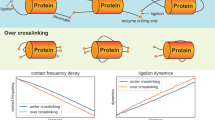
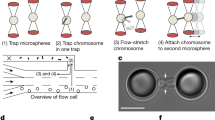
Abstract
The chromosome conformation capture method and its derivatives, such as circularized chromosome conformation capture, carbon copy chromosome conformation capture, high-throughput chromosome conformation capture and capture high-throughput chromosome conformation capture, have pioneered our understanding of the principles of chromosome folding in the nucleus. These technical advances, however, cannot precisely quantitate interaction frequency in very small input samples. Here we describe a protocol for the Nodewalk assay, which is based on converting chromosome conformation capture DNA samples to RNA and subsequently to cDNA using strategically placed primers. This pipeline enables the quantitative analyses of chromatin fiber interactions without compromising its sensitivity down to <300 cells, making it suitable for MiSeq analyses of higher-order chromatin structures in biopsies, circulating tumor cells and transitional cell states, for example. Importantly, the quality of the Nodewalk sample can be assessed before sequencing to avoid unnecessary costs. Moreover, it enables analyses from hundreds of different restriction enzyme fragment viewpoints within the same initial small input sample to uncover complex, genome-wide networks. Following optimization of the different steps, the entire protocol can be completed within 2 weeks.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
All processed Nodewalk data have been deposited in the Gene Expression Omnibus (GSE76049). The ChIP and DamId-seq data were retrieved from the Gene Expression Omnibus as follows: cLADs (GSE22428), H3K9me2 (GSE58534) and H3K4me1 (GSM1240111). Source data for Figs. 6, 7, 8 and 3, 4 are available online in refs. 24,25, respectively. The images in Fig. 2 represent unprocessed original Bioanalyzer files.
Code availability
The analysis pipeline is available at https://github.com/Anita-Rolf-lab/Nodewalk.
References
Hansen, A. S., Cattoglio, C., Darzacq, X. & Tjian, R. Recent evidence that TADs and chromatin loops are dynamic structures. Nucleus 9, 20–32 (2018).
Lieberman-Aiden, E. et al. Comprehensive mapping of long-range interactions reveals folding principles of the human genome. Science 326, 289–293 (2009).
Dixon, J. R. et al. Topological domains in mammalian genomes identified by analysis of chromatin interactions. Nature 485, 376–380 (2012).
Brant, L. et al. Exploiting native forces to capture chromosome conformation in mammalian cell nuclei. Mol. Syst. Biol. 12, 891 (2016).
Beagrie, R. A. et al. Complex multi-enhancer contacts captured by genome architecture mapping. Nature 543, 519–524 (2017).
Bintu, B. et al. Super-resolution chromatin tracing reveals domains and cooperative interactions in single cells. Science 362, eaau1783 (2018).
Quinodoz, S. A. et al. Higher-order inter-chromosomal hubs shape 3D genome organization in the nucleus. Cell 174, 744–757 e724 (2018).
Tan, L., Xing, D., Chang, C. H., Li, H. & Xie, X. S. Three-dimensional genome structures of single diploid human cells. Science 361, 924–928 (2018).
Ulianov, S. V. et al. Order and stochasticity in the folding of individual Drosophila genomes. Nat. Commun. 12, 41 (2021).
Chang, L. H., Ghosh, S. & Noordermeer, D. TADs and their borders: free movement or building a wall? J. Mol. Biol. 432, 643–652 (2020).
Lupianez, D. G. et al. Disruptions of topological chromatin domains cause pathogenic rewiring of gene–enhancer interactions. Cell 161, 1012–1025 (2015).
Osterwalder, M. et al. Enhancer redundancy provides phenotypic robustness in mammalian development. Nature 554, 239–243 (2018).
Darrow, E. M. et al. Deletion of DXZ4 on the human inactive X chromosome alters higher-order genome architecture. Proc. Natl Acad. Sci. USA 113, E4504–E4512 (2016).
Ay, F. et al. Identifying multi-locus chromatin contacts in human cells using tethered multiple 3C. BMC Genomics 16, 121 (2015).
Olivares-Chauvet, P. et al. Capturing pairwise and multi-way chromosomal conformations using chromosomal walks. Nature 540, 296–300 (2016).
de Laat, W. & Grosveld, F. Spatial organization of gene expression: the active chromatin hub. Chromosome Res. 11, 447–459 (2003).
Allahyar, A. et al. Enhancer hubs and loop collisions identified from single-allele topologies. Nat. Genet. 50, 1151–1160 (2018).
Oudelaar, A. M. et al. Single-allele chromatin interactions identify regulatory hubs in dynamic compartmentalized domains. Nat. Genet. 50, 1744–1751 (2018).
Fudenberg, G. et al. Formation of chromosomal domains by loop extrusion. Cell Rep. 15, 2038–2049 (2016).
Zhao, Z. et al. Circular chromosome conformation capture (4C) uncovers extensive networks of epigenetically regulated intra- and interchromosomal interactions. Nat. Genet. 38, 1341–1347 (2006).
Zhao, H. et al. PARP1- and CTCF-mediated interactions between active and repressed chromatin at the lamina promote oscillating transcription. Mol. Cell 59, 984–997 (2015).
Gondor, A., Rougier, C. & Ohlsson, R. High-resolution circular chromosome conformation capture assay. Nat. Protoc. 3, 303–313 (2008).
Paulsen, J. et al. Long-range interactions between topologically associating domains shape the four-dimensional genome during differentiation. Nat. Genet. 51, 835–843 (2019).
Sumida, N. et al. MYC as a driver of stochastic chromatin networks: implications for the fitness of cancer cells. Nucleic Acids Res. 48, 10867–10876 (2020).
Scholz, B. A. et al. WNT signaling and AHCTF1 promote oncogenic MYC expression through super-enhancer-mediated gene gating. Nat. Genet. 51, 1723–1731 (2019).
Chachoua, I. et al. Canonical WNT signaling-dependent gating of MYC requires a noncanonical CTCF function at a distal binding site. Nat. Commun. 13, 204 (2022).
Dekker, J., Rippe, K., Dekker, M. & Kleckner, N. Capturing chromosome conformation. Science 295, 1306–1311 (2002).
Timp, W. et al. Large hypomethylated blocks as a universal defining epigenetic alteration in human solid tumors. Genome Med. 6, 61 (2014).
Lewenshtein, B. Binary codes capable of correcting deletions, insertions, and reversals. Sov. Phys. Dokl. 10, 707–710 (1965).
Williams, J. & Mackinnon, D. P. Resampling and distribution of the product methods for testing indirect effects in complex models. Struct. Equ. Modeling 15, 23–51 (2008).
Acknowledgements
This work was supported by the Swedish Research Council (VR 03108), the Swedish Childhood Cancer Fund (PR2017-0132), the Swedish Cancer Society (CAN 708), the Lundberg Foundation (2018-0138), Karolinska Institutet, the Novo Nordisk Foundation (NNF16OC0021512), the Cancer Society in Stockholm (2018–2021) and the KA Wallenberg Foundation (KAW 2017.0077). We thank A. F. Woodbridge for his efforts in the initial development of the Nodewalk pipeline and R. Ohlsson for valuable discussions.
Author information
Authors and Affiliations
Contributions
J.V. updated the Nodewalk protocol and contributed to manuscript writing; N.S. contributed to the invention of the Nodewalk assay, developed the entire protocol and contributed to the development of the analysis pipeline and manuscript writing; R.M. and D.B. designed the submitted state of pipeline and implemented the Python code; D.B. designed the manual and tested the code; S.W. contributed to the pipeline code integration and A.G. contributed to the invention of the Nodewalk assay and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
D.B. has formed a bioinformatics company, Genomiki Solution Ltd., that analyses high throughput data, including chromatin fibre interaction maps.
Peer review
Peer review information
Nature Protocols thanks Argyris Papantonis and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Sumida, N. et al. Nucleic Acids Res. 48, 10867–10876 (2020): https://doi.org/10.1093/nar/gkaa817
Scholz, B. A. et al. Nat. Genet. 51, 1723–1731 (2019): https://doi.org/10.1038/s41588-019-0535-3
Supplementary information
Supplementary Information (download PDF )
Supplementary Note.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vestlund, J., Sumida, N., Mehmood, R. et al. The Nodewalk assay to quantitate chromatin fiber interactomes in very small cell populations. Nat Protoc 18, 755–782 (2023). https://doi.org/10.1038/s41596-022-00774-8
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41596-022-00774-8