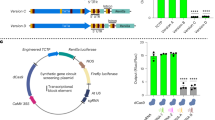
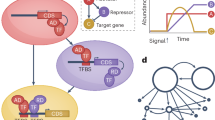
Abstract
Synthetic gene circuits are powerful tools for precisely programming gene expression and introducing novel cellular functions. However, their development and application in plants has lagged behind other systems, due mainly to the limited availability of modular genetic parts. We recently developed a CRISPR interference (CRISPRi)-based synthetic gene circuit system for programming gene expression in plants. Using a robust and high-throughput protoplast-based dual luciferase assay, we demonstrated the development, testing and functionality of these circuits in various plant species. Here we detail the key design principles and considerations for building and testing programmable and reversible CRISPRi-based gene circuits in plants. We also provide detailed procedures for isolating protoplasts from multiple plant species, including Arabidopsis thaliana, Brassica napus, Triticum aestivum and Physcomitrium patens. Furthermore, we provide step-by-step instructions for the 96-well plate-based protoplast transfection assay for testing genetic parts and synthetic circuits, using a dual luciferase assay. The detailed descriptions of these developed systems will enhance the efficiency and reproducibility of the construction, testing, and implementation of synthetic gene circuits in a variety of plant species. This protocol enables the design and testing of CRISPRi-based gene circuits in plants within ~4 weeks.
Key points
-
The protocol describes design principles for CRISPRi-based genetic circuits in plants, and outlines procedures for isolating protoplasts from different plant species and tissues, and for testing genetic parts and gene circuits using a plate-based, high-throughput, dual luciferase protoplast transfection assay.
-
CRISPRi-based circuits can integrate different signals to achieve programmable gene expression in plants, while the high-throughput protoplast-based assay permits rapid and scalable testing of circuit components across different species.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data presented in this protocol is available in the primary supporting paper4, except for Fig. 5d, which is provided as Source data. The DNA sequences for the no-input and input sgRNA-A constructs used to generate Fig. 5d are the exact same constructs used in Fig. 3h of the primary supporting paper. All plasmid DNA sequences used in this protocol are available in the Zenodo repository36, and have also been deposited to Addgene for public access under plasmid IDs 239869-239946. Source data are provided with this paper.
References
Iuchi, S. et al. Regulation of drought tolerance by gene manipulation of 9-cis-epoxycarotenoid dioxygenase, a key enzyme in abscisic acid biosynthesis in Arabidopsis. Plant J. 27, 325–333 (2001).
Hancock, J. F. A framework for assessing the risk of transgenic Crops. Bioscience 53, 512–519 (2003).
He, R. et al. Overexpression of 9-cis-epoxycarotenoid dioxygenase cisgene in grapevine increases drought tolerance and results in pleiotropic effects. Front. Plant Sci. 9, 970 (2018).
Khan, M. A. et al. CRISPRi-based circuits to control gene expression in plants. Nat. Biotechnol. 43, 416–430 (2024).
Brophy, J. A. N. et al. Synthetic genetic circuits as a means of reprogramming plant roots. Science 377, 747–751 (2022).
Lloyd, J. P. B. et al. Synthetic memory circuits for stable cell reprogramming in plants. Nat. Biotechnol. https://doi.org/10.1038/s41587-022-01383-2 (2022).
Khan, A. & Lister, R. Synthetic gene circuits in plants: recent advances and challenges. Quant. Plant Biol. 6, e6 (2025).
Lloyd, J. P. B., Khan, A. & Lister, R. The switch-liker’s guide to plant synthetic gene circuits. Plant J. 121, e70090 (2025).
Brophy, J. A. N. & Voigt, C. A. Principles of genetic circuit design. Nat. Methods 11, 508–520 (2014).
Kiani, S. et al. CRISPR transcriptional repression devices and layered circuits in mammalian cells. Nat. Methods 11, 723–726 (2014).
Gander, M. W., Vrana, J. D., Voje, W. E., Carothers, J. M. & Klavins, E. Digital logic circuits in yeast with CRISPR–dCas9 NOR gates. Nat. Commun. 8, 15459 (2017).
Vazquez-Vilar, M., Selma, S. & Orzaez, D. The design of synthetic gene circuits in plants: new components, old challenges. J. Exp. Bot. 74, 3791–380 (2023).
Wang, Y. & Demirer, G. S. Synthetic biology for plant genetic engineering and molecular farming. Trends Biotechnol. 41, 1182–1198 (2023).
Gaber, R. et al. Designable DNA-binding domains enable construction of logic circuits in mammalian cells. Nat. Chem. Biol. 10, 203–208 (2014).
Weinberg, B. H. et al. Large-scale design of robust genetic circuits with multiple inputs and outputs for mammalian cells. Nat. Biotechnol. 35, 453–462 (2017).
Santos-Moreno, J., Tasiudi, E., Stelling, J. & Schaerli, Y. Multistable and dynamic CRISPRi-based synthetic circuits. Nat. Commun. 11, 2746 (2020).
Kar, S. et al. Orthogonal control of gene expression in plants using synthetic promoters and CRISPR-based transcription factors. Plant Methods 18, 42 (2022).
Guiziou, S., Maranas, C. J., Chu, J. C. & Nemhauser, J. L. An integrase toolbox to record gene-expression during plant development. Nat. Commun. 14, 1844 (2023).
Schaumberg, K. A. et al. Quantitative characterization of genetic parts and circuits for plant synthetic biology. Nat. Methods 13, 94 (2015).
Ming, M. et al. CRISPR–Cas12b enables efficient plant genome engineering. Nat. Plants 6, 202–208 (2020).
Fujikawa, Y. et al. Split luciferase complementation assay to detect regulated protein-protein interactions in rice protoplasts in a large-scale format. Rice 7, 11 (2014).
Rigoulot, S. B. et al. Automated, high-throughput protoplast transfection for gene editing and transgene expression studies. Methods Mol. Biol. 2653, 129–149 (2023).
Moreno-Giménez, E., Selma, S., Calvache, C. & Orzáez, D. GB_SynP: a modular dCas9-regulated synthetic promoter collection for fine-tuned recombinant gene expression in plants. ACS Synth. Biol. 11, 3037–3048 (2022).
Calvache, C., Vazquez-Vilar, M., Moreno-Giménez, E. & Orzaez, D. A quantitative autonomous bioluminescence reporter system with a wide dynamic range for plant synthetic biology. Plant Biotechnol. J. 22, 37–47 (2024).
Rademacher, T. et al. Plant cell packs: a scalable platform for recombinant protein production and metabolic engineering. Plant Biotechnol. J. 17, 1560–1566 (2019).
Jusiak, B. et al. Synthetic gene circuits. In Encyclopedia of Molecular Cell Biology and Molecular Medicine Vol. 4 (ed. Meyers, R. A.) 1–56 (Wiley–VCH, 2014).
Jusiak, B., Cleto, S., Perez-Piñera, P. & Lu, T. K. Engineering synthetic gene circuits in living cells with CRISPR technology. Trends Biotechnol. 34, 535–547 (2016).
Haurwitz, R. E., Jinek, M., Wiedenheft, B., Zhou, K. & Doudna, J. A. Sequence- and structure-specific RNA processing by a CRISPR endonuclease. Science 329, 1355–1358 (2010).
Goodstein, D. M. et al. Phytozome: a comparative platform for green plant genomics. Nucleic Acids Res. 40, D1178–D1186 (2012).
Shahmuradov, I. A., Umarov, R. K. & Solovyev, V. V. TSSPlant: a new tool for prediction of plant Pol II promoters. Nucleic Acids Res. 45, e65 (2017).
Lai, H.-Y. et al. IProEP: a computational predictor for predicting promoter. Mol. Ther. Nucleic Acids 17, 337–346 (2019).
Umarov, R., Kuwahara, H., Li, Y., Gao, X. & Solovyev, V. Promoter analysis and prediction in the human genome using sequence-based deep learning models. Bioinformatics 35, 2730–2737 (2019).
Gibson, D. G. et al. Enzymatic assembly of DNA molecules up to several hundred kilobases. Nat. Methods 6, 343–345 (2009).
Engler, C. et al. A golden gate modular cloning toolbox for plants. ACS Synth. Biol. 3, 839–843 (2014).
Cove, D. J. et al. The moss Physcomitrella patens: a novel model system for plant development and genomic studies. Cold Spring Harb. Protoc. 2009, db.emo115 (2009).
Khan, M. A. et al. CRISPRi-based circuits to control gene expression in plants. Zenodo https://doi.org/10.5281/zenodo.11108565 (2024).
Swain, T. et al. A modular dCas9-based recruitment platform for combinatorial epigenome editing. Nucleic Acids Res. 52, 474–491 (2024).
Cano-Rodriguez, D. et al. Writing of H3K4Me3 overcomes epigenetic silencing in a sustained but context-dependent manner. Nat. Commun. 7, 12284 (2016).
Konermann, S. et al. Transcriptome engineering with RNA-targeting type VI-D CRISPR effectors. Cell 173, 665–676.e14 (2018).
Garcia-Bloj, B. et al. Waking up dormant tumor suppressor genes with zinc fingers, TALEs and the CRISPR–dCas9 system. Oncotarget 7, 60535–60554 (2016).
Shechner, D. M., Hacisuleyman, E., Younger, S. T. & Rinn, J. L. Multiplexable, locus-specific targeting of long RNAs with CRISPR-Display. Nat. Methods 12, 664–670 (2015).
Amabile, A. et al. Inheritable silencing of endogenous genes by hit-and-run targeted epigenetic editing. Cell 167, 219–232.e14 (2016).
Gilbert, L. A. et al. CRISPR-mediated modular RNA-guided regulation of transcription in eukaryotes. Cell 154, 442–451 (2013).
Maeder, M. L. et al. CRISPR RNA-guided activation of endogenous human genes. Nat. Methods 10, 977–979 (2013).
Qi, L. S. et al. Repurposing CRISPR as an RNA-guided platform for sequence-specific control of gene expression. Cell 152, 1173–1183 (2013).
Bikard, D. et al. Programmable repression and activation of bacterial gene expression using an engineered CRISPR-Cas system. Nucleic Acids Res. 41, 7429–7437 (2013).
Ohta, M., Matsui, K., Hiratsu, K., Shinshi, H. & Ohme-Takagi, M. Repression domains of class II ERF transcriptional repressors share an essential motif for active repression. Plant Cell 13, 1959–1968 (2001).
Hiratsu, K., Matsui, K., Koyama, T. & Ohme-Takagi, M. Dominant repression of target genes by chimeric repressors that include the EAR motif, a repression domain, in Arabidopsis. Plant J. 34, 733–739 (2003).
Liu, H. et al. CRISPR-P 2.0: an improved CRISPR–Cas9 tool for genome editing in plants. Mol. Plant 10, 530–532 (2017).
Xie, X. et al. CRISPR-GE: a convenient software toolkit for CRISPR-based genome editing. Mol. Plant 10, 1246–1249 (2017).
Park, J., Bae, S. & Kim, J.-S. Cas-Designer: a web-based tool for choice of CRISPR–Cas9 target sites. Bioinformatics 31, 4014–4016 (2015).
Tian, F., Yang, D.-C., Meng, Y.-Q., Jin, J. & Gao, G. PlantRegMap: charting functional regulatory maps in plants. Nucleic Acids Res. 48, D1104–D1113 (2020).
Fornes, O. et al. JASPAR 2020: update of the open-access database of transcription factor binding profiles. Nucleic Acids Res. 48, D87–D92 (2020).
Chow, C.-N. et al. PlantPAN3.0: a new and updated resource for reconstructing transcriptional regulatory networks from ChIP-seq experiments in plants. Nucleic Acids Res. 47, D1155–D1163 (2019).
Bae, S., Park, J. & Kim, J.-S. Cas-OFFinder: a fast and versatile algorithm that searches for potential off-target sites of Cas9 RNA-guided endonucleases. Bioinformatics 30, 1473–1475 (2014).
Acknowledgements
This work was supported by the following grants to R.L.: Australian Research Council (ARC) Center of Excellence in Plants for Space (CE230100015), ARC Discovery Project grants (DP210103954 and DP240103385), and National Health and Medical Research Council Investigator Grants (GNT1178460 and GNT2035042).
Author information
Authors and Affiliations
Contributions
A.K. and R.L. designed the study, conceived and developed the CRISPRi-based gene circuits system and wrote the manuscript. A.K. optimized the procedure for isolating protoplasts from A. thaliana leaves, and established the 96-well protoplast transfection protocol. A.K. and G.H. optimized protoplast isolation from T. aestivum and B. napus leaves, while G.H. and M.P. refined the protocol for isolating protoplasts from A. thaliana roots. J.Y.Z. optimized protoplast isolation from P. patens. All authors reviewed, contributed to and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Protocols thanks Alexander Leydon and the other, anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key references
Khan, M. A., Herring, G., Zhu, J. Y. et al. Nat. Biotechnol. 43, 416–430 (2025): https://doi.org/10.1038/s41587-024-02236-w
Lloyd, J. P. B., Ly, F., Gong, P. et al. Nat. Biotechnol. 40, 1862–1872 (2022): https://doi.org/10.1038/s41587-022-01383-2
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–3, Video 1 and Description of stock solutions.
Supplementary Video 1 (download MOV )
Leaf peeling procedure for protoplast isolation.
Source data
Source Data Fig. 5 (download XLSX )
Statistical source data for Fig. 5d.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Khan, A., Herring, G., Zhu, J.Y. et al. Designing and testing CRISPRi-based synthetic gene circuits in plants. Nat Protoc (2026). https://doi.org/10.1038/s41596-025-01312-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41596-025-01312-y