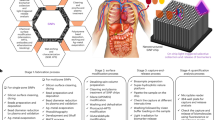
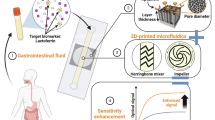
Abstract
There is an urgent requirement to improve accessibility to diagnostic tools in remote areas. This requires assays that are easy to use, are cost-effective and produce rapid results. Important public health applications include early disease diagnosis, real-time monitoring, epidemic control and medical cost control. This protocol describes the fabrication of all-printed photonic crystal (PC) biochips for point-of-care testing of biomarkers. The photonic crystal material is prepared by the self-assembly of latex nanospheres that are printed onto a polyethylene terephthalate substrate. Photonic crystals composed of latex nanospheres of different sizes enhance the fluorescent signal emitted at different wavelengths, resulting in remarkably higher detection sensitivity. PC microarrays enable mass-printed preparation (up to 2,700 pieces can be printed per hour by one printer), and each microarray can be stored for a long time (>6 months) after heating. Biomarker specificity is achieved by the biofunctionalization of the nanospheres, for example, attaching capture antibodies. The detection involves the use of a fluorescently labeled detection antibody and a simple point-of-care detection device. This universal approach can be applied to the detection of many biomarkers, and the simultaneous detection of multiple biomarkers is also possible. Here we demonstrate describing how to prepare a chip that can be used to detect three inflammatory biomarkers in 10 min at low sample volumes at a cost of less than 3 CNY (~US$0.41) per PC codetection biochip. The biofunctionalization process including capture antibody coupling and blocking takes 3–4 h, and the detection process takes 20 min.
Key points
-
In this protocol, photonic crystal biochips composed of self-assembled core–shell latex nanospheres printed on a polyethylene terephthalate substrate are functionalized with capture antibodies for the detection of specific biomarkers.
-
The printed biochips are inexpensive, and the biomarkers can be detected in small volumes of biofluids using a simple point-of-care testing device. This approach can be used to produce diagnostic tools for public health applications in remote areas.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The authors declare that the main data supporting the findings of this study are available within the article and previous publications44,45,46,47,48,49. Experimental data are available via Figshare at https://doi.org/10.6084/m9.figshare.28692044 (ref. 60) and https://doi.org/10.6084/m9.figshare.29117594.v1 (ref. 50).
References
Bodaghi, A., Fattahi, N. & Ramazani, A. Biomarkers: promising and valuable tools towards diagnosis, prognosis and treatment of Covid-19 and other diseases. Heliyon 9, e13323 (2023).
Atkinson, A. J. et al. Biomarkers and surrogate endpoints: preferred definitions and conceptual framework. Clin. Pharmacol. Ther. 69, 89–95 (2001).
Hartmann, M., Roeraade, J., Stoll, D., Templin, M. & Joos, T. Protein microarrays for diagnostic assays. Anal. Bioanal. Chem. 393, 1407–1416 (2009).
Yang, J. et al. An integrative review on the applications of 3D printing in the field of in vitro diagnostics. Chin. Chem. Lett. 33, 2231–2242 (2022).
Vashist, S. K., Luppa, P. B., Yeo, L. Y., Ozcan, A. & Luong, J. H. T. Emerging technologies for next-generation point-of-care testing. Trends Biotechnol. 33, 692–705 (2015).
Luppa, P. B., Müller, C., Schlichtiger, A. & Schlebusch, H. Point-of-care testing (POCT): current techniques and future perspectives. Trends Anal. Chem. 30, 887–898 (2011).
Guo, S., Schlecht, W., Li, L. & Dong, W. J. Paper-based cascade cationic isotachophoresis: multiplex detection of cardiac markers. Talanta 205, 120112 (2019).
Garms, D. S. D. et al. The role of urinary biomarkers as diagnostic and prognostic predictors of acute kidney injury associated with vancomycin. Front. Pharmacol. 12, 705636 (2021).
Sharfstein, J. M., Becker, S. J. & Mello, M. M. Diagnostic testing for the novel coronavirus. JAMA 323, 1437–438 (2020).
Yuan, X. et al. Current and perspective diagnostic techniques for COVID-19. ACS Infect. Dis. 6, 1998–2016 (2020).
Egilmezer, E. et al. Systematic review of the impact of point-of-care testing for influenza on the outcomes of patients with acute respiratory tract infection. Rev. Med. Virol. 28, e1995 (2018).
Allin, K. H. & Nordestgaard, B. G. Elevated C-reactive protein in the diagnosis, prognosis, and cause of cancer. Crit. Rev. Clin. Lab. Sci. 48, 155–170 (2011).
Meisner, M. Update on procalcitonin measurements. Ann. Lab. Med. 34, 263–273 (2014).
Zhang, Y., Zhang, J., Sheng, H., Li, H. & Wang, R. Acute phase reactant serum amyloid A in inflammation and other diseases. Adv. Clin. Chem. 90, 25–80 (2019).
Chen, Y. T. et al. Review of integrated optical biosensors for point-of-care applications. Biosensors 10, 209 (2020).
Kim, S., Chun, H. J., Lee, K. W. & Yoon, H. C. Smartphone-integrated urinary CTX-II immunosensor based on wavelength filtering from chromogenic reaction. Biosens. Bioelectron. 150, 111932 (2020).
Xu, L. Z. et al. Optical, electrochemical and electrical (nano)biosensors for detection of exosomes: a comprehensive overview. Biosens. Bioelectron. 161, 112222 (2020).
Parihar, A. et al. Next-generation intelligent MXene-based electrochemical aptasensors for point-of-care cancer diagnostics. Nano-Micro Lett. 14, 100 (2022).
Hang, Y. J., Wang, A. Y. & Wu, N. Q. Plasmonic silver and gold nanoparticles: shape- and structure-modulated plasmonic functionality for point-of-caring sensing, bio-imaging and medical therapy. Chem. Soc. Rev. 53, 2932–2971 (2024).
Jiang, S. Y. et al. A point-of-care testing device utilizing graphene-enhanced fiber optic SPR sensor for real-time detection of infectious. Pathog. Biosens. 13, 1029 (2023).
Wang, H. D., Jian, M. H., Fan, J. W., He, Y. Q. & Wang, Z. X. Scalable synthesis of Au@CeO2 nanozyme for development of colorimetric lateral flow immunochromatographic assay to sensitively detect heart-type fatty acid binding protein. Talanta 273, 125852 (2024).
Hang, Y. J., Boryczka, J. & Wu, N. Q. Visible-light and near-infrared fluorescence and surface-enhanced Raman scattering point-of-care sensing and bio-imaging: a review. Chem. Soc. Rev. 51, 329–375 (2022).
Semeniak, D., Cruz, D. F., Chilkoti, A. & Mikkelsen, M. H. Plasmonic fluorescence enhancement in diagnostics for clinical tests at point-of-care: a review of recent technologies. Adv. Mater. 35, 2107986 (2023).
Rasheed, S., Ikram, M., Ahmad, D., Abbas, M. N. & Shafique, M. Advancements in colorimetric and fluorescent-based sensing approaches for point-of-care testing in forensic sample analysis. Microchem J. 206, 111438 (2024).
Sauer, U. Analytical protein microarrays: advancements towards clinical applications. Sensors 17, 256 (2017).
Liu, Y., Guo, C. X., Hu, W., Lu, Z. & Li, C. M. Sensitive protein microarray synergistically amplified by polymer brush-enhanced immobilizations of both probe and reporter. J. Colloid Interface Sci. 360, 593–599 (2011).
Menendez-Miranda, M., Costa-Fernández, J. M., Encinar, J. R., Parak, W. J. & Carrillo-Carrion, C. Determination of the ratio of fluorophore/nanoparticle for fluorescence-labelled nanoparticles. Analyst 141, 1266–1272 (2016).
Zhou, S., Yuan, L., Hua, X., Xu, L. & Liu, S. Signal amplification strategies for DNA and protein detection based on polymeric nanocomposites and polymerization: a review. Analytica Chim. Acta 877, 19–32 (2015).
Jun, J. V., Chenoweth, D. M. & Petersson, E. J. Rational design of small molecule fluorescent probes for biological applications. Org. Biomol. Chem. 18, 5747–5763 (2020).
Michie, M. S. et al. Cyanine conformational restraint in the far-red range. J. Am. Chem. Soc. 139, 12406–12409 (2017).
Bassolino, G. et al. Photolabile coumarins with improved efficiency through azetidinyl substitution. Chem. Sci. 9, 387–391 (2018).
Hirabayashi, K. et al. Analysis of chemical equilibrium of silicon-substituted fluorescein and its application to develop a scaffold for red fluorescent probes. Anal. Chem. 87, 9061–9069 (2015).
Grimm, J. B. et al. A general method to optimize and functionalize red-shifted rhodamine dyes. Nat. Methods 17, 815–821 (2020).
Sungwienwong, I. et al. Improving the fluorescent probe acridonylalanine through a combination of theory and experiment. J. Phys. Org. Chem. 31, e3813 (2018).
Ma, F., Li, C.-c & Zhang, C.-y Development of quantum dot-based biosensors: principles and applications. J. Mat. Chem. B 6, 6173–6190 (2018).
Ding, T., Mertens, J., Lombardi, A., Scherman, O. A. & Baumberg, J. J. Light-directed tuning of plasmon resonances via plasmon-induced polymerization using hot electrons. ACS Photonics 4, 1453–1458 (2017).
Zhao, D. et al. Highly sensitive microfluidic detection of carcinoembryonic antigen via a synergetic fluorescence enhancement strategy based on the micro/nanostructure optimization of ZnO nanorod arrays and in situ ZIF-8 coating. Chem. Eng. J. 383, 123230 (2020).
Joannopoulos, J. D., Johnson, S. G., Winnand, J. N. & Meade, R. D. Photonic Crystals: Molding the Flow of Light (Princeton Univ. Press, 2008).
Inan, H. et al. Photonic crystals: emerging biosensors and their promise for point-of-care applications. Chem. Soc. Rev. 46, 366–388 (2017).
Hou, J. et al. Four-dimensional screening anti-counterfeiting pattern by inkjet printed photonic crystals. Chem. Asian J. 11, 2680–2685 (2016).
Yang, M., Liu, Y. & Jiang, X. Barcoded point-of-care bioassays. Chem. Soc. Rev. 48, 850–884 (2019).
Zhang, Y., Zhang, G., Zeng, Z. & Pu, K. Activatable molecular probes for fluorescence-guided surgery, endoscopy and tissue biopsy. Chem. Soc. Rev. 51, 566–593 (2022).
Zheng, S., Zhang, H., Ross, E., Le, T. V. & Wirth, M. J. Silica colloidal crystals for enhanced fluorescence detection in microarrays. Anal. Chem. 79, 3867–3872 (2007).
Chi, J. et al. Fast and sensitive detection of protein markers using an all-printing photonic crystal microarray via fingertip blood. ACS Sens 8, 1742–1749 (2023).
Chi, J. et al. All-printed point-of-care immunosensing biochip for one drop blood diagnostics. Lab Chip 22, 3008–3014 (2022).
Lian, Z. et al. At-home COVID-19 rapid antigen test down to 0.03 pg ml−1 of nucleocapsid protein. Small 19, 2301162 (2023).
Lin, X. et al. One-droplet saliva detection on photonic crystal-based competitive immunoassay for precise diagnosis of migraine. SmartMat 5, e1252 (2024).
Liu, Y. et al. Rapid and sensitive detection of urinary KIM-1 using fully printed photonic crystal microarrays. Nano Res 17, 4329–4337 (2024).
Liu, Z. et al. Rapid differential diagnosis of periprosthetic joint infection by microdroplet reaction on portable biochip and indicator. Chem. Eng. J. 494, 153034 (2024).
Lian, Z. et al. Combined point-of-care-testing (POCT) of three inflammatory biomarkers. Figshare https://doi.org/10.6084/m9.figshare.29117594.v1 (2025).
Wang, J. et al. Simple fabrication of full color colloidal crystal films with tough mechanical strength. Macromol. Chem. Phys. 207, 596–604 (2006).
John, S. & Quang, T. Spontaneous emission near the edge of a photonic band gap. Phys. Rev. A 50, 1764–1769 (1994).
Li, Z.-Y., Lin, L.-L. & Zhang, Z.-Q. Spontaneous emission from photonic crystals: full vectorial calculations. Phys. Rev. Lett. 84, 4341–4344 (2000).
Bechger, L., Lodahl, P. & Vos, W. L. Directional fluorescence spectra of laser dye in opal and inverse opal photonic crystals. J. Phys. Chem. B 109, 9980–9988 (2005).
Richel, A., Johnson, N. P. & McComb, D. W. Observation of Bragg reflection in photonic crystals synthesized from air spheres in a titania matrix. Appl. Phys. Lett. 76, 1816–1818 (2000).
Busch, K. & John, S. Photonic band gap formation in certain self-organizing systems. Phys. Rev. E 58, 3896–3908 (1998).
Huang, Y. et al. Colloidal photonic crystals with narrow stopbands assembled from low-adhesive superhydrophobic substrates. J. Am. Chem. Soc. 134, 17053–17058 (2012).
Park, J. & Moon, J. Control of colloidal particle deposit patterns within picoliter droplets ejected by ink-jet printing. Langmuir 22, 3506–3513 (2006).
Cui, L. Y. et al. Fabrication of large-area patterned photonic crystals by ink-jet printing. J. Mater. Chem. 19, 5499–5502 (2009).
Lian, Z. et al. Data of Printed photonic crystal biochips for rapid and sensitive detection of biomarkers in various body fluids. Figshare https://doi.org/10.6084/m9.figshare.28692044 (2025).
Vanderschueren, S. et al. Extremely elevated C-reactive protein. Eur. J. Intern. Med. 17, 430–433 (2006).
Stankovic Stojanovic, K. et al. Concordance between CRP and SAA in familial Mediterranean fever during attack-free period: a study of 218 patients. Clin. Biochem. 50, 206–209 (2017).
Ledue, T. B. et al. Analytical evaluation of particle-enhanced immunonephelometric assays for C-reactive protein, serum amyloid A and mannose-binding protein in human serum. Ann. Clin. Biochem. 35, 745–753 (1998).
Yamada, T. Serum amyloid A (SAA): a concise review of biology assay methods and clinical usefulness. Clin. Chem. Lab. Med. 37, 381–388 (1999).
Mieke De, B. et al. Structure and expression of different serum amyloid A (SAA) variants and their concentration-dependent functions during host insults. Curr. Med. Chem. 23, 1725–1755 (2016).
Zaki, H. A. et al. Accuracy of procalcitonin for diagnosing sepsis in adult patients admitted to the emergency department: a systematic review and meta-analysis. Syst. Rev. 13, 37 (2024).
Wacker, C., Prkno, A., Brunkhorst, F. M. & Schlattmann, P. Procalcitonin as a diagnostic marker for sepsis: a systematic review and meta-analysis. Lancet Infect. Dis. 13, 426–435 (2013).
Lippi, G. & Sanchis-Gomar, F. Procalcitonin in inflammatory bowel disease: drawbacks and opportunities. World J. Gastroenterol. 23, 8283–8290 (2017).
Ganesh, N. et al. Enhanced fluorescence emission from quantum dots on a photonic crystal surface. Nat. Nanotechnol. 2, 515–520 (2007).
Mathias, P. C., Ganesh, N. & Cunningham, B. T. Application of photonic crystal enhanced fluorescence to a cytokine immunoassay. Anal. Chem. 80, 9013–9020 (2008).
Shen, W. et al. Highly effective protein detection for avidin–biotin system based on colloidal photonic crystals enhanced fluoroimmunoassay. Biosens. Bioelectron. 26, 2165–2170 (2011).
Acknowledgements
This work was supported by the National Key R&D Program of China (grant no. 2023YFE0111500), the National Natural Science Foundation of China (grant nos T2394480, T2394484, 52321006, 52222313 and 22075296) and the Beijing National Laboratory for Molecular Sciences (grant no. BNLMS-CXXM-202005).
Author information
Authors and Affiliations
Contributions
Y.S. and M.S. proposed the project. Z.L. and W.W. contributed to the printing procedure. Z.L., J.C., X.Y., L.C., D.X., Z.T., S.C. and Y. Yibulayimu contributed to the biofunctionalization procedure, detection procedure and measurement procedure. Z.L. and Y. Yun analyzed the data. Z.L., Y.S. and M.S. cowrote the manuscript. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics statement
This study was approved by the Ethics Committee of Chinese PLA General Hospital (approval nos: S2022-803-01, S2023-283-01 and 2023KY103-KS001). All participants have signed an informed consent form.
Peer review
Peer review information
Nature Protocols thanks Judith Su, Francesco Michelotti and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key references
Chi, J. et al. Lab Chip 22, 3008–3014 (2022): https://doi.org/10.1039/D2LC00385F
Lian, Z. et al. Small 19, 2301162 (2023): https://doi.org/10.1002/smll.202301162
Lin, X. et al. SmartMat 5, e1252 (2024): https://doi.org/10.1002/smm2.1252
Liu, Y. et al. Nano Res. 17, 4329–4337 (2024): https://doi.org/10.1007/s12274-023-6335-1
Liu, Z. et al. Chem. Eng. J. 494, 153034 (2024): https://doi.org/10.1016/j.cej.2024.153034
Extended data
Extended Data Fig. 1 PC microarrays preparation.
The Jetting valves dispensing systems for preparation of PC microarrays.
Extended Data Fig. 2 Preparation of cleaning the nozzle in step 4 of the Printing Procedure.
Photograph of the installed syringe above the nozzle.
Extended Data Fig. 3 The step to power on the PICO Toμch controller in step 5 of the Printing Procedure.
Click the buttons in the red box in order.
Extended Data Fig. 4 The step to clean the nozzle in step 6 of the Printing Procedure.
a. Photograph of the barometer. b. Press and hold the clean button in the red box to clean the nozzle.
Extended Data Fig. 5 The step to add PC ink in step 7 of the Printing Procedure.
a. Photograph of a new syringe and a piston. b. Photograph of the syringe filled with PC ink.
Extended Data Fig. 6 The step to set-up the PICO Toμch controller in step 9 of the Printing Procedure.
Click the buttons in the red box in order. The proper range of pulse frequency is 0.45 ms-0.60 ms, and the proper range of stroke is 47-55 μm.
Extended Data Fig. 7 The step to set-up the portable fluorescence detector in step 18A of the Measurement Procedure.
a. Full view of the measurement software interface. b. The step to connect the portable fluorescence detector. Click the buttons in the red box to select the correct serial port. The pattern in the white box is displayed after successful connection. c. The step to set the cut-off value. Insert a blank test card, enter hCxE and click the button in the red box to set the cut-off value, where x is the cut-off value. The cut-off value in the white box is displayed after successful set-up. The cut-off value can be set from 0 to 65536.
Extended Data Fig. 8 The step to set-up the commercial fluorescence detector in step 18B of the Measurement Procedure.
click the buttons in the red box in order.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–10 and Table 1.
Supplementary Video 1 (download MP4 )
Print procedure.
Supplementary Video 2 (download MP4 )
Qualitative detection by portable fluorescence detector.
Supplementary Video 3 (download MP4 )
Qualitative detection by commercial fluorescence detector.
Supplementary Video 4 (download MP4 )
Detection procedure and measurement procedure.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lian, Z., Chi, J., Yang, X. et al. Printed photonic crystal biochips for rapid and sensitive detection of biomarkers in various body fluids. Nat Protoc 20, 3783–3807 (2025). https://doi.org/10.1038/s41596-025-01215-y
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41596-025-01215-y