Báez-Mendoza, R., Mastrobattista, E. P., Wang, A. J. & Williams, Z. M. Social agent identity cells in the prefrontal cortex of interacting groups of primates. Science 374, eabb4149 (2021).
Padilla-Coreano, N. et al. Cortical ensembles orchestrate social competition through hypothalamic outputs. Nature 603, 667–671 (2022).
Kingsbury, L. et al. Correlated neural activity and encoding of behavior across brains of socially interacting animals. Cell 178, 429–446 (2019).
Kingsbury, L. et al. Cortical representations of conspecific sex shape social behavior. Neuron 107, 941–953 (2020).
Chen, P. & Hong, W. Neural circuit mechanisms of social behavior. Neuron 98, 16–30 (2018).
Gangopadhyay, P., Chawla, M., Dal Monte, O. & Chang, S. W. C. Prefrontal-amygdala circuits in social decision-making. Nat. Neurosci. 24, 5–18 (2021).
Li, S. W. et al. Frontal neurons driving competitive behaviour and ecology of social groups. Nature 603, 661–666 (2022).
Yang, J., Zhang, H., Ni, J., De Dreu, C. K. W. & Ma, Y. Within-group synchronization in the prefrontal cortex associates with intergroup conflict. Nat. Neurosci. 23, 754–760 (2020).
Zhang, Z. et al. Estrogen-sensitive medial preoptic area neurons coordinate torpor in mice. Nat. Commun. 11, 1–14 (2020).
Cannon, B. & Nedergaard, J. Nonshivering thermogenesis and its adequate measurement in metabolic studies. J. Exp. Biol. 214, 242–253 (2011).
Lim, S. et al. Cold-induced activation of brown adipose tissue and adipose angiogenesis in mice. Nat. Protoc. 7, 606–615 (2012).
Felix-Ortiz, A. C. et al. Prefrontal regulation of safety learning during ethologically relevant thermal threat. eNeuro 11, ENEURO.0140-23.2024 (2024).
Gordon, C. J. Temperature Regulation in Laboratory Rodents (Cambridge Univ. Press, 2009).
Kaneko, T. & Kibayashi, K. Mild hypothermia facilitates the expression of cold-inducible RNA-binding protein and heat shock protein 70.1 in mouse brain. Brain Res. 1466, 128–136 (2012).
Lu, X. et al. The early metabolomic response of adipose tissue during acute cold exposure in mice. Sci. Rep. 7, 1–11 (2017).
Zhao, Z. et al. Body temperature is a more important modulator of lifespan than metabolic rate in two small mammals. Nat. Metab. 4, 320–326 (2022).
Grady, F., Peltekian, L., Iverson, G. & Geerling, J. C. Direct parabrachial-cortical connectivity. Cereb. Cortex 30, 4811–4833 (2020).
Tan, C. L. et al. Warm-sensitive neurons that control body temperature. Cell 167, 47–59 (2016).
Landen, J. G., Vandendoren, M., Killmer, S., Bedford, N. L. & Nelson, A. C. Huddling substates in mice facilitate dynamic changes in body temperature and are modulated by Shank3b and Trpm8 mutation. Commun. Biol. 7, 1186 (2024).
Sotelo, M. I. et al. Neurophysiological and behavioral synchronization in group-living and sleeping mice. Curr. Biol. 34, 132–146 (2024).
Kim, J. et al. A bird’s-eye view of brain activity in socially interacting mice through mobile edge computing (MEC). Sci. Adv. 6, eabb9841 (2020).
Zhao, Y. et al. Social rank-dependent effects of testosterone on huddling strategies in mice. iScience 26, 106516 (2023).
Acknowledgements
We thank N. Ramesh for technical assistance, T. Pereira and the Pereira lab for assistance with SLEAP tracking, and all members of the Hong Lab for feedback and support. T.R. thanks F. Arbab for early conversations that inspired this research direction. Funding sources: NIH grant nos. T32-NS048044 (T.R.), F32-MH123049 (T.R.), and K99-MH133159 (T.R.). Brain and Behavior Research Foundation Young Investigator Grant (T.R.). NIH grant no. R01-AG066821 (S.M.C.). NIH grant nos. R01-MH130941 (W.H.), R01-NS113124 (W.H.), R01-MH132736 (W.H.) and RF1-NS132912 (W.H.). Packard Fellowship in Science and Engineering (W.H.). Vallee Scholar Award (W.H.). Mallinckrodt Scholar Award (W.H.).
Author information
Authors and Affiliations
Contributions
W.H. and T.R. jointly conceived of the project and designed the experiments. T.R. and K.Y.L. performed stereotactic surgeries. T.R. carried out all behavior and imaging experiments, with assistance from G.M.E and L.G. T.R. generated the SLEAP model for multi-animal pose estimation. Q.L. created the automated behavior analysis pipeline and pipelines for annotations and analysis of thermal behavioral videos. T.R., Q.L. and L.G. analyzed the behavioral and calcium imaging data. T.R., G.M.E. and J.Y.T. performed behavior annotations. G.M.E. performed histological verification. X.Z. created the BehaviorAnnotator software. N.P.S. provided assistance with thermal bio-loggers. S.M.C. provided valuable input and equipment. T.R. designed and edited figures with support from K.Y.L. T.R. and W.H. wrote the paper. W.H. supervised all aspects of the study.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Neuroscience thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Additional data related to huddle analysis pipeline and titration of ambient temperature.
a. Pipeline for automated detection of huddle size. Raw frames are binarized into black and white pixels. Erosion and dilation are performed to removed tails and fecal artifacts. Edge detection is performed to identify connected groups of animals. b. Percent accuracy of detected huddle state compared to manual human annotation c. Automated Identification of huddle membership is achieved by tracking raw behavior videos with a trained neural network (Social Leap Estimates Animal Poses) to identify individual nodes and identities. Tracked poses and identities are overlayed on top of detected huddles to identify the membership. d. Example raster plot for one group demonstrating membership configurations for huddles of three throughout one behavior session. e. Frequency of group states observed when titrating ambient temperature at 20 °C, 15 °C, 10 °C, or 5 °C during thermal challenge assay (n = 6 groups of 4 individuals). f. Mean group state duration in seconds observed at 20 °C, 15 °C, 10 °C, or 5 °C during thermal challenge assay (n = 6 groups of 4 individuals). g. Rolling average (mean ± SEM) of percent time of all five group states plotted over time at 10 °C (n = 6 groups of 4 individuals). h. Rolling average (mean ± SEM) of percent time of all five group states plotted over time at 15 °C (n = 6 groups of 4 individuals). i. Rolling average (mean ± SEM) of percent time of all five group states plotted over time at 20 °C (n = 6 groups of 4 individuals). j. Individual animal’s total percent time spent in solitude in groups of four vs groups of two (n = 24 individuals from 6 groups). k. Comparison of observed huddling data versus a probabilistic model that calculates the likelihood of huddle sizes in a group of 4 based on the observed huddling in pairs. Box plots: center line–median; box limits–upper/lower quartiles; whiskers–min/max. Statistical tests: one-way (j) and two-way (e,f,k) ANOVA with Bonferroni post-hoc. *P < .05, **P < .01, ***P < .001, ****P < .0001. See Supplementary Table 1 for statistical details.
Extended Data Fig. 2 Additional data related to females and core body temperature measurements.
a. Schematics illustrating 5 unique group states derived via automated SLEAP pose estimation and identity tracking, ranging from most dispersed to most aggregated. b. Frequency of group states observed at 20 °C, 15 °C, 10 °C, or 5 °C during thermal challenge assay in females (n = 6 groups of 4 individuals). c. Mean group state duration in seconds observed at 20 °C, 15 °C, 10 °C, or 5 °C during thermal challenge assay in females (n = 6 groups of 4 individuals). d. Schematic illustrating behavioral thermotaxis assay. Animals are placed in a behavioral chamber at 5 °C with free access to a 30 °C warm corner. e. Comparison of percent time spent in warm corner during thermotaxis assay in males and females (n = 8 males, 8 females). f. Schematic measurement of core body temperature in pairs vs groups. g. Average change in core body temperature (°C) in pair and group conditions (n = 12 animals). h. Area under the curve (°C) from 5-30 min for pair and group conditions (n = 12 animals). i. Area under the curve (°C) during first and last minute of session for pair and group conditions (n = 12 animals). j. Comparison of total locomotion (cm) when animals are exposed to thermal challenge alone or in groups (n = 12 animals). k,l. Correlation between total locomotion and change in core body temperature in alone (k) and group (l) conditions (n = 12 animals). Box plots: center line–median; box limits–upper/lower quartiles; whiskers–min/max. Statistical tests: two-way ANOVA with Bonferroni post-hoc (b,c,i), two-tailed Wilcoxon matched pairs tests (e,h,j), and linear regressions (k,l). *P < .05, **P < .01, ***P < .001, ****P < .0001. See Supplementary Table 1 for statistical details.
Extended Data Fig. 3 Graphical user interface for BehaviorAnnotator, a novel software for behavior annotation and analysis of multi-animal behavior.
The graphic user interface of the annotator has 3 panels. Panel 1 displays the annotation streams containing user defined behaviors for all four animals, and a fifth stream which denotes the aggregate huddle size when a huddle is present. Panel 2 displays the behavior video(s). Panel 3 displays the list of user-defined behaviors and labeled behavior epochs.
Extended Data Fig. 4 Individual variability in active and passive decisions.
a. Percent of entry decisions (mean ± SEM) that are active plotted for all four individuals in six groups. b. Percent of exiting decisions (mean ± SEM) that are active plotted for all four individuals in six groups. c. Correlation between percent of entry decisions and percent of exiting decisions that are active (n = 24 individuals from 6 groups). d. Raster plot illustrating active and passive entry events throughout full behavioral session (n = 24 individuals from 6 groups). e. Raster plot illustrating active and passive exiting events throughout full behavioral session (n = 24 individuals from 6 groups). f. Schematic illustrating experimental protocol for tube dominance test. g-i. Correlation between percent tube test wins and percent of time huddling (g), percent of entry (h) and exit (i) decisions that are active. j. Frequency of group states observed in cages of cagemate vs. non-cagemate animals (n = 8 groups of 4 individuals). k. Correlation (per individual) between percent of entry decisions that are active between cagemate and non-cagemate conditions. l. Correlation (per individual) between percent of exit decisions that are active between cagemate and non-cagemate conditions. Statistical tests: linear regression (c,g,h,I,k,l) and two-way ANOVA with Bonferroni post-hoc (j). *P < .05, **P < .01, ***P < .001, ****P < .0001. See Supplementary Table 1 for statistical details. Illustrations in f created in BioRender; Raam, T. https://BioRender.com/s2f9lnx (2025).
Extended Data Fig. 5 Cold ambient temperature does not alter general social preference or dmPFC encoding of social stimuli.
a. Schematic illustrating pencil cup social preference assay. Animals were tested for 30 min at room temperature or 5 °C to determine preference for wired pencil cup containing a conspecific vs a toy. b. Quantification of investigation time directed towards social cup vs toy cup at room temperature (RT) or 5 °C (n = 10 animals). c. Venn diagram showing dmPFC cells responsive to social and toy investigation at room temperature. Total # of imaged cells = 4937 from animals. d. Venn diagram showing dmPFC cells responsive to social and toy investigation at room temperature. Total # of imaged cells = 4884 from 10 animals. e. Percent of dmPFC cells that are social responsive, toy responsive, or mixed responsive at room temperature (n = 10 animals). f. Percent of dmPFC cells that are social responsive, toy responsive, or mixed responsive at 5 °C (n = 10 animals). g. Support vector machine (SVM) decoder performance to decode social vs toy investigation at room temperature (n = 10 animals). h. Support vector machine (SVM) decoder performance to decode social vs toy investigation at 5 °C (n = 10 animals). Box plots: center line–median; box limits–upper/lower quartiles; whiskers–min/max. Statistical tests: one-way (e-f) and two-way (b) ANOVA with Bonferroni post-hoc, and two-tailed Wilcoxon matched pairs tests (g-h). *P < .05, **P < .01, ***P < .001, ****P < .0001. See Supplementary Table 1 for statistical details.
Extended Data Fig. 6 Additional data related to analysis of calcium imaging.
a. Matrix showing number of cells activated by behaviors on x and y axis. Percentages correspond to percent of total imaged cells. b. Matrix showing number of cells suppressed by behaviors on x and y axis. Percentages correspond to percent of total imaged cells. c. Matrix showing number of all cells responsive to behaviors on x and y axis. Percentages correspond to percent of total imaged cells. Total cells imaged = 5,141 from 11 animals. d. Venn diagrams showing overlap and event-triggered averages (mean ± SEM) of cells suppressed during huddling. e. Venn diagrams showing overlap and event-triggered averages (mean ± SEM) of cells modulated by both active entry and active exit, and their overlap with running cells.
Extended Data Fig. 7 Additional decoding of active and passive decisions, huddle size, and huddle membership.
a-d. Performance of SVM decoders (mean ± SEM) trained to classify active entry from speed-matched running (a), active exit from speed-matched running (b), passive entry from rest (c), and passive exit from rest (d). e,f. Cross-decoding performance of SVM decoders trained on active entry predicting active exit (e), or trained on active exit predicting active entry (f) g,h. Cross-decoding performance of SVM decoders trained on passive entry predicting passive exit (g), or trained on passive exit predicting passive entry (h). i,j. Cross-decoding performance of SVM decoders trained on active entry predicting passive entry (i), or trained on active exit predicting passive exit (j). k,l. Cross-decoding performance of SVM decoders trained on passive entry predicting active entry (k), or trained on passive exit predicting active exit (l). m. Example raster plot of huddling behavior for all four animals in one session, color coded by huddle size. n. Example pie charts showing proportion of time for various huddle configurations for huddles of two and three for one group. o. Matrix showing proportion of subject animal’s (x-axis) total huddle time with partner animals (y-axis) for one session. Sum of proportions for one animal can exceed 1 because subjects can huddle with multiple animals simultaneously. p. Partner preference index (maximum preferred partner – minimum preferred partner) for real data vs. shuffled data in which binary vectors containing individual huddle behaviors are circularly shifted relative to each other. q. Schematic of potential huddle memberships for huddles of two during miniscope imaging. r. Performance of multi-class LDA decoders trained to classify huddle membership for huddles of two from dmPFC population activity. Baseline is 33% (three possible memberships). s. Schematic of potential huddle sizes during miniscope imaging. t-v. Performance of SVM decoders trained to classify huddle size of 2 vs. 3 (t), 2 vs. 4 (u), and 3 vs. 4 (v). Box plots: center line–median; box limits–upper/lower quartiles; whiskers–min/max. Statistical tests: two-way ANOVA with Bonferroni post-hoc (a-d) and two-tailed Wilcoxon matched pairs tests (e-l,p,r,t-v). *P < .05, **P < .01, ***P < .001, ****P < .0001. See Supplementary Table 1 for statistical details.
Extended Data Fig. 8 Additional data related to chemogenetic silencing.
a. Schematic illustrating potential composition of membership for huddles of 2: SAL-SAL, SAL-CNO, and CNO-CNO during thermal challenge in 2C,2S condition. b. Percent of total time observed for possible membership compositions for huddles of two (n = 24 individuals from 6 groups). c. Within animal comparison of percent of entry decisions that are passive during 4S, 2C, and 2S conditions (n = 24 individuals from 6 groups). d. Within animal comparison of percent of exiting decisions that are passive during 4S, 2C, and 2S conditions (n = 24 individuals from 6 groups). e. Within group comparison of percent of entry decisions that are active for 2C, 2S condition (n = 24 individuals from 6 groups). Data in main figure shown as within animal comparisons. f. Within group comparison of percent of exiting decisions that are active for 2C, 2S condition (n = 24 individuals from 6 groups). Data in main figure shown as within animal comparisons. g. Individual animals’ total locomotion during null windows when no active, passive, or huddle behaviors are annotated during 4S, 2C, and 2S conditions (n = 24 individuals from 6 groups). h. Representative open field test trajectories at room temperature and 5 °C after SAL or CNO injection. Box plots: center line–median; box limits–upper/lower quartiles; whiskers–min/max. Statistical tests: one-way ANOVA with Bonferroni post-hoc (b-d,g) and two-tailed Mann-Whitney tests (e-f). *P < .05, **P < .01, ***P < .001, ****P < .0001. See Supplementary Table 1 for statistical details.
Extended Data Fig. 9 Chemogenetic silencing of all four animals does not alter huddling behaviors.
a. Example image showing AAV-hM4Di-mCherry expression in the dmPFC Scale bar, 500 µm. b. Schematic illustrating experimental paradigm for DREADD inhibition of dmPFC during thermal challenge. 4 SAL refers to condition in which all four animals are injected with saline. 4 CNO refers to condition in which two animals are injected with CNO. c-g. Percent time in huddle states observed for all five group states during 4S and 4 C conditions (n = 6 groups). h. Individual animal’s total percent time spent huddling in 4S and 4 C conditions (n = 24 individuals from 6 groups). i. Within animal comparison of percent of entry decisions that are active during 4S and 4 C conditions (n = 24 individuals from 6 groups). j. Total number of entry decisions per group (active and passive from all four animals) during 4S and 4 C conditions (n = 24 individuals from 6 groups). k. Within animal comparison of percent of exiting decisions that are active during 4S and 4 C conditions (n = 24 individuals from 6 groups). l. Total number of exiting decisions per group (active and passive from all four animals) during 4S and 4 C conditions (n = 24 individuals from 6 groups). m. Coronal sections illustrating histological verification of hm4Di-mCherry viral targeting and spread in dmPFC. Box plots: center line–median; box limits–upper/lower quartiles; whiskers–min/max. Statistical tests: two-tailed Wilcoxon matched pairs tests (c-l). *P < .05, **P < .01, ***P < .001, ****P < .0001. See Supplementary Table 1 for statistical details.
Extended Data Fig. 10 mCherry controls for chemogenetic silencing experiments.
a. Example image showing AAV-mCherry expression in the dmPFC. Scale bar, 500 µm. b. Schematic illustrating experimental paradigm for mCherry chemogenetic control during thermal challenge. 4 SAL refers to condition in which all four animals are injected with saline. 2CNO, 2 SAL refers to condition in which two animals are injected with CNO, and two with saline. c-g. Percent time in huddle states observed for all five group states during 4S and 2C,2S conditions (n = 5 groups). h. Individual animal’s total percent time spent huddling in 4S, 2C, and 2S conditions (n = 20 individuals from 5 groups). i,k. Within animal comparison of percent of entry or exit decisions that are active during 4S, 2C, and 2S conditions (n = 20 individuals from 5 groups). j,l. Total number of entry or exit decisions per group (active and passive from all four animals) during 4S and 2C,2S conditions (n = 20 individuals from 5 groups). m. Schematic illustrating open field test at room temperature (RT) and 5 °C. n-o. Within animal comparison of total locomotion or time in center during open field test at both room temperature and 5 °C after SAL or CNO injection (n = 20 animals). p. Representative infrared thermal images demonstrating temperature above BAT (brown adipose tissue, black circles) after SAL or CNO injection at room temperature. q. Quantification of thermography images in regions above BAT after SAL or CNO injection (n = 20 animals). r. Schematic illustrating behavioral thermotaxis assay. s. Within animal comparison of percent time spent in warm corner versus the average of three cold corners after SAL or CNO injection (n = 20 animals). Box plots: center line–median; box limits–upper/lower quartiles; whiskers–min/max. Statistical tests: one-way (h,i,k) and two-way (n,o,s) ANOVA with Bonferroni post-hoc, and two-tailed Wilcoxon matched pairs tests (c-g,j,l,q) tests. *P < .05, **P < .01, ***P < .001, ****P < .0001. See Supplementary Table 1 for statistical details.
Supplementary information
Supplementary Information (download PDF )
Supplementary Methods.
Supplementary Table 1 (download XLSX )
Detailed information for statistical analyses.
Source data
Source Data Fig. 1 (download XLSX )
Source data for plots and graphs.
Source Data Fig. 2 (download XLSX )
Source data for plots and graphs.
Source Data Fig. 3 (download XLSX )
Source data for plots and graphs.
Source Data Fig. 4 (download XLSX )
Source data for plots and graphs.
Source Data Fig. 5 (download XLSX )
Source data for plots and graphs.
Source Data Fig. 6 (download XLSX )
Source data for plots and graphs.
Source Data Extended Data Fig. 1 (download XLSX )
Source data for plots and graphs.
Source Data Extended Data Fig. 2 (download XLSX )
Source data for plots and graphs.
Source Data Extended Data Fig. 4 (download XLSX )
Source data for plots and graphs.
Source Data Extended Data Fig. 5 (download XLSX )
Source data for plots and graphs.
Source Data Extended Data Fig. 6 (download XLSX )
Source data for plots and graphs.
Source Data Extended Data Fig. 7 (download XLSX )
Source data for plots and graphs.
Source Data Extended Data Fig. 8 (download XLSX )
Source data for plots and graphs.
Source Data Extended Data Fig. 9 (download XLSX )
Source data for plots and graphs.
Source Data Extended Data Fig. 10 (download XLSX )
Source data for plots and graphs.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Raam, T., Li, Q., Gu, L. et al. Cortical regulation of collective social dynamics during environmental challenge. Nat Neurosci (2026). https://doi.org/10.1038/s41593-026-02224-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41593-026-02224-0