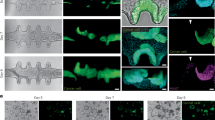
Abstract
Tumor initiation remains one of the least understood events in cancer biology, largely due to the challenge of dissecting the intricacy of the tumorigenic process in laboratory settings. The insufficient biological complexity of conventional in vitro systems makes animal models the primary experimental approach to study tumorigenesis. Despite providing valuable insights, these in vivo models function as experimental black boxes with limited spatiotemporal resolution of cellular dynamics during oncogenesis. In addition, their use raises ethical concerns, further underscoring the need for alternative ex vivo systems. Here we provide a detailed protocol to integrate state-of-the-art microfabrication, tissue engineering and optogenetic approaches to generate topobiologically complex miniature colons (‘mini-colons’) capable of undergoing tumorigenesis in vitro. We describe the key methodology for the generation of blue light-inducible oncogenic cells, the establishment of hydrogel-based mini-colon scaffolds within microfluidic devices, the development of mini-colons and the induction of spatiotemporally controlled tumorigenesis. This protocol enables the formation and long-term culture of complex cancerous tissues that capture in vivo-like tumoral biology while offering real-time and single-cell resolution analyses. It can be implemented in 4–6 weeks by researchers with prior experience in 3D cell culture techniques. We anticipate that these methodological guidelines will have a broad impact on the cancer research community by opening new avenues for tumorigenesis studies.
Key points
-
This protocol details the generation of topobiologically complex miniature colons capable of undergoing tumorigenesis in vitro. This approach uses key methodologies for the generation of blue light-inducible oncogenic cells, the establishment of hydrogel-based mini-colon scaffolds within microfluidic devices, the development of mini-colons and the induction of spatiotemporally controlled tumorigenesis.
-
The spatiotemporal resolution of cellular dynamics revealed by this approach makes it a versatile in vitro alternative to the animal models.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The laser ablation template used for the generation of hydrogel mini-colon scaffolds is available at GitHub (https://github.com/LorenzoLF/Mini-colon_bioengineering) and Zenodo35. All other relevant data can be accessed through the original publication describing the development of mini-colons for tumorigenic studies7.
References
Kapałczyńska, M. et al. 2D and 3D cell cultures—a comparison of different types of cancer cell cultures. Arch. Med. Sci. 14, 910–919 (2018).
Herms, A. et al. Self-sustaining long-term 3D epithelioid cultures reveal drivers of clonal expansion in esophageal epithelium. Nat. Genet. 56, 2158–2173 (2024).
Lorenzo-Martín, L. F. et al. VAV2 signaling promotes regenerative proliferation in both cutaneous and head and neck squamous cell carcinoma. Nat. Commun. 11, 4788 (2020).
Kim, J., Koo, B. K. & Knoblich, J. A. Human organoids: model systems for human biology and medicine. Nat. Rev. Mol. Cell Biol. 21, 571–584 (2020).
Drost, J. & Clevers, H. Organoids in cancer research. Nat. Rev. Cancer 18, 407–418 (2018).
O’Rourke, K. P. et al. Transplantation of engineered organoids enables rapid generation of metastatic mouse models of colorectal cancer. Nat. Biotechnol. 35, 577–582 (2017).
Lorenzo-Martín, L. F. et al. Spatiotemporally resolved colorectal oncogenesis in mini-colons ex vivo. Nature 629, 450–457 (2024).
Nikolaev, M. et al. Homeostatic mini-intestines through scaffold-guided organoid morphogenesis. Nature 585, 574–578 (2020).
Lorenzo-Martín, L. F. et al. Patient-derived mini-colons enable long-term modeling of tumor-microenvironment complexity. Nat. Biotechnol. 43, 727–736 (2025).
Mitrofanova, O. et al. Bioengineered human colon organoids with in vivo-like cellular complexity and function. Cell Stem Cell 31, 1175–1186.e1177 (2024).
Shih, C., Shilo, B. Z., Goldfarb, M. P., Dannenberg, A. & Weinberg, R. A. Passage of phenotypes of chemically transformed cells via transfection of DNA and chromatin. Proc. Natl Acad. Sci. USA 76, 5714–5718 (1979).
Thorne, C. A. et al. Enteroid monolayers reveal an autonomous WNT and BMP circuit controlling intestinal epithelial growth and organization. Dev. Cell 44, 624–633.e624 (2018).
Krotenberg Garcia, A. et al. Active elimination of intestinal cells drives oncogenic growth in organoids. Cell Rep. 36, 109307 (2021).
Weiswald, L. B. et al. A short-term colorectal cancer sphere culture as a relevant tool for human cancer biology investigation. Br. J. Cancer 108, 1720–1731 (2013).
Lo, Y. H., Karlsson, K. & Kuo, C. J. Applications of organoids for cancer biology and precision medicine. Nat. Cancer 1, 761–773 (2020).
van de Wetering, M. et al. Prospective derivation of a living organoid biobank of colorectal cancer patients. Cell 161, 933–945 (2015).
Carvalho, M. R. et al. Colorectal tumor-on-a-chip system: a 3D tool for precision onco-nanomedicine. Sci. Adv. 5, eaaw1317 (2019).
Brassard, J. A., Nikolaev, M., Hübscher, T., Hofer, M. & Lutolf, M. P. Recapitulating macro-scale tissue self-organization through organoid bioprinting. Nat. Mater. 20, 22–29 (2021).
Sun, H. et al. Prediction of clinical precision chemotherapy by patient-derived 3D bioprinting models of colorectal cancer and its liver metastases. Adv. Sci. 11, e2304460 (2024).
Shin, W. & Kim, H. J. 3D in vitro morphogenesis of human intestinal epithelium in a gut-on-a-chip or a hybrid chip with a cell culture insert. Nat. Protoc. 17, 910–939 (2022).
Costello, C. M. et al. Microscale bioreactors for in situ characterization of GI epithelial cell physiology. Sci. Rep. 7, 12515 (2017).
Costello, C. M. et al. 3-D intestinal scaffolds for evaluating the therapeutic potential of probiotics. Mol. Pharm. 11, 2030–2039 (2014).
Samsa, L. A., Williamson, I. A. & Magness, S. T. Quantitative analysis of intestinal stem cell dynamics using microfabricated cell culture arrays. Methods Mol. Biol. 1842, 139–166 (2018).
Jalili-Firoozinezhad, S. et al. A complex human gut microbiome cultured in an anaerobic intestine-on-a-chip. Nat. Biomed. Eng. 3, 520–531 (2019).
Cho, K. R. & Vogelstein, B. Genetic alterations in the adenoma–carcinoma sequence. Cancer 70, 1727–1731 (1992).
Meador, K. et al. Achieving tight control of a photoactivatable Cre recombinase gene switch: new design strategies and functional characterization in mammalian cells and rodent. Nucleic Acids Res. 47, e97 (2019).
Kawano, F., Okazaki, R., Yazawa, M. & Sato, M. A photoactivatable Cre-loxP recombination system for optogenetic genome engineering. Nat. Chem. Biol. 12, 1059–1064 (2016).
Duplus-Bottin, H. et al. A single-chain and fast-responding light-inducible Cre recombinase as a novel optogenetic switch. eLife https://doi.org/10.7554/eLife.61268 (2021).
Sato, T. et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459, 262–265 (2009).
Drost, J. et al. Sequential cancer mutations in cultured human intestinal stem cells. Nature 521, 43–47 (2015).
Louis, P., Hold, G. L. & Flint, H. J. The gut microbiota, bacterial metabolites and colorectal cancer. Nat. Rev. Microbiol. 12, 661–672 (2014).
Bernstein, C. et al. Carcinogenicity of deoxycholate, a secondary bile acid. Arch. Toxicol. 85, 863–871 (2011).
Wu, X. et al. Effects of the intestinal microbial metabolite butyrate on the development of colorectal cancer. J. Cancer 9, 2510–2517 (2018).
Dmitrieva-Posocco, O. et al. β-Hydroxybutyrate suppresses colorectal cancer. Nature 605, 160–165 (2022).
Lorenzo-Martín, L. F., Hübscher, T., Langer, J., Nikolaev, M. & Lutolf, M. P. Mini-colon_bioengineering. Zenodo https://doi.org/10.5281/zenodo.17120748 (2025).
Wong, C. C. & Yu, J. Gut microbiota in colorectal cancer development and therapy. Nat. Rev. Clin. Oncol. 20, 429–452 (2023).
Castejón, M. et al. Energy restriction and colorectal cancer: a call for additional research. Nutrients https://doi.org/10.3390/nu12010114 (2020).
Acknowledgements
We thank A. Bowler and F. Radtke for the inducible A, AK and AKP mouse colon organoids; M. Thomson for the original light-inducible plasmids; M. Wernig and G. Neumayer for the initial idea and work on the doxycycline- and light-inducible system; A. Aebi and B. Schneider for lentivirus production; C. Baur for discussing, designing and building the custom illumination device; A. Chrisnandy for assistance on photomask fabrication; O. Mitrofanova, B. Elçi and Y. Tinguely for assistance on microfluidic device fabrication; L. Tillard for technical support; and J. Prébandier for administrative assistance. We acknowledge support from the following EPFL core facilities: CMi, CPG, PTBTG, HCF, BIOP, FCCF, BSF and GECF.
Author information
Authors and Affiliations
Contributions
L.F.L.-M. conceived the study, designed experiments, carried out the experimental work, analyzed the data, performed artwork design and wrote the manuscript. T.H. generated the OptoCre module and blue light-associated systems, performed experimental work and analyzed data. J.L. produced the microfluidic devices and optimized hydrogel scaffolds. M.N. designed and developed the first mini-gut system. M.P.L. conceived the work, designed experiments and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The EPFL has filed for patent protection (nos. EP16199677.2, PCT/EP2017/079651, US20190367872A1) on the scaffold-guided organoid technology used here, and M.P.L. and M.N. are named as inventors on those patents. M.P.L. and M.N. are employees of Roche. The other authors declare no competing interests.
Peer review
Peer review information
Nature Protocols thanks Mehdi Nikkhah, Xiling Shen and Yuan Wang for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key references
Nikolaev, M. et al. Nature 585, 574–578 (2020): https://doi.org/10.1038/s41586-020-2724-8
Lorenzo-Martín, L. F. et al. Nature 629, 450–457 (2024): https://doi.org/10.1038/s41586-024-07330-2
Lorenzo-Martín, L. F. et al. Nat. Biotechnol. 43, 727–736 (2025): https://doi.org/10.1038/s41587-024-02301-4
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lorenzo-Martín, L.F., Hübscher, T., Langer, J. et al. Bioengineering mini-colons for ex vivo colorectal cancer research. Nat Protoc (2025). https://doi.org/10.1038/s41596-025-01292-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41596-025-01292-z