Abstract
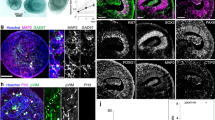
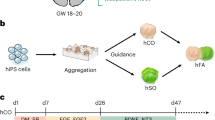
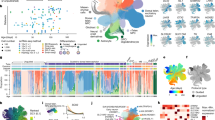
We have developed a method for generating human telencephalic organoids from stem cell-derived isolated single neural rosettes. The use of single neural rosettes for generating organoids offers several important advantages. First, it mimics the development of neural tissue from a singular neural tube in vivo. Second, single neural rosette-derived organoids exhibit a relatively consistent and reproducible composition of telencephalic neural cells. Finally, single neural rosette-derived organoids demonstrate predictable organization of the identified neural cells around a single neural rosette-derived lumen and contain a large proportion of functionally mature neurons that generate action potentials and receive both excitatory and inhibitory synaptic inputs. These unique features of our protocol enable the study of the specification and organization of different neural cells in the developing human telencephalon, as well as modeling of neurodevelopmental disorders associated with disrupted neural networks. Here, we describe our protocols for generating CRISPR–Cas9-engineered human stem cells with a hemizygous SHANK3 deletion, stem cell-derived single neural rosettes and telencephalic brain organoids. We also offer insights on how to conduct single-cell RNA sequencing, immunohistochemistry and slice patch-clamp electrophysiology on these organoids. Completion of the protocols takes 5–6 months and requires experience working with cultured cells. We expect this protocol will prove useful for studies of human brain development and disease, as well as for advancing the development of new organoid-based biocomputers.
Key points
-
This protocol describes the generation and characterization of human telencephalic brain organoids from stem cell-derived single neural rosettes. Procedures for studying the organization of the different neural cells, as well as generating models of neurodevelopmental disorders are included.
-
Compared with other organoids that develop form multiple rosettes, single rosette-derived organoids have a predictable organization with a reproducible cellular composition and functional neural networks.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
Data availability
The data related to this protocol are included in the original paper43. Interactive visualization of our transcriptomic and electrophysiological data is provided in our online browser: http://organoid.chpc.utah.edu.
Code availability
We developed and open- sourced the Shiny Single Cell Browser software in R to build the single-cell RNA-seq browser, code available: https://github.com/yueqiw/shiny_cell_browser. The electrophysiology browser was developed in Python, code available at https://github.com/yueqiw/ephys_analysis.
References
Rakic, P. Evolution of the neocortex: a perspective from developmental biology. Nat. Rev. Neurosci. 10, 724–735 (2009).
Vanderhaeghen, P. & Polleux, F. Developmental mechanisms underlying the evolution of human cortical circuits. Nat. Rev. Neurosci. 24, 213–232 (2023).
Wallace, J. L. & Pollen, A. A. Human neuronal maturation comes of age: cellular mechanisms and species differences. Nat. Rev. Neurosci. 25, 7–29 (2023).
Lui, J. H., Hansen, D. V. & Kriegstein, A. R. Development and evolution of the human neocortex. Cell 146, 18–36 (2011).
Silbereis, J. C., Pochareddy, S., Zhu, Y., Li, M. & Sestan, N. The cellular and molecular landscapes of the developing human central nervous system. Neuron 89, 248–268 (2016).
Peng, Y. et al. Directed and acyclic synaptic connectivity in the human layer 2–3 cortical microcircuit. Science 384, 338–343 (2024).
Beaulieu-Laroche, L. et al. Enhanced dendritic compartmentalization in human cortical neurons. Cell 175, 643–651.e14 (2018).
Chiola, S., Edgar, N. U. & Shcheglovitov, A. iPSC toolbox for understanding and repairing disrupted brain circuits in autism. Mol. Psychiatry https://doi.org/10.1038/s41380-021-01288-7 (2021).
Yang, G. & Shcheglovitov, A. Probing disrupted neurodevelopment in autism using human stem cell-derived neurons and organoids: an outlook into future diagnostics and drug development. Dev. Dyn. https://doi.org/10.1002/dvdy.100 (2019).
Hansen, D. V., Lui, J. H., Parker, P. R. L. & Kriegstein, A. R. Neurogenic radial glia in the outer subventricular zone of human neocortex. Nature 464, 554–561 (2010).
Letinic, K., Zoncu, R. & Rakic, P. Origin of GABAergic neurons in the human neocortex. Nature 417, 645–649 (2002).
Pollen, A. A. et al. Molecular identity of human outer radial glia during cortical development. Cell 163, 55–67 (2015).
Nowakowski, T. J. et al. Spatiotemporal gene expression trajectories reveal developmental hierarchies of the human cortex. Science 358, 1318–1323 (2017).
Gandal, M. J. et al. Broad transcriptomic dysregulation occurs across the cerebral cortex in ASD. Nature 611, 532–539 (2022).
Velmeshev, D. et al. Single-cell genomics identifies cell type-specific molecular changes in autism. Science 364, 685–689 (2019).
Mathys, H. et al. Single-cell transcriptomic analysis of Alzheimer’s disease. Nature 570, 332–337 (2019).
Espuny-Camacho, I. et al. Pyramidal neurons derived from human pluripotent stem cells integrate efficiently into mouse brain circuits in vivo. Neuron 77, 440–456 (2013).
Chambers, S. M. et al. Highly efficient neural conversion of human ES and iPS cells by dual inhibition of SMAD signaling. Nat. Biotechnol. 27, 275–280 (2009).
Liu, Y., Wang, J., Südhof, T. C. & Wernig, M. Efficient generation of functional neurons from mouse embryonic stem cells via neurogenin-2 expression. Nat. Protoc. 18, 2954–2974 (2023).
Krencik, R. & Zhang, S. C. Directed differentiation of functional astroglial subtypes from human pluripotent stem cells. Nat. Protoc. 6, 1710–1717 (2011).
Abud, E. M. et al. iPSC-derived human microglia-like cells to study neurological diseases. Neuron 94, 278–293.e9 (2017).
Yang, N. et al. Generation of pure GABAergic neurons by transcription factor programming. Nat. Methods 14, 621–628 (2017).
Douvaras, P. & Fossati, V. Generation and isolation of oligodendrocyte progenitor cells from human pluripotent stem cells. Nat. Protoc. 10, 1143–1154 (2015).
Eiraku, M. et al. Self-organized formation of polarized cortical tissues from ESCs and its active manipulation by extrinsic signals. Cell Stem Cell 3, 519–532 (2008).
Kadoshimaa, T. et al. Self-organization of axial polarity, inside-out layer pattern, and species-specific progenitor dynamics in human ES cell-derived neocortex. Proc. Natl Acad. Sci. USA 110, 20285–20289 (2013).
Mariani, J. et al. Modeling human cortical development in vitro using induced pluripotent stem cells. Proc. Natl Acad. Sci. USA 109, 12770–12775 (2012).
Lancaster, M. A. & Knoblich, J. A. Generation of cerebral organoids from human pluripotent stem cells. Nat. Protoc. 9, 2329–2340 (2014).
Paşca, A. M. et al. Functional cortical neurons and astrocytes from human pluripotent stem cells in 3D culture. Nat. Methods 12, 671–678 (2015).
Qian, X. et al. Generation of human brain region-specific organoids using a miniaturized spinning bioreactor. Nat. Protoc. 13, 565–580 (2018).
Quadrato, G., Brown, J. & Arlotta, P. The promises and challenges of human brain organoids as models of neuropsychiatric disease. Nat. Med. 22, 1220–1228 (2016).
Marton, R. M. & Pașca, S. P. Organoid and assembloid technologies for investigating cellular crosstalk in human brain development and disease. Trends Cell Biol. https://doi.org/10.1016/j.tcb.2019.11.004 (2020).
Thomas, C. A. et al. Modeling of TREX1-dependent autoimmune disease using human stem cells highlights L1 accumulation as a source of neuroinflammation. Cell Stem Cell 21, 319–331.e8 (2017).
Trujillo, C. A. & Muotri, A. R. Brain organoids and the study of neurodevelopment. Trends Mol. Med. 24, 982–990 (2018).
Andrews, M. G. & Kriegstein, A. R. Challenges of organoid research. Annu. Rev. Neurosci. 45, 23–39 (2022).
Eichmüller, O. L. & Knoblich, J. A. Human cerebral organoids—a new tool for clinical neurology research. Nat. Rev. Neurol. 18, 661–680 (2022).
Zhou, Y., Song, H. & Ming, G. L. Genetics of human brain development. Nat. Rev. Genet. 25, 26–45 (2023).
Quadrato, G. et al. Cell diversity and network dynamics in photosensitive human brain organoids. Nature 545, 48–53 (2017).
Bhaduri, A. et al. Cell stress in cortical organoids impairs molecular subtype specification. Nature 578, 142–148 (2020).
Qian, X. et al. Sliced human cortical organoids for modeling distinct cortical layer formation. Cell Stem Cell 26, 766–781.e9 (2020).
Trujillo, C. A. et al. Complex oscillatory waves emerging from cortical organoids model early human brain network development. Cell Stem Cell 25, 558–569.e7 (2019).
Watanabe, M. et al. Self-organized cerebral organoids with human-specific features predict effective drugs to combat zika virus infection. Cell Rep. 21, 517–532 (2017).
Landry, C. R. et al. Electrophysiological and morphological characterization of single neurons in intact human brain organoids. J. Neurosci. Methods 394, 109898 (2023).
Wang, Y. et al. Modeling human telencephalic development and autism-associated SHANK3 deficiency using organoids generated from single neural rosettes. Nat. Commun. https://doi.org/10.1038/s41467-022-33364-z (2022).
Zhang, S.-C., Wernig, M., Duncan, I. D., Brüstle, O. & Thomson, J. A. In vitro differentiation of transplantable neural precursors from human embryonic stem cells. Nat. Biotechnol. 19, 1129–1133 (2001).
Elkabetz, Y. et al. Human ES cell-derived neural rosettes reveal a functionally distinct early neural stem cell stage. Genes Dev. 22, 152–165 (2008).
Perrier, A. L. et al. Derivation of midbrain dopamine neurons from human embryonic stem cells. Proc. Natl Acad. Sci. USA 101, 12543–12548 (2004).
Schoenwolf, G. C., Bleyl, S. B., Brauer, P. R. & Francis-West, P. H. Larsen’s Human Embryology (Elsevier, 2021).
Wippold, F. J. & Perry, A. Neuropathology for the neuroradiologist: rosettes and pseudorosettes. Am. J. Neuroradiol. 27, 488–492 (2006).
Ladewig, J., Koch, P. & Brüstle, O. Auto-attraction of neural precursors and their neuronal progeny impairs neuronal migration. Nat. Neurosci. 17, 24–26 (2014).
Li, X. J. et al. Specification of motoneurons from human embryonic stem cells. Nat. Biotechnol. 23, 215–221 (2005).
Shi, Y., Kirwan, P. & Livesey, F. J. Directed differentiation of human pluripotent stem cells to cerebral cortex neurons and neural networks. Nat. Protoc. https://doi.org/10.1038/nprot.2012.116 (2012).
Siegenthaler, J. A. et al. Retinoic acid from the meninges regulates cortical neuron generation. Cell 139, 597–609 (2009).
Kalani, M. Y. S. et al. Wnt-mediated self-renewal of neural stem/progenitor cells. Proc. Natl Acad. Sci. USA 105, 16970–16975 (2008).
Chenn, A. & Walsh, C. A. Regulation of cerebral cortical size by control of cell cycle exit in neural precursors. Science 297, 365–369 (2002).
Gunhaga, L. et al. Specification of dorsal telencephalic character by sequential Wnt and FGF signaling. Nat. Neurosci. 6, 701–707 (2003).
Campbell, K. Dorsal–ventral patterning in the mammalian telencephalon. Curr. Opin. Neurobiol. 13, 50–56 (2003).
Li, X. J. et al. Coordination of sonic hedgehog and Wnt signaling determines ventral and dorsal telencephalic neuron types from human embryonic stem cells. Development 136, 4055–4063 (2009).
Backman, M. et al. Effects of canonical Wnt signaling on dorso-ventral specification of the mouse telencephalon. Dev. Biol. 279, 155–168 (2005).
Tropepe, V. et al. Distinct neural stem cells proliferate in response to EGF and FGF in the developing mouse telencephalon. Dev. Biol. 208, 166–188 (1999).
Gregg, C. & Weiss, S. Generation of functional radial glial cells by embryonic and adult forebrain neural stem cells. J. Neurosci. 23, 11587–11601 (2003).
Ziv, O. et al. Quantitative live imaging of human embryonic stem cell derived neural rosettes reveals structure-function dynamics coupled to cortical development. PLoS Comput. Biol. 11, 1–21 (2015).
McAllister, A. K., Katz, L. C. & Lo, D. C. Neurotrophins and synaptic plasticity. Annu. Rev. Neurosci. 22, 295–318 (1999).
Ledda, F., Paratcha, G., Sandoval-Guzmán, T. & Ibá̃ez, C. F. GDNF and GFRα1 promote formation of neuronal synapses by ligand-induced cell adhesion. Nat. Neurosci. 10, 293–300 (2007).
Amiri, A. et al. Transcriptome and epigenome landscape of human cortical development modeled in organoids. Science 362, eaat6720 (2018).
Tidball, A. M. et al. Deriving early single-rosette brain organoids from human pluripotent stem cells. Stem Cell Rep. 18, 2498–2514 (2023).
Lancaster, M. A. et al. Cerebral organoids model human brain development and microcephaly. Nature 501, 373–379 (2013).
Qian, X. et al. Brain-region-specific organoids using mini-bioreactors for modeling ZIKV exposure. Cell 165, 1238–1254 (2016).
Kadoshima, T. et al. Self-organization of axial polarity, inside-out layer pattern, and species-specific progenitor dynamics in human ES cell-derived neocortex. Proc. Natl Acad. Sci. USA 110, 20284–20289 (2013).
Knight, G. T. et al. Engineering induction of singular neural rosette emergence within hPSC-derived tissues. eLife 7, 1–23 (2018).
Karzbrun, E. et al. Human neural tube morphogenesis in vitro by geometric constraints. Nature 599, 268–272 (2021).
Yang, G. et al. Neurite outgrowth deficits caused by rare PLXNB1 mutation in pediatric bipolar disorder. Mol. Psychiatry https://doi.org/10.1038/s41380-023-02035-w (2023).
La Manno, G. et al. RNA velocity in single cells. Nature https://doi.org/10.1101/206052 (2018).
Xiang, Y., Cakir, B. & Park, I. H. Deconstructing and reconstructing the human brain with regionally specified brain organoids. Semin. Cell Dev. Biol. 111, 40–51 (2021).
Miura, Y. et al. Engineering brain assembloids to interrogate human neural circuits. Nat. Protoc. 17, 15–35 (2022).
Atamian, A., Birtele, M., Hosseini, N. & Quadrato, G. Generation and long-term culture of human cerebellar organoids from pluripotent stem cells. Nat. Protoc. https://doi.org/10.1038/s41596-024-01093-w (2024).
Reumann, D. et al. In vitro modeling of the human dopaminergic system using spatially arranged ventral midbrain–striatum–cortex assembloids. Nat. Methods 20, 2034–2047 (2023).
Sloan, S. A., Andersen, J., Pașca, A. M., Birey, F. & Pașca, S. P. Generation and assembly of human brain region-specific three-dimensional cultures. Nat. Protoc. 13, 2062–2085 (2018).
Cai, H. et al. Brain organoid reservoir computing for artificial intelligence. Nat. Electron. 6, 1032–1039 (2023).
Mansour, A. A. et al. An in vivo model of functional and vascularized human brain organoids. Nat. Biotechnol. 36, 432–441 (2018).
Schafer, S. T. et al. An in vivo neuroimmune organoid model to study human microglia phenotypes. Cell 186, 2111–2126.e20 (2023).
Revah, O. et al. Maturation and circuit integration of transplanted human cortical organoids. Nature 610, 319–326 (2022).
Jgamadze, D. et al. Structural and functional integration of human forebrain organoids with the injured adult rat visual system. Cell Stem Cell 30, 137–152.e7 (2023).
Ludwig, T. E. et al. ISSCR standards for the use of human stem cells in basic research. Stem Cell Rep. 18, 1744–1752 (2023).
Patzke, C. & Südhof, T. C. The conditional KO approach: Cre/Lox technology in human neurons. Rare Dis. 4, e1131884 (2016).
Yi, F. et al. Autism-associated SHANK3 haploinsufficiency causes Ih channelopathy in human neurons. Science 2669, 1–22 (2016).
Huang, Y.-W. A., Zhou, B., Wernig, M. & Südhof, T. C. ApoE2, ApoE3, and ApoE4 differentially stimulate APP transcription and Aβ secretion. Cell https://doi.org/10.1016/j.cell.2016.12.044 (2017).
Pak, C. H. et al. Human neuropsychiatric disease modeling using conditional deletion reveals synaptic transmission defects caused by heterozygous mutations in NRXN1. Cell Stem Cell 17, 316–328 (2015).
Bertucci, T. et al. Improved protocol for reproducible human cortical organoids reveals early alterations in metabolism with MAPT mutations. Preprint at bioRxiv (2023).
Bischofberger, J., Engel, D., Li, L., Geiger, J. R. P. & Jonas, P. Patch-clamp recording from mossy fiber terminals in hippocampal slices. Nat. Protoc. 1, 2075–2081 (2006).
Davie, J. T. et al. Dendritic patch-clamp recording. Nat. Protoc. 1, 1235–1247 (2006).
Ting, J. T. et al. Preparation of acute brain slices using an optimized N-Methyl-D-glucamine protective recovery method. J. Vis. Exp. 2018, 1–13 (2018).
Edwards, F. A. & Konnerth, A. Patch-clamping cells in sliced tissue preparations. Methods Enzymol. 207, 208–222 (1992).
Velasco, S. et al. Individual brain organoids reproducibly form cell diversity of the human cerebral cortex. Nature 570, 523–527 (2019).
Ataman, B. et al. Evolution of Osteocrin as an activity-regulated factor in the primate brain. Nature 539, 242–247 (2016).
Rolland, T. et al. Phenotypic effects of genetic variants associated with autism. Nat. Med. 29, 1671–1680 (2023).
Singh, T. et al. Rare coding variants in ten genes confer substantial risk for schizophrenia. Nature 604, 509–516 (2022).
Kubanek, J. et al. Stem cell-derived brain organoids for controlled studies of transcranial neuromodulation. Heliyon 9, e18482 (2023).
Ran, F. A. et al. Genome engineering using the CRISPR–Cas9 system. Nat. Protoc. https://doi.org/10.1038/nprot.2013.143 (2013).
Acknowledgements
The authors are thankful to current and former members of the Shcheglovitov Lab for their contribution to the protocol development and to J. Cui for help with gRNA design and validation. Cartoons used in Figs. 1, 4a and 6d were created with BioRender.com. The study was supported by the National Institute of Mental Health (R01MH113670), NINDS (R01NS123849 and R21NS104963), Utah Neuroscience Initiative, and Utah Genome Project grants (to A.S.) and the NHGRI T32 Genomic Medicine Training Grant (T32HG008962) to H.M.A.U.
Author information
Authors and Affiliations
Contributions
H.M.A.U., Q.H., S.C. and Y.W. performed data acquisition and analyses. A.S. conceived the protocol idea and wrote the manuscript. All authors commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Protocols thanks Alysson Muotri, In-Hyun Park, Clive Svendsen, Zhexing Wen, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key references
Wang, Y. et al. Nat Commun. 13, 5688 (2022): https://doi.org/10.1038/s41467-022-33364-z
Yang, G. et al. Mol. Psychiatry 28, 2525–2539 (2023): https://doi.org/10.1038/s41380-023-02035-w
Kubanek, J. et al. Heliyon 9 (8), e18482 (2023): https://doi.org/10.1016/j.heliyon.2023.e18482
Supplementary information
Supplementary Information (download PDF )
Supplementary Fig. 1.
Supplementary Video 1 (download MOV )
Preparation of glass hook from Pasteur pipette.
Supplementary Video 2 (download MOV )
Isolation of SNR using glass hook.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ullah, H.M.A., Huang, Q., Chiola, S. et al. Generating and characterizing human telencephalic brain organoids from stem cell-derived single neural rosettes. Nat Protoc 21, 718–748 (2026). https://doi.org/10.1038/s41596-025-01197-x
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41596-025-01197-x