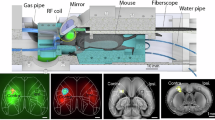
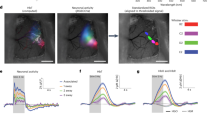
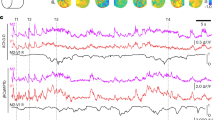
Abstract
Neuromodulatory transmitters have vasoactive properties. Therefore, the impulse response function (IRF) linking spontaneous neuronal activity with hemodynamics may depend on neuromodulation. To test this hypothesis, we used optical imaging to measure norepinephrine (NE) or acetylcholine (ACh), calcium (Ca2+) activity of cortical neurons and hemodynamics in cerebral cortex in awake mice. We show that modeling of hemodynamics as a weighted sum of Ca2+-specific and NE-specific IRFs (IRFCa2+ and IRFNE) convolved with the respective time courses dramatically improved the model performance compared to using IRFCa2+ alone. In contrast to NE, ACh was largely redundant with Ca2+ and, therefore, did not improve the hemodynamic estimation. Because NE covaried with arousal, we observed instances of the diminished hemodynamic coherence between cortical regions during high arousal despite coherent behavior of the underlying neuronal Ca2+ activity. We conclude that, without accounting for noradrenergic neuromodulation, diminished hemodynamic coherence can be falsely interpreted as neuronal desynchronizations in neuroimaging studies.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
All data were uploaded to Distributed Archives for Neurophysiology Data Integration (https://doi.org/10.48324/dandi.001543/0.260130.1715)82. Source data are provided with this paper.
Code availability
All MATLAB code used to analyze wide-field data and to load the uploaded NWB files is available from GitHub (https://github.com/NIL-NeuroScience/Neuromodulation).
References
Chen, N., Sugihara, H. & Sur, M. An acetylcholine-activated microcircuit drives temporal dynamics of cortical activity. Nat. Neurosci. 18, 892–902 (2015).
Breton-Provencher, V. & Sur, M. Active control of arousal by a locus coeruleus GABAergic circuit. Nat. Neurosci. 22, 218–228 (2019).
Thiele, A. & Bellgrove, M. A. Neuromodulation of attention. Neuron 97, 769–785 (2018).
Hamel, E. Perivascular nerves and the regulation of cerebrovascular tone. J. Appl. Physiol. (1985) 100, 1059–1064 (2006).
Renden, R. B. et al. Modulatory effects of noradrenergic and serotonergic signaling pathway on neurovascular coupling. Commun. Biol. 7, 287 (2024).
Biswal, B. et al. Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magn. Reson. Med. 34, 537–541 (1995).
Grimm, C. et al. Tonic and burst-like locus coeruleus stimulation distinctly shift network activity across the cortical hierarchy. Nat. Neurosci. 27, 2167–2177 (2024).
Oyarzabal, E. A. et al. Chemogenetic stimulation of tonic locus coeruleus activity strengthens the default mode network. Sci. Adv. 8, eabm9898 (2022).
Zerbi, V. et al. Rapid reconfiguration of the functional connectome after chemogenetic locus coeruleus activation. Neuron 103, 702–718 (2019).
Pais-Roldan, P. et al. Contribution of animal models toward understanding resting state functional connectivity. Neuroimage 245, 118630 (2021).
Turchi, J. et al. The basal forebrain regulates global resting-state fMRI fluctuations. Neuron 97, 940–952 (2018).
Leopold, D. A. & Maier, A. Ongoing physiological processes in the cerebral cortex. Neuroimage 62, 2190–2200 (2012).
Zaldivar, D. et al. Two distinct profiles of fMRI and neurophysiological activity elicited by acetylcholine in visual cortex. Proc. Natl Acad. Sci. USA 115, E12073–E12082 (2018).
Feng, J. et al. Monitoring norepinephrine release in vivo using next-generation GRABNE sensors. Neuron 112, 1930–1942 (2024).
Jing, M. et al. A genetically encoded fluorescent acetylcholine indicator for in vitro and in vivo studies. Nat. Biotechnol. 36, 726–737 (2018).
Jing, M. et al. An optimized acetylcholine sensor for monitoring in vivo cholinergic activity. Nat. Methods 17, 1139–1146 (2020).
Sabatini, B. L. & Tian, L. Imaging neurotransmitter and neuromodulator dynamics in vivo with genetically encoded indicators. Neuron 108, 17–32 (2020).
Doran, P. R. et al. Widefield in vivo imaging system with two fluorescence and two reflectance channels, a single sCMOS detector, and shielded illumination. Neurophotonics 11, 034310 (2024).
Shahsavarani, S. et al. Cortex-wide neural dynamics predict behavioral states and provide a neural basis for resting-state dynamic functional connectivity. Cell Rep. 42, 112527 (2023).
Lohani, S. et al. Spatiotemporally heterogeneous coordination of cholinergic and neocortical activity. Nat. Neurosci. 25, 1706–1713 (2022).
Reimer, J. et al. Pupil fluctuations track rapid changes in adrenergic and cholinergic activity in cortex. Nat. Commun. 7, 13289 (2016).
Pickel, V. M., Segal, M. & Bloom, F. E. A radioautographic study of the efferent pathways of the nucleus locus coeruleus. J. Comp. Neurol. 155, 15–42 (1974).
Wang, Q. et al. The Allen Mouse Brain Common Coordinate Framework: a 3D reference atlas. Cell 181, 936–953 (2020).
Korte, N. et al. Noradrenaline released from locus coeruleus axons contracts cerebral capillary pericytes via α2 adrenergic receptors. J. Cereb. Blood Flow Metab. 43, 1142–1152 (2023).
Devor, A. et al. Suppressed neuronal activity and concurrent arteriolar vasoconstriction may explain negative blood oxygenation level-dependent signal. J. Neurosci. 27, 4452–4459 (2007).
Uhlirova, H. et al. Cell type specificity of neurovascular coupling in cerebral cortex. eLife 5, e14315 (2016).
Vo, T. T. et al. Parvalbumin interneuron activity drives fast inhibition-induced vasoconstriction followed by slow substance P-mediated vasodilation. Proc. Natl Acad. Sci. USA 120, e2220777120 (2023).
Cerri, D. H. et al. Distinct neurochemical influences on fMRI response polarity in the striatum. Nat. Commun. 15, 1916 (2024).
Uhlirova, H. et al. The roadmap for estimation of cell-type-specific neuronal activity from non-invasive measurements. Philos. Trans. R. Soc. Lond. B Biol. Sci. 371, 20150356 (2016).
Cauli, B. & Hamel, E. Revisiting the role of neurons in neurovascular coupling. Front. Neuroenergetics 2, 9 (2010).
Drew, P. J. Vascular and neural basis of the BOLD signal. Curr. Opin. Neurobiol. 58, 61–69 (2019).
Toussay, X. et al. Locus coeruleus stimulation recruits a broad cortical neuronal network and increases cortical perfusion. J. Neurosci. 33, 3390–3401 (2013).
Raichle, M. E. et al. Central noradrenergic regulation of cerebral blood flow and vascular permeability. Proc. Natl Acad. Sci. USA 72, 3726–3730 (1975).
Segal, S. S., Damon, D. N. & Duling, B. R. Propagation of vasomotor responses coordinates arteriolar resistances. Am. J. Physiol. 256, H832–H837 (1989).
O’Donnell, J. et al. Norepinephrine: a neuromodulator that boosts the function of multiple cell types to optimize CNS performance. Neurochem. Res. 37, 2496–2512 (2012).
Labarrera, C. et al. Adrenergic modulation regulates the dendritic excitability of layer 5 pyramidal neurons in vivo. Cell Rep. 23, 1034–1044 (2018).
Deitcher, Y. et al. Nonlinear relationship between multimodal adrenergic responses and local dendritic activity in primary sensory cortices. Preprint at bioRxiv https://doi.org/10.1101/814657 (2019).
Hauglund, N. L. et al. Norepinephrine-mediated slow vasomotion drives glymphatic clearance during sleep. Cell 188, 606–622 (2025).
Sun, L. et al. Globally patterned locus coeruleus–norepinephrine neuron–pericyte coupling orchestrates brain-wide vascular dynamics. Neuron 114, 287–306 (2026).
Chao, T. H. et al. Computing hemodynamic response functions from concurrent spectral fiber-photometry and fMRI data. Neurophotonics 9, 032205 (2022).
Zhang, W. T. et al. Spectral fiber photometry derives hemoglobin concentration changes for accurate measurement of fluorescent sensor activity. Cell Rep. Methods 2, 100243 (2022).
Ma, Z. et al. Gaining insight into the neural basis of resting-state fMRI signal. Neuroimage 250, 118960 (2022).
Tong, C. et al. Differential coupling between subcortical calcium and BOLD signals during evoked and resting state through simultaneous calcium fiber photometry and fMRI. Neuroimage 200, 405–413 (2019).
Drew, P. J. Neurovascular coupling: motive unknown. Trends Neurosci. 45, 809–819 (2022).
Jenkins, B. G. Pharmacologic magnetic resonance imaging (phMRI): imaging drug action in the brain. Neuroimage 62, 1072–1085 (2012).
Smialowska, M., Gastol-Lewinska, L. & Tokarski, K. The role of α-1 adrenergic receptors in the stimulating effect of neuropeptide Y (NPY) on rat behavioural activity. Neuropeptides 26, 225–232 (1994).
Tian, P. et al. Monte Carlo simulation of the spatial resolution and depth sensitivity of two-dimensional optical imaging of the brain. J. Biomed. Opt. 16, 016006 (2011).
Major, G., Larkum, M. E. & Schiller, J. Active properties of neocortical pyramidal neuron dendrites. Annu. Rev. Neurosci. 36, 1–24 (2013).
Lacroix, A. et al. COX-2-derived prostaglandin E2 produced by pyramidal neurons contributes to neurovascular coupling in the rodent cerebral cortex. J. Neurosci. 35, 11791–11810 (2015).
Lecrux, C. et al. Pyramidal neurons are ‘neurogenic hubs’ in the neurovascular coupling response to whisker stimulation. J. Neurosci. 31, 9836–9847 (2011).
Nishimura, N. et al. Limitations of collateral flow after occlusion of a single cortical penetrating arteriole. J. Cereb. Blood. Flow Metab. 30, 1914–1927 (2010).
Granger, A. J. et al. Cortical ChAT+ neurons cotransmit acetylcholine and GABA in a target- and brain-region-specific manner. eLife 9, e57749 (2020).
Obermayer, J. et al. Prefrontal cortical ChAT-VIP interneurons provide local excitation by cholinergic synaptic transmission and control attention. Nat. Commun. 10, 5280 (2019).
Knudstrup, S. G. et al. Visual stimulation drives retinotopic acetylcholine release in the mouse visual cortex. Preprint at bioRxiv https://doi.org/10.1101/2024.02.04.578821 (2024).
Schneider, M. et al. Spontaneous pupil dilations during the resting state are associated with activation of the salience network. Neuroimage 139, 189–201 (2016).
DiNuzzo, M. et al. Brain networks underlying eye’s pupil dynamics. Front. Neurosci. 13, 965 (2019).
Yellin, D., Berkovich-Ohana, A. & Malach, R. Coupling between pupil fluctuations and resting-state fMRI uncovers a slow build-up of antagonistic responses in the human cortex. Neuroimage 106, 414–427 (2015).
Gozzi, A. & Schwarz, A. J. Large-scale functional connectivity networks in the rodent brain. Neuroimage 127, 496–509 (2016).
Grandjean, J. et al. Common functional networks in the mouse brain revealed by multi-centre resting-state fMRI analysis. Neuroimage 205, 116278 (2020).
Xu, N. et al. Functional connectivity of the brain across rodents and humans. Front. Neurosci. 16, 816331 (2022).
Whitesell, J. D. et al. Regional, layer, and cell-type-specific connectivity of the mouse default mode network. Neuron 109, 545–559 (2021).
Hayden, B. Y., Smith, D. V. & Platt, M. L. Electrophysiological correlates of default-mode processing in macaque posterior cingulate cortex. Proc. Natl Acad. Sci. USA 106, 5948–5953 (2009).
van den Heuvel, M. P. & Sporns, O. A cross-disorder connectome landscape of brain dysconnectivity. Nat. Rev. Neurosci. 20, 435–446 (2019).
Hutchison, R. M. et al. Dynamic functional connectivity: promise, issues, and interpretations. Neuroimage 80, 360–378 (2013).
Benisty, H. et al. Rapid fluctuations in functional connectivity of cortical networks encode spontaneous behavior. Nat. Neurosci. 27, 148–158 (2024).
Higley, M. J. & Cardin, J. A. Spatiotemporal dynamics in large-scale cortical networks. Curr. Opin. Neurobiol. 77, 102627 (2022).
Shine, J. M. Neuromodulatory influences on integration and segregation in the brain. Trends Cogn. Sci. 23, 572–583 (2019).
Pais-Roldán, P. et al. Indexing brain state-dependent pupil dynamics with simultaneous fMRI and optical fiber calcium recording. Proc. Natl Acad. Sci. USA 117, 6875–6882 (2020).
Sobczak, F. et al. Decoding the brain state-dependent relationship between pupil dynamics and resting state fMRI signal fluctuation. eLife 10, e68980 (2021).
Drew, P. J. et al. Ultra-slow oscillations in fMRI and resting-state connectivity: neuronal and vascular contributions and technical confounds. Neuron 107, 782–804 (2020).
Halnes, G. et al., Electric Brain Signals: Foundations and Applications of Biophysical Modeling (Cambridge University Press, 2024).
Dana, H. et al. Sensitive red protein calcium indicators for imaging neural activity. eLife 5, e12727 (2016).
Chan, K. Y. et al. Engineered AAVs for efficient noninvasive gene delivery to the central and peripheral nervous systems. Nat. Neurosci. 8, 1172–1179 (2017).
Goldey, G. J. et al. Removable cranial windows for long-term imaging in awake mice. Nat. Protoc. 9, 2515–2538 (2014).
Kilic, K. et al. Chronic cranial windows for long term multimodal neurovascular imaging in mice. Front. Physiol. 11, 612678 (2020).
Turner, K. L. et al. Neurovascular coupling and bilateral connectivity during NREM and REM sleep. eLife 9, e62071 (2020).
Ma, Y. et al. Resting-state hemodynamics are spatiotemporally coupled to synchronized and symmetric neural activity in excitatory neurons. Proc. Natl Acad. Sci. USA 113, E8463–E8471 (2016).
Kobat, D. et al. Deep tissue multiphoton microscopy using longer wavelength excitation. Opt. Express 17, 13354–13364 (2009).
OʼShea, T. M. et al. Foreign body responses in mouse central nervous system mimic natural wound responses and alter biomaterial functions. Nat. Commun. 11, 6203 (2020).
Stringer, C. et al. Spontaneous behaviors drive multidimensional, brainwide activity. Science 364, eaav7893 (2019).
Bokil, H. et al. Chronux: a platform for analyzing neural signals. J. Neurosci. Methods 192, 146–151 (2010).
Rauscher, B. et al. Neurovascular impulse response function (IRF) during spontaneous activity differentially reflects intrinsic neuromodulation across cortical regions. DANDI https://doi.org/10.48324/dandi.001543/0.260130.1715 (2026).
Acknowledgements
We thank Y. Li for providing the latest GRAB constructs. We thank A. Arkhipov, L.-H. Tsai and X. Han for helpful discussions and T. O’Shea for help with immunostainings. We gratefully acknowledge support from the National Institutes of Health (NIH; Brain Research Through Advancing Innovative Neurotechnologies (BRAIN) Initiative U19NS123717, BRAIN Initiative R01NS122742, R01DA050159 and T32NS136080), the Boston University Neurophotonics Center and the Boston University Kilachand Fund. B.C.R. was supported by a Ruth L. Kirschstein Predoctoral Fellowship F31NS145737 and Boston University Neurophotonics Center fellowship. P.R.D. was supported by a Ruth L. Kirschstein Predoctoral Fellowship F31NS118949. K.E.H. was supported by a Ruth L. Kirschstein Predoctoral Fellowship F31NS141509 and Boston University Neurophotonics Center CAN-DO award. N.X.C. was supported by a Garry Goldwater Undergraduate Fellowship and Boston University UROP award. P.F.B. was supported by the NIH (T32NS131178) and National Science Foundation Graduate Research Fellowship Program (2234657). The funders had no role in study design, data collection and analysis, decision to publish or preparation of the paper.
Author information
Authors and Affiliations
Contributions
B.C.R. and N.F.-H., investigation, formal analysis, conceptualization, methodology, visualization, writing—original draft and writing—review and editing. S.K., formal analysis, methodology and writing—review and editing. P.R.D., methodology, software, writing—original draft and writing—review and editing. P.D.P., D.B. and P.F.B., formal analysis, conceptualization and writing—review and editing. N.X.C., F.A.F. and A.G., investigation, formal analysis and writing—review and editing. K.E.H., investigation, visualization, resources, writing—original draft and writing—review and editing. K.K., E.A.M. and J.X.J., resources and writing—review and editing. R.P.T. and S.G.K., investigation and writing—review and editing. J.P.G., M.L., D.K., M.E.H., L.D.L., E.P.S., D.A.B., L.T., G.M. and S.S., conceptualization, methodology, supervision and writing—review and editing. M.T., conceptualization, methodology, software, supervision, writing—original draft and writing—review and editing. A.D., conceptualization, funding acquisition, supervision, writing—original draft and writing—review and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Neuroscience thanks Alessandro Gozzi, Yen-Yu Shih, Valerio Zerbi and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Accuracy of the HbT models in the GRABNE cohort.
a) Maps of HbT prediction accuracy for the Global IRF model for each mouse in the GRABNE cohort. The ‘standard FOV’ is outlined in black/gray. b-e) Same as (a) but for the SSp IRF, Variant IRF, Linear regression, and Double IRF model, respectively. f-g) Maps of the Ca2+ (f) and NE (g) coefficients (A and B, respectively) estimated in the Linear regression model for each mouse.
Extended Data Fig. 2 Collinearity between optical measures and behavioral readouts.
a) Normalized power spectra for Ca2+ (red), NE (green), ACh (orange), and HbT (blue; n = 16,8,8,16 subjects; mean ± SEM). b) Lag cross-correlation between ACh vs. Ca2+ (orange; n = 8 subjects; mean ± SEM) and NE vs. Ca2+ (green; n = 8 subjects; mean ± SEM). c) Left: average correlation between the eye pupil diameter (purple), whisking (cyan), and movement (black) with ACh, NE, Ca2+, and HbT (n = 8,8,16,16 subjects; mean ± SEM; r = 0.37 ± 0.05, 0.29 ± 0.04, 0.26 ± 0.07, 0.56 ± 0.04, 0.15 ± 0.02, 0.23 ± 0.02, 0.44 ± 0.03, 0.30 ± 0.02, 0.35 ± 0.04, 0.08 ± 0.05, 0.12 ± 0.02, 0.09 ± 0.03). Each point represents an average across runs for one subject. Right: correlation matrix across all seven measurements. d) Correlation map between low pass filtered ACh and low pass filtered Ca2+ ( < 0.5 Hz; n = 8 subjects). Space-averaged performance across subjects is shown on the right (n = 8 subjects; mean ± SEM; r = 0.70 ± 0.03). e) Coherence between Ca2+ and ACh (orange) and phase (black; n = 8 subjects; mean ± SEM). f) Example Ca2+ and ACh time-courses derived from the SSp-ll and SSp-bfd. g) Ca2+ averaged within the SSp-ll region plotted against the average ACh (left) and NE (right) within the same region for a single 10-min run. Correlation value displayed in red. h) Same as (d) but for NE vs. Ca2+ (n = 8 subjects; mean ± SEM; r = 0.47 ± 0.06). i) Same as (e) but for NE vs. Ca2+. j) Direct IRF estimation using matrix division between NE and Ca2+ (left) and prediction accuracy map calculated as the correlation between the experimental and predicted NE (right; n = 8 subjects; mean ± SEM). Statistics across subjects is shown on the right (r = 0.61 ± 0.04). k) Average connectivity matrix of NE showing the correlation across cortical regions (n = 8 subjects). l) Coherence along with phase lag between Ca2+ (left), NE (middle), and ACh (right) vs. HbT (n = 16,8,8 subjects, respectively; mean ± SEM). m) Lag cross-correlation between ACh (orange), NE (green), and Ca2+ (red) vs. HbT (n = 8,8,16 subjects, respectively; mean ± SEM).
Extended Data Fig. 3 Linear Regression and Double IRF prediction models with respect to HbO and HbR.
a) Maps of the weights, A (left) and B (right), in the Linear regression model for HbO (n = 8 subjects). b) HbO timing coefficients for Ca2+ and NE (tA, tB); each point corresponds to an average across runs for one subject (n = 8 subjects; mean ± SEM; tA = 0.39 ± 0.08, tB = 0.30 ± 0.07). c) Linear Regression model performance map quantified as the correlation between the experimental and predicted HbO (n = 8 subjects). d) Difference between HbO Linear Regression model performance and HbT Linear Regression model performance (n = 8 subjects). e-h) Same as (a-d) but for HbR. For (f), tA = 0.28 ± 0.08, tB = 0.52 ± 0.17. i, k, l, m, o, p) Same as (a, c, d, e, g, h) but for the Double IRF model. j, n) Estimated IRFCa2+ and IRFNE for the HbO and HbR model (n = 8 subjects; mean ± SEM). q) Side-by-side model comparison. For each subject and model, the model performance (r) was averaged across space. Each point represents one subject (*p < 0.01 two-sample, two-sided Kolmogorov-Smirnov Test; mean ± SEM; r = 0.32 ± 0.02, 0.29 ± 0.03, 0.39 ± 0.02, 0.60 ± 0.03, 0.59 ± 0.03, 0.69 ± 0.02, 0.67 ± 0.03, 0.67 ± 0.01, 0.65 ± 0.02; n = 16 subjects for Global, SSp, and Variant models; n = 8 subjects for Double IRF and Linear regression models). P-values are provided in figure source data.
Extended Data Fig. 4 Two-photon imaging of Ca2+, NE, and arteriolar diameter.
a) Imaging setup depicts Ti:sapph laser set to 990 traveling through an electro-optic modulator (EOM) to excite GRABNE, jRGECO1a and Alexa 680; D900LP, D685LP and D575LP—long-pass dichroic mirrors with a cutoff at 900, 685 and 575 nm, respectively; GaAsP and multialkaline—PMT tubes; 900SP—short-pass optical filter with a cutoff at 900 nm; Em525/70, Em617/70, Em736/128—bandpass emission filters. Absorption plot at the top right indicates the emission spectra of our desired fluorophores. b) Left: An example average intensity projection of a low magnification image stack across the top 300 µm showing the vasculature labeled with Alexa 680. Yellow rectangle indicates the ROI chosen for imaging of brain activity. Right: A field of view 170 µm below the cortical surface with a diving arteriole labeled with Alexa 680(blue), neurons labeled with jRGECO1a (red), and GRABNE (green). Time-courses at the bottom show spontaneous activity acquired at 10 Hz. c) Cross-correlation of the NE signal and arteriolar diameter for different arteries (ROIs) across three cortical regions: MOs, SSp and VISp. d) Average across ROIs for the three cortical regions. e) Cross-correlation of the NE signal and HbT derived from mesoscale imaging (n = 8 subjects; mean ± SEM).
Extended Data Fig. 5 Effects of HbT filtering and NE reshuffling.
a) Maps of the weights, A (left) and B (right), in the Linear regression model using unfiltered HbT (n = 8 subjects). b) Timing coefficients for Ca2+ and NE (tA, tB) for each subject using unfiltered HbT; each dot shows an average across runs for one subject (n = 8 subjects; mean ± SEM; tA = 0.73 ± 0.08, tB = -0.04 ± 0.05). c) Linear Regression model performance map quantified as the correlation between experimental and predicted HbT using unfiltered HbT (n = 8 subjects). d) Difference between unfiltered and filtered Linear Regression model performance (n = 8 subjects). e, g, h) Same as (a, c, d) but for the Double IRF model. f) Estimated IRFCa2+ and IRFNE using unfiltered HbT (n = 8 subjects; mean ± SEM). i) Maps of the weights, A (left) and B (right), in the Linear regression model after shuffling the NE data (n = 8 subjects). j) Timing coefficients for Ca2+ and NE (tA, tB) for each subject after shuffling NE; each dot shows an average across runs for one subject (n = 8 subjects; mean ± SEM; tA = 0.63 ± 0.05, tB = 0.09 ± 0.02). k) Difference between shuffled NE Linear Regression model performance and Global IRF model performance (n = 8 subjects). l, n) Same as (i, k) but for the Double IRF model. m) Estimated IRFCa2+ and IRFNE after shuffling NE (n = 8 subjects; mean ± SEM). o) Side-by-side model comparison. For each subject and model, the model performance (r) was averaged across space. Each point represents one subject (*p < 0.01 two-sample, two-sided Kolmogorov-Smirnov Test; mean ± SEM; r = 0.32 ± 0.02, 0.29 ± 0.03, 0.39 ± 0.02, 0.60 ± 0.03, 0.59 ± 0.03, 0.52 ± 0.02, 0.53 ± 0.02, 0.37 ± 0.03, 0.37 ± 0.03 for the Global, SSp, and variant IRF models, the Linear Regression and Double IRF models, the unfiltered Linear Regression and Double IRF models, and the shuffled Linear Regression and Double IRF models; n = 16 subjects for the Global, SSp, and variant IRF models n = 8 subjects for all models using NE). P-values are provided in figure source data.
Extended Data Fig. 6 Blockade of a- and b-adrenergic receptors.
a) Schematic of the experimental paradigm. A cocktail of prazosin, propranolol, and atipamezole (PPA) was delivered IP. b-c) Example Ca2+ and HbT time courses along with behavior readouts recorded during the baseline (pre-PPA, b) and post-injection (post-PPA, c). d) Coherence between Ca2+ and HbT pre- (orange) and post-PPA (cyan) coherence (n = 3 subjects; mean ± SEM). e) Left: Coherence in the <0.1 Hz and 0.1-0.5 Hz frequency band for each cortical region comparing pre- and post-PPA. Right: difference between the pre- and post-PPA (averaged across 3 subjects). f) Lag cross-correlation between Ca2+ and HbT comparing pre- (orange) and post-PPA (cyan; averaged across 3 subjects; mean ± SEM). g) Estimated IRFCa2+ using the Variant IRF model from SSp-tr and SSp-ll (top) and MOs (bottom) comparing pre- (orange) and post-PPA (cyan; averaged across 3 subjects; mean ± SEM). h) Accuracy of the Variant IRF model comparing pre- and post-PPA along with the difference between the two (n = 3 subjects). i) Average Variant IRF performance pre- and post-PPA (orange and cyan, respectively) in SSp-bfd. Each dot shows an average across runs for one subject (*p < 0.05 two-sample, two-sided Kolmogorov-Smirnov Test; n = 3 subjects; mean ± SEM; r = 0.30 ± 0.01, 0.53 ± 0.04 for pre- and post-PPA, respectively, p = 3.3e-2).
Extended Data Fig. 7 Comparison of FC results for high and low frequency Ca2+ activity and HbT.
a) Low frequency ( < 0.1 Hz) Ca2+ connectivity matrices, averaged across subjects, comparing connectivity during low NE ( < 30th percentile; left) and high NE ( > 70th percentile; middle). Right: Difference between the high and low NE (‘+’ indicates p < 0.05, two-sided linear mixed-effects model, comparisons between low and high NE were performed separately for each region pair, n = 123 sessions, 8 subjects). P-values are provided in figure source data. b-c) Same as (a) but for 0.1-0.5 Hz and 0.5-5 Hz frequency bands. d-e) Same as (a-b) but for HbT. f) Low frequency ( < 0.1 Hz) Ca2+ seed correlation maps, averaged across subjects, comparing connectivity during low NE and high NE for MOs, SSp-ll, and VISp regions (n = 8 subjects). g-h) Same as (f) but for 0.1-0.5 Hz and 0.5-5 Hz frequency bands. i-j) Same as (f-g) but for HbT.
Extended Data Fig. 8 Spectral analysis of Ca2+ and HbT during low and high NE.
a) The average of detected global NE “events” (see Methods) (n = 301 events accumulated from 8 subjects; mean ± SEM). b) Averaged event-triggered global Ca2+ spectrogram corresponding to the NE events from (a) (n = 301 events, 8 subjects). The power is multiplied by frequency to correct for the 1/f relationship. c) Same as (b) but for global HbT (n = 301 events, 8 subjects). d) Comparison between subject-averaged, global Ca2+ and HbT spectra (top and bottom, respectively) during periods of low NE ( < 30th percentile; cyan) and high NE ( > 70th percentile; orange) (n = 8 subjects; mean ± SEM). The power is multiplied by frequency to correct for the 1/f relationship. e) Maps of the average Ca2+ spectral power (corrected for 1/f) during periods of low (top) and high (middle) NE. Bottom: The difference between high and low NE; n = 8 subjects). f) Same as (e) but for HbT (n = 8 subjects).
Extended Data Fig. 9 Regression of NE from HbT increases the similarity between FCCa and FCHbT.
a) Excerpt from Fig. 3a: Example Ca2+, NE, and HbT time-courses derived from two cortical regions, MOs and SSp-ll, along with behavior readouts. The dashed vertical lines indicate several NE peaks to facilitate visual inspection. b) Top: Excerpt from Fig. 3b: Dynamic functional connectivity between MOs and SSp-ll for Ca2+ (red) and HbT (blue) computed as the correlation between the respective time-courses using a 30-s sliding window. Bottom: Dynamic functional connectivity between MOs and SSp-ll for HbT after regression of the NE signal using the Linear regression model weights and lag. c) Left: Subject-averaged correlation between Ca2+ and HbT connectivity time courses (see Methods). Middle: the same after regression of region-specific NE signal from HbT using the Linear regression model. Right: the same as the left panel after direct regression of global NE from HbT (n = 8 subjects, n = 123 sessions, 8 subjects). d) Average HbT connectivity matrices comparing connectivity during low NE ( < 30th percentile; left) and high NE ( > 70th percentile; middle) after regression of region-specific NE signal using the Linear regression model. Right: Difference between the high and low NE (‘+’ indicates p < 0.05, two-sided linear mixed-effects model, comparisons between low and high NE were performed separately for each region pair, n = 123 sessions, 8 subjects). P-values are provided in figure source data. e) Same as (d) but regressing global NE from HbT.
Extended Data Fig. 10 The eye pupil diameter measurement, illumination, and hemodynamic correction.
a-b) Agreement between automated and manual eye pupil measurements. Example time-course of estimated pupil diameter using the algorithm described in Methods (a) and manual eye pupil measurement (b) corresponding to the time points marked in (a). The video frames in (b) (179 and 243 s) show manual annotations of the eye and eye pupil diameter and the calculated ratio. c) Variability in Ca2+ signal amplitude does not follow variation in the illumination intensity. Left: An example map of the average raw jRGECO fluorescent intensity (F) for a single run reflecting variation in the illumination intensity. Right: A map of the standard deviation of the Ca2+ ∆F/F signal for the same run. d) Example time courses of Ca2+ ∆F/F from the MOs and SSp-bfd. e) The pixel-by-pixel standard deviation of the Ca2+ ∆F/F signal across time plotted against the average raw jRGECO fluorescent intensity for the same pixels within the exposure outlined in (c). f) Top: A 2-photon imaging field of view 230 µm below the cortical surface with a diving arteriole labeled with Alexa 680 (blue) and GRABNE-mutant (gray). Time-courses at the bottom show spontaneous activity acquired at ~15 Hz. g) Example widefield HbT (blue), uncorrected GRABNE-mutant signal (red), and corrected GRABNE-mutant signal (gray) time-courses from SSp-ll. h) Average cross-correlation function between 2-photon GRABNE-mutant vs. vessel diameter for the imaging session shown in (f). i) Subject-averaged cross-correlation functions between widefield GRABACh, GRABNE, and GRABNE-mutant vs. HbT (n = 8,8,3 subjects, respectively; mean ± SEM).
Supplementary information
Supplementary Information (download PDF )
Supplementary Fig. 1, and Tables 1 and 2.
Supplementary Video 1 (download AVI )
Simultaneous mesoscopic imaging of Ca2+ and NE fluorescence and HbT. a–c, Mesoscopic video of Ca2+ (top), NE release (mid) and HbT (bottom). d, Video recording of the mouse using an infrared behavior CCD camera. e, The Ca2+, NE and HbT signals averaged within the SSp-ll (top) and SSp-bfd (bottom) regions. f, The estimated pupil diameter, whisking and movement.
Supplementary Video 2 (download AVI )
SSp IRF model performance as a function of NE and eye pupil diameter. a, Example video showing the SSp IRF model performance over time. b, Simultaneous measurements of NE and eye pupil diameter.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 6 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 7 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 8 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 9 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 10 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rauscher, B.C., Fomin-Thunemann, N., Kura, S. et al. The neurovascular impulse response function differentially reflects intrinsic neuromodulation across cortical regions. Nat Neurosci (2026). https://doi.org/10.1038/s41593-026-02239-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41593-026-02239-7