Abstract
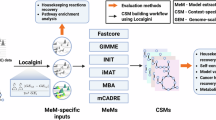
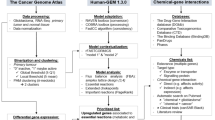
Constraint-based modeling can mechanistically simulate the behavior of a biochemical system, permitting hypothesis generation, experimental design and interpretation of experimental data, with numerous applications, especially the modeling of metabolism. Given a generic model, several methods have been developed to extract a context-specific, genome-scale metabolic model by incorporating information used to identify metabolic processes and gene activities in each context. However, the existing model extraction algorithms are unable to ensure that a context-specific model is thermodynamically flux consistent. Here we introduce XomicsToModel, a semiautomated pipeline that integrates bibliomic, transcriptomic, proteomic and metabolomic data with a generic genome-scale metabolic reconstruction, or model, to extract a context-specific, genome-scale metabolic model that is stoichiometrically, thermodynamically and flux consistent. One of the key advantages of the XomicsToModel pipeline is its ability to seamlessly incorporate omics data into metabolic reconstructions, ensuring not only mechanistic accuracy but also physicochemical consistency. This functionality enables more accurate metabolic simulations and predictions across different biological contexts, enhancing its utility in diverse research fields, including systems biology, drug development and personalized medicine. The XomicsToModel pipeline is exemplified for extraction of a specific metabolic model from a generic metabolic model; it enables omics data integration and extraction of physicochemically consistent mechanistic models from any generic biochemical network. It can be implemented by anyone who has basic MATLAB programming skills and the fundamentals of constraint-based modeling.
Key points
-
XomicsToModel is a semi-automated pipeline that integrates bibliomic, transcriptomic, proteomic and metabolomic data with a generic genome-scale metabolic reconstruction or model.
-
It enables the seamless incorporation of multiomics datasets into metabolic reconstructions, ensuring mechanistic accuracy and physicochemical consistency.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Constraint-based reconstruction, modeling and analysis was implemented in MATLAB (MathWorks). Open-source computer code enabling the reproduction of all computational steps is available within the COBRA Toolbox34 version 3.4+. This includes code for the model generation XomicsToModel.m, thermodynamically feasible model extraction thermoKernel.m, debugging the performance of XomicsToModel debugXomicsToModel.m, the comparison of multiple models compareXomicsToModel.m and for the maximization of flux entropy, with the option of quadratic penalization of deviation from measured experimental fluxes entropicFBA.m; it is available via GitHub at https://github.com/opencobra/cobratoolbox/tree/master/src/dataIntegration/XomicsToModel (ref. 51). An executable narrative tutorial demonstrating the use of the XomicsToModel pipleine is also available, tutorial_XomicsToModel.mlx, with the example data for generating a dopaminergic neuronal metabolic model; it is available via GitHub at https://github.com/opencobra/COBRA.tutorials/blob/master/dataIntegration/XomicsToModel/tutorial_XomicsToModel.mlx (ref. 52). An HTML version of this tutorial is accessible via GitHub at https://github.com/opencobra/COBRA.tutorials/blob/master/dataIntegration/XomicsToModel/tutorial_XomicsToModel.html. Linear and quadratic optimization problems may be solved with open source solvers, for example, GLPK, or using an industrial solver, such as Gurobi 9.1 (Gurobi). Nonlinear convex optimization problems, for example, entropic optimization, may be solved with the open source solver primal-dual interior method for convex objectives PDCO implemented in MATLAB (MathWorks) or with an industrial solver, using the exponential cone solver within Mosek 10.0.30 (Mosek ApS). Note that PDCO may require specialist numerical optimization parameters to be tailored to each model, whereas Mosek is more robust with respect to the numerical characteristics of input models. Each of the aforementioned solvers are interfaced with the COBRA Toolbox and are suited for models that are not numerically ill-scaled.
References
Palsson, B. Ø. Systems Biology: Constraint-Based Reconstruction and Analysis. (Cambridge Univ. Press, 2015).
Thiele, I. & Palsson, B. Ø A protocol for generating a high-quality genome-scale metabolic reconstruction. Nat. Protoc. 5, 93–121 (2010).
Machado, D. et al. Fast automated reconstruction of genome-scale metabolic models for microbial species and communities. Nucleic Acids Res. 46, 7542–7553 (2018).
Norsigian, C. J. et al. A workflow for generating multi-strain genome-scale metabolic models of prokaryotes. Nat. Protoc. 15, 1–14 (2020).
Wang, H. et al. Genome-scale metabolic network reconstruction of model animals as a platform for translational research. Proc. Natl Acad. Sci. USA https://doi.org/10.1073/pnas.2102344118(2021).
Opdam, S. et al. A systematic evaluation of methods for tailoring genome-scale metabolic models. Cell Syst. 4, 318–329.e6 (2017).
Jerby, L., Shlomi, T. & Ruppin, E. Computational reconstruction of tissue-specific metabolic models: application to human liver metabolism. Mol. Syst. Biol. https://doi.org/10.1038/msb.2010.56 (2010).
Vinnakota, K. C. et al. Network modeling of liver metabolism to predict plasma metabolite changes during short-term fasting in the laboratory rat. Front. Physiol. 10, 161 (2019).
Preciat, G. et al. Mechanistic model-driven exometabolomic characterisation of human dopaminergic neuronal metabolism. Preprint at bioRxiv https://doi.org/10.1101/2021.06.30.450562 (2021).
Haraldsdóttir, H. S. et al. CHRR: coordinate hit-and-run with rounding for uniform sampling of constraint-based models. Bioinformatics 33, 1741–1743 (2017).
Fleming, R. M. T. & Thiele, I. Mass conserved elementary kinetics is sufficient for the existence of a non-equilibrium steady state concentration. J. Theor. Biol. 314, 173–181 (2012).
Vlassis, N., Pacheco, M. P. & Sauter, T. Fast reconstruction of compact context-specific metabolic network models. PLoS Comput. Biol. 10, e1003424 (2014).
Fleming, R. M. T. et al. A variational principle for computing nonequilibrium fluxes and potentials in genome-scale biochemical networks. J. Theor. Biol. 292, 71–77 (2011).
Orth, J. D., Thiele, I. & Palsson, B. Ø What is flux balance analysis? Nat. Biotechnol. 28, 245–248 (2010).
Thiele, I., Vlassis, N. & Fleming, R. M. T. fastGapFill: efficient gap filling in metabolic networks. Bioinformatics 30, 2529–2531 (2014).
Beard, D. A., Liang, S.-D. & Qian, H. Energy balance for analysis of complex metabolic networks. Biophys. J. 83, 79–86 (2002).
Henry, C. S., Broadbelt, L. J. & Hatzimanikatis, V. Thermodynamics-based metabolic flux analysis. Biophys. J. 92, 1792–1805 (2007).
Dill, K. & Bromberg, S. Molecular Driving Forces Statistical Thermodynamics in Biology, Chemistry, Physics, and Nanoscience (Garland Science, 2011).
Boyd, S. P. & Vandenberghe, L. Convex Optimization (Cambridge Univ. Press, 2004).
Desouki, A. A. et al. CycleFreeFlux: efficient removal of thermodynamically infeasible loops from flux distributions. Bioinformatics 31, 2159–2165 (2015).
Becker, S. A. & Palsson, B. O. Context-specific metabolic networks are consistent with experiments. PLoS Comput. Biol. 4, e1000082 (2008).
Zur, H., Ruppin, E. & Shlomi, T. iMAT: an integrative metabolic analysis tool. Bioinformatics 26, 3140–3142 (2010).
Wang, Y., Eddy, J. A. & Price, N. D. Reconstruction of Genome-Scale Metabolic Models for 126 Human Tissues Using mCADRE. BMC Syst. Biol. 6, 153 (2012).
Brunk, E. et al. Recon3D enables a three-dimensional view of gene variation in human metabolism. Nat. Biotechnol. 36, 272 (2018).
Luo, X. et al. Constraint-based modeling of bioenergetic differences between synaptic and non-synaptic components of human dopaminergic neurons. Front. Comput. Neurosci. 19, 1594330 (2025).
Luo, X. et al. Identification of metabolites reproducibly associated with Parkinson’s disease via meta-analysis and computational modelling. NPJ Parkinsons Dis. 10, 126 (2024).
Zagare, A. et al. Omics data integration suggests a potential idiopathic Parkinson’s disease signature. Commun. Biol. 6, 1179 (2023).
Fleming, R. M. T. et al. Conserved moiety fluxomics. Preprint at bioRxiv https://doi.org/10.1101/2024.11.21.624666 (2024).
Huang, L. et al. fluxTrAM: integration of tracer-based metabolomics data into atomically resolved genome-scale metabolic networks for metabolic flux analysis. Preprint at bioRxiv https://doi.org/10.1101/2024.11.26.625485 (2024).
Kümmel, A., Panke, S. & Heinemann, M. Putative regulatory sites unraveled by network-embedded thermodynamic analysis of metabolome data. Mol. Syst. Biol. 2, 2006.0034 (2006).
Thiele, I. et al. Functional characterization of alternate optimal solutions of Escherichia coli’s transcriptional and translational machinery. Biophys. J. 98, 2072–2081 (2010).
Medlock, G. L., Moutinho, T. J. & Papin, J. A. Medusa: software to build and analyze ensembles of genome-scale metabolic network reconstructions. PLoS Comput. Biol. 16, e1007847 (2020).
Balestrino, R. & Schapira, A. H. V. Parkinson disease. Eur. J. Neurol. 27, 27–42 (2020).
Heirendt, L. et al. Creation and analysis of biochemical constraint-based models using the COBRA toolbox v.3.0. Nat. Protoc. 14, 639 (2019).
Thiele, I. et al. Personalized whole-body models integrate metabolism, physiology, and the gut microbiome. Mol. Syst. Biol. 16, e8982 (2020).
Pacheco, M. P. et al. scFASTCORMICS: a contextualization algorithm to reconstruct metabolic multi-cell population models from single-cell RNAseq data. Metabolites 12, 1211 (2022).
Vieira, V., Ferreira, J. & Rocha, M. A pipeline for the reconstruction and evaluation of context-specific human metabolic models at a large-scale. PLoS Comput. Biol. 18, e1009294 (2022).
Estévez, S. R. & Nikoloski, Z. Generalized framework for context-specific metabolic model extraction methods. Front. Plant Sci. 5, 491 (2014).
Gopalakrishnan, S. et al. Guidelines for extracting biologically relevant context-specific metabolic models using gene expression data. Metab. Eng. 75, 181–191 (2023).
Tefagh, M. & Boyd, S. P. SWIFTCORE: a tool for the context-specific reconstruction of genome-scale metabolic networks. BMC Bioinform. 21, 140 (2020).
Akbari, A. et al. The quantitative metabolome is shaped by abiotic constraints. Nat. Commun. 12, 3178 (2021).
Mayr, E. What Makes Biology Unique?: Considerations on the Autonomy of a Scientific Discipline (Cambridge Univ. Press, 2007).
Haraldsdóttir, H. S., Thiele, I. & Fleming, R. M. T. Quantitative assignment of reaction directionality in a multicompartmental human metabolic reconstruction. Biophys. J. 102, 1703–1711 (2012).
Sahin, A., Weilandt, D. R. & Hatzimanikatis, A. Optimal enzyme utilization suggests that concentrations and thermodynamics determine binding mechanisms and enzyme saturations. Nat. Commun. 14, 2618 (2023).
Nath, S. S. Evaluation of thermodynamic consistency of kinetic parameters in cyclic enzyme-catalyzed reaction networks. Chem. Phys. Lett. 804, 139890 (2022).
Kenney, I. M. & Beckstein, O. Thermodynamically consistent determination of free energies and rates in kinetic cycle models. Biophys. Rep. 3, 100120 (2023).
Gevorgyan, A., Poolman, M. G. & Fell, D. A. Detection of stoichiometric inconsistencies in biomolecular models. Bioinformatics 24, 2245–2251 (2008).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Shah, V. P. et al. Bioanalytical method validation—a revisit with a decade of progress. Pharm. Res. 17, 1551–1557 (2000).
Agren, R. et al. Reconstruction of genome-scale active metabolic networks for 69 human cell types and 16 cancer types using INIT. PLoS Comput. Biol. 8, e1002518 (2012).
cobratoolbox. GitHub https://github.com/opencobra/cobratoolbox/tree/master/src/dataIntegration/XomicsToModel (2025).
COBRA.tutorials. GitHub https://opencobra.github.io/cobratoolbox/stable/tutorials/tutorial_XomicsToModel.html (2025).
Agren, R. et al. Identification of anticancer drugs for hepatocellular carcinoma through personalized genome-scale metabolic modeling. Mol. Syst. Biol. 10, 721 (2014).
Gudmundsson, S. & Thiele, I. Computationally efficient flux variability analysis. BMC Bioinform. 11, 489 (2010).
Noronha, A. et al. The virtual metabolic human database: integrating human and gut microbiome metabolism with nutrition and disease. Nucleic Acids Res. 47, D614–D624 (2019).
Acknowledgements
We thank H. Leegwater for her helpful feedback while revising the manuscript. We acknowledge funding from the European Union's Horizon 2020 Programme (668738), Horizon Europe Framework Programme (101080997), European Research Council (101125633) and Dutch Research Council NWO project 184.034.019.
Author information
Authors and Affiliations
Contributions
G.P.: conceptualization, methodology, software, tutorial and writing—original draft; A.W.: methodology, software, data curation and writing—review and editing; X.L.: methodology and software; I.T.: conceptualization, methodology, software and funding acquisition; T.H.: resources, supervision and funding acquisition; and R.M.T.F.: conceptualization, methodology, software, writing—original draft, writing—review and editing, supervision and funding acquisition.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Protocols thanks Miguel Rocha and the other, anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key references
Luo, X., El Assal, D. C., Liu, Y., Ranjbar, S. & Fleming, R. M. T. Constraint-based modeling of bioenergetic differences between synaptic and non-synaptic components of human dopaminergic neurons. Front. Comput. Neurosci. 19, 594330 (2025): https://doi.org/10.3389/fncom.2025.1594330
Luo, X., Liu, Y., Balck, A., Klein, C. & Fleming, R. M. T. Identification of metabolites reproducibly associated with Parkinson’s disease via meta-analysis and computational modelling. NPJ Parkinsons Dis. 10, 126 (2024): https://doi.org/10.1038/s41531-024-00732-z
Zagare, A. et al. Omics data integration suggests a potential idiopathic Parkinson’s disease signature. Commun. Biol. 6, 1179 (2023): https://doi.org/10.1038/s42003-023-05548-w
Supplementary information
Supplementary Information (download PDF )
This includes five additional functions for use with XomicsToModel. preprocessingOmicsModel: prepares context-specific data for integration into XomicsToModel. modelPredictiveCapacity: assesses the model’s ability to predict fluxes and metabolic activities. XomicsToMultipleModels: generates an ensemble of models with variations in conditions, genetics or data. debugXomicsToModel: analyzes debug files to track changes in genes, reactions and metabolites. compareXomicsModels: identifies overlapping metabolites, reactions and genes across models. plotOverlapResults: visualizes overlaps with heat maps.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Preciat, G., Wegrzyn, A.B., Luo, X. et al. XomicsToModel: omics data integration and generation of thermodynamically consistent metabolic models. Nat Protoc (2025). https://doi.org/10.1038/s41596-025-01288-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41596-025-01288-9
This article is cited by
-
Constraint-based modelling of metabolic dysregulation in Gaucher disease: mitochondrial dysfunction and disrupted cholesterol homeostasis
Orphanet Journal of Rare Diseases (2026)